Abstract
Increased plant productivity and decreased microbial respiratory C loss can potentially mitigate increasing atmospheric CO2, but we currently lack effective means to achieve these goals. Soil microbes may play critical roles in mediating plant productivity and soil C/N dynamics under future climate scenarios of elevated CO2 (eCO2) through optimizing functioning of the root-soil interface. By using a labeling technique with 13C and 15N, we examined the effects of plant growth-promoting Pseudomonas fluorescens on C and N cycling in the rhizosphere of a common grass species under eCO2. These microbial inoculants were shown to increase plant productivity. Although strong competition for N between the plant and soil microbes was observed, the plant can increase its capacity to store more biomass C per unit of N under P. fluorescens addition. Unlike eCO2 effects, P. fluorescens inoculants did not change mass-specific microbial respiration and accelerate soil decomposition related to N cycling, suggesting these microbial inoculants mitigated positive feedbacks of soil microbial decomposition to eCO2. The potential to mitigate climate change by optimizing soil microbial functioning by plant growth-promoting Pseudomonas fluorescens is a prospect for ecosystem management.
Similar content being viewed by others
Introduction
Increased plant productivity and decreased microbial respiratory C loss could potentially mitigate increasing atmospheric CO2 concentrations, but we currently lack effective means to achieve these goals1,2,3. The accumulation of ecosystem C is controlled by the balance between plant productivity versus heterotrophic respiration through soil organic matter (SOM) decomposition4,5. Numerous studies have reported that elevated CO2 (eCO2) promotes plant growth and increases photosynthetic C input to soils6,7,8,9. These increased C inputs can stimulate microbial growth and N demand, which can limit soil N availability and plant N uptake under eCO210. On the other hand, positive feedbacks of soil microbial communities under eCO2 may accelerate SOM decomposition and potentially result in soil net C losses11,12,13. It is clear that microbial dynamics play an important role in regulating net ecosystem C storage under future climates, but potentially counteracting responses make it difficult to determine their net effect.
Some beneficial microbial inoculants have been shown to increase plant growth by improving soil N availability in many managed ecosystems under ambient CO214,15,16. However, stoichiometric homeostasis theory suggests that increased plant C fixation under eCO2 can accelerate soil decomposition related to N mineralization by increasing the allocation of resources towards the microbial production of enzymes that degrade N-rich substrates17,18,19. Recent studies suggest that eCO2 promotes SOM decomposition associated with microbial activity through increased rhizosphere priming effects (RPEs)20,21. Increased microbial utilization of C exudate under eCO2 can induce increased N mineralization and create positive nutrient feedbacks to mitigate soil N limitation8,21,22,23. However, in order to increase the efficiency by which microbes metabolize and transform plant detritus under eCO2, microbial inoculants should increase plant productivity, without accelerating SOM decomposition rates and microbial respiratory C loss. For example, applications of arbuscular mycorrhizal fungi (AMF) were generally shown to facilitate plant growth and stimulate soil carbon storage under ambient CO224,25. However, AMF can speed up the turnover of recently fixed photosynthetic C associated with accelerated N cycling in spite of increased plant net primary productivity in forests exposed to eCO2, limiting soil C accumulation20,21.
Many isolated strains have been identified as plant growth-promoting rhizobacteria (PGPR), which could be used to stimulate plant growth under rising atmospheric CO226,27. Numerous studies have shown that PGPR can effectively increase plant performance in nutrient-limited environments, through positively influencing root growth and morphology and promoting other beneficial plant–microbe symbioses28,29. In various natural terrestrial ecosystems, application of PGPR inoculants has emerged as a technology to facilitate grassland management30, ecosystem restoration29 and reforestation31. In contrast to soil saprotrophic bacteria, PGPR appear to have high substrate use efficiencies32. Under future climate conditions, eCO2 may increase PGPR dominance as these plant-associated microbes enhance plant success under eCO226,33. Nevertheless, our understanding of how plants interact with PGPR inoculants to regulate SOM decomposition and soil N availability in the rhizosphere is still limited, despite the potential of PGPB to alleviate environmental stresses imposed by eCO226,27. The effective application of these PGPB in natural terrestrial ecosystems requires understanding the traits that may enhance ecological performance in the rhizosphere3,16.
Here, we used a dual-isotope labeling technique with 13C and 15N to evaluate the effect of Pseudomonas fluorescens inoculation on plant productivity and soil C/N cycling under eCO2. P. fluorescens, which is common in soils, improves plant growth through several different mechanisms, such as the suppression of plant diseases and enhancement of essential metal uptake15,28,31. The objectives of the current study were to test whether using P. fluorescens as a soil microbial inoculum (1) improves plant productivity and photosynthetic C input to soils, (2) promotes plant N use and (3) mitigates C lost through microbial respiration.
Results
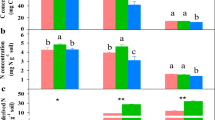
Bacteria and eCO2 had positive, cumulative effects on plant productivity in terms of total plant biomass C (Figure 1a). The highest plant productivity occurred in the combined bacteria and eCO2 treatment (Figure 1a). In addition, bacteria and eCO2 increased the plant C:N ratio as individual treatments, but demonstrated a synergistic effect when combined (Figure 1b), suggesting that plants can assimilate more C per unit of N in tissue under eCO2 with these P. fluorescens inoculants. Increased plant root surface area associated with bacterial inoculants and eCO2 (Table S1) demonstrated a strong positive relationship with plant tissue C:N (Figure 2a). Combined bacterial inoculant and eCO2 treatments demonstrated significantly lower total soil enzyme C:N acquisition activity ratios (Table S1); and decreases in enzyme C:N were strongly correlated with higher plant tissue C:N (Figure 2b), suggesting a strong competition for N between plant and soil microbes.
Plant C pool size per pot (a) and biomass C:N ratio (b).
Control: ambient CO2 and without bacteria addition; B: ambient CO2 and with bacteria addition; eCO2: elevated CO2 and without bacteria addition; B + eCO2: elevated CO2 and with bacteria addition. Error bars show standard error of the mean (n = 6). The same letters denote non-significant differences between treatments (P > 0.05).
Linear relationships of plant C:N ratio with root surface area (a) and total C:N enzyme ratio (b) across all treatments.
Control: ambient CO2 and without bacteria addition; B: ambient CO2 and with bacteria addition; eCO2: elevated CO2 and without bacteria addition; B + eCO2: elevated CO2 and with bacteria addition.
We took advantage of the distinct δ15N values to estimate the relative sources of plant N (N from the original inorganic pool vs. N mineralized from SOM). eCO2 significantly increased the δ15N value in plant biomass (Figure 3a). The higher δ15N in soil organic N compared to soil inorganic N (see δ15N values of two pools in Methods) indicates that eCO2 increased plant uptake of mineralized N from SOM relative to soil inorganic N. However, P. fluorescens inoculants did not change plant δ15N values at each CO2 level (Fig. 3a), suggesting these inoculants did not accelerate soil decomposition related to N mineralization.
δ15N value in plant biomass (a) and linear relationships of plant δ15N value with rhizosphere priming effects (RPEs) (b) and total root length (c) across all treatments.
Control: ambient CO2 and without bacteria addition; B: ambient CO2 and with bacteria addition; eCO2: elevated CO2 and without bacteria addition; B + eCO2: elevated CO2 and with bacteria addition. Error bars show standard error of the mean (n = 6). The same letters denote non-significant differences between treatments (P > 0.05).
Although high variability in rhizosphere priming effects was observed among the treatments, priming of SOM decomposition was positively related with plant δ15N (Figure 3b), suggesting that rhizosphere exudation (priming) was induced by plants in order to facilitate N mineralization from SOM. Likewise, total root length (which determines root system expansion) was positively related with plant δ15N (Figure 3c).
Compared to the control, all treatments induced higher soil C inputs from the plant-derived C (Figure 4a). However, significantly higher mass-specific microbial respiration (lower C use efficiency) was observed only under eCO2 (Figure 4b).
Plant-derived C inputs to soil (a) and specific microbial respiration (b).
Control: ambient CO2 and without bacteria addition; B: ambient CO2 and with bacteria addition; eCO2: elevated CO2 and without bacteria addition; B + eCO2: elevated CO2 and with bacteria addition. Error bars show standard error of the mean (n = 6). The same letters denote non-significant differences between treatments (P > 0.05).
Discussion
The capability of plants and soil microbes to successfully sequester atmospheric C in terrestrial ecosystems largely depends on plant productivity along with microbial decomposition and mineralization feedbacks within the rhizosphere8,10,11,12. Advancing current efforts to mitigate effects of climate change could minimize harmful effects of elevated CO21,26. Here, for the first time, we show that addition of a microbial inoculant has the potential to promote plant productivity while mitigating positive feedbacks of microbial decomposition to increased plant C inputs that typically accompany eCO2. Our results also demonstrated that P. fluorescens inoculation led to increased plant tissue C:N under eCO2, resulting in an increased capacity to store C per unit of N in plant tissue. Therefore, this soil microbial inoculant may be a useful tool to mitigate climate change.
The results from this study are consistent with numerous field and growth chamber experiments showing that P. fluorescens inoculants can increase plant production under ambient CO214,15,31 and show that these largely stimulating effects could be additive when combined with eCO2. Although it was impossible to exclude potential fertilization effects on plant available N through bacterial cell addition, the increases in total soil N pool by bacterial cell addition (0.016%) is likely negligible in comparison with the increase in plant growth by an average of 42%. In addition, chemical adjustments to litter C:N may contribute to reduced quality and decomposability of plant litter under eCO29,34 which may reduce soil decomposition rates. In comparison to our previous study, overall plant C:N ratios during the rapid vegetative growth stage were lower than during the reproductive stage35.
Plant roots play an essential role in regulating acquisition of soil nutrients. Nevertheless, little is known about the relationship of root functional traits with plant N use strategies. Root surface area is generally correlated to plant nutrient uptake rates36. P. fluorescens inoculants significantly increased root surface area under eCO2 (Table S1), suggesting that P. fluorescens inoculants could enhance the potential for plant roots to acquire N under N-limited eCO2 condition. Moreover, our results demonstrated that root surface area positively correlated with plant C:N ratio across experimental treatments (Figure 2a), further indicating that the plants' ability to acquire N from the soil could be influenced by soil N availability.
Coupled C and N processes in the rhizosphere play a critical role in maintaining the sustainability of ecosystems34,37. Recent studies suggest that plant productivity slows when plant N demand decouples from soil N cycling under climate change or other ecological disturbances38,39. In our previous study conducted in the same ecosystem as this present work, we found that eCO2 increased microbial biomass N immobilization and decreased soil N availability19. Likewise, this study revealed that eCO2 significantly decreased soil enzyme C:N stoichiometry (Table S1), indicating greater microbial demand for soil N under eCO240. N limitation could ultimately dampen ecosystem C sequestration in terms of the eCO2 fertilization effect on plant productivity10,34. However, P. fluorescens inoculant did not directly affect soil enzyme C:N stoichiometry (Table S1). Moreover, the negative relationship between plant biomass C:N and enzyme C:N (Figure 2b) suggests that plants can continue to grow through increase in their capacity to store C per unit of N in response to changes in soil N availability, which is mediated by soil microbial activities.
It is well known that plants can alter their N uptake rates to cope with plant physiological and environmental changes41,42. By using the 15N isotopic method, we observed that eCO2 increased the importance of N mineralized from SOM, indicating that the positive effects of eCO2 on soil N enzyme activities increased soil N availability (Figure 3a and Table S1). However, P. fluorescens inoculants had no detectable effects on δ15N values of plant biomass (Figure 3a) or on soil N enzyme activities (Table S1). This suggests that P. fluorescens does not facilitate N mineralization under eCO2 conditions. We note that N in P. fluorescens cells is mostly in organic form with the δ15N value of 4.2‰ (the δ15N value of organic N in SOM is 587.5‰). If plants took up mineralized N from dead P. fluorescens cells, the δ15N value of plant biomass should be lower than plant uptake. However, even if all bacterial N was absorbed by plants, the N in P. fluorescens cells would only contribute from 0.8% to 2.2% of the total plant N pool. In addition, the δ15N value of organic N corrected by bacterial cells (587.4‰) was still much higher than inorganic N (445.7‰).
eCO2-induced rhizosphere priming effects and subsequent microbial N mineralization could influence the magnitude of plant growth8,21. Previous studies have identified several plant and microbial traits related to RPE8,21,43, but the direct evidence of priming-related effects on plant N availability has not been well documented. Our results, for the first time, clearly demonstrated that priming of SOM decomposition was positively related to plant N availability (Figure 3b), suggesting that priming made soil N more available to the plant for uptake. Increased root length was also observed in this study as an important root functional trait related to plant N uptake adaptations associated with microbial plant growth-promoting properties under eCO2 conditions. These results add to a growing body of evidence that plants could increase N availability through rhizosphere priming and development of root systems to alleviate nitrogen limitation under eCO28,9,21,43.
P. fluorescens inoculants and eCO2 were expected to increase plant C inputs to soil (Figure 4a). However, a synergistic effect of bacteria and eCO2 on plant-derived C was not observed (Figure 4a). This may be due to the use of planting pots which may have constrained root growth in this experiment. Results from a meta-analysis suggest that CO2-induced increases in belowground biomass are stronger in plants grown in open fields relative to closed pots9. In spite of higher rates of new C inputs, P. fluorescens inoculants and eCO2 demonstrated contrasting effects on heterotrophic respiration due to microbial activities. eCO2 increased mass-specific microbial respiration (Figure 4b)35, consistent with previous observations of climate-induced positive feedbacks12,40. P. fluorescens inoculants did not change mass-specific microbial respiration under ambient CO2 but mitigated positive microbial feedbacks under eCO2 conditions (Figure 4b). Thus, these findings suggest that P. fluorescens inoculants may potentially decrease soil C losses via heterotrophic respiration.
Our results indicate that P. fluorescens inoculants may optimize soil microbial functioning and potentially be implemented as a strategy for increasing plant productivity while mitigating positive feedbacks of microbial decomposition to eCO2. If the benefits of P. fluorescens inoculants can be scaled from the growth chamber and applied in natural ecosystems in a high-CO2 world, the potential for terrestrial C sequestration may increase to mitigate rising atmospheric CO2. Further assessment is needed to extend these findings to field experiments and to formulate economical methods of inoculation for field deployment. Additional experiments should be performed to assess rhizosphere colonization by P. fluorescens inoculants across a range of plant species.
Methods
Experimental setup
The C4, perennial grass Bouteloua gracilis was selected in this experiment because it is a widespread grass in North America and accounts for most ecosystem net primary productivity in the shortgrass prairie of the central and southern Great Plains44. We collected soils for this experiment from the USDA-ARS Central Plains Experimental Range, Colorado, USA. To trace sources of plant N uptake in our experiment, we took advantage of 15N-enriched soil from a prior experiment43. About 15 years after 0.5 g m−2 15N was added (and 9 years after the completion of the original experiment), soil was collected from the top 15-cm and sieved (mesh size 2 mm) to remove roots and homogenize the soil. The soil is a Remmit fine sandy loam (Ustollic camborthids) with 0.8% organic C in the top 15 cm. To reduce soil nutrient availability, we leached the soil with DI water in large buckets with small waterspouts. After that, the soil was air-dried and passed through a 2-mm sieve to further remove plant residues, soil fauna and other coarse materials and then homogenized to attain a composite sample. 600 g soil (dry weight) was packed into each plastic pot at a similar bulk density to field conditions. The pots were capped at the bottom and no leaching occurred during the experiment. Before our experiment, the initial soil inorganic N (NH4+ + NO3−) and organic N (total soil N minus soil inorganic N) content were 0.14 ± 0.002 and 0.41 ± 0.03 mg g−1, respectively (n = 8; t-statistic < 0.0001). The δ15N values of inorganic and organic N were 445.7 ± 12.5 and 587.5 ± 10.8‰, respectively (n = 8; t-statistic < 0.0001). Inorganic δ15N was determined using the diffusion method45. Organic N was estimated as the difference between total and inorganic N and organic δ15N was determined by mass balance. The δ15C value of soil organic matter was −20.4‰ and no inorganic C was present.
We performed our experiment in climate-controlled growth chambers (Percival PGC-9/2, Percival Scientific, Perry, IN, USA). The chamber systems we used were shown to have high reliability and stability during comparative studies of plant genetics and eco-physiology46,47. To simulate field conditions during the growing season, the growth chambers were set to a 14 h daytime period with light intensity of 700 μmol m−2 s−1. The daytime and night-time temperatures were 25°C and 18°C, respectively. We used a Li-250 light meter (LI-COR, Lincoln, NE, USA) and Telaire 7001 meters (Telaire, Goleta, CA, USA) to ascertain the reliability of light intensities and temperatures of the chambers every day. To achieve continuous 13C-labeling of plant tissues, the chambers were modified to receive an influx of 13C-depleted CO2 (δ13C = −33.1‰) combined with an external air input which had been scrubbed by a 70-L gas tight soda lime column. The CO2 concentrations inside the chambers were calibrated by infrared CO2 sensors (GMM220, Vaisala, Helsinki, Finland) and continuously monitored by Telaire 7001 meters (ambient CO2 concentration: 371.9 ± 2.1 ppm (mean ± se)); elevated CO2 concentration: 702.9 ± 8.7 ppm). The δ13C values of CO2 inside chambers were continuously monitored by a Picarro G2101i 13CO2 analyzer (Picarro, Sunnyvale, CA, USA). Throughout the experiment, the δ13C values of CO2 were stable (ambient CO2: −25.0 ± 0.2‰; elevated CO2: −24.9 ± 0.2‰) and there was no significant daily difference in the δ13C values of experimental chambers.
We used 24 planted pots with six replicates for each treatment: ambient CO2 without bacteria addition (Control), ambient CO2 and with bacteria addition (B), elevated CO2 and without bacteria addition (eCO2); elevated CO2 and with bacteria addition (B + eCO2). Correspondingly, another 24 unplanted pots were set up with six replicates for each treatment. Three seedlings were transferred to each planted pot after the emergence of the first euphylla on moist filter paper in glass Petri dishes. During the first week of the experiment, all pots (including unplanted plots) were rewetted to 25% gravimetric soil moisture content to enhance seedling growth. After that, the gravimetric water content in each pot was maintained at 15% (approximately 50% water holding capacity) using DI water, with no fertilizer additions. On day 10, we inoculated 1.5 × 109 cells of P. fluorescens isolated from soil to 24 randomly assigned pots (12 planted and 12 unplanted), which we refer to as bacterial treatments. The strains were grown in liquid LB medium (Sigma, USA) at room temperature on a rotary shaker (200 rpm). After 48 h the cells were harvested by centrifugation for 10 min at 7000 rpm at 4°C and re-suspended in sterile DI water. Following two additional washes, they were re-suspended in sterile DI water. To obtain 1.5 × 109 cells, the cell density was adjusted based upon optical density measured at 600 nm48. Bacterial cell addition only increased the total soil C and N pool sizes by 0.006% and 0.016%, respectively. The δ13C and δ15N values of bacterial cells were −24.9‰ and 4.2‰, respectively. After bacterial cell addition, therefore, the δ13C value of soil organic matter and δ15N value of soil organic N was −20.4‰ and 587.4‰, respectively.
Measurements
We harvested 30 days after planting, because plants have high rates of interactions with soil processes during the rapid vegetative growth stage35,49. We placed each pot (planted and unplanted pots) in an opaque, capped PVC chamber (45-cm height, 20-cm diameter)50. Briefly, we sealed the bottom of chamber by placing it on a plastic dish containing water to impede gas loss and removed CO2 inside each chamber by circulating air through a gas tight in-line soda-lime scrubber for 30 min. Then we immediately collected an initial 30 ml air sample with a plastic syringe to ensure CO2 scrubbing was complete. After 3 h we collected a final CO2 sample. Half of the final gas sample was analyzed for δ13C by a Thermo Finnigan Delta Plus XP isotope ratio mass spectrometer (Thermo Finnigan, Bremen, Germany) and the other was analyzed for CO2 concentration by a Li-Cor 820 (LICOR Inc. Lincoln, NE) calibrated with 4 standard gases.
After CO2 trapping, we immediately separated plants into shoots and roots and homogenized soils. Traits of fresh roots (root length and surface area) were analyzed by a WinRHIZO system (Regent Instruments, Montreal, QC, Canada). Then each plant tissue and part of each soil sample were dried, weighed, ground and analyzed for C/N concentrations and 13C/15N by a Thermo Finnigan Delta Plus XP isotope ratio mass spectrometer.
The continuous 13C-labeling of plant tissues allowed us to separate total soil respiration (Ctotal) into SOM-C (SOC) decomposition (CSOC) and root respiration using the following model51:

where δ13Croot, δ13Ctotal and δ13CSOC are the δ13C values of the root respiration, the total soil respiration in the planted treatments and the mean value of soil respiration in the corresponding unplanted treatments, respectively. For each CO2 treatment, the δ13C value of the root respiration was determined by growing plants in a SOM-free sand.
We calculated rhizosphere priming effects (RPEs) using following equation:

We calculated the amount of new soil C (Cnew) from plant-derived C through rhizodeposition during the experiment using the following model52:

where Cend is total amount of SOC at the end of the experiment, δ13Cstart (corrected by the C in the inoculants if needed) and δ13Cend each are the δ13C values of SOC at the start and end of the experiment and δ13Croot biomass is the δ13C values of root biomass.
A subsample of fresh soil from each pot was used to assess microbial community attributes. To normalize activity to the size of the microbial community, specific microbial respiration was calculated as the ratio of SOC decomposition rate (CSOC) to microbial biomass C (MBC)35. MBC was determined by the fumigation–extraction method and the factor for MBC calculation was 0.4553. To stoichiometrically link plant nutrient availability and microbial-mediated SOM decomposition, we measured soil enzyme activities involved in the cycling of C (β-Glucosidase and β-D-Cellubiosidase) and N (N-acetyl-β-Glucosaminidase and Leucine amino peptidase) cycling. The enzyme activities were measured using a 4-methylumbelliferyl (MUB) substrate yielding the highly fluorescent cleavage products MUB upon hydrolysis40.
Statistical analyses
To determine the effects of eCO2 and bacteria on plant productivity and soil C/N cycling, we used a two-way ANOVA with eCO2 and bacteria as fixed effects by SPSS 13.0. Post hoc means were determined using least squares means separation by SPSS. The significance level (P value) of post-hoc LSD (least significant difference) test was set to 0.05. Data not meeting assumptions of normality and homogeneity of variance were log-transformed before statistical testing. Simple regression was performed to evaluate relationships underlying rhizosphere processes by Sigma Plot 10.0. Significant effects are reported at P < 0.05 unless otherwise stated.
References
Lal, R. Soil carbon sequestration to mitigate climate change. Geoderma 123, 1–22 (2004).
Cramer, W. et al. Global response of terrestrial ecosystem structure and function to CO2 and climate change: results from six dynamic global vegetation models. Glob. Change Biol. 7, 357–373 (2001).
Hart, K. M. et al. Tracking the fate of microbially sequestered carbon dioxide in soil organic matter. Environ. Sci. Technol. 47, 5128–5137 (2013).
Luo, Y. & Zhou, X. Soil respiration and the environment. (Elsevier Academic Press, 2006).
Schlesinger, W. H. & Andrews, J. A. Soil respiration and the global carbon cycle. Biogeochemistry 48, 7–20 (2000).
Morgan, J. A. et al. C4 grasses prosper as carbon dioxide eliminates desiccation in warmed semi-arid grassland. Nature 476, 202–205 (2011).
Pendall, E., Mosier, A. R. & Morgan, J. A. Rhizodeposition stimulated by elevated CO2 in a semiarid grassland. New Phytol. 162, 447–458 (2004).
Phillips, R. P., Finzi, A. C. & Bernhardt, E. S. Enhanced root exudation induces microbial feedbacks to N cycling in a pine forest under long-term CO2 fumigation. Ecol. Lett. 14, 187–194 (2011).
Nie, M., Lu, M., Bell, J., Raut, S. & Pendall, E. Altered root traits due to elevated CO2: a meta-analysis. Glob. Ecol. Biogeogr. 22, 1095–1105 (2013).
Luo, Y. et al. Progressive nitrogen limitation of ecosystem responses to rising atmospheric carbon dioxide. Bioscience 54, 731–739 (2004).
Carney, K. M., Hungate, B. A., Drake, B. G. & Megonigal, J. P. Altered soil microbial community at elevated CO2 leads to loss of soil carbon. Proc. Natl. Acad. Sci. U. S. A. 104, 4990–4995 (2007).
Nie, M. et al. Positive climate feedbacks of soil microbial communities in a semi-arid grassland. Ecol. Lett. 16, 234–241 (2013).
Pendall, E. et al. Warming reduces carbon losses from grassland exposed to elevated atmospheric carbon dioxide. Plos One 8, e71921 (2013).
Kloepper, J. W., Leong, J., Teintze, M. & Schroth, M. N. Enhanced plant growth by siderophores produced by plant growth-promoting rhizobacteria. Nature 286, 885–886 (1980).
Berg, G. Plant-microbe interactions promoting plant growth and health: perspectives for controlled use of microorganisms in agriculture. Appl. Environ. Microbiol. 84, 11–18 (2009).
King, G. M. Enhancing soil carbon storage for carbon remediation: potential contributions and constraints by microbes. Trends Microbiol. 19, 75–84 (2011).
Bell, C. et al. Rhizosphere stoichiometry: are C:N:P ratios of plants, soils and enzymes conserved at the plant species-level? New Phytol. 201, 505–517 (2014).
Sterner, R. W. & Elser, J. J. in Ecological stoichiometry: The biology of elements from molecules to the biosphere (eds Sterner, R. W. & Elser, J. J.) 1–43 (Princeton University Press, 2002).
Dijkstra, F. A. et al. Contrasting effects of elevated CO2 and warming on nitrogen cycling in a semiarid grassland. New Phytol. 187, 426–437 (2010).
Cheng, L. et al. Arbuscular mycorrhizal fungi increase organic carbon decomposition under elevated CO2 . Science 337, 1084–1087 (2012).
Phillips, R. P. et al. Roots and fungi accelerate carbon and nitrogen cycling in forests exposed to elevated CO2 . Ecol. Lett. 15, 1042–1049 (2012).
Martin-Olmedo, P., Rees, R. M. & Grace, J. The influence of plants grown under elevated CO2 and N fertilization on soil nitrogen dynamics. Glob. Change Biol. 8, 643–657 (2002).
Levine, J. M., Pachepsky, E., Kendall, B. E., Yelenik, S. G. & HilleRisLambers, J. Plant-soil feedbacks and invasive spread. Ecol. Lett. 9, 1005–1014 (2006).
Hooker, J. E. & Black, K. E. Arbuscular mycorrhizal fungi as components of sustainable soil-plant systems. Crit. Rev. Biotechnol. 15, 201–212 (1995).
Hamel, C. & Strullu, D. G. Arbuscular mycorrhizal fungi in field crop production: potential and new direction. Can. J. Plant Sci. 86, 941–950 (2006).
Compant, S., van der Heijden, M. G. A. & Sessitsch, A. Climate change effects on beneficial plant-microorganism interactions. FEMS Microb. Ecol. 73, 197–214 (2010).
Drigo, B., Kowalchuk, G. A. & van Veen, J. A. Climate change goes underground: effects of elevated atmospheric CO2 on microbial community structure and activities in the rhizosphere. Biol. Fert. Soils 44, 667–679 (2008).
Vessey, J. K. Plant growth promoting rhizobacteria as biofertilizers. Plant Soil 255, 571–586 (2003).
Requena, N., Perez-Solis, E., Azcon-Aguilar, C., Jeffries, P. & Barea, J. M. Management of indigenous plant-microbe symbioses aids restoration of desertified ecosystems. Appl. Environ. Microbiol. 67, 495–498 (2001).
van der Heijden, M. G. A. et al. Symbiotic bacteria as a determinant of plant community structure and plant productivity in dune grassland. FEMS Microb. Ecol. 56, 178–187 (2006).
Chanway, C. P. Inoculation of tree roots with plant growth promoting soil bacteria: an emerging technology for reforestation. Forest Sci. 43, 99–112 (1997).
Fan, T. W. M., Bird, J. A., Brodie, E. L. & Lane, A. N. 13C-Isotopomer-based metabolomics of microbial groups isolated from two forest soils. Metabolomics 5, 108–122 (2009).
Marilley, L., Hartwig, U. A. & Aragno, M. Influence of an elevated atmospheric CO2 content on soil and rhizosphere bacterial communities beneath Lolium perenne and Trifolium repens under field conditions. Microb. Ecol. 38, 39–49 (1999).
Pendall, E. et al. Below-ground process responses to elevated CO2 and temperature: a discussion of observations, measurement methods and models. New Phytol. 162, 311–322 (2004).
Carrillo, Y., Dijkstra, F., Pendall, E., LeCain, D. & Tucker, C. Plant rhizosphere influence on microbial C metabolism: the role of elevated CO2, N availability and root stoichiometry. Biogeochemistry, 2–3, 229–240 (2014).
Tachibana, Y. & Ohta, Y. Root surface area, as a parameter in relation to water and nutrient uptake by cucumber plant. Soil Sci. Plant Nutr. 29, 387–392 (1983).
Kulmatiski, A., Beard, K. H., Stevens, J. R. & Cobbold, S. M. Plant–soil feedbacks: a meta-analytical review. Ecol. Lett. 11, 980–992 (2008).
Yelenik, S. G. & D'Antonio, C. M. Self-reinforcing impacts of plant invasions change over time. Nature 503, 517–520 (2013).
Reich, P. B. & Hobbie, S. E. Decade-long soil nitrogen constraint on the CO2 fertilization of plant biomass. Nature Clim. Change 3, 278–282 (2013).
Nie, M., Pendall, E., Bell, C. & Wallenstein, M. D. Soil aggregate size distribution mediates microbial climate change feedbacks. Soil Biol. Biochem. 68, 357–365 (2014).
Nasholm, T., Kielland, K. & Ganeteg, U. Uptake of organic nitrogen by plants. New Phytol. 182, 31–48 (2009).
Schimel, J. P. & Bennett, J. Nitrogen mineralization: challenges of a changing paradigm. Ecology 85, 591–602 (2004).
Dijkstra, F. A. et al. Long-term enhancement of N availability and plant growth under elevated CO2 in a semi-arid grassland. Funct. Ecol. 22, 975–982 (2008).
Alward, R. D., Detling, J. K. & Milchunas, D. G. Grassland vegetation changes and nocturnal global warming. Science 283, 229–231 (1999).
Brookes, P. D., Stark, J. M., Mclnteer, B. B. & Preston, T. Diffusion method to prepare soil extracts for automated nitrogen-15 analysis. Soil Sci. Soc. Am. J. 53, 1707–1711 (1989).
Haselhorst, M. S. H., Edwards, C. E., Rubin, M. J. & Weinig, C. Genetic architecture of life history traits and environment-specific trade-offs. Mol. Ecol. 20, 4042–4058 (2011).
Edwards, C. E. et al. The genetic architecture of ecophysiological and circadian traits in Brassica rapa. Genetics 189, 375–390 (2011).
Ravnskov, S., Nybroe, O. & Jakobsen, I. Influence of an arbuscular mycorrhizal fungus on Pseudomonas fluorescens DF57 in rhizosphere and hyphosphere soil. New Phytol. 142, 113–122 (1999).
Nie, M. et al. Understanding plant-microbe interactions for phytoremediation of petroleum-polluted soil. Plos One 6, e17961 (2011).
Dijkstra, F. A., Morgan, J. A., Blumenthal, D. & Follett, R. F. Water limitation and plant inter-specific competition reduce rhizosphere-induced C decomposition and plant N uptake. Soil Biol. Biochem. 42, 1073–1082 (2010).
Zhu, B. A. & Cheng, W. X. Rhizosphere priming effect increases the temperature sensitivity of soil organic matter decomposition. Glob. Change Biol. 17, 2172–2183 (2011).
Dijkstra, F. A. & Cheng, W. X. Interactions between soil and tree roots accelerate long-term soil carbon decomposition. Ecol. Lett. 10, 1046–1053 (2007).
Vance, E. D., Brookes, P. C. & Jenkinson, D. S. An extraction method for measuring soil microbial biomass C. Soil Biol. Biochem. 19, 703–707 (1987).
Acknowledgements
We thank Drs. Yolima Carrillo and Feike Dijkstra for providing experimental facilities. We also thank Dr. Marcus Brock and Mark Schimelpfenig for laboratory assistance. This material is based upon work supported by the US Department of Agriculture, US Department of Energy's Office of Science (BER), through the Terrestrial Ecosystem Science program and by the National Science Foundation (DEB# 1021559). Any opinions, findings and conclusions or recommendations expressed in this material are those of the author(s) and do not necessarily reflect the views of the National Science Foundation.
Author information
Authors and Affiliations
Contributions
M.N. and E.P. conceived and designed the experiments. M.N. and C.B. performed the experiments. M.N., C.B. and E.P. analyzed the data. M.N., C.B., M.D.W. and E.P. wrote the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Supplementary Information
Table S1
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article's Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder in order to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Nie, M., Bell, C., Wallenstein, M. et al. Increased plant productivity and decreased microbial respiratory C loss by plant growth-promoting rhizobacteria under elevated CO2. Sci Rep 5, 9212 (2015). https://doi.org/10.1038/srep09212
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep09212
This article is cited by
-
Multifarious Responses of Forest Soil Microbial Community Toward Climate Change
Microbial Ecology (2023)
-
Temperature effect on water dynamics in tetramer phosphofructokinase matrix and the super-arrhenius respiration rate
Scientific Reports (2021)
-
Bacillus atrophaeus HAB-5 secretion metabolites preventing occurrence of systemic diseases in tobacco plant
European Journal of Plant Pathology (2020)
-
Role of biochar and plant growth promoting rhizobacteria to enhance soil carbon sequestration—a review
Environmental Monitoring and Assessment (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.