Abstract
Each oomycete pathogen encodes a large number of effectors. Some effectors can be used in crop disease resistance breeding, such as to accelerate R gene cloning and utilisation. Since cytoplasmic effectors may cause acute physiological changes in host cells at very low concentrations, we assume that some of these effectors can serve as functional genes for transgenic plants. Here, we generated transgenic Nicotiana benthamiana plants that express a Phytophthora sojae CRN (crinkling and necrosis) effector, PsCRN115. We showed that its expression did not significantly affect the growth and development of N. benthamiana, but significantly improved disease resistance and tolerance to salt and drought stresses. Furthermore, we found that expression of heat-shock-protein and cytochrome-P450 encoding genes were unregulated in PsCRN115-transgenic N. benthamiana based on digital gene expression profiling analyses, suggesting the increased plant defence may be achieved by upregulation of these stress-related genes in transgenic plants. Thus, PsCRN115 may be used to improve plant tolerance to biotic and abiotic stresses.
Similar content being viewed by others
Introduction
Effectors are molecules secreted by pathogens that manipulate host cell function, thereby facilitating infection or triggering defence responses1. The dual functions of effectors have been extensively reported in many plant-microbial pathosystems2. In the presence of the corresponding resistance (R) protein, the effector is recognised as an avirulence (AVR) factor, resulting in a defence response often associated with a hypersensitive response (HR) that restricts the spread of pathogens from the infection site. It is believed that effectors contribute to virulence in a susceptible host. Indeed, the virulence functions of many effectors have been characterised3,4.
Plant-pathogenic oomycetes cause devastating diseases on many different host plants and have a huge impact on agriculture. For example, Phytophthora infestans is responsible for the Irish potato famine; P. sojae causes root and stem rot of soybean (Glycine max), which is one of the most important pathogens of soybean5. Bioinformatic analyses of the genome sequences of several oomycete pathogens have identified a large number of candidate effector proteins6,7. Among them, RxLR and CRN effectors are two major classes of well-studied cytoplasmic effectors. RxLR effectors are defined by a conserved motif in the N-terminus, termed RxLR (for Arg, any amino acid, Leu, Arg), which mediates translocation of effectors into host cells8,9. To date, nearly all avirulence genes identified from oomycete plant pathogens belong to RxLR effectors10. Virulence functions of several RxLR effectors have been characterised, including the avirulent proteins PiAvr3a, PiAvrblb2, PsAvr3b, HaATR1 and HaATR1310. Interestingly, recent work on P. capsici correlated non-host resistance (NHR) in a range of Nicotiana species with HR elicited by a single RxLR effector, PcAvr3a-like11.
CRNs were first identified in P. infestans and their transient expression resulted in a leaf-crinkling and cell-death phenotype in plants12. The CRN effector family shows extensive expansion in all sequenced Phytophthora species. Analogous to the RxLR effectors, the N-terminus of CRN contains a conserved FLAK (for Phe, Leu, Ala, Lys) motif required for effector translocation13. In contrast to the N-terminus, CRN C-terminal domains have high levels of variation and control virulence. Our previous studies revealed a role of some CRN C-termini towards P. sojae virulence on soybean14. It has also been shown that the P. infestans CRN8 C-terminus contains a kinase-like domain and exhibits kinase activity, suggestive of a role in modification of host cell signalling pathways during infection15. Recent studies showed that the expression of CRN effectors rarely leads to cell death16,17. Interestingly, we showed previously that many CRNs from P. sojae suppress cell death induced by PAMPs or other elicitors, suggestive of diverse activities16. However, only one CRN from P. capsici has a positive effect on pathogen virulence in a screening assay17, suggesting that CRNs play a more subtle role in plant-pathogen interactions than believed previously. The majority of CRN effectors are localised to plant nuclei, indicating that CRNs target and perturb host nuclear processes to exert effector activity17. However, the functions and molecular mechanisms of most CRN effectors remain unclear.
Plants are constantly exposed to a variety of biotic (i.e. pathogen infection and insect herbivory) and abiotic (i.e. high temperature, drought and high salinity) stresses throughout their life cycles. These stresses negatively impact plant growth and development and cause considerable losses in crop yield worldwide. Many attempts have been made to improve resistance to pathogens and increase tolerance to abiotic stress. One commonly used strategy is to overexpress plant genes that are induced by biotic or abiotic stresses, such as mitogen-activated protein kinases18,19, transcriptional factors20 and disease-related genes21. Another important strategy is to introduce heterologous genes that confer resistance to pathogens or abiotic stress22,23.
It has been shown that the expression of pathogen-derived elicitors induces plant immune responses and improves plant resistance to pathogens or insects24,25,26. Elongation factor (EF-Tu), a highly conserved elicitor in bacteria, can induce defence responses in plants. The N-terminal 18 amino acids of EF-Tu can trigger plant immune responses24. Flagellin is another well-characterised elicitor in bacteria. Expression of a flagellin gene from the bacterial pathogen Acidovorax avenae in rice enhances disease resistance against the fungal rice blast pathogen Magnaporthe grisea27. However, transgenic plants showed small chlorotic spots on leaves, indicating that the constitutive activation of flagellin-mediated immune responses causes detrimental effects on plants. Harpins (Hrps), another class of bacterial-pathogen-derived elicitors, can also be used to improve plant defence. Interestingly, Hrp protein treatment not only improves plant tolerance to drought stress28 but also promotes plant root growth29. Expression of a functional fragment of harpin protein Hpa1 increased resistance to fungal pathogens and insects30,31. Interestingly, although Hpa10-42 expression represses root development, it enhances the growth of aerial parts of plants30.
PsCRN115 and PsCRN63 were previously identified from P. sojae and share high sequence similarity, but exhibit contrasting activities on host plants. PsCRN63 triggers cell death, while PsCRN115 suppresses cell death. Silencing of PsCRN115 and PsCRN63 simultaneously reduces P. sojae virulence14; however, the roles of PsCRN115 and PsCRN63 during pathogen infection remain unclear. To evaluate the effects of PsCRN115 expression on plant defence, we transferred PsCRN115 into the model plant Nicotiana benthamiana. We found that expression of PsCRN115 did not lead to obvious developmental changes in N. benthamiana. Unexpectedly, PsCRN115 expression increased plant resistance to Phytophthora pathogens and improved plant tolerance to salt and drought stresses. Upregulation of heat-shock-protein (HSP) and cytochrome-P450 genes may contribute to biotic and abiotic stress resistance in PsCRN115-transgenic N. benthamiana. Our results suggest that the oomycete effectors can be used to improve plant tolerance to biotic and abiotic stresses.
Results
PsCRN115 expression does not significantly affect the growth and development of Nicotiana benthamiana
We showed previously that silencing of PsCRN63 and PsCRN115 jointly reduced P. sojae virulence14. To further explore the function of PsCRN115, we generated a GFP:PsCRN115 fusion construct driven by the CaMV 35S promoter and introduced it into N. benthamiana by Agrobacterium-mediated leaf disc transformation32. A total of 34 independent PsCRN115-expressing lines were obtained using kanamycin resistance selection. Transgene integration and expression was further confirmed based on genomic PCR and RT-PCR (Supplementary Fig. S1). Furthermore, Western blot was conducted to confirm that GFP:PsCRN115 fusion protein was properly expressed in N. benthamiana at the expected size in T1 transgenic plants (Supplementary Fig. S1). Four representative lines (#13, #18, #27 and #34) were selected for further characterisation because PsCRN115 was correctly expressed. T3 progeny plants of these lines were confirmed by Western blot (Fig. 1a) and used for further phenotypic characterisation.
Characterisation of PsCRN115-transgenic Nicotiana benthamiana.
(a) Western blot analysis of total proteins from leaf extracts of transgenic N. benthamiana lines of T3 generation. Total proteins were extracted from 40-day-old leaf tissues of PsCRN115 transgenic N. benthamiana lines (#13, #18, #27 and #34) and the GFP transgenic N. benthamiana line (GFP). The monoclonal antibodies against GFP were used for protein expression detection. PS is Ponceau stain. (b) Seed germination of N. benthamiana plants on MS medium 12 days after sowing. (c) The post-germination growth of transgenic seedlings on MS medium. The root length (10 seedlings for each transgenic line) was recorded 14 days after sowing. (d) The developmental phenotypes of transgenic plants at 7 (above) and 10 weeks (below) post-germination. WT, wild-type N. benthamiana used as recipient; GFP, GFP-transgenic N. benthamiana; #13, a PsCRN115-transgenic line used as an example.
It was previously shown that PsCRN115 is localised to the plant nucleus using Agrobacterium-mediated transient expression14. We confirmed the nuclear localisation of PsCRN115 in stable transgenic lines under normal and stress conditions (Supplementary Fig. S2). This result suggested that PsCRN115 targets plant nuclei to exert its biological function. To determine whether the transgenes affect plant development, we observed growth and development of the transgenic lines at different stages. Germination rate of the PsCRN115-transgenic lines were similar to the wild-type and GFP lines 12 d after sowing on MS plates (Fig. 1b). At the same time, no significant differences in root lengths (Fig. 1c) and no visible morphological changes were observed in transgenic plants compared to wild-type and GFP lines (Fig. 1d).
CRNs are conserved in oomycete pathogens and could be classified as pathogen-associated molecular patterns (PAMPs)33. To preliminarily determine whether PsCRN115 functions as a PAMP in N. benthamiana, callose deposits were detected in PsCRN115-transgenic lines. Callose deposits were clearly observed in a positive control for staining in which wild type plants were treated with a well-known PAMP molecule flg22. However, no signals of callose deposits were detected in wild type, GFP- and PsCRN115-transgenic lines when flg22 was absent (Supplementary Fig. S3). Thus, we suggest that expression of PsCRN115 does not cause visible reinforcement of cell wall and not significantly affect the development of transgenic N. benthamiana under normal growth conditions.
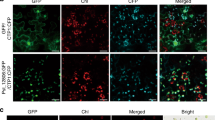
PsCRN115 expression suppresses cell death in N. benthamiana
We showed previously that PsCRN115 suppresses plant cell death triggered by PsCRN63 and PsojNIP by Agrobacterium-mediated transient expression14. To determine whether GFP:PsCRN115 fusion affects the function of PsCRN115, we conducted the cell-death-suppression assay in PsCRN115-transgenic lines. Cell death triggered by PsCRN63 and PsojNIP was suppressed in PsCRN115-expressing plants. However, cell death triggered by other elicitors was not (Fig. 2a), which was consistent with previous results based on transient expression assay14. This result indicated that the GFP:PsCRN115 fusion does not affect the function of PsCRN115. To determine whether the cell-death elicitor proteins were unstable in transgenic plants, Western blot analysis was conducted. The results showed that the cell death inducers were equally expressed in PsCRN115-transgenic plants compared to GFP lines (Fig. 2b), indicating that cell death suppression was caused by PsCRN115 expression in N. benthamiana.
Suppression of cell death in PsCRN115-expressing plants.
(a) Suppression of elicitor-triggered cell death in PsCRN115-transgenic N. benthamiana. The leaves were infiltrated with Agrobacterium GV3101 cells containing a PVX vector carrying PsAvh241, Bax, Avr3a/R3a, PsojNIP or PsCRN63. The photographs were taken 5 d after infiltration. (b) Confirmation of protein expression of the cell-death elicitors using Western blot analysis. Mouse monoclonal antibodies against the HA-epitope tag were used to detect expression of the cell-death elicitors fused to HA tags. (c) Heat-induced programmed cell death in transgenic N. benthamiana. Leaves of GFP- and PsCRN115-transgenic plants were detached and floated on water at room temperature or 50 °C for 40 min in the dark. The photographs were taken at 0, 12 and 24 h after treatment. The treated leaves were stained with trypan blue to visualise cell death. The results were reproducible in each transgenic line in at least three replicates and #13 was shown as an example.
Since PsCRN115 suppresses cell death triggered by specific elicitors, we explored whether it also suppressed programmed cell death (PCD) induced by other cell‐death‐inducing stress. Exposure to heat shock can lead to PCD in a number of species, including Arabidopsis and tobacco34. The detached leaves were floated on water at high temperature (50 °C) for 40 min and transferred to room temperature and incubated for 12 hours or 24 hours. All the PsCRN115‐transgenic line showed reduced PCD compared to control plants as determined by trypan blue staining (Fig. 2c). Thus, PsCRN115 may protect plant cells from PCD induced by heat‐shock stress and pathogen elicitors.
PsCRN115 expression enhances plant resistance to Phytophthora pathogens
Since PCD plays a key role in plant immunity35, we evaluated the effects of PsCRN115 expression on plant resistance. We previously showed that transient expression of PsCRN115 increased plant resistance to P. capsici36,37. Here we challenged the stable PsCRN115-transgenic plants with two oomycete pathogens (P. capsici and P. parasitica) using two methods. Detached leaves of transgenic plants were first inoculated with Phytophthora zoospores. All inoculated leaves showed water-soaked lesions at 36 hours post-inoculation (hpi). However, the lesion diameters were significantly smaller in PsCRN115-transgenic lines compared to GFP lines (Fig. 3a, b). We then inoculated the transgenic plants with zoospores using the root-dip inoculation method. Although both PsCRN115- and GFP-transgenic plants showed symptoms of wilting and stunting (Fig. 3c), the PsCRN115-expressing plants showed reduced disease severity and significantly increased survival rate (Fig. 3d). Taken together, these findings suggest that expression of PsCRN115 improves plant resistance to two Phytophthora pathogens.
Enhanced resistance to Phytophthora pathogens in PsCRN115-transgenic plants.
(a) Leaf phenotypes upon Phytophthora infection. Detached leaves of GFP- and PsCRN115-transgenic plants were inoculated with P. capsici or P. parasitica zoospores. The representative photographs were taken at 36 hpi. (b) Average lesion diameters of the inoculated leaves. Error bars represent standard errors calculated from at least 10 independent biological replicates. Asterisks indicate significant differences determined using Dunnett’s test (P < 0.01). (c) Phenotypes of the inoculated plants. A total of 5 mL of P. capsici or P. parasitica zoospores (100 μL−1) were dipped into the soils. The photographs were taken at 4 dpi. (d) Survival rates of the inoculated plants. The rates were measured at 4 dpi. The experiments were repeated four times with similar results. A total of 20 plants for each line were used for inoculation assays. Bars represent the standard errors. Different letters above bars indicate statistical significance (**, P < 0.01, Dunnett’s test).
Upregulated defence-related genes and promoted accumulation of H2O2 in PsCRN115-expressing N. benthamiana
To preliminarily explore the resistance to Phytophthora pathogens in transgenic plants, expression of defence-related genes were analysed using real-time qPCR. Since the salicylic acid (SA)-signalling pathway contributes positively to resistance against hemibiotrophic Phytophthora pathogens, expression levels of PR1b and PR2b, two marker genes in this pathway, were analysed. Expression levels of these defence-related genes were significantly higher in PsCRN115-transgenic N. benthamiana during P. capsici infection compared to in GFP lines (Fig. 4a). These results suggested that expression of PsCRN115 might improve plant resistance to Phytophthora pathogens by upregulating the expression levels of defence-related genes.
PsCRN115 mediates upregulation of pathogenesis-related genes and H2O2 accumulation in N. benthamiana.
(a) Expression of PR1b and PR2b genes. Samples were collected at the indicated time points upon infection with P. capsici zoospores. The relative expression levels were standardized to the EF1α gene. Error bars represent standard errors (**, P < 0.01, Dunnett’s test). (b) Increased H2O2 accumulation in PsCRN115-transgenic plants. H2O2 accumulation was visualized by DAB in transgenic N. benthamiana leaves at 12 h after inoculation with water (mock) or P. capsici zoospores. Detached leaves were stained with DAB solution as described in the Methods section. Microscopic observations of the DAB-stained leaves of N. benthamiana are shown at the bottom. (c) Relative intensity of DAB staining in P. capsici-infected N. benthamiana. Values are means ± SE of four biological replicates. Asterisks indicate significant differences between GFP- and PsCRN115-transgenic plants (**, P < 0.01, Dunnett’s test). (d) Expression levels of ROS-producing genes RbohA and RbohB. Asterisks indicate significant differences between GFP- and PsCRN115-transgenic plants (**, P < 0.01, Dunnett’s test).
To increase our understanding of the mechanism that leads to resistance to Phytophthora pathogens, we analysed the accumulation of H2O2 in plant leaves using the diamino-benzidine (DAB) staining method38. Very weak staining was observed in both PsCRN115- and GFP- transgenic plants under mock treatment with water and no significant differences in staining strength were observed (Fig. 4b). By contrast, dark staining was observed in both P. capsici-infected PsCRN115- and GFP-transgenic plants (Fig. 4b). However, DAB staining was significantly stronger in PsCRN115-transgenic N. benthamiana compared to GFP-expressing plants challenged with P. capsici zoospores (Fig. 4b,c). To investigate the possible mechanisms underlying the elevated H2O2 accumulation in PsCRN115-transgenic plants, we detected the expression levels of genes encoding ROS-producing proteins; namely, respiratory burst oxidase homologues (RbohA and RbohB). As shown in Fig 4d, there was a minor but significant increase in expression of ROS-producing genes in PsCRN115-transgenic lines compared to GFP-expressing lines. This result suggested that the increased levels of H2O2 accumulation in PsCRN115-transgenic plants might be caused by upregulation of ROS-producing genes. Taken together, these results suggested that improved resistance to Phytophthora pathogens in PsCRN115-transgenic plants is likely caused by upregulation of defence-related genes and promotion of H2O2 accumulation.
PsCRN115 expression improves plant tolerance to salt stress
Biotic and abiotic stress responses converge in the stress signalling networks39 and PCD is also important for abiotic stresses40. We evaluated whether PsCRN115 expression affected stress tolerance to salt and drought. No significant differences in seed germination were observed between PsCRN115 and GFP lines on normal MS medium without NaCl (Fig. 5a and Supplementary Fig. S4). However, a significant increase in germination rates was observed in PsCRN115 lines compared with WT and GFP lines in the presence of 100 mM NaCl (Fig. 5a,b). No seeds of WT and GFP lines germinated on the MS medium containing 150 mM NaCl (Fig. 5a and Supplementary Fig. S4); by contrast, about 10% of the seeds of the PsCRN115 lines germinated 12 d after sowing (Fig. 5a and Supplementary Fig. S4). To confirm the increased tolerance to salt stress, the post-germination growth of the transgenic plants were tested. Seeds of the control and transgenic lines were allowed to germinate on normal MS plates for 4 d and then transferred onto MS medium containing different NaCl concentrations. The growth of seedlings was monitored by measuring their root lengths. The root lengths were significantly longer in PsCRN115-transgenic lines than control plants in the presence of 100 and 150 mM NaCl (Fig. 5c,d). Furthermore, we observed that the growth of the control plants (8-week-old) was considerable slower than the PsCRN115-transgenic lines 2 weeks after NaCl treatment (Fig. 5e). Nearly 80% of control plants died 3 weeks after treatment. In contrast, more than 70% of the PsCRN115-transgenic lines survived after treatment (Fig. 5f). These results indicated that expression of PsCRN115 improved plant salt tolerance in N. benthamiana.
Increased salt tolerance in transgenic plants overexpressing PsCRN115.
(a) Seed germination in the presence of specific NaCl concentrations 12 days after sowing. (b) Germination rates of the plants under normal and NaCl treatments. Germination rate was scored at the indicated days. Data represent the means ± SE of three independent experiments (**, P < 0.01, Dunnett’s test). (c) The post-germination seedling growth of plants. Seeds were germinated on MS medium for 4 d and transferred to MS medium supplemented with 100 or 150 mM NaCl. The photographs were taken 12 d after germination. (d) Root length of the seedlings 12 d after germination. Data represent the means ± SE of three independent measurements (**, P < 0.01, Dunnett’s test). (e) Effect of salt stress on plant growth in soil. Plants were treated with 150 mM NaCl for 2 weeks and photographed at 15 weeks after germination. (f) Survival rates of plants. Data represent the means ± SE of three independent measurements (**, P < 0.01, Dunnett’s test).
PsCRN115 expression enhances drought tolerance in plants
To assess whether the expression of PsCRN115 affects plant responses to other abiotic stresses, we sowed seeds of PsCRN115 lines and control plants onto MS agar medium containing 100 or 200 mM mannitol, a widely used condition to mimic drought stress treatment41. The germination of control plant seeds was severely inhibited with increasing mannitol concentrations (Fig. 6a, Supplementary Fig. S5 online). However, the germination rate of PsCRN115-transgenic plants was significantly higher than control plants at 12 d after sowing on MS ager medium containing 100 or 200 mM mannitol (Fig. 6b and Supplementary Fig. S5). After 12 days of drought treatment of 8-week-old plants, WT and GFP lines were completely wilted, whereas the PsCRN115-transgenic plants were less affected (Fig. 6c). Two days after resumption of watering, the PsCRN115-transgenic plants recovered more rapidly than control plants (Fig. 6c). The survival rate of PsCRN115-transgenic plants was significantly higher than control plants after drought treatment (Fig. 6d). In addition, the rate of water loss from the detached leaves of PsCRN115-expressing plants was lower than that from detached leaves of WT and GFP plants under dehydration conditions (Fig. 6e). These results indicated that the PsCRN115-transgenic plants were more tolerant to drought stress.
PsCRN115 expression enhances drought tolerance in transgenic plants.
(a) Seed germination at 12 days after sowing. (b) Germination rates. The data was scored and calculated at the indicated time points when treated with mannitol (**, P < 0.01, Dunnett’s test). (c) Phenotypes of plants submitted to drought stress at the vegetable stage. Water was withheld from 8-week-old plants for 12 d, after which the plants were watered for 2 d to allow them to recover. BD, before drought treatment; RW, re-watering. (d) Survival rates of plants under drought stress. Data represent the means ± SE of three independent experiments (**, P < 0.01, Dunnett’s test). (e) Water loss from the detached leaves of the indicated plants. The rate of water loss was calculated based on the loss of fresh weight in the samples. Bars indicate the standard error of 10 biological replicates.
Genome-wide expression analysis in transgenic lines
To explore the mechanisms of biotic and abiotic stress tolerance underlying PsCRN115-expressing N. benthamiana, digital gene expression (DGE) tag profiling was performed to determine the differential gene expression between PsCRN115- and GFP- transgenic lines in leaves of 8-week-old plants under normal growth conditions. We observed 273 genes were significantly up- or down-regulated by over four-fold in PsCRN115-transgenic lines compared to GFP lines (Supplementary Table S1). This result suggested that PsCRN115 had a significant impact on global gene expression profiles in N. benthamiana.
Among the upregulated genes, more than 10% belong to heat shock protein genes and cytochrome P450 genes (Table 1), which was not observed in downregulated genes (Supplementary Table S1). Eight HSPs were upregulated by at least six-fold in PsCRN115-transgenic plants compared to GFP lines (Table 1), suggesting that PsCRN115-trangenic plants acquire abiotic tolerance by accumulating more HSPs before stress conditions. Five cytochrome P450 genes were upregulated by at least four-fold in PsCRN115-expressing plants compared to GFP lines (Table 1), suggesting the P450 may play a role in plant stress resistance. Interestingly, a Bcl-2 binding anthanogene-1 gene, which functions in regulating apoptosis-like processes during pathogen attack and abiotic stress42, was upregulated by 5.31-fold in PsCRN115-trangenic plants compared to GFP lines (Supplementary Table S1). To evaluate the quality of the sequencing results, expression levels of 3 HSP genes (NbS00034783g0004.1, NbC25233207g0001.1, NbS00009579g0009.1) and 3 P450 genes (NbC25673618g0001.1, NbC24805505g0002.1 and NbS00038435g0004.1) in Table 1 were determined using quantitative RT-PCR (Fig. 7). The RT-PCR analyses confirmed the results of DGE analysis.
Furthermore, we performed Blast2GO annotations to gain a general picture of the functions of genes identified by DGE approach. The upregulated unigenes are categorized into 20 functional groups in the three ontologies. The categories “cell part”, “organelle”, “intracellular” and “response to stress” are dominant (Supplementary Fig. S6). By contrast, The downregulated unigenes are categorized into 17 functional groups and “cell part”, “ion binding” and “cellular metabolic process” are dominant (Supplementary Fig. S6). These results suggest that the increased stress tolerance in PsCRN115-transgenic N. benthamiana was achieved by modulation of plant stress responses and modification of cellular metabolism and functions.
Discussion
Genome-wide catalogues of effectors are becoming available for many plant pathogens and effectors have been used to assist in plant breeding43. In addition to applying effectors as molecular markers for marker-assisted selection, effectors have been exploited to accelerate R gene cloning and specificity profiling43. However, these strategies are dependent on knowledge of the interaction between R and Avr genes. Can we use effectors directly to improve plant resistance? In this study, we showed that expression of a CRN effector PsCRN115 significantly improved plant disease resistance and abiotic stress tolerance in N. benthamiana.
Recent study on P. capsici showed that non-host resistance (NHR) is correlated with HR elicited by a single RxLR effector11. N. benthamiana is not a host of P. sojae pathogen and expression of a P. sojae effector may elicit NHR. However, effector-triggered immunity (ETI) tends to be associated with HR and constitutive activation of ETI may lead to fitness costs44. In this study, we did not observe obvious developmental changes in PsCRN115-expressing N. benthamiana, suggesting that improvement of plant defence by PsCRN115 in N. benthamiana is likely achieved by other mechanisms and not due to perception by an unknown R protein.
CRNs are conserved in oomycete pathogens and share conserved domains17, therefore, they could be classified as pathogen-associated molecular patterns (PAMPs)33. PAMP-triggered immunity (PTI) is generally characterised by the induction of a reactive oxygen species (ROS) burst, callose deposition and expression of defence-related genes without any signs of PCD45. In this study, we observed that ROS accumulation and expression of defence-related genes were upregulated in PsCRN115-transgenic lines compared to the control. These defence responses were similar to PTI. However, treatment with PAMPs leads to constitutive activation of defence responses, which usually causes severe growth reduction46. We found that expression of PsCRN115 did not result in an obvious growth reduction while the defence-related genes were weakly induced in transgenic plants under normal growth conditions. Therefore, PsCRN115 may function as a weak “PAMP” to activate plant defence. However, constitutive expression of PsCRN115 in N. benthamiana did not lead to visible callose deposition, which is a marker of PTI. These results further suggest that the enhanced stress tolerance in transgenic plants may be achieved by other unknown mechanisms.
DGE analyses showed that many HSPs and cytochrome P450 genes were upregulated in PsCRN115-transgenic plants. It has been shown HSPs and cytochrome P450 play a role in biotic and abiotic stress responses47,48,49. Heat shock proteins (HSPs) can assist in protein refolding under stress conditions47 and play a crucial role in protecting plants against stress by re-establishing normal protein conformation and cellular homeostasis. Timely expression of HSPs before severe heat conditions is vital for plants to acquire thermotolerance48. This result suggests that PsCRN115 expression renders the plant more resilient to abiotic stress by upregulating many HSPs. Cytochrome P450s play crucial roles in the biosynthesis of a variety of endogenous lipophilic compounds, such as fatty acids, phytoalexins, brassinolides and gibberellins. Expression of cytochrome P450 genes was induced by various biotic and abiotic stresses, indicating that they may be involved in the regulation of plant defence49. This result suggests that PsCRN115 improves plant disease resistance and abiotic stress tolerance by upregulating expression of HSPs and cytochrome P450.
The majority of CRNs are localised to plant nuclei13,17 and we found that PsCRN115 also targets the plant nucleus under biotic and abiotic stresses, indicating that CRNs may target and perturb host nuclear processes. However, it remains unclear whether PsCRN115 regulates expression of plant genes by directly binding to plant genomic DNA or indirectly by interfering with other proteins, such as histone proteins and transcriptional factors. Moderate levels of ROS may function as signalling molecules to promote cell survival, whereas severe increases in ROS can trigger cell death50. However, biotic and abiotic stresses usually lead to the overexpression of ROS, which are toxic and cause damage to plants51,52. We recently showed that PsCRN115 interacts with plant catalases, essential enzymes for ROS-scavenging. PsCRN115 may enhance the ability of plants to scavenge reactive oxygen species by stabilising catalases36. This may explain why PsCRN115 can improve plant tolerance to biotic and abiotic stresses.
PCD is involved in responses to biotic and abiotic stress stimuli35,40. We showed that PsCRN115 not only suppressed cell death triggered by some elicitors, but also suppressed PCD triggered by heat shock treatment. Interestingly, a Bcl-2 binding anthanogene-1 gene was upregulated in PsCRN115-transgenic lines based on DGE analyses. It was reported that this gene regulates apoptosis-like processes during pathogen attack and abiotic stress42. These results suggest that PsCRN115 improves biotic and abiotic stress tolerances by suppressing cell death triggered by these conditions.
In conclusion, we showed that expression of a CRN effector, PsCRN115, from P. sojae enhanced plant tolerance to biotic and abiotic stresses, suggesting that oomycete effectors can be used directly to improve plant defence against biotic and abiotic stresses.
Methods
Plant material and growth conditions
Nicotiana benthamiana seeds were surface sterilised and planted on Murashige and Skoog (MS) medium for germination under greenhouse conditions. Three-leaf stage N. benthamiana seedlings were transferred to soil and maintained under greenhouse conditions at 25 ± 1 °C with a 16 h light/8 h dark cycle.
Vector construction and genetic transformation
The PsCRN115 gene (lacking the predicted secretory signal peptide) was amplified using specific primers (Supplementary Table S2) and inserted into the binary vector pBinGFP2 via BamH I and Xba I. The recombinant plasmid was introduced into Agrobacterium tumefaciens strain EHA105 for N. benthamiana transformation using the leaf disc method, as reported previously53. The transformants were screened for kanamycin resistance (100 mg/L) and further verified by PCR. Plants transformed with the pBinGFP2 vector were used as controls.
Protein extraction and Western blot analyses
N. benthamiana leaf tissues were ground in liquid nitrogen and mixed with protein extraction buffer (50 mM HEPS, 150 mM KCl, 1 mM EDTA, 0.1% triton X-100, adjust pH to 7.5 with KOH) supplemented with 1 mM DTT and 1 × protease inhibitor mixture (Cocktail, Roche). Suspensions were mixed and centrifuged at 12000 × g for 15 min at 4 °C. Total proteins were separated on 12% SDS-polyacrylamide gels and transferred to Immobi-lon-PSQ polyvinylidene difluoride membranes. The membranes were washed with PBST (PBS with 0.1% Tween 20) for 3 min and then blocked in 5% non-fat milk for 1 h. Mouse monoclonal anti-GFP or -HA antibody (Sigma-Aldrich) was added at a ratio of 1:5000 and incubated for 2 h, followed by three washes with PBST. The membranes were then incubated with goat anti-mouse IRDye 800CW (Odyssey, Li-Cor) at a ratio of 1:10000 at room temperature for 40 min with shaking. After three washes with PBST, the membranes were visualised using an Odyssey imaging system with excitation at 700 and 800 nm.
Confocal microscopy
N. benthamiana leaves were cut into small squares and immersed into PBS buffer containing 5 μg/mL DAPI for staining of the nuclei for 5 min. Fluorescence was visualised with a Zeiss LSM 710 confocal laser-scanning microscope (CLSM). The excitation wavelength used for GFP was 488 nm and 405 nm for DAPI. Leaf tissues of GFP-transgenic lines were used as controls.
Callose deposition assay
To induce callose deposition, 40 μM flg22 was infiltrated into 6-week-old N. benthamiana leaves. Leaf discs of the leaves were destained in 95% ethanol and then incubated at 60 °C until chlorophyll was removed. The cleared leaf discs were washed with 70% ethanol and then rinsed with distilled water. The leaf discs were then immersed in 0.1% aniline blue in 150 mM K2HPO4, pH 9.5 and incubated in the dark for 1 h. The stained leaf discs were rinsed with distilled water and mounted in 50% glycerol and examined under a UV epifluorescence microscope (Olympus BX71).
Transient protein expression in planta
A.tumefaciens GV3101 was used to deliver T-DNA constructs into N. benthamiana leaves. Overnight A.tumefaciens cultures were harvested by centrifugation at 3500 × g for 5 min, washed with 10 mM MgCl2 three times and resuspended in infiltration buffer (10 mM MgCl2, 10 mM MES, pH 5.6 and 150 μM acetosyringone) to an OD600 of 0.1 prior to infiltration into the entire leaf or leaf sections.
Trypan blue staining
Cell death in N. benthaminana leaf tissues was detected using trypan blue staining. Trypan blue solution contains 10 g of phenol, 10 mL of lactic acid, 10 mL of glycerol, 10 mL of distilled water and 20 mg of trypan blue (Sigma-Aldrich). After 12 h or 24 h recovery from heat shock treatment, leaf tissues were soaked in boiling trypan blue solution for 3 min and incubated for 5 h. Samples were then destained in chloral hydrate solution (250%, w/v) and photographed.
Phytophthora infection assays
Phytophthora parasitica Pp025 and Phytophthora capsici Pc35 were routinely grown on 10% (v/v) V8 juice agar plates at 25 °C in the dark. Zoospores were prepared as reported previously53,54. For detached leaves, Phytophthora infection assays were performed using droplet inoculations of 10 μL of zoospore suspensions with a concentration of 50 zoospores/mL on the abaxial surface. The phenotype was monitored within 48 hours and photographs were taken at 36 hours post-inoculation. For whole seedlings, the infection assays were performed by dipping roots into zoospore suspensions. The GFP-transgenic lines were used as controls. The inoculated plants were maintained in a moist chamber and disease progression was monitored within 10 days. At least three independent experiments were performed for this assay. Dunnett’s test was used for statistical analysis (P < 0.01).
RNA extraction and quantitative RT-PCR
Total RNA of N. benthamiana leaves was extracted using the RNAsimple Total RNA Kit (Tiangen, China) according to the manufacturer’s instructions. The cDNA was generated using the PrimeScript™ RT reagent Kit (TaKaRa). Real-time quantitative PCR was performed with SYBR Green fluorescence detection in a quantitative PCR thermal cycler (ABI PRISM 7300, Applied Biosystems). Each reaction was prepared using 2 μL of cDNA, 10 μL of SYBR® Premix ExTaq (TaKaRa), 0.2 μM gene-specific primers (primer sequences in Table S2) and 0.4 μL of ROX reference dye in a total volume of 20 μL. The cycling conditions were as follows: 95 °C for 30 s, 40 cycles of 95 °C for 5 s and 60 °C for 31 s to calculate cycle threshold values, followed by a dissociation program of 95 °C for 15 s, 60 °C for 1 min and 95 °C for 15 s to obtain melt curves. The N. benthamiana EF1α gene was used as an internal reference gene to calculate relative transcriptional levels.
DAB staining
H2O2 was detected in situ using DAB staining38 with minor modifications. Briefly, N. benthamiana leaves were detached and inoculated with P. parasitica zoospores. The infected leaves 8 h after inoculation were soaked in DAB solution and maintained for 8 h at 25 °C. The leaf tissues were fixed in 95% ethanol and photographed.
Salt and drought stress analyses
For salt treatment, T3-generation PsCRN115- and GFP-transgenic seeds were surfaced-sterilised and sown on MS agar medium supplemented with 0, 100, or 150 mM NaCl. The germination rate was measured daily after sowing. The root length was measured at 12 d after germination. For drought treatment, the seed germination rate was measured using the method described above. To evaluate drought tolerance during vegetative growth, water was withheld completely from 8-week-old PsCRN115- and GFP-transgenic lines for 12 d, after which the plants were watered for 2 d to allow them to recover. For water loss measurements, detached leaves of transgenic plants were incubated at 37 °C and weighed on an electronic balance at the indicated time points. The rate of water loss was plotted as the loss in leaf weight over time. Salt and drought stress analyses were repeated at least three times. Dunnett’s test was used for statistical analysis (P < 0.01).
Digital gene expression profiling analysis
Total RNA was isolated from PsCRN115- and GFP- transgenic N. benthamiana, respectively, followed by Illumina sequencing using a HiSeq2000 to produce 100 bp paired-end data. All obtained clean reads were mapped to N. benthamiana reference gene sequences using Tophat55 with default parameters. The normalised gene expression level for each gene was calculated using the reads per kilo bases per million reads (RPKM) method56. The statistical significance of the differentially expressed genes was determined using GFOLD software (GFOLD > 1 or GFOLD < −1; log2 (fold change) >2 or log2 (fold change) < −2)57. For functional annotation, distinct sequences were BLAST against the NCBI NR database with an E-value cut-off of 10−5.
Additional Information
How to cite this article: Zhang, M. et al. A Phytophthora sojae cytoplasmic effector mediates disease resistance and abiotic stress tolerance in Nicotiana benthamiana. Sci. Rep. 5, 10837; doi: 10.1038/srep10837 (2015).
References
Kamoun, S. A catalogue of the effector secretome of plant pathogenic oomycetes. Annu Rev Phytopathol 44, 41–60 (2006).
Alfano, J.R. & Collmer, A. Type III secretion system effector proteins: double agents in bacterial disease and plant defense. Annu Rev Phytopathol 42, 385–414 (2004).
Feng, F. & Zhou, J.M. Plant-bacterial pathogen interactions mediated by type III effectors. Curr Opin Plant Biol 15, 469–476 (2012).
Dou, D.L. & Zhou, J.M. Phytopathogen effectors subverting host immunity: different foes, similar battleground. Cell Host Microbe 12, 484–495 (2012).
Tyler, B.M. Phytophthora sojae: root rot pathogen of soybean and model oomycete. Mol Plant Pathol 8, 1–8 (2007).
Tyler, B.M. et al. Phytophthora genome sequences uncover evolutionary origins and mechanisms of pathogenesis. Science 313, 1261–1266 (2006).
Haas, B.J. et al. Genome sequence and analysis of the Irish potato famine pathogen Phytophthora infestans. Nature 461, 393–398 (2009).
Whisson, S.C. et al. A translocation signal for delivery of oomycete effector proteins into host plant cells. Nature 450, 115–118 (2007).
Dou, D.L. et al. RXLR-mediated entry of Phytophthora sojae effector Avr1b into soybean cells does not require pathogen-encoded machinery. Plant Cell 20, 1930–1947 (2008).
Jiang, R.H.Y. & Tyler, B.M. Mechanisms and evolution of virulence in oomycetes. Annu Rev Phytopathol 50, 295–318 (2012).
Vega-Arreguin, J.C., Jalloh, A., Bos, J.I. & Moffett, P. Recognition of an Avr3a homologue plays a major role in mediating nonhost resistance to Phytophthora capsici in Nicotiana species. Mol Plant Microbe In 27, 770–780 (2014).
Torto, T.A. et al. EST mining and functional expression assays identify extracellular effector proteins from the plant pathogen Phytophthora. Genome Res 13, 1675–1685 (2003).
Schornack, S. et al. Ancient class of translocated oomycete effectors targets the host nucleus. P Natl Acad Sci USA 107, 17421–17426 (2010).
Liu, T.L. et al. Two host cytoplasmic effectors are required for pathogenesis of Phytophthora sojae by suppression of host defenses. Plant Physiol 155, 490–501 (2011).
van Damme, M. et al. The Irish potato famine pathogen Phytophthora infestans translocates the CRN8 kinase into host plant cells. PLoS Pathog 8, e1002875 (2012).
Shen, D.Y. et al. Gene duplication and fragment recombination drive functional diversification of a superfamily of cytoplasmic effectors in Phytophthora sojae. PLoS ONE 8, e70036 (2013).
Stam, R. et al. Identification and characterisation CRN effectors in Phytophthora capsici shows modularity and functional diversity. PLoS ONE 8, e59517 (2013).
Piao, H.L., Lim, J.H., Kim, S.J., Cheong, G.W. & Hwang, I. Constitutive over-expression of AtGSK1 induces NaCl stress responses in the absence of NaCl stress and results in enhanced NaCl tolerance in Arabidopsis. Plant J 27, 305–314 (2001).
Zhang, S.Q. & Liu, Y.D. Activation of salicylic acid-induced protein kinase, a mitogen-activated protein kinase, induces multiple defense responses in tobacco. Plant Cell 13, 1877–1889 (2001).
Park, J.M. et al. Overexpression of the tobacco Tsi1 gene encoding an EREBP/AP2-Type transcription factor enhances resistance against pathogen attack and osmotic stress in tobacco. Plant Cell 13, 1035–1046 (2001).
Deak, M. et al. Plants ectopically expressing the iron-binding protein, ferritin, are tolerant to oxidative damage and pathogens Nat Biotechnol 17, 393–393 (1999).
Dana, M.D., Pintor-Toro, J.A. & Cubero, B. Transgenic tobacco plants overexpressing chitinases of fungal origin show enhanced resistance to biotic and abiotic stress agents. Plant Physiol 142, 722–730 (2006).
Lorito, M. et al. Genes from mycoparasitic fungi as a source for improving plant resistance to fungal pathogens. P Natl Acad Sci USA 95, 12734–12734 (1998).
Kunze, G. et al. The N terminus of bacterial elongation factor Tu elicits innate immunity in Arabidopsis plants. Plant Cell 16, 3496–3507 (2004).
Medeira, C. et al. Cryptogein and capsicein promote defence responses in Quercus suber against Phytophthora cinnamomi infection. Eur J Plant Pathol 134, 145–159 (2012).
Kawamura, Y. et al. INF1 elicitin activates jasmonic acid- and ethylene-mediated signalling pathways and induces resistance to bacterial wilt disease in tomato. J Phytopathol 157, 287–297 (2009).
Takakura, Y. et al. Expression of a bacterial flagellin gene triggers plant immune responses and confers disease resistance in transgenic rice plants. Mol Plant Pathol 9, 525–529 (2008).
Dong, H.P. et al. The ABI2-dependent abscisic acid signalling controls HrpN-induced drought tolerance in Arabidopsis. Planta 221, 313–327 (2005).
Ren, X.Y. et al. Root growth of Arabidopsis thaliana is regulated by ethylene and abscisic acid signaling interaction in response to HrpNEa, a bacterial protein of harpin group. Plant Mol Biol Rep 26, 225–240 (2008).
Fu, M.Q. et al. Transgenic expression of a functional fragment of harpin protein Hpa1 in wheat induces the phloem-based defence against English grain aphid. J Exp Bot 65, 1439–1453 (2014).
Wang, D.F. et al. Transgenic expression of the functional fragment Hpa110-42 of the harpin protein Hpa1 imparts enhanced resistance to powdery mildew in wheat. Plant Dis 98, 448–455 (2014).
Regner, F. et al. Coat protein mediated resistance to Plum Pox Virus in Nicotiana clevelandii and N. benthamiana. Plant Cell Rep 11, 30–33 (1992).
Thomma, B.P.H.J., Nurnberger, T. & Joosten, M.H.A.J. Of PAMPs and effectors: the blurred PTI-ETI dichotomy. Plant Cell 23, 4–15 (2011).
Swidzinski, J.A., Sweetlove, L.J. & Leaver, C.J. A custom microarray analysis of gene expression during programmed cell death in Arabidopsis thaliana. Plant J 30, 431–446 (2002).
Coll, N.S., Epple, P. & Dangl, J.L. Programmed cell death in the plant immune system. Cell Death Differ 18, 1247–1256 (2011).
Zhang, M.X. et al. Two cytoplasmic effectors of Phytophthora sojae regulate plant cell death via interactions with plant catalases. Plant Physiol 167, 164–175 (2015).
Liu, T.L., Ru, Y.Y., Liu, L., Liu, P.H. & Dou, D.L. The mechanism analysis of induced disease resistance in Nicotiana benthamiana triggered by effector PsCRN115 of Phytophthora sojae. J Nanjing Agric Univ 35, 65–68 (2012).
Thordal-Christensen, H., Zhang, Z.G., Wei, Y.D. & Collinge, D.B. Subcellular localization of H2O2 in plants. H2O2 accumulation in papillae and hypersensitive response during the barley-powdery mildew interaction. Plant J 11, 1187–1194 (1997).
Fujita, M. et al. Crosstalk between abiotic and biotic stress responses: a current view from the points of convergence in the stress signaling networks. Curr Opin Plant Biol 9, 436–442 (2006).
Shabala, S. Salinity and programmed cell death: unravelling mechanisms for ion specific signalling. J Exp Bot 60, 709–711 (2009).
Knight, H., Brandt, S. & Knight, M.R. A history of stress alters drought calcium signalling pathways in Arabidopsis. Plant J 16, 681–687 (1998).
Doukhanina, E.V. et al. Identification and functional characterization of the BAG protein family in Arabidopsis thaliana. J Biol Chem 281, 18793–18801 (2006).
Vleeshouwers, V.G.A.A. & Oliver, R.P. Effectors as tools in disease resistance breeding against biotrophic, hemibiotrophic and necrotrophic plant pathogens. Mol Plant Microbe In 27, 196–206 (2014).
Tian, D., Traw, M.B., Chen, J.Q., Kreitman, M. & Bergelson, J. Fitness costs of R-gene-mediated resistance in Arabidopsis thaliana. Nature 423, 74–77 (2003).
Boller, T. & Felix, G. A renaissance of elicitors: perception of microbe-associated molecular patterns and danger signals by pattern-recognition receptors. Annu Rev Plant Biol 60, 379–406 (2009).
Oome, S. et al. Nep1-like proteins from three kingdoms of life act as a microbe-associated molecular pattern in Arabidopsis. Proc Natl Acad Sci U S A 111, 16955–16960 (2014).
Wang, W.X., Vinocur, B., Shoseyov, O. & Altman, A. Role of plant heat-shock proteins and molecular chaperones in the abiotic stress response. Trends Plant Sci 9, 244–252 (2004).
Hua, J. From freezing to scorching, transcriptional responses to temperature variations in plants. Curr Opin Plant Biol 12, 568–573 (2009).
Narusaka, Y. et al. Crosstalk in the responses to abiotic and biotic stresses in Arabidopsis: analysis of gene expression in cytochrome P450 gene superfamily by cDNA microarray. Plant Mol Biol 55, 327–342 (2004).
Trachootham, D., Lu, W.Q., Ogasawara, M.A., Valle, N.R.D. & Huang, P. Redox regulation of cell survival. Antioxid Redox Sign 10, 1343–1374 (2008).
Gill, S.S. & Tuteja, N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol Bioch 48, 909–930 (2010).
Torres, M.A. ROS in biotic interactions. Physiol Plantarum 138, 414–429 (2010).
Rajput, N.A. et al. Phytophthora sojae effector PsCRN70 suppresses plant defenses in Nicotiana benthamiana. PLoS ONE 9 (2014).
Zhang, M.X. et al. PnPMA1, an atypical plasma membrane H+-ATPase, is required for zoospore development in Phytophthora parasitica. Fungal Biol 116, 1013–1023 (2012).
Trapnell, C., Pachter, L. & Salzberg, S.L. TopHat: discovering splice junctions with RNA-Seq. Bioinformatics 25, 1105–1111 (2009).
Mortazavi, A., Williams, B.A., Mccue, K., Schaeffer, L. & Wold, B. Mapping and quantifying mammalian transcriptomes by RNA-Seq. Nat Methods 5, 621–628 (2008).
Feng, J.X. et al. GFOLD: a generalized fold change for ranking differentially expressed genes from RNA-seq data. Bioinformatics 28, 2782–2788 (2012).
Acknowledgements
We thank Professor Brett Tyler (Oregon State University) for comments on the manuscript and professor Weixing Shan (Northwest A&F University) for providing P. parasitica strain Pp025. This work was supported by National Science and Technology Major Projects (2014ZX0800910B) and NSFC (31301613, 31371894).
Author information
Authors and Affiliations
Contributions
D.D. and M.Z. designed the experiments. M.Z., N.A.R., D.S., P.S., W.Z., T.L. and J.J.M. performed the experiments. D.D., M.Z. and N.A.R. discussed the results. D.D. and M.Z. wrote the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Zhang, M., Ahmed Rajput, N., Shen, D. et al. A Phytophthora sojae cytoplasmic effector mediates disease resistance and abiotic stress tolerance in Nicotiana benthamiana. Sci Rep 5, 10837 (2015). https://doi.org/10.1038/srep10837
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep10837
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.