Abstract
Primary aldosteronism (PA) is a common form of secondary hypertension and has significant cardiovascular consequences. Mutated channelopathy due to the activation of calcium channels has been recently described in aldosterone-producing adenoma (APA). The study involved 148 consecutive PA patients, (66 males; aged 56.3 ± 12.3years) who received adrenalectomy and were collected from the Taiwan PA investigator (TAIPAI) group. A high rate of somatic mutation in APA was found (n = 91, 61.5%); including mutations in KCNJ5 (n = 88, 59.5%), ATP1A1 (n = 2, 1.4%) and ATP2B3 (n = 1, 0.7%); however, no mutations in CACNA1D were identified. Mutation-carriers were younger (<0.001), had lower Cyst C (p = 0.042), pulse wave velocity (p = 0.027), C-reactive protein (p = 0.042) and a lower rate of proteinuria (p = 0.031) than non-carriers. After multivariate adjustment, mutation carriers had lower serum CRP levels than non-carriers (p = 0.031. Patients with mutation also had a greater chance of recovery from hypertension after operation (p = 0.005). A high incidence of somatic mutations in APA was identified in the Taiwanese population. Mutation-carriers had lower CRP levels and a higher rate of cure of hypertension after adrenalectomy. This raises the possibility of using mutation screening as a tool in predicting long-term outcome after adrenalectomy.
Similar content being viewed by others
Introduction
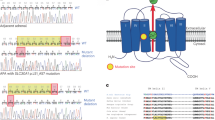
Primary aldosteronism (PA), characterized by the autonomous production of aldosterone, is a common potentially curable cause of secondary hypertension1. It affects 5–13% of patients with hypertension in specialized centers2,3. PA patients have a high incidence of cardiovascular events in comparison with essential hypertension (EH) patients4,5. Somatic mutations in the selectivity filter of the KCNJ5 channel in APA result in a loss of selectivity for potassium and the entry of sodium, resulting in membrane depolarization, calcium mobilization, increased CYP11B2 expression and aldosteronism6. Further exome sequencing of aldosterone producing adenoma (APA) has identified ATPase mutations in ATP1A1 and ATP2B3, which could lead to the opening of voltage-dependent calcium (Ca2+) channels7,8. The CACNA1D mutation induced a shift in voltage-dependent gating to a more negative voltage, again implicating gain-of-function Ca2+ channel mutations in APAs9,10. Therefore, aldosterone secretion is regulated by the augmentation of intracellular calcium levels via channelopathies and pumps, which lead to depolarization of zona glomerulosa cells in the adrenal cortex11,12.
Although aldosterone is now recognized as an increasingly important contributor to cardiometabolic pathology via informatory non-genomic pathways, in addition to its classically described genomic role in sodium and volume-related regulation13,14, its effect on cardio-metabolic factors in patients harboring these mutations has yet to be characterized. Previous findings are contradictory with regard to whether KCNJ5 mutations translate into a specific phenotype15. In this study, we have analyzed 148 APAs, diagnosed and surgically removed by the Taiwan Primary Aldosteronism investigator (TAIPAI) study group, for somatic mutations in KCNJ5, ATP1A1, ATP2B3 and CACNA1D. We searched for clinical correlations and determined the impact on patients who harbored these mutations, focusing on their cardio-metabolic properties.
Materials and methods
Ethics Statement
The study complied with the Declaration of Helsinki and was approved by the institutional review board of National Taiwan University Hospital (Taipei, Taiwan) (No. 200611031R). All participants signed the informed consent form before inclusion in the study. All of the methods were carried out in “accordance” with the approved guidelines ( http://www.nature.com/srep/policies/index.html#experimental-subjects).
Identification of PA
The present study was based on the Taiwan Primary Aldosteronism Investigation (TAIPAI) database and tissue bank16,17,18. The database was constructed from June 2008 to March 2011 for quality assurance at 2 medical centers and their 3 affiliated hospitals and two local hospitals in different cities in Taiwan19. Before confirmatory tests, all antihypertensive medications were discontinued for at least 21 days. Diltiazem and/or doxazosin were administered to control markedly high blood pressure when required1. Patients with an abnormal aldosterone-renin ratio (ARR) were confirmed with PA by saline infusion tests and subsequently underwent imaging studies for subtype identification (figures S1 and S2).
PA confirmation and subtype studies were established in hypertensive patients according to the standard protocol of TAIPAI59, including adrenal venous sampling and NP-59 scintigraphy with SPECT-CT imaging16,17,18,20 (see the supplementary file). Patients who were diagnosed with family type I (FH-I)/GRA were excluded via long-range polymerase chain reaction, as described previously21.
Functional survey
The aldosterone concentration was measured by radioimmunoassay using a commercial kit (Aldosterone Maia Kit, Adaltis Italia S.P.A., Bologna, Italy)22 and PRA was measured by the generation of angiotensin I in vitro using a commercially available radioimmunoassay kit (DiaSorin, Stillwater, MN, USA)19. The amount of daily protein loss was defined as the urinary microalbumin-to-creatinine ratio (mg/mg); the cardiovascular marker cystatin C11 was measured using a particle-enhanced immunonephelometric assay (N Latex Cystatin C; Siemens, Berlin, Germany) with a nephelometer (BNII; Siemens)23. The pulse wave velocity (PWV) was measured with the subject in a supine position after a 15-min rest using an automatic waveform analyzer (Colin VP-2000, Omeron Inc., Japan), as previously reported24.
A Hewlett-Packard Sonos 5500 ultrasound system equipped with an S3 transducer was used for Echocardiography, including two-dimensional, M-mode and Doppler ultrasound recordings. Left atrial diameter and left ventricular ejection fraction (M-mode) were measured via the parasternal long-axis view, as in our previous report25. Left ventricular mass (LVM) index (LVMI) was calculated according to the method of Devereux et al.26.
Adrenalectomy
The indication for adrenalectomy is according to AVS (n = 65) or NP 59 (n = 88) lateralization (supplementary file). All of the operations were performed using the lateral trans-peritoneal approach and were performed by a single experienced laparoscopic surgeon to ensure that the principles of adrenal gland surgery were strictly followed27.
Sequencing
Nucleic acid extraction
The adrenal tumors obtained were fresh-frozen and stored at −72 oC. Genomic DNA was extracted from 148 paired peritumoral adrenal cortices in addition to each peripheral DNA sample. Tumor DNA was extracted using a QIAamp DNA mini kit (Qiagen, Hilden, Germany); total RNA was isolated from frozen tissue using Trizol (Invitrogen, Carlsbad, Ca, USA) and then cleaned-up using the GENEzol TriRNA Pure Kit (Geneaid, New Taipei City, Taiwan). After DNaseI treatment (Invitrogen, Carlsbad, CA, USA), 500 ng of total RNA were reverse-transcribed using Moloney Murine Leukemia Virus Reverse Transcriptase (M-MLV RT) (Promega, Madison, WI, USA) and random hexamers (Promega, Madison, WI, USA) according to the manufacturer’s instructions.
Somatic mutation sequencing
The coding region of the genomic DNA was investigated by exome sequencing. The entire coding sequence (exons 2–3) and flanking regions of KCNJ5 were amplified and sequenced using gene-specific primers, as previously reported28. Accordingly, the PCR primers used to amplify fragments for direct sequencing of ATP1A1/ATP2B3 and CACNAD1 also followed previous reports7,9,29 (listed in table S1). The annealing temperature was 58 °C. Direct sequencing of PCR products was performed using The BigDye® Terminator v3.1 Cycle Sequencing Kit (Applied Biosystems, Foster City, USA) with a 3730 DNA Analyzer (Applied Biosystems, Foster City, USA).
Outcome measurement
Patients were followed monthly for the first three months after the operation and then every three months. Assessment of cure rate of hypertension has been described previously30. Briefly, hypertension was considered cured if 75% of their recorded systolic BP was <140 mmHg and their diastolic BP was <90 mmHg with no use of anti-hypertensive medications for at least 1 year after adrenalectomy30,31.
Statistical analysis
Statistical analyses were performed with R software, version 2.8.1 (Free Software Foundation, Inc., Boston, MA, U.S.A.). A normal distribution was attained by the appropriate transformations of skewed variables such as C-reactive protein (CRP) and ARR. Logistic regression analysis with a stepwise variable selection procedure was applied using available variables (body mass index (BMI), diabetes mellitus, preoperative aldosterone, potassium blood pressure, kidney function, CRP, proteinuria, somatic mutation result) to identify important factors associated with postoperative residual hypertension. The goodness-of-fit (GOF) of the fitted multiple logistic regression model was assessed by the estimated area under the receiver operating characteristic (ROC) curve, the adjusted generalized R2 and the Hosmer-Lemeshow GOF test. Furthermore, LOESS curves32 were drawn to evaluate the relationship between age with CRP and PWV.
Because of dis-equivalent ages and CRP between groups and to display the implications of variables for individual patients, a generalized additive model (GAM) (with spline) incorporating the subject-specific (longitudinal) random effects was plotted, adjusted for the factors listed in Table 1. We used a transformation of the β coefficients [100* (eβ −1)] to obtain the percentage increase in the outcomes per SD increase in variables. This approach permits adjustments for possible nonlinear effects of continuous variables24,33.
All data were expressed as the mean ± standard deviation (SD). A p-value of <0.05 was considered significant.
Results
Patient characteristics
Demographics of Patients (Table 1)
Of the 148 patients (66 males; aged 56.3 ± 12.3years) identified who received adrenalectomy, a high rate of somatic mutation was found (n = 91, 61.5%), most of which were KCNJ5 mutations (n = 88, 59.5%).
Sequencing of adenoma samples demonstrated the occurrence of the p.Gly151Arg (c.451G > A or c.451G > C) (n = 45), the p.Leu168Arg (c.503T > G)(n = 41), p.Ile157del (c.470_472delTCA) (n = 1) and p.Thr158Ala(c.472A > G) (n = 1) mutations in the heterozygous state. Two adenomas had nucleotide substitutions in ATP1A1 (p.Leu104Arg [c.311 T > G]) and one in ATP2B3 (p.Tyr410Asp [c. 1228 T > G]) (supplementary table 2).
There was no CACNA1D mutation found in our specimens. The absence of KCNJ5 mutations in all peripheral DNA samples and paired peritumoral cortices confirmed the somatic nature of the genetic alteration.
Mutation carriers were younger (p < 0.001) compared with their mutation-free counterparts. The basal levels of serum PAC and the aldosterone to renin ratio (ARR) were higher, while potassium was lower in mutation-carriers than in non-carriers (all p < 0.001). Patients with these mutations were more likely to demonstrate a higher glomerular filtration rate (p = 0.001), estimated by the CKD-Epi formula, than mutation-free patients.
Cardiometabolic parameters
Cyst C (p = 0.042), PWV (p = 0.027), CRP (p = 0.042) and proteinuria (p = 0.031) were lower in mutation-carriers than in the non-carriers group. However, the preoperative LV mass and LVEF were not significantly different between the groups. To compare these vascular parameters further between the mutation-carriers and non-carriers, we generated Loess plots stratified by chronological age with objects. In terms of the relationship of CRP to each age, the mutation-carriers had a lower CRP than the non-carriers after multivariate correlation (p = 0.031, Fig. 1). In the almost linear relationship between age and PWV, there was a steep increase in PWV up to 50 years in both groups. The relationship between PWV or Cystatin C and age showed no difference between the mutation-carriers and non-carriers after multivariate adjustments (all p > 0.05).
Scatter plots with an adjusted spline of age with (a) C reactive protein (CPR), ( p = 0.031 between mutation-carriers and non-carriers after multivariate adjustments) (b) Cystatin C (Cys C) and (c) pulse wave velocity (PWV). The plots were incorporated with the subject-specific (longitudinal) random effects to predict the association. The probability of proteinuria was constructed with chronological age (d) and is centered to have an average of zero over the range of the data as constructed with the generalized additive model (GAM). *Adjusted by sex, diabetes mellitus, body mass index (BMI), serum aldosterone, potassium, estimated glomerular filtration rate, mean blood pressure. **All plots were constructed and stratified by patients with somatic mutation and non-mutation.
The probability of proteinuria associated with the chronological age of the aldosteronism patients at diagnosis was constructed with the GAM. The prevalence of proteinuria was also not different between the two groups after adjustment. Age did not relate to having proteinuria in either group (all p > 0.05).
Factors predicting post-operative residual hypertension
After unilateral adrenalectomy, hypertension was cured in 125 (84.5%) patients. Most (n = 100, 80%) of the cured patients became normotensive within 6 months after surgery; twenty patients (16%) became normotensive after 9 months and 5 took up to 1 year. Of the 23 patients with residual hypertension, 5 continued using the same antihypertensive agents.
Compared with wild type individuals, the mutation-carriers had a greater chance of recovery from hypertension (92.3% vs 71.9%, p = 0.002) after a one year follow-up. Adjusted for possible variables, being a mutation-carrier [OR, 0.08, 95% CI, 0.01– 0.47, p = 0.005] was an independent factor predicting postoperative cure of hypertension (Table 2). However, preoperative logCRP [OR, 8.12, 95% CI, 1.12– 58.99, p = 0.039] and proteinuria [OR, 8.12, 95% CI, 1.02– 24.34, p = 0.048] were risks predicting postoperative residual hypertension. The final multiple logistic regression model had a high discriminatory power (estimated area under the curve of receiver operating characteristics [eAUC-ROC] = 0.807) and fitted the observed binary data well (adjusted generalized R2 = 0.349 and Hosmer-Lemeshow GOF test p = 0.074). The GAM curve was further plotted to show the differentiation of residual hypertension between mutation-carriers and non-carriers. Stratified by chronological age at diagnosis, non-carriers had a greater probability of residual hypertension than patients with a mutation after adrenalectomy in the model (Fig. 2a). This result was consistent in patients stratified by baseline CRP level; patients without a mutation had a higher probability of residual hypertension (Fig. 2b).
Generalized additive model (GAM) with the spline of (a) chronological age at diagnosis and (b) baseline c-reactive protein (CRP) level, incorporating the subject-specific (longitudinal) random effects, were plotted to predict the residual hypertension after adrenalectomy. The curve was centered to have an average of zero over the range of the data. *Adjusted by sex, diabetes mellitus, body mass index (BMI), serum aldosterone, potassium, estimated glomerular filtration rate, mean blood pressure. Figures 2a, the GAM figures of correlation with age and residual hypertension after adrenalectomy. Figures 2b, the GAM figures of correlation with CRP and outcome effects. (The response scale reflects natural transformation).
Discussion
The main finding of this multicenter study is that nearly two thirds of these Taiwanese APA patients had candidate somatic mutations, almost all in KCNJ5 and the prevalence was higher than previously reported in Caucasian subjects29,34. There were no gender differences in mutation carriers or non-carriers. Our results reinforce the view that patients with somatic mutations are younger; however, mutation carriers and non-carriers had similar degrees of arterial stiffness, proteinuria and left ventricular hypertrophy after multivariate adjustment, suggesting that, despite mutation carriers having an earlier onset of the disease, this does not translate into a unique cardiovascular phenotype35,36. Most importantly, we demonstrated that those patients with somatic mutation have a lower level of pro-inflammatory status independent of chronological age and are more likely to be cured post-operatively.
Mutation Carriers
The prevalence of being a mutation carrier was 61.5% among adenomas, most of which were in KCNJ5 (59.5%); this is a higher rate than previously reported29,36. We showed that KCNJ5 mutations are the most frequent genetic cause of excessive aldosterone production in Eastern Asian patients with APA. Heterozygous mutations in the conservative filter of KCNJ5 at p.Gly151Arg were the most common channelopathy in our cohort. There was no mutated CACNA1D identified. To date, the three categories of mutation in adenoma have been mutually exclusive. Likewise, nearly three-fifths of our patients had a KCNJ5 mutation, meaning that it was less likely that there would be other mutations. In contrast, the use of AVS in a relative minority of patients with PA (43.9%) could have been responsible for the extremely low prevalence of CACNA1D mutations in our cohort. In fact, smaller nodular dimensions were found in patients harboring CACNA1D mutations10.
Indeed, both the current and previous studies have shown that mutation carriers had higher preoperative plasma aldosterone levels and lower potassium compared with patients without mutation35,36,37. This may explain why mutation carriers were likely to be diagnosed at a younger age. However, our findings could not evaluate the possibility that the etiology of PA diagnosed at a younger age may be attributed to mutations. We found no significant differences in preoperative blood pressure, body mass or tumor size between the two groups. This suggests that harboring a mutation brings the presentation of aldosteronism forward and may be indicative of a more florid phenotype.
However, non-somatic mutation carriers had higher C-reactive protein levels than mutation carriers. Although recently identified mutations in the Kir3.4 (GIRK4) gene coding for KCNJ5 channels were shown to represent a key mechanism for the aldosterone excess in part of APA patients, the mechanism underlying the aldosterone overproduction in non-mutation carriers is still not clear. The overproduction of oxidative stress in aldosteronism38,39 is not only attributed to cardiovascular injury but could have an impact on redox-activation of G-protein-activated inward rectifier K+ channels40 in non-mutation carriers, at least partially as a key pathophysiology in oxidative stress-associated aldosterone secretion. It was found that G-protein-activated, inwardly rectifying K+ channel subunit GIRK is a potential target for oxidative stress40. To date, a variety of membrane-bound proteins like Na+/K+-ATPase or adenylate cyclase are known to be affected by reactive oxygen species (ROS) and are probably involved in noxious Ca2+ overload41,42. In particular, K+-selective channels are prone to operational modulation by ROS and redox regulation of K+ channels as a mechanism that provides a link between cellular metabolism and excitability43,44. The mechanism of high CRP in non-mutation carriers and its impact on channelopathy is still not clear; therefore, the hypothesis that oxidative stress is a causative factor for the generation of autonomous aldosterone production still needs further study.
Factors related to residual hypertension after adrenalectomy
Importantly, we found that mutation-carriers had better odds of recovery from hypertension after adrenalectomy, a finding which is in concordance with two recent smaller studies from Western Norway45 and a center in Australia46. Patients with KCNJ5 mutations were reported to have a higher likelihood of achieving regression of LVH35. Our results showed that among younger patients receiving adrenalectomy, mutation carriers were more likely to be cured of hypertension, while non-carriers often had residual hypertension, despite adjusting for age (Fig. 2a. GAM plot). This result was especially true for younger ages and was therefore not only age-related essential hypertension. We have further shown that patients who have proteinuria will have a lower chance of recovery from hypertension after adrenalectomy. Heavy proteinuria in PA patients, which is associated with less renin suppression47, showed a lower possibility of recovery from kidney damage after adrenalectomy47.
Cardiometabolic parameters
Patients harboring mutations had lower cystatin C, PWV and CRP levels and a lower percentage of proteinuria than non-carriers. However, there were no differences in cystatin C, proteinuria and PWV between groups after adjustments in our final model. These findings contradict the contention that PA mutations do not translate into a specific phenotype36. However, in the multivariate model, non-carriers had higher CRP than mutation carriers and CRP could predict post-operative residual hypertension. It has been noted that high pro-inflammatory status is related to vascular hypertrophy48,49, endothelial dysfunction50 and cardio-metabolic syndrome51.
Several mechanisms have been proposed to explain residual hypertension after adrenalectomy, such as endothelial dysfunction, vascular damage and arteriolosclerosis52,53. In a European report, non-carriers in the APA were less likely to achieve regression of left ventricle hypertrophy35, partially reflecting their long-term exposure to a higher pro-inflammation status. It has been shown that aldosterone promotes oxidative stress through a non-genomic effect and is mineralocorticoid receptor (MR)-independent54, i.e. cannot be attenuated by the classic MR-blocker spironolactone. Importantly, the effect of mutated channelopathy and pumps on CRP levels persisted after adjustments for level of PAC and potassium level. Thus, it appears that the possibility of identifying APA patients who are at risk of developing a pro-inflammatory status early is attractive because the cardiovascular risks could be reversed and hypertension cured by further targeted management in the early stages of disease. Future studies should determine the long-term cardiometabolic events in patients with or without mutations, especially the effect of non-genomic actions.
Higher prevalence of mutation-carriers
In contrast to previous studies conducted in other ethnic groups, mutations were detected in a relatively higher percentage of Taiwanese patients; also, the patterns of mutations were shown to be different. The largest cohort from Asia in this study and also that in Japanese reports37,55,56 with a higher prevalence of mutation-carriers showed no gender differences, which is different to Caucasian reports6,29,36. Although a plausible explanation for the difference in the phenotype between sexes has been reported to be the androgen-regulated compensatory expression of TASK3 channels, our results did not show a sex difference phenotype in Eastern Asian patients. This suggests the presence of epidemiological differences between Asian and Western populations. This high frequency of KCNJ5 mutations may partially contribute to the increased prevalence of adenoma compared with hyperplasia in Asian individuals17,55,57. The variations in phenotypes resulted from different mutations in KCNJ5 also have implications for the potential channelopathy of PA15. Under the standard diagnostic implementation criteria and possessing the same ethnic background, the patients enrolled and sample collections in this study were the same between the centers involved. Using standard methods, with the higher percentage of APA identified17, the higher rate of KCNJ5 mutations in our adenomas is less likely to be related to differences in the approach and treatment methods17.
Although unilateral adrenalectomy represents the treatment of choice for lateralized PA, it is tempting to speculate that channelopathy and pumps may become interesting new drug targets for some subgroups of APAs that are not eligible for surgery, e.g. blocked by verapamil58, especially in areas with a higher prevalence of mutation-carriers.
There are some limitations of this study. As most of the mutations were in KCNJ5, with a small number of patients with ATPase mutations, but no CACNA1D mutations in our samples, analysis of clinical and biological correlates of these rare mutations could not be performed. The role of sex hormones such as estrogen and progesterone on channelopathy and pumps in PA patients warrants further study. Prospective studies evaluating the predictive value of mutation screening for outcomes in patients undergoing adrenalectomy for PA will also be necessary.
Conclusions
We sought to provide a genotype-phenotype perspective in correlating mutated status with clinical outcome and cardio-metabolic parameters used in APA patients from an area with high mutation rates. Lower CRP levels in mutation-carriers relates to a higher rate of cure of hypertension after adrenalectomy. It also raises the possibility of using mutation screening as tool in determining long-term outcome after surgery.
Additional Information
How to cite this article: Wu, V.-C. et al. Prevalence and clinical correlates of somatic mutation in aldosterone producing adenoma -Taiwanese population. Sci. Rep. 5, 11396; doi: 10.1038/srep11396 (2015).
References
Wu, K. D. et al. Preoperative diagnosis and localization of aldosterone-producing adenoma by adrenal venous sampling after administration of metoclopramide. J Formos Med Assoc 100, 598–603 (2001).
Young, W. F., Jr. Minireview: primary aldosteronism–changing concepts in diagnosis and treatment. Endocrinology 144, 2208–2213 (2003).
Rossi, G. P. et al. A prospective study of the prevalence of primary aldosteronism in 1,125 hypertensive patients. Journal of the American College of Cardiology 48, 2293–2300 (2006).
Yang, N. P., Chan, C. L., Yu, I. L., Lee, C. Y. & Chou, P. Estimated prevalence of orthopaedic fractures in Taiwan–A cross-sectional study based on nationwide insurance data. Injury 41, 1266–1272 (2010).
Petramala, L. et al. Bone and mineral metabolism in patients with primary aldosteronism. International journal of endocrinology 2014, 836529 (2014).
Choi, M. et al. K+ channel mutations in adrenal aldosterone-producing adenomas and hereditary hypertension. Science (New York, N.Y 331, 768–772 (2011).
Beuschlein, F. et al. Somatic mutations in ATP1A1 and ATP2B3 lead to aldosterone-producing adenomas and secondary hypertension. Nature genetics 45, 440–444 (2013).
Gomez-Sanchez, C. E., Kuppusamy, M. & Gomez-Sanchez, E. P. Somatic mutations of the ATP1A1 gene and aldosterone-producing adenomas. Molecular and cellular endocrinology 14, S0303–7207 (2014).
Scholl, U. I. et al. Somatic and germline CACNA1D calcium channel mutations in aldosterone-producing adenomas and primary aldosteronism. Nature genetics 45, 1050–1054 (2013).
Azizan, E. A. et al. Somatic mutations in ATP1A1 and CACNA1D underlie a common subtype of adrenal hypertension. Nature genetics 45, 1055–1060 (2013).
Angelidis, C. et al. Cystatin C: an emerging biomarker in cardiovascular disease. Current topics in medicinal chemistry 13, 164–179 (2013).
Al-Salameh, A., Cohen, R. & Desailloud, R. Overview of the genetic determinants of primary aldosteronism. The application of clinical genetics 7, 67–79 (2014).
Williams, J. S. Evolving research in nongenomic actions of aldosterone. Curr Opin Endocrinol Diabetes Obes 20, 198–203 (2013).
Monticone, S., Else, T., Mulatero, P., Williams, T. A. & Rainey, W. E. Understanding primary aldosteronism: impact of next generation sequencing and expression profiling. Molecular and cellular endocrinology 399, 311–320 (2015).
Scholl, U. I. et al. Hypertension with or without adrenal hyperplasia due to different inherited mutations in the potassium channel KCNJ5. Proceedings of the National Academy of Sciences of the United States of America 109, 2533–2538 (2012).
Wu, V. C. et al. Primary Aldosteronism: Diagnostic Accuracy of the Losartan and Captopril Tests. Am J Hypertens 22, 821–827 (2009).
Kuo, C. C. et al. Verification and evaluation of aldosteronism demographics in the Taiwan Primary Aldosteronism Investigation Group (TAIPAI Group). J Renin Angiotensin Aldosterone Syst 12, 348–357 (2011).
Chao, C. T. et al. Diagnosis and management of primary aldosteronism: an updated review. Annals of medicine 45, 375–383 (2013).
Wu, C. H. et al. Comparison of 24-h urinary aldosterone level and random urinary aldosterone-to-creatinine ratio in the diagnosis of primary aldosteronism. PloS one 8, e67417 (2013).
Sechi, L. A. et al. Long-term renal outcomes in patients with primary aldosteronism. Jama 295, 2638–2645 (2006).
Mulatero, P. et al. Recombinant CYP11B genes encode enzymes that can catalyze conversion of 11-deoxycortisol to cortisol, 18-hydroxycortisol and 18-oxocortisol. J Clin Endocrinol Metab 83, 3996–4001 (1998).
Wu, V. C. et al. Diagnosis of primary aldosteronism: Comparison of post-captopril active renin concentration and plasma renin activity. Clin Chim Acta 411, 657–663 (2010).
Wu, V. C. et al. Primary aldosteronism: changes in cystatin C-based kidney filtration, proteinuria and renal duplex indices with treatment. J Hypertens 29, 1778–1786 (2011).
Wu, V. C. et al. Endothelial Progenitor Cells in Primary Aldosteronism: A Biomarker of Severity for Aldosterone Vasculopathy and Prognosis. J Clin Endocrinol Metab 96, 3175–3183 (2011).
Lin, Y. H. et al. The association of serum potassium level with left ventricular mass in patients with primary aldosteronism. European journal of clinical investigation 41, 743–750 (2011).
Devereux, R. B. & Reichek, N. Echocardiographic determination of left ventricular mass in man. Anatomic validation of the method. Circulation 55, 613–618 (1977).
Chen, S. F. et al. Clinical Outcomes in Patients Undergoing Laparoscopic Adrenalectomy for Unilateral Aldosterone Producing Adenoma: Partial Versus Total Adrenalectomy. Journal of endourology / Endourological Society 28, 1103–1106 (2014).
Azizan, E. A. et al. Somatic Mutations Affecting the Selectivity Filter of KCNJ5 Are Frequent in 2 Large Unselected Collections of Adrenal Aldosteronomas. Hypertension (2012).
Fernandes-Rosa, F. L. et al. Genetic spectrum and clinical correlates of somatic mutations in aldosterone-producing adenoma. Hypertension 64, 354–361 (2014).
Wu, V. C. et al. Association of kidney function with residual hypertension after treatment of aldosterone-producing adenoma. Am J Kidney Dis 54, 665–673 (2009).
Catena, C. et al. Long-term cardiac effects of adrenalectomy or mineralocorticoid antagonists in patients with primary aldosteronism. Hypertension 50, 911–918 (2007).
Cleveland, W. S. Robust locally weighted regression and smoothing scatterplots. J Am Stat Assoc 74, 829–836 (1979).
Woo, M. J., Reiter, J. P. & Karr, A. F. Estimation of propensity scores using generalized additive models. Statistics in medicine 27, 3805–3816 (2008).
Mulatero, P. et al. Prevalence and characteristics of familial hyperaldosteronism: the PATOGEN study (Primary Aldosteronism in TOrino-GENetic forms). Hypertension 58, 797–803 (2011).
Rossi, G. P. et al. KCNJ5 gene somatic mutations affect cardiac remodelling but do not preclude cure of high blood pressure and regression of left ventricular hypertrophy in primary aldosteronism. J Hypertens 32, 1514–1522 (2014).
Boulkroun, S. et al. Prevalence, clinical and molecular correlates of KCNJ5 mutations in primary aldosteronism. Hypertension 59, 592–598 (2012).
Taguchi, R. et al. Expression and mutations of KCNJ5 mRNA in Japanese patients with aldosterone-producing adenomas. J Clin Endocrinol Metab 97, 1311–1319 (2012).
Calo, L. A. et al. Oxidative stress-related proteins in a Conn’s adenoma tissue. Relevance for aldosterone’s prooxidative and proinflammatory activity. Journal of endocrinological investigation 33, 48–53 (2010).
Petramala, L. et al. Oxidative stress in patients affected by primary aldosteronism. J Hypertens 32, 2022–2029 (2014).
Jeglitsch, G. et al. The cardiac acetylcholine-activated, inwardly rectifying K+-channel subunit GIRK1 gives rise to an inward current induced by free oxygen radicals. Free Radic Biol Med 26, 253–259 (1999).
Shattock, M. J. & Matsuura, H. Measurement of Na(+)-K+ pump current in isolated rabbit ventricular myocytes using the whole-cell voltage-clamp technique. Inhibition of the pump by oxidant stress. Circulation research 72, 91–101 (1993).
Tan, C. M., Xenoyannis, S. & Feldman, R. D. Oxidant stress enhances adenylyl cyclase activation. Circulation research 77, 710–717 (1995).
Ruppersberg, J. P. et al. Regulation of fast inactivation of cloned mammalian IK(A) channels by cysteine oxidation. Nature 352, 711–714 (1991).
Duprat, F. et al. Susceptibility of cloned K+ channels to reactive oxygen species. Proc Natl Acad Sci U S A 92, 11796–11800 (1995).
Arnesen, T. et al. Outcome after surgery for primary hyperaldosteronism may depend on KCNJ5 tumor mutation status: a population-based study from Western Norway. Langenbeck’s archives of surgery / Deutsche Gesellschaft fur Chirurgie 398, 869–874 (2013).
Ip, J. C. et al. Mutations in KCNJ5 determines presentation and likelihood of cure in primary hyperaldosteronism. ANZ journal of surgery 10, [Epub ahead of print] (2013).
Catena, C. et al. Relationships of plasma renin levels with renal function in patients with primary aldosteronism. Clin J Am Soc Nephrol 2, 722–731 (2007).
Takahashi, T. et al. Ischemia- and cytokine-induced mobilization of bone marrow-derived endothelial progenitor cells for neovascularization. Nature medicine 5, 434–438 (1999).
Rossi, G. P. et al. Vascular remodeling and duration of hypertension predict outcome of adrenalectomy in primary aldosteronism patients. Hypertension 51, 1366–1371 (2008).
Wu, V. C. et al. Diagnosis and Management of Primary Aldosteronism. Acta Nephrologica 26, 111–120 (2012).
Fadini, G. P. et al. Peripheral blood CD34+KDR+ endothelial progenitor cells are determinants of subclinical atherosclerosis in a middle-aged general population. Stroke; a journal of cerebral circulation 37, 2277–2282 (2006).
Horita, Y. et al. Cause of residual hypertension after adrenalectomy in patients with primary aldosteronism. Am J Kidney Dis 37, 884–889 (2001).
Pierce, G. L. et al. Weight loss alone improves conduit and resistance artery endothelial function in young and older overweight/obese adults. Hypertension 52, 72–79 (2008).
Callera, G. E. et al. Aldosterone activates vascular p38MAP kinase and NADPH oxidase via c-Src. Hypertension 45, 773–779 (2005).
Monticone, S. et al. Effect of KCNJ5 mutations on gene expression in aldosterone-producing adenomas and adrenocortical cells. J Clin Endocrinol Metab 97, E1567–1572 (2012).
Kitamoto, T. et al. Comparison of Cardiovascular Complications in Patients with and without KCNJ5 Gene Mutations Harboring Aldosterone-producing Adenomas. Journal of atherosclerosis and thrombosis 22, 191–200 (2015).
Nishikawa, T., Saito, J. & Omura, M. Prevalence of primary aldosteronism: should we screen for primary aldosteronism before treating hypertensive patients with medication? Endocrine journal 54, 487–495 (2007).
Lu, K. C., Wu, C. C., Yen, J. F. & Liu, W. C. Vascular calcification and renal bone disorders. TheScientificWorldJournal 2014, 637065 (2014).
Lin, J. H. et al. Eligibility for Statin Therapy According to New Cholesterol Guidelines on Primary Aldosteronism. J Clin Endocrinol Metab [epublish] (2015).
Author information
Authors and Affiliations
Contributions
V.C. wrote the main manuscript text. K.D. conceived and designed the experiments. K.H., K.Y., Y.C., C.H., S.M., S.Y., L.Y., C.C., Y.H., S.L. and T.S. collected the specimens. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Wu, VC., Huang, KH., Peng, KY. et al. Prevalence and clinical correlates of somatic mutation in aldosterone producing adenoma-Taiwanese population. Sci Rep 5, 11396 (2015). https://doi.org/10.1038/srep11396
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep11396
This article is cited by
-
Identifying primary aldosteronism patients who require adrenal venous sampling: a multi-center study
Scientific Reports (2023)
-
Fat mass as an important predictor of persistent hypertension in patients with primary aldosteronism after adrenalectomy
Hypertension Research (2023)
-
Update on the Genetics of Primary Aldosteronism and Aldosterone-Producing Adenomas
Current Cardiology Reports (2022)
-
Evolution of the cardiometabolic profile of primary hyperaldosteronism patients treated with adrenalectomy and with mineralocorticoid receptor antagonists: results from the SPAIN-ALDO Registry
Endocrine (2022)
-
Genetic causes of primary aldosteronism
Experimental & Molecular Medicine (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.