Abstract
Many animals avoid attack from predators through toxicity or the emission of repellent chemicals. Defensive mimicry has evolved in many species to deceive shared predators, for instance through colouration and other morphological adaptations, but mimicry hardly ever seems to involve multi-trait similarities. Here we report on a wingless parasitoid wasp that exhibits a full spectrum of traits mimicing ants and affording protection against ground-dwelling predators (wolf spiders). In body size, morphology and movement Gelis agilis (Ichneumonidae) is highly similar to the black garden ant (Lasius niger) that shares the same habitat. When threatened, G. agilis also emits a volatile chemical that is similar to an ant-produced chemical that repels spiders. In bioassays with L. niger, G. agilis, G. areator, Cotesia glomerata and Drosophila melanogaster, ants and G. agilis were virtually immune to spider attack, in contrast the other species were not. Volatile characterisation with gas chromatography-mass spectrometry identified G. agilis emissions as 6-methyl-5-hepten-2-one, a known insect defence semiochemical that acts as an alarm pheromone in ants. We argue that multi-trait mimicry, as observed in G. agilis, might be much more common among animals than currently realized.
Similar content being viewed by others
Introduction
Mimicry is widespread amongst plants and animals and involves the resemblance of one species to another, at least to the benefit of the mimic1,2,3,4. For instance, the Malaysian orchid mantis Hymenopus coronatus visually mimics flowers such that it attracts more pollinator prey than the flowers it resembles5. To avoid attack from shared predators, prey species may also mimic morphological features of a co-existing unpalatable or toxic model species3, as has long been observed and studied in hoverflies, butterflies and snakes2,6,7,8,9. In hoverflies, which visually resemble stinging bees or wasps, colour mimicry can coincide with behavioural mimicry, such as mock stinging, wing wagging and leg waving10,11 or activity patterns2. Predators and prey can also be deceived via chemical mimicry: some spiders attract their lepidopteran prey through the emission of moth sex pheromones12 but chemical mimicry appears to be less frequently adopted as an anti-predator strategy13,14,15.
Whilst behavioural mimicry often coincides with morphological similarity, mimicry only rarely seems to require convergence of a greater number of traits13,16.The secondary hyperparasitoid Gelis agilis could represent an exception, as it shows several distinct similarities to sympatric ant species. Gelis agilis is a small (3–5 mm long) wingless, asexually reproducing parasitoid wasp that attacks several host species, including the pupae of other parasitoids17,18. Adult G. agilis first paralyze the host with venom and then oviposit a single egg onto the exterior of the host's body18. After hatching, the G. agilis larva feeds on the moribund host, eventually consuming all of it prior to pupation. Gelis agilis is extremely abundant in grassy habitats across much of Eurasia18 and co-occurs and shares predators with several ant species. Its potential predators include wolf spiders, which are visually foraging diurnal hunters that attack a wide range of prey types on the ground19,20. In morphology (body size, colour) and behaviour, G. agilis (Fig. 1a) closely resembles several species of ants that occur in the same habitat, including the common black garden ant, Lasius niger (Fig. 1b). Moreover, when alarmed the parasitoid releases a pungent and distinctive odour. Here, we test the hypotheses that ant-mimicry by G. agilis acts as a defensive strategy to reduce attack by wolf spiders and that chemical mimicry is facilitated by the emission of an ant-like alarm pheromone21.
Results
To test whether ant-mimicry by Gelis agilis reduces predator attack rates, wolf spiders were exposed to adults of the ant Lasius niger, Gelis agilis, Gelis areator (another secondary hyperparasitoid), Cotesia glomerata (a primary parasitoid ) and Drosophila melanogaster (fruit fly) in choice and non-choice bioassays performed in closed arenas over 18 hour periods. When given no choice, spiders killed and consumed virtually all D. melanogaster, which, in spite of possessing wings, were highly susceptible to attack. Cotesia glomerata and G. areator suffered significantly higher predation than G. agilis and L. niger (χ2 = 191.7, DF = 4, P < 0.0001; Fig. 2a–b). Similar patterns were obtained in choice experiments (Fig. 2a–b). Moreover, virtually no insects died from natural causes; they were either alive at the end of the observation period or consumed by the spiders.
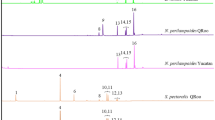
Gelis agilis also emitted a single volatile compound when manually agitated. This volatile emission was not detected in the atmosphere of the experimental environment prior to or after agitation. Analysis of the volatile by atmospheric-pressure chemical ionization-mass spectrometry (APCI-MS) generated spectra displaying two major peaks with the molecular weights of 127 and 109 (Fig. 3). Spectral comparisons with published literature and spectral databases of known compounds (Massbank, Massbank Project) indicated a consistency with 6-methyl-5-hepten-2-one, an unsaturated terpenoid22, also known as sulcatone. Fragmentation data of this emission generated by gas chromatography-mass spectrometry (GC-MS) exhibited a high degree of similarity with the spectral fragmentation and retention time of a standard preparation of 6-methyl-5-hepten-2-one (Sigma-Aldrich) injected during analysis (Figs. 4 &5).
Representative APCI-MS spectrum of G. agilis volatile emissions.
This soft ionization technique resulted in a fragmentation pattern consistent with that of 6-methyl-5-hepten-2-one12.
GC-MS confirmation of G. agilis volatile emissions.
GC-MS spectrum the main peak observed (7.94 min) during G. agilis agitation. GC-MS spectrum of main peak observed (8.04 min) during analysis of a prepared 6-methyl-5-hepten-2-one standard. The two spectra display a high level of consistency in both fragmentation and retention time.
Discussion
Ants have long been known to be important drivers of selection for defensive traits in other organisms23. Ant species are often extremely abundant and form large colonies that occur in close proximity to one another. Many species of predators co-occur with ants; cursorial spiders are among the most abundant24. The parasitoid Gelis agilis shows remarkable morphological and behavioural similarity to sympatric ant species that share the same predators. When crushed, G. agilis and another wingless congener, G. acarorum, produce pungent odours that are easily detectable by human nasal olfaction. These odours are not perceived by human olfaction in other related winged hyperparasitoids that attack the same host, including Gelis areator, Lysibia nana (Hym: Ichneumonidae) and Acrolyta nens (Hym: Ichneumonidae) (Pers. obs. M.M, B.V. & J.A.H.). In choice tests, G. agilis and L. niger suffered little from predator attack, indicating that G. agilis is an effective mimic of L. niger. Comparisons of attack rates on the non-pungent G. areator (which is similar in appearance to G. agilis), C. glomerata and the more distantly related D. melanogaster further revealed that morphological and behavioural mimicry by G. areator also may substantially reduce spider attack. Spider repellence is, however, only as effective as that of L. niger when behavioural, morphological and chemical mimicry are all employed, as is the case for G. agilis.
The chemical, 6-methyl-5-hepten-2-one (sulcatone), that affords predator repellence in G. agilis is the same as that employed as an alarm pheromone by some ant species25,26,27. In some ants, alarm pheromones are employed to recruit colony members for defence (Iridomyrmex purpureus)25 and as a result ants are often avoided by arthropod predators, which move away from them when visual and/or olfactory contact is made28,29. Sulcatone also occurs across a broad array of insect and plant species30, functioning variously as an aggregation pheromone (moths31, bed bugs32), a sex pheromone (ambrosia beetles33) or as an allomone inducing panic alarm among predatory ants (rove beetles27). On encountering G. agilis, spiders thus likely respond as if encountering an ant capable of recruiting assistance from members of its colony.
Chemical mimicry has been observed in other parasitoid species, for instance those that attack aphids being tended by ants. Lysiphlebus cardui mimics the cuticular hydrocarbon profile of its ant-attended host, thereby avoiding ant aggression34,35. Similar to G. agilis, Alloxysta brevis, a hyperparasitoid of aphid parasitoids, releases several compounds, including sulcatone, from its mandibular glands. These releases inhibit aggressive behaviour when the parasitoid is attacked by L. niger and repel attacks in subsequent encounters36. The compounds produced by A. brevis also confer protection against some spider species21. Hence these mandibular gland releases protect against aphid-attending ants and shared predators. Sulcatone is thus exploited by different, distantly related parasitoid species to avoid aggression from predatory spiders. What is important to stress is that chemical mimicry of ants requires an intimate evolutionary history with them. In wingless gelines, as well as in aphid primary parasitoids and hyperparasitoids, the importance of ants as selective agents is clear.
Mimicry is often imperfect, where mimics only superficially resemble their model37. Such limited resemblance may evolve, for instance, when selection on high similarity between mimic and model is reduced or relaxed, as in small hoverflies that are less favourable as prey items38. Whilst imperfect mimicry seems to suffice in some cases, similarity in only a single trait may confer little advantage, particularly when resemblance between mimic and model is limited. Our results show that the morphological and behavioural ant-mimic G. areator suffers less from spider attacks compared to the non-mimetic species but is still attacked at a considerably higher rate than G. agilis which is a morphological, behavioural and chemical mimic (hence, our use of the term ‘full spectrum mimicry’). Mimicry in morphology often coincides with similarity in behavioural traits2,8, but mimicry in more than two traits is rare . Studies of mimicry have focused on characters that are easily observable to the human eye, but as we have shown mimicry may involve more subtle morphological, behavioural, olfactory and acoustic signals. Another case of multi-trait mimicry was recently found in a viperine snake (Natrix maura) that mimics the venomous asp viper (Vipera aspis) in terms of body size, shape, colouration, patterning and acoustic emissions39.We expect that the phenomenon of multi-trait mimicry, as observed in G. agilis and Natrix maura, might be much more common among animals than has been thus far realized.
Methods
All insects were reared at 23°C and a 16 h light: 8 h dark photoperiod. Gelis agilis and G. areator (both Hymenoptera: Ichneumonidae) were reared on cocoons of C. glomerata (Hymenoptera: Braconidae) that were reared on caterpillars of the cabbage butterfly, Pieris brassicae (Lepidoptera: Pieridae), on cabbage plants at the Netherlands Institute of Ecology. Adult C. glomerata wasps were maintained in groups of ~200 wasps in rearing cages. Cabbage leaves infested with L1 caterpillars of P. brassicae were presented to wasps in the rearing cages for parasitism. Parasitized caterpillars were reared in cages with 3–4 cabbage plants. D. melanogaster (Diptera: Drosophilidae) flies were reared on a baker's yeast suspension. A laboratory colony of L. niger (Hymenoptera: Formicidae) was established from single queens and several workers supplied by Antstore, Berlin, Germany. These colonies were supplemented by cocoons of workers collected in the field from wild colonies. Newly emerged workers are immediately ‘conditioned’ to ant pheromones of the host colony, in order to recognize members belonging to the same colony.
Choice and non-choice bioassays were conducted in closed Petri dishes (Petri dishes Ø 12 cm). For bioassays, wolf spiders were kept in individual Petri dishes containing water absorbed into cotton wool but were starved for 2–3 days after collection from the field. In the choice experiments, 2 species-combinations of insects were introduced in pairs into single Petri dishes with an individual spider. For non-choice bioassays, 3 individuals of a single species were introduced into Petri dishes with an individual spider. The dishes were left for approximately 24 h and then they were checked for evidence of predation. Predation was recorded only as insects that had been visibly killed and eaten, where only cuticular rudiments remained. Some insects died but were not visibly attacked by the spiders; this included very few ants and G. agilis wasps. These insects were excluded from the analyses, as death may have been due to natural mortality. Spiders only eat freshly killed prey and thus only those insects that were clearly attacked by the spiders were included in the analyses.
Statistical analyses
The number of prey of each species consumed in the no-choice experiment was compared using a Kruskal-Wallis test based on ranked data. To compare individual prey species, the ranked data were analyzed using a one-way analysis of variance (ANOVA) followed by a Tukey HSD post-hoc test.
The number of prey consumed of each of the two species in the choice experiments was analyzed using a Monte Carlo permutation test with 199 permutations. In each test we randomly allocated the consumed prey in each of the replicates (n = 61–121) to one of the two species. The consumption of each of the two species in the experiment (realized) was then compared to the consumption calculated in the random permutations and a P-value was determined based on the number of times that the difference in consumption between the two species in the random permutations was more distinct than the realized difference.
Chemical analyses
Chemical analysis took place at the University of Nottingham, School of Biosciences. For initial analysis, volatile chemical releases of G. agilis were monitored in real-time using an APCI-MS40 . Five adults were placed individually in 20 ml glass scintillation vials and deliberately agitated for 1 minute with a paintbrush whilst positioned adjacent to the APCI-MS sampling point40. Agitation comprised of deliberately restricting the movement of individual wasps by pressing them against the edge of the vial. The APCI-MS sampling point draws a continuous stream of air, set up at 25 ml min−1, into a heated transfer line (160°C) through a deactivated silica tube (1 m × 0.53 mm ID) before entering the APCI source. Volatiles then entered the source and were ionized by a positive ion corona discharge (4 kV), which typically forms the adduct ion M+H+. Spectra were recorded using a Platform II mass spectrometer (Waters, Manchester, UK) across a mass range of 25–250 Da, with the cone voltage set to 18 V. Two major ions with the m/z of 108 and 127 respectively were observed, consistent with the fragmentation pattern of an unsaturated terpenoid with a molecular mass of M = 126 (127).
In order to confirm the identity of the chemical released, individual G. agilis were placed in a 20 ml flask and deliberately agitated for 1 minute under the same protocol as the APCI analysis. Flasks were then sealed with a PTFE lined septum. Volatile compounds were transferred for GC-MS analysis using a SPME fibre (50/30 mm, assembly Divinylbenzene/Carboxen/Polydimethylsiloxane; Supelco, Bellefonte, USA), which was exposed in the flask headspace for 0.2 min at 22°C. Desorption of volatile compounds attached to the fibre occurred in the injector at 250°C for 2 min. Volatile compounds were transferred to the column (30 m × 0.25 mm ID, BP-5, 1.0 mM film thickness; SGE, Milton Keynes, UK) and the gas chromatogram temperature programme initialised. The GC (Trace GC 1300, Thermo, Austin, USA) temperature programme held at a temperature of 40°C for 1 min before increasing at a rate of 8°C min−1 to 200°C. Mass spectra were recorded using an ISQ mass spectrometer (Thermo) at 2 scans s−1 from between 20–200 m/z.
References
Müller, F. Ituna and Thyridia: a remarkable case of mimicry in butterflies. Trans. Entomol. Soc. Lond 8, 20–29 (1897).
Howarth, B., Edmunds, M. & Gilbert, F. Does the abundance of hoverfly (Syrphidae) mimics depend on the numbers of their hymenopteran models? Evolution 58, 367–375 (2004).
Bates, H. W. XXXII. Contributions to an Insect Fauna of the Amazon Valley. Lepidoptera: Heliconidæ. Trans. Linn. Soc. Lon 23, 495–566 (1862).
Huang, J. N., Cheng, R. C., Li, D. & Tso, I. M. Salticid predation as one potential driving force of ant mimicry in jumping spiders. Proc. R. Soc. B 278, 1356–1364 (2010).
O'Hanlon, J. C., Holwell, G. I. & Herberstein, M. E. Pollinator deception in the orchid mantis. Am. Nat 183, 126–132 (2014).
Clarke, C. A. & Sheppard, P. M. The evolution of mimicry in the butterfly Papilio dardanus. Heredity 14, 163–173 (1960).
Kunte, K. et al. Doublesex is a mimicry supergene. Nature 507, 229–232 (2014).
Greene, H. W. & McDiarmid, R. W. Coral snake mimicry: does it occur. Science 213, 1207–1212 (1981).
Kikuchi, D. W., Seymoure, B. M. & Pfennig, D. W. Mimicry's palette: widespread use of conserved pigments in the aposematic signals of snakes. Evol. & Dev 16, 61–67 (2014).
Penney, H. D., Hassall, C., Skevington, J. H., Lamborn, B. & Sherratt, T. N. The relationship between morphological and behavioral mimicry in hover flies (Diptera: Syrphidae). Am. Nat 183, 281–289 (2014).
Johnstone, R. A. The evolution of inaccurate mimics. Nature 418, 524–526 (2002).
Eberhard, W. G. Aggressive chemical mimicry by a bolas spider. Science 198, 1173–1175 (1977).
Rettenmeyer, C. W. Insect mimicry. Annu. Rev. Entomol 15, 43–74 (1970).
Dettner, K. & Liepert, C. Chemical mimicry and camouflage. Annu. Rev. Entomol 39, 129–154 (1994).
Lorenzi, M. C., Bagnères, A. G. & Clement, J. L. The role of cuticular hydrocarbons in social insects: is it the same in paper-wasps. [178–189] (Oxford University Press, Oxford, 1996).
Pasteur, G. A classification review of mimicry systems. Annu. Rev. Ecol. Sys 13, 169–199 (1982).
Lei, G. C. & Hanski, I. Metapopulation structure of Cotesia melitaearum, a specialist parasitoid of the butterfly Melitaea cinxia. Oikos 78, 91–100 (1997).
Harvey, J. A. Comparing and contrasting development and reproductive strategies in the pupal hyperparasitoids Lysibia nana and Gelis agilis (Hymenoptera: Ichneumonidae). Evol. Ecol 22, 153–166 (2008).
Nentwig, W. & Wissel, C. A comparison of prey lengths among spiders. Oecologia 68, 595–600 (1986).
Oelbermann, K. & Scheu S. Stable isotope enrichment (δ15N and δ13C) in a generalist predator (Pardosa lugubris, Araneae: Lycosidae): effects of prey quality. Oecologia 130, 337–344 (2002).
Hübner, G. & Dettner, K. Hyperparasitoid defense strategies against spiders: the role of chemical and morphological protection. Entomol. Ex. App 97, 67–74 (2000).
Raspotnig, G., Schaider, M., Stabentheiner, E., Leis, H.-J. & Karaman, I. On the enigmatic scent glands of dyspnoan harvestmen (Arachnida, Opiliones): first evidence for the production of volatile secretions. Chemoecology 24, 43–55 (2014).
Hölldobler, B. The ants. Harvard University Press, United States. (1990).
Samu, F., Szirányi, A. & Kiss, B. Foraging in agricultural fields: local ‘sit-and-move’ strategy scales up to risk-averse habitat use in a wolf spider. Animal behavior 66, 939–947 (2003).
Allan, R. A., Elgar, M. A. & Capon, R. Mimicry of host cuticular hydrocarbons by salticid spider Cosmophasis bitaeniata that preys on larvae of tree ants Oecophylla smaragdina. J. Proc. R. Soc. B 263, 69–73 (1996).
Moore, B. P. & Brown, W. V. Identification of warning odour components, bitter principles and antifeedants in an aposematic beetle, Metriorrhynchus rhipidius (Coleoptera: Lycidae). Insect Biochem 11, 493–499 (1981).
Stoeffler, M., Maier, T. S., Tolasch, T. & Steidle, J. L. Foreign-language skills in rove-beetles? Evidence for chemical mimicry of ant alarm pheromones in myrmecophilous Pella beetles (Coleoptera: Staphylinidae). J. Chem. Ecol 33, 1382–1392 (2007).
Mestre, L., Bucher, R. & Entling, M. H. Trait-mediated effects between predators: ant chemical cues induce spider dispersal. J. Zool 293, 119–125 (2014).
Halaj, J., Ross, D. W. & Moldenke, A. R. Negative effects of ant foraging on spiders in Douglas-fir canopies. Oecologia 109, 313–322 (1997).
Schiestl, F. P. The evolution of floral scent and insect chemical communication. Ecol. Lett 13, 643–656 (2010).
Jumean, Z., Gries, R., Unruh, T., Rowland, E. & Gries, G. Identification of the larval aggregation pheromone of codling moth, Cydia pomonella. J. Chem. Ecol 31, 911–924 (2005).
Siljander, E., Gries, R., Khaskin, G. & Gries, G. Identification of the airborne aggregation pheromone of the common bed bug, Cimex lectularius. J. Chem. Ecol 34, 708–718 (2008).
Gatti, P., Zerba, E. & Gonzalez-Audino, P. Anatomical site of pheromone accumulation and temporal pattern of pheromone emission in the ambrosia beetle Megaplatypus mutatus. Physiol. Entom 36, 201–207 (2011).
Liepert, C. & Dettner, K. Recognition of aphid parasitoids by honeydew-collecting ants: the role of cuticular lipids in a chemical mimicry system. J. Chem. Ecol 19, 2143–2153 (1993).
Liepert, C. & Dettner, K. Role of cuticular hydrocarbons of aphid parasitoids in their relationship to aphid-attending ants. J. Chem. Ecol 22, 695–707 (1996).
Völkl, W., Hübner, G. & Dettner, K. Interactions between Alloxysta brevis (Hymenoptera, Cynipoidea, Alloxystidae) and honeydew-collecting ants: How an aphid hyperparasitoid overcomes ant aggression by chemical defense. J. Chem. Ecol 20, 2901–2915 (1994).
Sherratt, T. N. The evolution of imperfect mimicry. Behav. Ecol 13, 821–826 (2002).
Penney, H. D., Hassall, C., Skevington, J. H., Abbott, K. R. & Sherratt, T. N. A comparative analysis of the evolution of imperfect mimicry. Nature 483, 461–464 (2012).
Aubret, F. & Mangin, A. The snake hiss: potential acoustic mimicry in a viper–colubrid complex. Biol. J. Linn. Soc 113, 1095–8312 (2014).
Goubault, M., Batchelor, T. P., Linforth, R. S., Taylor, A. J. & Hardy, I. C. Volatile emission by contest losers revealed by real-time chemical analysis. Proc. R. Soc. B 273, 2853–2859 (2006).
Acknowledgements
We thank Roel Wagenaar and Gregor Disveld for rearing Cotesia glomerata and providing supplementary materials. We also thank Rob Linforth for aid in MS instrument operation and volatile chemical characterization. Photos of the species were kindly provided by Rui Andrade and Alex Kraus. The research leading to these results has received funding from the People Programme (Marie Curie Actions) of the European Union's Seventh Framework Programme (FP7/2007–2013) under REA grant agreement no 298457.
Author information
Authors and Affiliations
Contributions
M.M., J.A.H. and B.V. designed and analyzed the experiment and wrote the initial manuscript; M.B. performed the bioassay; C.S. and I.C.W.H. analyzed the chemicals of G. agilis; T.M.B. carried out the data analysis. All authors contributed to revising the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License. The images or other third party material in this article are included in the article's Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder in order to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/
About this article
Cite this article
Malcicka, M., Bezemer, T., Visser, B. et al. Multi-trait mimicry of ants by a parasitoid wasp. Sci Rep 5, 8043 (2015). https://doi.org/10.1038/srep08043
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep08043
This article is cited by
-
Southeast Asian clearwing moths buzz like their model bees
Frontiers in Zoology (2021)
-
Drosophila parasitoid wasps bears a distinct DNA transposon profile
Mobile DNA (2018)
-
Ant-like Traits in Wingless Parasitoids Repel Attack from Wolf Spiders
Journal of Chemical Ecology (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.