Abstract
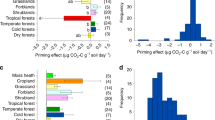
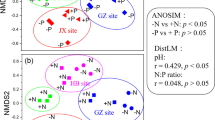
The soil priming effect (PE), defined as the modification of soil organic matter decomposition by labile carbon (C) inputs, is known to influence C storage in terrestrial ecosystems. However, how chronic nutrient addition, particularly in leguminous and non-leguminous forests, will affect PE through interaction with nutrient (e.g., nitrogen and phosphorus) availability is still unclear. Therefore, we collected soils from leguminous and non-leguminous subtropical plantations across a suite of historical nutrient addition regimes. We added 13C-labeled glucose to investigate how background soil nutrient conditions and microbial communities affect priming and its potential microbial mechanisms. Glucose addition increased soil organic matter decomposition and prompted positive priming in all soils, regardless of dominant overstory tree species or fertilizer treatment. In non-leguminous soil, only combined nitrogen and phosphorus addition led to a higher positive priming than the control. Conversely, soils beneath N-fixing leguminous plants responded positively to P addition alone, as well as to joint NP addition compared to control. Using DNA stable-isotope probing, high-throughput quantitative PCR, enzyme assays and microbial C substrate utilization, we found that positive PE was associated with increased microbial C utilization, accompanied by an increase in microbial community activity, nutrient-related gene abundance, and enzyme activities. Our findings suggest that the balance between soil available N and P effects on the PE, was dependent on rhizosphere microbial community composition. Furthermore, these findings highlight the roles of the interaction between plants and their symbiotic microbial communities in affecting soil priming and improve our understanding of the potential microbial pathways underlying soil PEs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
We are sorry, but there is no personal subscription option available for your country.
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The sequencing raw data are deposited in the NCBI Short Read Archive database under accession numbers PRJNA884088 and PRJNA884089.
References
Davidson EA, Janssens IA. Temperature sensitivity of soil carbon decomposition and feedbacks to climate change. Nature. 2006;440:165–73.
Lal R. Digging deeper: a holistic perspective of factors affecting soil organic carbon sequestration in agroecosystems. Glob Change Biol. 2018;24:3285–301.
Li J, Alaei S, Zhou MY, Bengtson P. Root influence on soil nitrogen availability and microbial community dynamics results in contrasting rhizosphere priming effects in pine and spruce soil. Funct Ecol. 2021;35:1312–24.
Rasul M, Cho J, Shin HS, Hur J. Biochar-induced priming effects in soil via modifying the status of soil organic matter and microflora: a review. Sci Total Environ. 2022;805:150304.
Cheng WX. Rhizosphere feedbacks in elevated CO2. Tree Physiol. 1999;19:313–20.
Kuzyakov Y, Friedel JK, Stahr K. Review of mechanisms and quantification of priming effects. Soil Biol Biochem. 2000;32:1485–98.
Bastida F, García C, Fierer N, Eldridge DJ, Bowker MA, Abades S, et al. Global ecological predictors of the soil priming effect. Nat Commun. 2019;10:1–9.
Huo CF, Luo YQ, Cheng WX. Rhizosphere priming effect: a meta-analysis. Soil Biol Biochem. 2017;111:78–84.
Guenet B, Camino-Serrano M, Ciais P, Tifafi M, Maignan F, Soong JL, et al. Impact of priming on global soil carbon stocks. Glob Change Biol. 2018;24:1873–83.
Morrissey EM, Mau RL, Schwartz E, McHugh TA, Dijkstra P, Koch BJ, et al. Bacterial carbon use plasticity, phylogenetic diversity and the priming of soil organic matter. ISME J. 2017;11:1890–9.
Chen RR, Senbayram M, Blagodatsky S, Myachina O, Dittert K, Lin XG, et al. Soil C and N availability determine the priming effect: microbial N mining and stoichiometric decomposition theories. Glob Change Biol. 2014;20:2356–67.
Feng J, Zhu B. Global patterns and associated drivers of priming effect in response to nutrient addition. Soil Biol Biochem. 2021;153:108118.
Li J, Zhou M, Alaei S, Bengtson P. Rhizosphere priming effects differ between Norway spruce (Picea abies) and Scots pine seedlings cultivated under two levels of light intensity. Soil Biol Biochem. 2020;145:107788.
Li J, Bengtson P. Comparative analysis of planted and unplanted controls for assessment of rhizosphere priming effect. Pedosphere. 2022;32:884–92.
Liu XJA, Finley BK, Mau RL, Schwartz E, Hungate BA. The soil priming effect: consistent across ecosystems, elusive mechanisms. Soil Biol Biochem. 2020;140:107617.
Blagodatskaya E, Kuzyakov Y. Mechanisms of real and apparent priming effects and their dependence on soil microbial biomass and community structure: critical review. Biol Fertil Soils. 2008;45:115–31.
Lv C, Wang C, Cai A, Zhou Z. Global magnitude of rhizosphere effects on soil microbial communities and carbon cycling in natural terrestrial ecosystems. Sci Total Environ. 2022;856:158961.
Razanamalala K, Razafimbelo T, Maron P-A, Ranjard L, Chemidlin N, Lelièvre M, et al. Soil microbial diversity drives the priming effect along climate gradients: a case study in Madagascar. ISME J. 2018;12:451–62.
Tao X, Feng J, Yang Y, Wang G, Tian R, Fan F, et al. Winter warming in Alaska accelerates lignin decomposition contributed by Proteobacteria. Microbiome. 2020;8:1–12.
Chen J, Sinsabaugh RL. Linking microbial functional gene abundance and soil extracellular enzyme activity: implications for soil carbon dynamics. Glob Chang Biol. 2021;27:1322–5.
Manoharan L, Kushwaha SK, Ahrén D, Hedlund K. Agricultural land use determines functional genetic diversity of soil microbial communities. Soil Biol Biochem. 2017;115:423–32.
Trivedi P, Delgado-Baquerizo M, Trivedi C, Hu H, Anderson IC, Jeffries TC, et al. Microbial regulation of the soil carbon cycle: evidence from gene–enzyme relationships. ISME J. 2016;10:2593–604.
Zhao F, Wang J, Li Y, Xu X, He L, Wang J, et al. Microbial functional genes driving the positive priming effect in forest soils along an elevation gradient. Soil Biol Biochem. 2022;165:108498.
Song M, Guo Y, Yu F, Zhang X, Cao G, Cornelissen JHC. Shifts in priming partly explain impacts of long-term nitrogen input in different chemical forms on soil organic carbon storage. Glob Chang Biol. 2018;24:4160–72.
Fang XM, Zhang XL, Chen FS, Zong YY, Bu W-S, Wan S-Z, et al. Phosphorus addition alters the response of soil organic carbon decomposition to nitrogen deposition in a subtropical forest. Soil Biol Biochem. 2019;133:119–28.
Liu W, Qiao C, Yang S, Bai W, Liu L. Microbial carbon use efficiency and priming effect regulate soil carbon storage under nitrogen deposition by slowing soil organic matter decomposition. Geoderma. 2018;332:37–44.
Hessen DO. Carbon sequestration in ecosystems: the role of stoichiometry. Ecology. 2004;85:1179–92.
Liebig JF. Animal Chemistry: Or, Organic chemistry in its applications to physiology and pathology. 1842. Johnson Reprint Corporation, New York, USA.
Camenzind T, Hättenschwiler S, Treseder KK, Lehmann A, Rillig MC. Nutrient limitation of soil microbial processes in tropical forests. Ecol Monogr. 2018;88:4–21.
Jobbágy EG, Jackson RB. The vertical distribution of soil organic carbon and its relation to climate and vegetation. Ecol Appl. 2000;10:423–36.
Schulte‐Uebbing L, de Vries W. Global‐scale impacts of nitrogen deposition on tree carbon sequestration in tropical, temperate, and boreal forests: a meta‐analysis. Glob Change Biol. 2018;24:e416–31.
Vitousek PM, Porder S, Houlton BZ, Chadwick OA. Terrestrial phosphorus limitation: mechanisms, implications, and nitrogen-phosphorus interactions. Ecol Appl. 2010;20:5–15.
Du E, Fenn ME, De Vries W, Ok YS. Atmospheric nitrogen deposition to global forests: status, impacts and management options. Envion Pollut. 2019;250:1044–8.
Elrys AS, Zhu Q, Jiang C, Liu J, Sobhy HHH, Shen Q, et al. Global soil nitrogen cycle pattern and nitrogen enrichment effects: tropical versus subtropical forests. Glob Chang Biol. 2023;29:1905–21.
Elser JJ, Bracken ME, Cleland EE, Gruner DS, Harpole WS, Hillebrand H, et al. Global analysis of nitrogen and phosphorus limitation of primary producers in freshwater, marine and terrestrial ecosystems. Ecol Lett. 2007;10:1135–42.
Wang S, Zhou K, Mori T, Mo J, Zhang W. Effects of phosphorus and nitrogen fertilization on soil arylsulfatase activity and sulfur availability of two tropical plantations in southern China. Ecol Manag. 2019;453:117316.
Lu J, Yang J, Keitel C, Yin L, Wang P, Cheng W, et al. Rhizosphere priming effects of Lolium perenne and Trifolium repens depend on phosphorus fertilization and biological nitrogen fixation. Soil Biol Biochem. 2020;150:108005.
Phillips RP, Fahey TJ. Tree species and mycorrhizal associations influence the magnitude of rhizosphere effects. Ecology. 2006;87:1302–13.
Bhattacharyya PN, Jha DK. Plant growth-promoting rhizobacteria (PGPR): emergence in agriculture. World J Microbiol Biotechnol. 2012;28:1327–50.
Kawaka F. Characterization of symbiotic and nitrogen fixing bacteria. AMB Express. 2022;12:11.
Zhang W, Zhu X, Luo Y, Rafique R, Chen H, Huang J, et al. Responses of nitrous oxide emissions to nitrogen and phosphorus additions in two tropical plantations with N-fixing vs. non-N-fixing tree species. Biogeosci Disc. 2014;11:1413–42.
Hicks LC, Leizeaga A, Rousk K, Michelsen A, Rousk J. Simulated rhizosphere deposits induce microbial N-mining that may accelerate shrubification in the subarctic. Ecology. 2020;101: e03094.
Rousk J, Smith AR, Jones DL. Investigating the long-term legacy of drought and warming on the soil microbial community across five European shrubland ecosystems. Glob Chang Biol. 2013;19:3872–84.
Lu RK. Analytical methods of soil agrochemistry. Chinese Agriculture Science and Technology Press, Beijing 1999.
Brookes PC, Landman A, Pruden G, Jenkinson DS. Chloroform fumigation and the release of soil-nitrogen-a rapid direct extraction method to measure microbial biomass nitrogen in soil. Soil Biol Biochem. 1985;17:837–42.
Frostegård Å, Bååth E, Tunlio A. Shifts in the structure of soil microbial communities in limed forests as revealed by phospholipid fatty acid analysis. Soil Biol Biochem. 1993;25:723–30.
Dumont MG, Murrell JC. Stable isotope probing-linking microbial identity to function. Nat Rev Microbiol. 2005;3:499–504.
Zhang K, Ni Y, Liu X, Chu H. Microbes changed their carbon use strategy to regulate the priming effect in an 11-year nitrogen addition experiment in grassland. Sci Total Environ. 2020;727:138645.
Neufeld JD, Vohra J, Dumont MG, Lueders T, Manefield M, Friedrich MW, et al. DNA stable-isotope probing. Nat Protoc. 2007;2:860–6.
Zhou J, Deng Y, Luo F, He Z, Yang Y. Phylogenetic molecular ecological network of soil microbial communities in response to elevated CO2. MBio. 2011;2:e00122–11.
Ihrmark K, Bödeker I, Cruz-Martinez K, Friberg H, Kubartova A, Schenck J, et al. New primers to amplify the fungal ITS2 region–evaluation by 454-sequencing of artificial and natural communities. FEMS Microbiol Ecol. 2012;82:666–77.
Zheng B, Zhu Y, Sardans J, Penuelas J, Su J. QMEC: a tool for high-throughput quantitative assessment of microbial functional potential in C, N, P, and S biogeochemical cycling. Sci China Life Sci. 2018;61:1451–62.
Campbell CD, Chapman SJ, Cameron CM, Davidson MS, Potts JM. A rapid microtiter plate method to measure carbon dioxide evolved from carbon substrate amendments so as to determine the physiological profiles of soil microbial communities by using whole soil. Appl Environ Microbiol. 2003;69:3593–9.
Marx MC, Wood M, Jarvis SC. A microplate fluorimetric assay for the study of enzyme diversity in soils. Soil Biol Biochem. 2001;33:1633–40.
Saiya-Cork KR, Sinsabaugh RL, Zak DR. The effects of long term nitrogen deposition on extracellular enzyme activity in an Acer saccharum forest soil. Soil Biol Biochem. 2002;34:1309–15.
Wild B, Li J, Pihlblad J, Bengtson P, Rütting T. Decoupling of priming and microbial N mining during a short-term soil incubation. Soil Biol Biochem. 2018;129:71–79.
Heuck C, Weig A, Spohn M. Soil microbial biomass C: N: P stoichiometry and microbial use of organic phosphorus. Soil Biol Biochem. 2015;85:119–29.
Widdig M, Schleuss P-M, Biederman LA, Borer ET, Crawley MJ, Kirkman KP, et al. Microbial carbon use efficiency in grassland soils subjected to nitrogen and phosphorus additions. Soil Biol Biochem. 2020;146:107815.
Kuzyakov Y, Blagodatskaya E. Microbial hotspots and hot moments in soil: concept and review. Soil Biol Biochem. 2015;83:184–99.
Bei S, Li X, Kuyper TW, Chadwick DR, Zhang J. Nitrogen availability mediates the priming effect of soil organic matter by preferentially altering the straw carbon-assimilating microbial community. Sci Total Environ. 2022;815:152882.
Mehnaz KR, Corneo PE, Keitel C, Dijkstra FA. Carbon and phosphorus addition effects on microbial carbon use efficiency, soil organic matter priming, gross nitrogen mineralization and nitrous oxide emission from soil. Soil Biol Biochem. 2019;134:175–86.
Wu W, Wang F, Xia A, Zhang Z, Wang Z, Wang K, et al. Meta-analysis of the impacts of phosphorus addition on soil microbes. Agric, Ecosyst Environ. 2022;340:108180.
Ilstedt U, Giesler R, Nordgren A, Malmer A. Changes in soil chemical and microbial properties after a wildfire in a tropical rainforest in Sabah, Malaysia. Soil Biol Biochem. 2003;35:1071–8.
Jing Z, Chen R, Wei S, Feng Y, Zhang J, Lin X. Response and feedback of C mineralization to P availability driven by soil microorganisms. Soil Biol Biochem. 2017;105:111–20.
Zhu Z, Ge T, Luo Y, Liu S, Xu X, Tong C, et al. Microbial stoichiometric flexibility regulates rice straw mineralization and its priming effect in paddy soil. Soil Biol Biochem. 2018;121:67–76.
Ilstedt, Singh S. Nitrogen and phosphorus limitations of microbial respiration in a tropical phosphorus-fixing acrisol (ultisol) compared with organic compost. Soil Biol Biochem. 2005;37:1407–10.
Delgado-Baquerizo M, Maestre FT, Reich PB, Jeffries TC, Gaitan JJ, Encinar D, et al. Microbial diversity drives multifunctionality in terrestrial ecosystems. Nat Commun. 2016;7:10541.
Fu X, Song Q, Li S, Shen Y, Yue S. Dynamic changes in bacterial community structure are associated with distinct priming effect patterns. Soil Biol Biochem. 2022;169:108671.
Zhou S, Wang J, Chen L, Wang J, Zhao F. Microbial community structure and functional genes drive soil priming effect following afforestation. Sci Total Environ. 2022;825:153925.
Blagodatskaya, Khomyakov N, Myachina O, Bogomolova I, Blagodatsky S, Kuzyakov Y. Microbial interactions affect sources of priming induced by cellulose. Soil Biol Biochem. 2014;74:39–49.
Yang H, Li Y, Wang S, Zhan J, Ning Z, Han D. The response of critical microbial taxa to litter micro-nutrients and macro-chemistry determined the agricultural soil priming intensity after afforestation. Front Microbiol. 2021;12:730117.
Xiao W, Chen X, Jing X, Zhu B. A meta-analysis of soil extracellular enzyme activities in response to global change. Soil Biol Biochem. 2018;123:21–32.
Zechmeister-Boltenstern S, Keiblinger KM, Mooshammer M, Peñuelas J, Richter A, Sardans J, et al. The application of ecological stoichiometry to plant–microbial–soil organic matter transformations. Ecol Monogr. 2015;85:133–55.
Lehmann J, Kleber M. The contentious nature of soil organic matter. Nature. 2015;528:60–8.
Acknowledgements
This study was supported by the National Natural Science Foundation of China (42207262 and 42177289) and the China Postdoctoral Science Foundation (2021M703135). JS was supported by Spanish Government grant. JP and JS were supported by the Spanish Government grants PID2020-115770RB-I00 and TED2021-132627B-I00 funded by MCIN, AEI/10.13039/501100011033 and the European NextGenerationEU/PRTR, the Fundación Ramón Areces grant CIVP20A6621, and the Catalan government project SGR2021-01333.
Author information
Authors and Affiliations
Contributions
ZFL, YGZ, and JL conceptualized the study and led the writing of the manuscript. JL, MKJ, WZ, and JZ performed the study and analyzed the data. HL, DFH, CL, DHW, JS, JP, DFP, and DWF contributed substantially to manuscript revisions. All authors reviewed and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, J., Liu, ZF., Jin, MK. et al. Microbial controls over soil priming effects under chronic nitrogen and phosphorus additions in subtropical forests. ISME J 17, 2160–2168 (2023). https://doi.org/10.1038/s41396-023-01523-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41396-023-01523-9