Abstract
Apterous A (apA), a member of the LIM-homeobox gene family, plays a critical role in the development of wing. The achaete-scute Complex (AS-C) encodes basic helix-loop-helix (bHLH) transcription factors and functions in bristle development. In the present study, we cloned apA (NlapA) and an achaete-scute homologue (NlASH) from N. lugens. Levels of NlapA and NlASH were higher in nymphs than adults, with particularly high expression in the thorax of nymphs. NlapA expressed more highly in nymphs of the macropterous strain (MS) than those of the brachypterous strain (BS) at 2nd and 4th instar. Knockdown of NlapA and NlASH in vivo generated similar phenotypic defects in the wing (loss-of-bristles, twisted or erect wing). Silencing of NlapA in nymphs of MS led to decreased wing size in adults. Moreover, depletion of NlapA suppressed expression of NlDl, Nlsal, Nlser, Nlvg and Nlwg, both in MS and BS, but induced differential responses of Nlubx and Nlnotch expression between MS and BS. Notably, expression of NlASH was regulated by NlapA. These results collectively indicate that NlapA is an upstream modulator of wing size, bristle formation and patterning. Further studies on DNA-protein and protein-protein interactions are required to elucidate NlapA-mediated regulation of wing development.
Similar content being viewed by others
Introduction
The acquisition of the ability to fly is one of the most important events in insect evolution and undoubtedly a major reason for their evolutionary success. The molecular mechanisms underlying wing developmental processes have attracted extensive research attention. Genes related to wing development of holometabolous Drosophila melanogaster have been comprehensively characterized to date. Before identification of wing patterning genes in ants and aphids, Abouheif and Wray (2002) and Brisson et al. (2010) deduced the presumptive gene network involved in wing patterning from D. melanogaster based on previous literatures1,2. While limited information is available on genes related to wing development in hemimetabolous insects. All wing patterning genes have been identified from hemimetabolous Acyrthosiphon pisum and the expression levels of 11 of these genes at embryogenesis and across nymphal instars between winged and wingless populations compared2. However, few reports have focused on the functions of wing patterning genes of hemimetabolous insects.
The Drosophila Apterous gene (ap) is a member of the LIM-homeobox gene family, which encodes a transcription factor and contains a DNA-binding homeodomain plus two cysteine-rich LIM domains3,4. Ap plays a crucial role in wing development. Restricted expression and activity of ap are responsible for development of the dorsal and ventral compartments of the Drosophila wing imaginal disc5,6,7. In ap mutants, the wing is lost6,7, possibly because of loss of expression of genes regulated by ap, such as Serrate, Fringe and integrins8,9. While several studies have characterized ap in holometabolous insects, its role in wing development of hemimetabolous insects remains unknown.
Genes of the achaete/scute (ASH) gene family encode basic helix-loop-helix (bHLH) transcription factors and are widely conserved throughout the animal kingdom10. The achaete-scute complex (AS-C) induces the formation of external sensory organs that function in mechano- and chemo-sensory reception. In Drosophila, ac/sc is expressed in the proneural clusters. Some of the cells are selected to develop into sensory organ precursors and subsequently, mature bristles11,12. The ac/sc double mutant leads to loss of the majority of bristles, while ectopic expression of ac and/or sc induces extra bristles in flies13,14,15,16. A 26 bp deletion within the Bm-ASH2 (ac/sc homologue of Bombyx mori) promoter is closely linked to the scaleless mutation and leads to loss of Bm-ASH2 expression and the scaleless-wing phenotype, indicating that Bm-ASH2 plays a critical role in scale formation in B. mori17. Selection of the sensory organ precursor is associated with high levels of ac/sc expression maintained by regulators, such as Sens, Pannier, Chip, dLMO, Ap, Wingless and Shaggy. These activators or suppressors act in concert to regulate expression of ASH18,19,20,21,22,23,24,25. At present, no reports on ASH identification and characterization in hemimetabolous insects are available in the literature.
The brown planthopper (BPH), Nilaparvata lugens (Stål) (Hemiptera: Delphacidae), is one of the most destructive insect pests of rice. BPH has two winged morphs, specifically, long-winged (migratory) and short-winged (non-migratory), leading to difficulty in its control. The effects of environmental and genetic factors on wing development and wing dimorphism of BPH are well documented26, but the combinatorial actions of genes responsible for wing development and morph differentiation remain unclear. The ecdysone receptor is known to control wing morphogenesis and melanization in N. lugens27. Disruption of Krüppel-homolog 1 expression via RNAi in BPH causes stunted wing development and malformation of both male and female external genitalia28. Downregulation of BPH Distal-less in 3rd-instar larvae leads to disrupted development of the leg and that of NlDll in 5th- instar larvae to abnormal wing formation29.
In the present study, we cloned the homologue of ap from N. lugens, investigated the expression patterns in the brachypterous strain (BS) and macropterous strain (MS) and determined its role in wing morph differentiation and development. The ac/sc homologue from N. lugens responsible for bristle formation of wing was additionally cloned and characterized. Expression levels of downstream target genes of ap, such as wingless (wg), Notch, ultrabithorax (ubx), vestigial (vg), spalt major (sal), Delta (Dl) and serrate (ser) of N. lugens, were quantified after knockdown of ap. To our knowledge, this is the first report on ap and ac/sc cloning and characterization in hemimetabolous N. lugens.
Results
Characterization of NlapA cDNA
Full-length cDNA of ap of N. lugens was isolated from wing pads of nymphs using the rapid amplification of cDNA ends (RACE) method. Clustal W analysis revealed that the ap homologue belongs to the apA class (NlapA) (Supplementary Fig. S1). Full-length NlapA cDNA (GenBank accession No: KC978728) was determined as 3004 bp, with a 5’ untranslated region (UTR) of 57 bp, 3’ UTR of 1678 bp and an open reading frame (ORF) of 1269 bp (Supplementary Fig. S2). NlapA encoded 422 amino acid residues with a predicted molecular weight of 46.6 kDa and pI of 8.93. The deduced amino acid sequence was predicted to contain two cysteine/histidine-rich domains known as ‘LIM domains’, located amino terminal to a homeodomain (Supplementary Fig. S2).
Phylogenetic analysis using the coding region of NlapA was conducted for determining the evolutionary pattern among insects. NlapA was most closely related to apA of Tribolium castaneum, whereby both appeared to originate from a common ancestor and most distantly related to ap of Apis floreal (Supplementary Fig. S3).
Comparison of the deduced amino acid sequence of NlapA with apA of A. pisum (XP_001946004.2), T. castaneum (NP_001139341.1), B. mori (BAK19079.1) and Danaus plexippus (EHJ74086.1) revealed the highest sequence identity (75.5%) with apA of T. castaneum, followed by A. pisum (57.1%), B. mori (50.6%) and D. plexippus (50.1%).
Characterization of NlASH cDNA
cDNA of an ASH homologue of N. lugens (NlASH, GenBank accession No: KM244736) was amplified from total RNA of wing pads of nymphs. EXPASY analysis indicated that NlASH includes a full-length coding sequence. The start and stop codons are shown in Supplementary Fig. S4. NlASH encoded 233 residues with a predicted molecular weight of 23.9 kDa and pI of 10.09. One basic helix-loop-helix (bHLH) motif was predicted from the deduced amino acid sequence (Supplementary Fig. S4).
Phylogenetic analysis was performed using the coding region of NlASH to determine the evolutionary pattern among insects. NlASH was most closely related to ASH of Apis mellifera and Nasonia vitripennis (all appeared to originate from a common ancestor) and most distantly related to ASH1 of Culex pipiens (Supplementary Fig. S5).
Comparison of the deduced amino acid sequence of NlASH with ASH of A. mellifera (NW_001252982), N. vitripennis (XP_003426998.1) and T. castaneum (NP_001034537.1), SC of D. melanogaster (NP _476803.1) and AC of D. melanogaster (NP_476824.1) revealed the highest sequence identity (47.4%) with AS-C of N. vitripennis, followed by AS-C of T. castaneum (44.1%), AS-C of A. mellifera (43.3%), SC of D. melanogaster (39.9%) and AC of D. melanogaster (36.1%).
Expression patterns of NlapA and NlASH
NlapA was expressed from 1st instar nymphs to 3 days after emergence. The NlapA level was significantly higher in nymphs of MS than those of BS at 2nd and 4th instar and higher in adults of BS than those of MS at 1 and 3 days after emergence (Fig. 1A). NlapA was expressed in the head, thorax, abdomen and leg of 2nd, 3rd, 4th and 5th instar nymphs in a tissue- and development-specific manner. In MS, high expression was detected in the head at 3rd and 4th instar, thorax at 4th and 5th instar and abdomen at 3rd and 4th instar (Fig. 1B). In BS, high expression was detected in the head at 3rd instar and thorax at 4th and 5th instar (Fig. 1C).
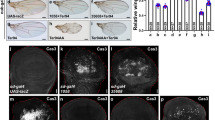
The expression pattern of NlapA and NlASH.
(A) The relative expression pattern of NlapA at different developmental stages of MS and BS; (B) The tissue-specific expression pattern of NlapA in nymphs of MS; (C) The tissue-specific expression pattern of NlapA in nymphs of BS; (D) The relative expression pattern of NlASH at different developmental stages of MS and BS; (E) The tissue-specific expression pattern of NlASH in nymphs of MS; (F) The tissue-specific expression pattern of NlASH in nymphs of BS. BPH actin1 was used as reference control. The expression level was quantified relative to the value of actin1. The average expression level was based on three biological replicates. Error bars indicate standard errors. Bars labeled with asterisk differed significantly between the BPHs of MS and BS, as determined using one way ANOVA (P < 0.05).
NlASH was also expressed from 1st instar nymphs to 3 days after emergence. There was no significant difference in NlASH expression level between insects of MS and BS at each development stage (Fig. 1D). Expression of NlASH during the larval stages was significantly higher than that at the adult stage and peaked at 1st instar (Fig. 1D). NlASH was expressed in the head, thorax, abdomen and leg of 2nd, 3rd, 4th and 5th instar nymphs, with highest expression in thorax. Highest expression was detected in the thorax of 3rd instar nymphs (P < 0.05) (Fig. 1E,F).
Effects of dsRNA microinjection on gene expression of NlapA and NlASH
Both in MS and BS, injection of dsNlapA into 3rd instar nymphs significantly suppressed the expression of endogenous NlapA mRNA as early as 2 d after treatment and at all four sampling points. From the second to 10th day after injection, transcript levels of NlapA were significantly decreased by 30.8%–78.7% and 23.6%–65.3% in MS and BS, respectively (P < 0.05, one-way ANOVA) (Fig. 2A,B). When nymphs at 3rd-instar were injected with dsNlASH, NlASH transcript levels were significantly decreased by 11.4%–69.8% from the second to 10th day after injection (P < 0.05, one-way ANOVA) (Fig. 2C). Treatment of nymphs at 4th instar with dsNlASH led to a significant decrease in the transcript level by 34.2%–58.1% from days 2 to 5 after injection (P < 0.05, Fig. 2D).
The relative expression levels of NlapA and NlASH after injection of dsRNA at 3rd-instar (400ng per nymph).
(A) The relative expression levels of NlapA in BPHs of MS after injection of dsNlapA; (B) The relative expression levels of NlapA in BPHs of BS after injection of dsNlapA; (C) The relative expression levels of NlASH after injection of dsNlASH into 3rd- instar nymphs; (D) The relative expression levels of NlASH after injection of dsNlASH into 4th-instar nymphs. Error bars indicate standard errors. The expression level was quantified relative to the value of the nymphs which were injected with dsGFP. Bars labeled with asterisk differed significantly between the treatments on the same day, as determined using one way ANOVA (P < 0.05). BPH actin1 was used as reference control. The average expression level was based on three biological replicates.
Effects of knockdown of NlapA and NlASH on wing development in vivo
The specific function of NlapA in wing development of N. lugens was investigated using in vivo knockdown. dsRNAs were injected into 3rd instar nymphs and when adults emerged, wing phenotypic defects were observed. The results clearly indicated a deleterious effect on wing development of N. lugens. Compared to dsGFP injection, adults in both MS and BS groups injected with dsNlapA displayed loss-of-bristle forewings (MS: 74.5 ± 3.9%; BS: 70.5 ± 4.9%. Fig. 3J,K,L,S,T,U) and twisted or erect wings (MS: 54.8 ± 1.7%; BS: 26.3 ± 3.2%; Fig. 3B,E).
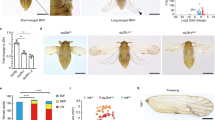
The wing phenotypes of BPHs adults emerged from nymphs injected with dsNlapAor dsNlASH (400ng per nymph) at 3rd-instar.
(A) (D) short-winged and long-winged adults developed from nymphs treated with dsGFP; (B) (E) short-winged and long-winged adults developed from nymphs treated with dsNlapA; (C) (F) short-winged and long-winged adults developed from nymphs treated with dsNlASH; (G) (H) (I) forewing of short-winged adults developed from nymphs treated with dsGFP (magnified 60 times, 100 times and 250 times respectively); (J) (K) (L) forewing of short-winged adults developed from nymphs treated with dsNlapA (magnified 60 times, 100 times and 250 times respectively); (M) (N) (O) forewing of short-winged adults developed from nymphs treated with dsNlASH (magnified 60 times, 100 times and 250 times respectively); (P) (Q) (R) forewing of long-winged adults developed from nymphs treated with dsGFP (magnified 60 times, 100 times and 250 times respectively); (S) (T) (U) forewing of long-winged adults developed from nymphs treated with dsNlapA (magnified 60 times, 100 times and 250 times respectively); (V) (W) (X) forewing of long-winged adults developed from nymphs treated with dsNlASH (magnified 60 times, 100 times and 250 times respectively).
Injection of dsNlASH into 3rd instar nymphs led to significant suppression of NlASH, as expected. Adults treated with dsNlASH exhibited similar phenotypic defects as those injected with dsNlapA, with loss-of-bristle forewings (89.4 ± 3.3%, Fig. 3M,N,O,V,W,X) and twisted or erect wings (28.1 ± 2.8%, Fig. 3C,F).
Effects of knockdown of NlapA on wing size in vivo
Wing sizes of adults emerging from nymphs injected with dsNlapA were measured to establish the role of NlapA in wing morph differentiation. The lengths of the fore- and hindwing from the base to tip of adults that emerged from 3rd-instar nymphs of MS injected with dsNlapA were significantly shorter, compared to those of adults treated with dsGFP, both in the female and male groups (Fig. 4). In contrast, the wing size of adults emerging from 3rd-instar nymphs of BS was not affected by dsNlapA injection. This finding indicates that NlapA is involved in wing morph differentiation to some extent.
The wing length of BPHs adults treated with dsNlapA and dsGFP (400ng per nymph) at 3rd-instar.
Error bars indicate standard errors. Bars labeled with asterisk differed significantly between the treatments, as determined using one way ANOVA (P < 0.05). M: macropterous adults; B: brachypterous adults; FW: forewing; HW: hindwing.
NlapA regulation of NlASH expression
The effect of in vivo knockdown of NlapA on expression of NlASH was investigated using qRT-PCR. Expression of NlASH decreased significantly from 4 to 7 d after dsNlapA injection in both MS and BS (Fig. 5A,B). However, knockdown of NlASH had no effect on expression of NlapA (Fig. 5C). Our results indicate that NlapA is an upstream regulator of NlASH, which is responsible for the development of forewing bristles and normal folding and stretching of wing.
The relative expression levels of NlapA and NlASH after injection of dsRNA (400ng per nymph).
(A) The relative expression levels of NlASH in MS after injection of dsNlapA at 3rd-instar; (B) The relative expression levels of NlASH in BS after injection of dsNlapA at 3rd-instar; (C) The relative expression levels of NlapA after injection of dsNlASH at 3rd-instar. Error bars indicate standard errors. Error bars indicate standard errors. The expression level was quantified relative to the value of the nymphs which were injected with dsGFP. Bars labeled with asterisk differed significantly between the treatments on the same day, as determined using one way ANOVA (P < 0.05). BPH actin1 was used as reference control. The average expression level was based on three biological replicates.
Effect of dsRNA injection on N. lugens survival
We observed no significant differences in survival rates between BPHs treated with dsGFP and dsNlapA at 2, 4, 7 and 10 d (Fig. 6A,B), indicating that dsNlapA has no lethal effects on N. lugens. Following treatment of nymphs at 3rd instar with dsNlASH, the survival rate was significantly decreased, compared to that in nymphs treated with dsGFP (P < 0.05, Fig. 6C), implying that dsNlASH causes lethality in this case. However, when nymphs at 4th instar were treated with dsNlASH, the survival rate was not affected (P < 0.05, Fig. 6D).
Survival response of N. lugens to dsNlapA and dsNlASH injection at 3rd-instar or 4th-instar.
(A) Survival response of BPHs of MS to dsNlapA injection at 3rd-instar; (B) Survival response of BPHs of BS to dsNlapA injection at 3rd-instar; (C) Survival response of BPHs to dsNlASH injection at 3rd-instar; (D) Survival response of BPHs to dsNlASH injection at 4th-instar. dsGFP injection was served as negative control. Each n = 3 groups of 45 nymphs. Survival responses to the dsGFP and dsNlASH were analyzed using Repeated Measure ANOVA: (A).F1,4 = 0.015; P = 0.908; (B). F1,4 = 0.002; P = 0.965; (C). F1,4 = 16.164; P = 0.016; (D). F1,4 = 1.021; P = 0.369. M: macropterous strain; B: brachypterous strain.
Effects of in vivo knockdown of NlapA on expression of other genes in the wing-patterning network
Within the deduced wing-patterning gene network of D. melanogaster, wg, Notch, Dl, ser, vg, sal and ubx are regulated by or interact with ap, with important roles in wing patterning1,2,30. The effects of in vivo knockdown of NlapA on expression of these genes in nymphs were investigated using qRT-PCR. In both MS and BS, knockdown of NlapA led to decrease in NlDl, Nlsal, Nlser, Nlvg and Nlwg expression (Fig. 7A,C,D,F,G). However, Nlubx and Nlnotch displayed different expression patterns between MS and BS in response to NlapA depletion. After dsNlapA injection, Nlnotch expression was suppressed at 2 d and enhanced at 4 d in MS, but upregulated both at 2 d and 4 d in BS (Fig. 7B). Depletion of NlapA resulted in downregulation of Nlubx, both at 2 d and 4 d in MS, while Nlubx expression was increased at 2 d and decreased at 4 d in BS (Fig. 7E).
The effects of in vivo knockdown of NlapA on expression levels of Nldl, Nlnotch,Nlsal,Nlser, Nlubx,Nlvg and Nlwg.
The expression level was quantified relative to the value of the insects which were injected with dsGFP. All error bars indicate the SE of the mean, as determined from three independent replicates. Bars labeled asterisk differed significantly between the treatments on the same day, as determined using one way ANOVA (P < 0.05). BPH actin1 was used as reference control. M: macropterous strain; B: brachypterous strain.
Effects of dsNlASH injection on expression of other genes in the wing-patterning network
In addition to loss-of-bristle forewings, knockdown of NlASH resulted in wing malformation. Accordingly, we examined the effects of in vivo knockdown of NlASH on expression of wg, Notch, Dl, ser, vg, decapentaplegic (dpp) and ubx. Data from qRT-PCR analysis showed that after microinjection of dsNlASH into 3rd instar nymphs, NlDl expression was not affected (Fig. 8A), while Nlnotch was downregulated (Fig. 8B) and Nldpp, Nlser, Nlubx, Nlvg and Nlwg were upregulated (Fig. 8C,D,E,F,G).
The expression levels of Nldelta, Nlnotch,Nldpp,Nlser, Nlubx,Nlvgand Nlwg after dsNlASH injection at 3rd-instar.
All error bars indicate the SE of the mean, as determined from three independent replicates. The expression level was quantified relative to the value of the insects which were injected with dsGFP. Bars labeled asterisk differed significantly between the treatments on the same day, as determined using one way ANOVA (P < 0.05). BPH actin1 was used as reference control.
Discussion
BPH is a significant insect pest of rice. The planthopper has macropterous and brachypterous morphs and can migrate over long distances. Although several reports have focused on the effects of environmental and genetic factors on wing development and dimorphism of BPH26, the underlying molecular mechanisms remain unclear. In the current study, a brachypterous pure strain (BS) and macropterous near-pure strain (MS) were obtained after 38-generation selection, providing relatively good material for the study of wing morph differentiation. In D. melanogaster and T. castaneum, the dorsal selector gene, ap, determines the activation of Notch signaling along the dorso-ventral boundary, in turn, inducing expression of wg and acts as a long-range morphogen, providing positional information along the dorso-ventral axis by triggering expression of vg at different thresholds31. Wing-patterning genes of D. melanogaster, including wg, Notch, Dl, ser, vg, sal and ubx, interact with or are directly regulated by ap1,2,30,32. In ap mutants of D. melanogaster, the wing is lost6,7. Here, we cloned full-length cDNA encoding apA from the hemipteran insect, N. lugens. NlapA is present throughout the body from the nymphal to adult stage and expressed highly in thorax. NlapA displayed different expression patterns in MS and BS. The gene was expressed more highly in nymphs of MS than those of BS at 2nd and 4th instar, while higher expression was detected in adults of BS than those of MS at 1 d and 3 d after emergence (Fig. 1A). dsNlapA injection into 3rd instar nymphs caused wing malformation (loss of bristles of forewing, erect or twisted wings), along with decreased expression of NlDl, Nlsal, Nlser, Nlvg and Nlwg in both MS and BS groups (Fig. 7A,C,D,F,G). However, Nlubx and Nlnotch responded differently to NlapA depletion between MS and BS (Fig. 7B,E). Knockdown of NlapA in nymphs of MS additionally led to decreased wing size of adults. These results clearly indicate that NlapA is involved in wing patterning of N. lugens and higher expression in nymphs is beneficial to generate macropterous adults. Significantly higher expression of ap1 has been reported in winged, relative to unwinged morphs at 1st and 2nd instar nymphs in another hemipteran insect, A. pisum2, similar to our results. As an upstream transcription factor in the wing patterning gene network, ap may be an active participant in wing morph differentiation. In T. castaneum, injection of Tcap dsRNA into the early to late last larval stage induced deletion, marginal truncation and ventralization of the dorsal appendages31. In the present study, injection of dsNlapA into 3rd instar nymphs of N. lugens caused phenotypic defects in wings, but no other appendages of treated insects. The issue of whether proliferation in other appendages occurs earlier than the 3rd instar stage and the effects of NlapA knockdown require further investigation.
ASH encodes bHLH transcription factors and induces the formation of external sensory organs that function in mechano- and chemosensory reception. In Drosophila, an ASH double-mutant lost the majority of bristles, while ectopic expression of ac and/or sc induced extra bristles13,14,15,16. We cloned the full coding sequence of an ASH homologue from the hemipteran insect, N. lugens. NlASH was detected throughout the body from the nymphal to adult stage, but with significantly higher expression at the nymphal stage, there was no significant difference in NlASH expression level between insects of MS and BS at each development stage (Fig. 1D). Moreover, NlASH was expressed more highly in the thorax than other parts of the body (Fig. 1E,F). Adults of N. lugens developed from nymphs injected with dsNlASH displayed the phenotype of loss-of-bristle forewings, in accordance with the function of ac/sc in Drosophila. Additionally, knockdown of NlASH led to twisted or erect wing in N. lugens, which is not reported in Drosophila, signifying an additional role in the wing patterning process in this case. Injection of dsNlASH into 3rd instar nymphs resulted in increased expression of Nldpp, Nlser, Nlubx, Nlvg, Nlwg, decreased expression of Nlnotch and no alteration in expression of NlDl (Fig. 8). In vivo knockdown of NlapA and NlASH results in similar phenotyes, why they caused different expression trends of these wing patterning genes? No available literatures about regulation of ASH on these wing patterning genes could be found. Actually, according to presumptive gene network involved in wing patterning of D. melanogaster, ap is a fairly upstream regulator of wing patterning genes such as dpp, ser, vg, wg and notch, while ASH is a fairly downstream target gene of ap, dpp, ser, ubx, vg and wg1,2. Therefore, knockdown of NlapA directly regulated the expression of wing patterning genes such as dpp, ser, vg, wg and notch and depletion of NlASH could only indirectly regulate the expression of dpp, ser, ubx, vg and wg in a feedback way. This may be the reason for different expression trends of these wing patterning genes caused by in vivo knockdown of NlapA and NlASH.
Expression of ASH is regulated by many upstream modulators, including Ap. In Drosophila, Chip cooperates with Pannier in bridging the GATA factor, activates AS-C through enhancer binding and provides positional information for thorax sensory bristle patterning. Within the Pannier domain of expression, Pannier and Ap compete for binding to a common Chip cofactor. Overexpression of Pannier and absence of Ap promote the development of extra dorsocentral bristles. Thus, Ap antagonizes Pannier function and accurate stoichiometry between these three proteins is essential for both proneural prepattern and compartmentalization of the thorax23. Conversely, in the present study, knockdown of NlapA inhibited NlASH expression (Fig. 5A,B) and suppressed the development of wing bristles. Further research is needed to establish the factors underlying the differential mechanisms of ap regulation on ac-sc between Drosophila and N. lugens.
Our results showed that NlapA regulates the expression of NlASH and in vivo knockdown of NlapA and NlASH results in similar phenotyes. We are yet to establish why dsNlASH, but not dsNlapA, has lethal effects on N. lugens. Downregulation of NlASH was initiated at 4 d after in vivo knockdown of NlapA via dsNlapA injection into nymphs. At this time-point, nymphs were at 5th instar (Fig. 5A,B). However, when nymphs at 3rd instar were injected with dsNlASH, downregulation of NlASH was detected as early as 2 d after injection (at this time-point, nymphs were at 4th-instar) (Fig. 2C) and caused lethality (Fig. 6C). Thus, we assumed that knockdown of NlASH at 4th instar may be the reason for lethality. To confirm this hypothesis, we injected dsNlASH into nymphs at 4th instar instead of 3rd instar and examined the depletion efficiency and survival of N. lugens. Expression of NlASH was significantly decreased 2 d after dsNlASH injection (at this time-point, nymphs were at 5th instar) (Fig. 2D), but no lethality was observed (Fig. 6D). Based on these results, we propose that NlASH is critical at 4th instar and its depletion at this stage induces lethality. In contrast, treatment with dsNlapA did not result in depletion of NlASH at 4th instar and had no lethal effects on N. lugens.
Gene knockdown in planthopper may be conducted via dsRNA ingestion27 or injection33. In the current study, we injected 40 nL (10 μg/μL) dsRNA into 3rd instar nymphs to eliminate expression of NlapA and NlASH. Maximal depletion of NlapA and NlASH were ~79% and ~70%, respectively. RNAi efficiency was not high, compared to that for Spodoptera litura (95% inhibition for the aminopeptidase gene)34. According to previous literature, RNAi efficiency via dsRNA injection in BPH is no greater than 90%. Injection of 0.1 μg dsNlKr-h1 and dsNlDll into 3rd instar N. lugens nymphs resulted in 78% and 87% reduction in NlKr-h1 and NlDll expression, respectively28,29. After injection with 50 nL (5 μg/μL) dsRNA-calreticulin and dsRNA-cathepsin-B of BPH, maximal reduction values of calreticulin and cathepsin-B were reported as 43.8% and 36.4%, respectively33. RNAi efficiency in these reports was similar to that in the present study. RNAi efficiency is affected by various parameters and may be enhanced by increasing the injection volume and dsRNA concentration, which may, however, cause high mortality33,35,36. In this study, we selected a fairly high injection volume and dsRNA concentration (40 nL and 10 μg/μL, respectively) to eliminate expression of NlapA and NlASH. After 10 d of injection, unspecific mortality was about 30-40%, leading to the conclusion that it is not feasible to improve RNAi efficiency by increasing the dsRNA volume and concentration. Both NlapA and NlASH are critical genes for wing development, although they are expressed throughout the nymph and adult stages. since their expression levels were fairly low (only ~0.2–0.6% that of Nlactin1; Fig. 1), knockdown effects were not as significant as those for highly expressed genes, which may partially explain the mild RNAi efficiency observed in the present study.
Methods
Insects
Wild BPH populations were collected from paddy fields in Wuhan, Hubei Province, China and raised on TN1 (Taichuang Native 1, BPH-susceptible rice variety). Predominantly brachypterous strain (BS) and macropterous strain (MS) were selected for 38 successive generations, as described by Morooka and Tojo37. The percentage of brachypterous form (BS) was 100%, while the macropterous form (MS) was ~80%. Insects were raised in a growth chamber under conditions of 28 ± 1°C, 14 h light:10 h dark and 70 ± 5% relative humidity.
cDNA cloning of NlapA and NlASH
Total RNA was extracted from nymphs of N. lugens using TRIzol reagent (Invitrogen, USA). Sequences of NlapA and NlASH were obtained from the transcriptome of N. lugens wing pads. Expressed sequence tags (EST) of NlapA and NlASH were amplified from total RNA of N. lugens. A rapid amplification of cDNA ends (RACE) method was used to isolate full-length cDNA of NlapA. 3’-RACE and 5’-RACE were performed using the GenRace Core Kit (Invitrogen, USA), according to the manufacturer’s instructions. The primers used for EST cloning and RACE are shown in Supplementary Table S1.
In silico analysis
The sequence, start codon, stop codon, molecular weight and isoelectric point of the protein were predicted using Expasy (http://web.expasy.org/)38. Functional domains of the protein were predicted using InterProScan (http://www.ebi.ac.uk/interpro/)39. The deduced amino acid sequences were aligned with Clustal W to determine sequence identity40. Phylogenetic trees were built based on the amino acid sequences using MEGA 6 software with 1000 bootstraps41.
Synthesis of dsRNA
The dsRNA for gene knockdown of NlapA was located between positions 1427 and 1985 (supplementary Fig. S2). The sequence of dsRNA for gene knockdown of NlASH (dsNlASH) is shown in supplementary Fig. S4. DsRNA was synthesized using the T7 RiboMAX Express RNAi System (Promega, USA). The sequence of a T7 polymerase promoter was fused with gene-specific primers at the 5’-end, so that the resulting PCR products contained the T7 polymerase promoter site at both ends. Amplified products were purified using a QIAquickTM PCR purification kit (Qiagen, Germany) and used as the template for in vitro transcription. Sense and antisense strands were transcribed from the DNA template in the same reaction. DsRNAs were extracted with phenol: chloroform and precipitated with isopropanol. Precipitated dsRNA was dissolved in nuclease-free water, heated at 75 °C for 10 min and cooled at room temperature. pEGFP (GenBank accession No.U76561) dsRNA (dsGFP) served as a negative control. The sequences of primers used to synthesize dsRNA are presented in supplementary Table S1.
RNA interference (RNAi) using microinjection
Third instar or fourth instar nymphs (6–12 h) of N. lugens were collected for microinjection, which was carried out as described by Liu et al33. Before injection, a 1.2% agarose plate was prepared and placed on an ice tray. Nymphs were anesthetized with CO2 for 40 s at PCO2 = 1 mPa and placed using a small soft brush on the agarose plate with the abdomen uppermost. Approximately 400 ng dsNlapA, dsNlASH or dsGFP was microinjected into each nymphal conjunctive between the prothorax and mesothorax using a Nanoliter 2000 injector (WPI, USA). For each dsRNA injection, 150 nymphs were used per replicate, with a total of three replicates (i.e., 450 total insects were injected). Treated nymphs were placed in culture dishes with fresh rice seedlings to recover and transferred into glass tubes (3 cm diameter × 25 cm length) containing seven 15-day-old TN1 rice seedlings. Each tube contained five nymphs was sealed with a nylon cover. Tubes were placed in a growth chamber at 70 ± 5% RH at 28 ± 1 °C and a 14 h:10 h (light:dark) cycle for 1-10 days. Survival rates of BPHs were recorded at 2, 4, 7 and 10 days. Meanwhile, five larvae treated with dsRNA were collected randomly at 2, 4, 7 and 10 d after injection and total RNA extracted for qRT-PCR analysis. When adults emerged, wing phenotypes were observed under a stereomicroscope (Olympus szx16, Japan) and recorded. Twisted wings were unfolded by dipping in 5μL ddH2O on glass slides before measurement. Wing length was measured from the base to tip under a stereomicroscope using the cellSens Dimension 1.5 (Olympus szx10, Japan).
qRT-PCR
Total RNA was extracted using TRIzol reagent (Invitrogen, USA). DNA contamination was removed using RNase-free DNase (Ambion, USA). Actin1 (GenBank accession No.EU179846.1) transcript was used as the internal control gene. qRT-PCR reactions were conducted on an ABI Prism 7300 (Applied Biosystems, USA) using SYBR Premix Ex Taq (Perfect Real Time; Takara Biotechnology Corporation Co. Ltd, Dalian, China), according to the manufacturer’s instructions. Independent reactions were performed in triplicate for each RNA sample and the signal intensity of the target gene presented as the average value. Three biological replicates were set up for each treatment. The relative expression level of genes was calculated according to the method of Livak and Schmittgen34. Sequences of primers for qRT-PCR are presented in supplementary Table S1.
Data analysis
All data were analyzed using the general linear model procedure. One-way analysis of variance (ANOVA) was used to evaluate gene expression. Differences between means were examined using the Least Significant Difference (LSD) test at P < 0.05. Survival response to dsRNA microinjection was analyzed using Repeated Measures ANOVA. Percentage values were converted to arcsine before statistical analysis.
Additional Information
How to cite this article: Liu, F. et al. Apterous A modulates wing size, bristle formation and patterning in Nilaparvata lugens. Sci. Rep. 5, 10526; doi: 10.1038/srep10526 (2015).
References
Abouheif, E. & Wray, G. A. Evolution of the gene network underlying wing polyphenism in ants. Science 297, 249–252 (2002).
Brisson, J. A., Ishikawa, A. & Miura, T. Wing development genes of the pea aphid and differential gene expression between winged and unwinged morphs. Insect Mol Biol 19, 63–73 (2010).
Bourgouin, C., Lundgren, S. E. & Thomas, J. Apterous is a Drosophila LIM domain gene required for the development of a subset of embryonic muscles. Neuron 9, 549–561 (1992).
Cohen, B. et al. Apterous, a gene required for imaginal disc development in Drosophila encodes a member of the LIM family of developmental regulatory proteins. Genes Dev 6, 715–729 (1992).
Fernández-Fúnez, P. et al. The relative expression amounts of apterous and its co-factor dLdb/Chip are critical for dorso-ventral compartmentalization in the Drosophila wing. EMBO J 17, 6846–6853 (1998).
Butterworth, F. & King, R. The developmental genetics of apterous mutants of Drosophila melanogaster. Genetics 52, 1153–1174 (1965).
Williams, J. A., Paddock, S. W. & Carroll, S. B. Pattern formation in a secondary field: a hierarchy of regulatory genes subdivides the developing Drosophila wing disc into discrete subregions. Development 117, 571–584 (1993).
Blair, S. S. et al. The role of apterous in the control of dorsoventral compartmentalization and PS integrin gene expression in the developing wing of Drosophila. Development 120, 1805–1815 (1994).
Kim, J., Irvine, K. D. & Carroll, S. B. Cell recognition, signal induction and symmetrical gene activation at the dorsal-ventral boundary of the developing Drosophila wing. Cell 82, 795–802 (1995).
Bertrand, N., Castro, D. S. & Guillemot, F. Proneural genes and the specification of neural cell types. Nat Rev Neurosci 3, 517–530 (2002).
Skeath, J. & Carroll, S. B. Regulation of achaete-scute gene expression and sensory organ pattern formation in the Drosophila wing. Genes Dev 5, 984–995 (1991).
Carroll, S. B. Homeotic genes and the evolution of arthropods and chordates. Nature 376, 479–485 (1995).
Balcells, L., Modolell, J. & Ruiz-Gómez, M. A unitary basis for different Hairy-wing mutations of Drosophila melanogaster. EMBO J 7, 3899–3906 (1988).
Campuzano, S. et al. Excess function hairy-wing mutations caused by gypsy and copia insertions within structural genes of the achaete-scute locus of Drosophila. Cell 44, 303–312 (1986).
Rodríguez, I. et al. Competence to develop sensory organs is temporally and spatially regulated in Drosophila epidermal primordia. EMBO J 9, 3583–3592 (1990).
García-Bellido, A. Genetic analysis of the achaete-scute system of Drosophila melanogaster. Genetics 91, 491–520 (1979).
Zhou, Q. et al. Homology of dipteran bristles and lepidopteran scales: requirement for the Bombyx mori achaete-scute homologue ASH2. Genetics 183, 619–627 (2009).
Asmar, J., Biryukova, I. & Heitzler, P. Drosophila dLMO-PA isoform acts as an early activator of achaete/scute proneural expression. Dev Biol 316, 487–497 (2008).
García-García, M. J. et al. Different contributions of pannier and wingless to the patterning of the dorsal mesothorax of Drosophila. Development 126, 3523–3532 (1999).
Gómez-Skarmeta, J. L. et al. Cis-regulation of achaete and scute: shared enhancer-like elements drive their coexpression in proneural clusters of the imaginal discs. Genes Dev 9, 1869–1882 (1995).
Jafar-Nejad, H. et al. Senseless acts as a binary switch during sensory organ precursor selection. Genes Dev 17, 2966–2978 (2003).
Milán, M. & Cohen, S. M. Temporal regulation of Apterous activity during development of the Drosophila wing. Development 127, 3069–3078 (2000).
Ramain, P. et al. Interactions between chip and the achaete/scute–daughterless heterodimers Are required for pannier-driven proneural patterning. Mol cell 6, 781–790 (2000).
van Meyel, D. J., Thomas, J. B. & Agulnick, A. D. Ssdp proteins bind to LIM-interacting co-factors and regulate the activity of LIM-homeodomain protein complexes in vivo. Development 130, 1915–1925 (2003).
Yang, M., Hatton-Ellis, E. & Simpson, P. The kinase Sgg modulates temporal development of macrochaetes in Drosophila by phosphorylation of Scute and Pannier. Development 139, 325–334 (2012).
Denno, R. F. & Roderick, G. K. Population biology of planthoppers. Annu Rev Entomol 35, 489–520 (1990).
Li, J. et al. RNA interference in Nilaparvata lugens (Homoptera: Delphacidae) based on dsRNA ingestion. Pest Manage Sci 67, 852–859 (2011).
Lin, X. et al. Characterization of the Distal-less gene homologue, NlDll, in the brown planthopper, Nilaparvata lugens(Stål). Gene 535, 112–118 (2014).
JIN, M. et al. Molecular Characterization and Functional Analysis of Krüppel-homolog 1 (Kr-h1) in the Brown Planthopper, Nilaparvata lugens (Stål). J Integr Agric 13, 1972–1981 (2014).
Azpiazu, N. & Morata, G. Function and regulation of homothorax in the wing imaginal disc of Drosophila. Development 127, 2685–2693 (2000).
Tomoyasu, Y. et al. Repeated co-options of exoskeleton formation during wing-to-elytron evolution in beetles. Current Biol 19, 2057–2065 (2009).
Panganiban, G. & Rubenstein, J. L. Developmental functions of the Distal-less/Dlx homeobox genes. Development 129, 4371–4386 (2002).
Liu, S. et al. Gene knockdown by intro-thoracic injection of double-stranded RNA in the brown planthopper, Nilaparvata lugens. Mol Biol 40, 666–671 (2010).
Rajagopal, R. et al. Silencing of Midgut Aminopeptidase N of Spodoptera litura by Double-stranded RNA Establishes Its Role as Bacillus thuringiensis Toxin Receptor. J Biol Chem 277, 46849–46851 (2002).
Meyering-Vos, M. & Müller, A. RNA interference suggests sulfakinins as satiety effectors in the cricket Gryllus bimaculatus. J Insect Physiol 53, 840–848 (2007).
Shakesby, A. et al. A water-specific aquaporin involved in aphid osmoregulation. Mol Biol 39, 1–10 (2009).
Morooka, S. & Tojo, S. Maintenance and Selection of Strains Exhibiting Specific Wing Form and Body Colour under High Density Conditions in the Brown Plant-hopper, Nilaparvata lugens (Homoptera: Delphacidae). Appl Entomol Zool 27, 445–445 (1992).
Bairoch, A., Appel, R. D. & Peitsch, M. C. The ExPASy WWW Server-a tool for proteome research. Protein Data Bank Quart. Newsletter 81, 5–7 (1997).
Zdobnov, E. M. & Apweiler, R. InterProScan–an integration platform for the signature-recognition methods in InterPro. Bioinformatics 17, 847–848 (2001).
Thompson, J. D., Higgins, D. G., & Gibson, T. J. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22, 4673–4680 (1994).
Tamura, K. et al. MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance and maximum parsimony methods. Mol Biol Evol 28, 2731–2739 (2011).
Acknowledgements
This work was supported by a grant from the National Natural Science Foundation of China (No. 31171846).
Author information
Authors and Affiliations
Contributions
H.X.H. conceived the work and prepared the manuscript. F.Z.L., K.Y.L., D.B.H. and J.L performed the experiments. Y.L.Z. and Y.N.F. took the photographs. J.Z. and Y.P.H. analyzed the data.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Supplementary Information
Supplementary Figures 1-6
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Liu, F., Li, K., Li, J. et al. Apterous A modulates wing size, bristle formation and patterning in Nilaparvata lugens. Sci Rep 5, 10526 (2015). https://doi.org/10.1038/srep10526
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep10526
This article is cited by
-
Cloning and differential methylation of 28 S rRNA gene from two wing phenotypes of Sogatella furcifera, an important pest in rice
International Journal of Tropical Insect Science (2023)
-
Transcriptome dynamics during metamorphosis of imaginal discs into wings and thoracic dorsum in Apis mellifera castes
BMC Genomics (2021)
-
Differential DNA methylation between long-winged and short-winged adults of Nilaparvata lugens
3 Biotech (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.