Abstract
In the present work, it is shown that the friction stress of ethylene glycol can decrease by an order of magnitude to achieve superlubricity if there are hydrogen ions attached on the friction surfaces. An ultra-low friction coefficient (μ = 0.004) of ethylene glycol between Si3N4 and SiO2 can be obtained with the effect of hydrogen ions. Experimental result indicates that the hydrogen ions adsorbed on the friction surfaces forming a hydration layer and the ethylene glycol in the contact region forming an elastohydrodynamic film are the two indispensable factors for the reduction of friction stress. The mechanism of superlubricity is attributed to the extremely low shear strength of formation of elastohydrodynamic film on the hydration layer. This finding may introduce a new approach to reduce friction coefficient of liquid by attaching hydrogen ions on friction surfaces.
Similar content being viewed by others
Introduction
A significant portion of energy in machine is continuously lost because of high friction and wear in the sliding system1. Consequently, how to reduce friction coefficient and wear rate becomes one of the most important tasks for researchers in many fields. The common method is adding lubricant to separate the contacting surfaces and thus the friction and wear would decrease due to the lower shear stress of lubricant relative to the original friction surface. At present, the separation can be obtained by introducing a solid lubricant (e.g., graphite, DLC and MoS2)2,3,4, or a liquid lubricant (e.g., aqueous solution, oil and grease)5,6,7. When the sliding friction coefficient of them reduces to less than 0.01, it can be referred as superlubricity8,9. As for solid lubricants, the friction coefficient can reduce to less than 0.01 in some special conditions such as vacuum or nitrogen protection to achieve superlubricity. As for liquid lubricants, it is easy to achieve a friction coefficient between 0.01–0.1 if the hydrodynamic film is formed between two sliding surfaces. However, due to the limitation of viscosity, it is very difficult to obtain a friction coefficient in the magnitude order of 0.001 to achieve superlubricity.
During recent years, lots of efforts have been made to reduce friction coefficient of common liquid to a lower value (less than 0.01)10. One of the most typical examples is ceramic with water lubrication, which has been studied by Kato's group systematically11,12,13. They found that an ultra-low friction coefficient of 0.002–0.007 can be achieved after a running-in period. The mechanism is attributed to the tribochemical reaction between ceramic surfaces and water, which can produce a layer of colloidal silica for boundary lubrication and meanwhile form a smooth surface for hydrodynamic lubrication11. In addition to this, Matta et al. found that the DLC film (ta-C) under pure glycerol lubrication could realize a friction coefficient of 0.007 due to the easy sliding on the tribo-formed OH-terminated surfaces14. Spencer et al. obtained a rolling friction coefficient of 0.0001 under polyethylene glycol (PEG) lubrication due to the formation of molecular brushes15. Recently, our group found that the friction coefficient between glass and ceramic could decrease to 0.004 with the lubrication of phosphoric acid or mixture of polyhydroxy alcohol and acid16,17,18.
In comparison with these examples, they have a common feature, i.e., the friction stress of liquid confined between two sliding surfaces is extremely low. A simple method to obtain a low friction stress is to reduce the viscosity of confined liquid. However, it would produce another problem (difficult to form a full lubricating film between two sliding surfaces) that is harmful to the reduction of friction coefficient. Therefore, the above method almost has no effect on reducing friction coefficient of common liquid from the magnitude order of 0.01 to 0.001. Is there a method to reduce friction stress of common liquid to a lower value and meantime form a full lubricating film? In the present work, we would show a method to satisfy the two requirements above simultaneously; that is adding hydrogen ions in the confined liquid or attaching hydrogen ions on the friction surfaces. Here, the ethylene glycol (C2H6O2) was chosen as an example and the mechanism of hydrogen ions reducing friction stress of liquid was investigated.
Results
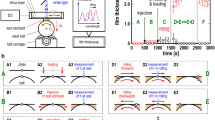
The friction coefficient of C2H6O2 (20%) between Si3N4 and SiO2 is shown in Fig. 1. It is found that the friction coefficient can reduce to 0.05 after a running-in period of 380 s. However, if C2H6O2 (20%) was mixed with H2SO4 (pH = 1), the final friction coefficient would reduce to 0.004 after a running-in period of 400 s, which is only about one-twelfth of its original value (μ = 0.05). Compared with the two results, it is obvious that adding H2SO4 in C2H6O2 solution can reduce its friction coefficient to the magnitude order of 0.001 (superlubricity). After the running-in period, the topography of friction surfaces was investigated by the optical microscope, as shown in Fig. 2. A round plane with a diameter of 260 μm appears on the top region of ball and many micro-pits and micro-furrow ditches are observed in the track on the glass substrate after the lubrication of mixture of C2H6O2 and H2SO4. However, after the lubrication of C2H6O2 (no acid), it is seen that the top region of ball becomes an elliptic plane with a max diameter of 260 μm and there are also several micro-pits and micro-furrow ditches in the middle of track. Although there are some differences in topography between the two cases, the wear degree and the contact area are almost the same for the two cases after the running-in period. Because the friction stress (τ) of confined liquid between two sliding surfaces can be described by τ = μp approximately, where μ is the friction coefficient and p is the average contact pressure, it can be concluded that the friction stress of C2H6O2 reduces to one-twelfth of its original value if it is mixed with H2SO4 solution.
In the running-in period, the viscosity increased with free water evaporating from the solution as well as the contact pressure reduced with the wear scar enlarging. As for C2H6O2 (no acid), the concentration increased to 83% after the running-in period, corresponding to a viscosity of 9.6 mPas (measured by a standard rheometer) and the diameter of contact region would increase to 260 μm (Fig. 2), corresponding to an average contact pressure of 57 MPa. In this case, it is easy to form an elastohydrodynamic (EHD) film between two sliding surfaces to realize a low friction coefficient of 0.05. As for mixture of C2H6O2 and H2SO4, the viscosity and the average contact pressure after the running-in period were almost the same as that of C2H6O2 (20%), which can also form an EHD film between two sliding surfaces. In comparison with the two results, it is interesting to find that the same lubrication conditions (surface, viscosity, speed and pressure) can lead to totally different friction coefficient only because a spot of H2SO4 is introduced in the contact region.
To investigate the mechanism of H2SO4 reducing friction stress of C2H6O2, it is necessary to determine which kind of ion in the acid solution plays the key role. Therefore, the friction coefficients of C2H6O2 (20%) mixed with other three kinds of acids (HCl, H2C2O4 and HNO3S, pH = 1), Na2SO4 (pH = 7.0) and NaOH (pH = 13) were all measured, as shown in Table 1. It is found that the friction coefficients of C2H6O2 mixed with the three kinds of acids can reduce to 0.003–0.004 after a running-in period, which is very close to the value of mixture of C2H6O2 and H2SO4. It indicates that all the acid solutions have the same effect on reducing friction stress of C2H6O2. However, the Na2SO4 and NaOH solutions almost have no effect on the reduction of friction stress of C2H6O2. It is seen that the friction coefficients of their mixtures are almost the same as that of C2H6O2 (no acid). These results suggest that the hydrogen ions in the mixed solution play the key role on reducing friction stress of C2H6O2.
Due to the mixture of C2H6O2 and acid being confined between two sliding surfaces, there are two kinds of interactions with hydrogen ions; one is the interaction between hydrogen ions and C2H6O2 and the other is the interaction between hydrogen ions and friction surfaces (Si3N4 and SiO2). To determine whether the interaction between hydrogen ions and C2H6O2 has influence on the reduction of friction stress, the friction pairs were changed to other three kinds of materials (to remove the interaction between hydrogen ions and surface) and then the friction coefficients of C2H6O2 (no acid) and mixture of C2H6O2 and H2SO4 were measured in the same test condition, as shown in Table 2. It is found that the superlubricity of mixture cannot be achieved (μ > 0.01) and the friction coefficient of it is almost the same as that of C2H6O2 (no acid). It indicates that the interaction between hydrogen ions and C2H6O2 has no effect on the reduction of friction stress of C2H6O2. Because there are only two kinds of interactions with hydrogen ions, it is inferred that the interaction between hydrogen ions and surfaces (Si3N4 and SiO2) plays the key role on reducing friction stress of C2H6O2.
To investigate how the interaction between hydrogen ions and friction surfaces influences friction stress of C2H6O2, an experiment based on separating hydrogen ions from mixture was designed as follows. First, the friction coefficient with the lubrication of H2SO4 (pH = 1) between Si3N4 and SiO2 was measured for about 250 s, as shown in Fig. 3(a). It is seen that the friction coefficient reduced to 0.03 after a running-in period of 160 s and then had no further reduction with time. Second, at the moment of t = 250 s, 20 μL of pure C2H6O2 (100%) was added in the contact region between two friction surfaces. It is seen that the friction coefficient decreased to less than 0.01 (μ = 0.007) suddenly once the pure C2H6O2 was added in the contact region. After that, the friction coefficient had a small reduction and then kept an ultra-low value of 0.004. However, if the experimental order was changed; that is measuring the friction coefficient of C2H6O2 (100%) for 250 s first and then adding 5 μL of H2SO4 (pH = 1) in the contact region, it is found that there was no drastic reduction in friction coefficient, as shown in Fig. 3(b). The final friction coefficient (μ = 0.044) was almost the same as that of C2H6O2 (no acid). It indicates that the interaction between hydrogen ions and surfaces is the prerequisite for the reduction of friction stress of C2H6O2.
(a) Friction coefficient of C2H6O2 (100%) after testing H2SO4 (pH = 1) for about 250 s. The dot line is the friction coefficient of H2SO4 (pH = 1) without adding C2H6O2 (100%) in the contact region. (b) Friction coefficient of H2SO4 (pH = 1) after testing C2H6O2 (100%) for about 250 s. The load is 3 N and the speed is 0.075 m/s.
Our previous work showed that all the acid solutions (pH ≤ 2) could lead to the friction coefficient reducing to 0.03–0.05 after a running-in period by tribochemical reaction between hydrogen ions and Si3N4/SiO2 surfaces19. During the running-in period, the original layer of surface is grinded off by rubbing action (wear), leading to the generation of many dangling bonds (S-O−). The dangling bonds (Si-O−) can react with hydrogen ions in acid solution (SiO− + 2H+ → SiOH2+) by surface protonation reaction, leading to many positively charged sites being formed on the friction surfaces. In addition, the rubbing action can increase the numbers of effective active sites of protonation reaction and meantime reduce the contact pressure as the wear scar enlarges. After the running-in period, these positively charge sites are neutralized by equal dissolved counter-ions to form the stern layer and the diffuse electrical double layer, leading to the friction coefficient reducing to 0.03–0.05 (see more details in Ref. 19). Due to the viscosity of H2SO4 solution being close to that of water, it is hard to form an EHD film in this test condition and thereby the boundary lubrication is the dominant mechanism for H2SO4 solution when its friction coefficient reduces to 0.03. At the moment, if adding C2H6O2 with a high viscosity in the contact region, it would form an EHD film to separate the two sliding surfaces apart further. In our experiment, the concentration of added C2H6O2 is 100%, corresponding to a viscosity of 17.5 mPas. According to Hamrock-Dowson (H-D) theory20, the film thickness between two sliding surfaces is about 32 nm. It indicates that the C2H6O2 solution forms an EHD film between two sliding surfaces with attached hydrogen ions when superlubricity appears.
However, if the experimental order was changed, the friction coefficient would reduce to 0.05 in a very short period (14 s), which indicates that an EHD film of C2H6O2 solution has been formed between two sliding surfaces. At the moment, if we added hydrogen ions in the contact region, the tribochemical reaction between hydrogen ions and friction surfaces would not occur because there is no rubbing action (the two surfaces are not in contact). Thus, there is only a small amount of hydrogen ions adsorbed on the two original friction surfaces by surface protonation reaction, which would lead to a high friction. In comparison with the two results, it can be inferred that the C2H6O2 solution forming an EHD film between two sliding surfaces with attached hydrogen ions (by tribochemical reaction) is the main factor for the reduction of friction stress of C2H6O2.
To confirm this inference further, the viscosity and speed (two main factors influencing EHD film) were changed to different values respectively to see whether there exists an EHD film when superlubricity appears. First, to change the viscosity of C2H6O2, three different concentrations (50%, 70% and 90%) of C2H6O2 were chose (the viscosities of them are 3.3 mPas, 6 mPas and 12.1 mPas, respectively). When the friction coefficient of H2SO4 (pH = 1) reduced to about 0.03, these C2H6O2 solutions with different viscosities (20 μL) were added in the contact region, as shown in Fig. 4. It is found that the friction coefficient would reduce suddenly once they were introduced in the contact region. Moreover, the reduced friction coefficient is lower if the viscosity is higher. After that, the friction coefficient would reduce further to 0.004 after a running-in period (the lower the viscosity, the longer the running-in period).
These results indicate that the lowest friction appears only when the viscosity of solution is high enough to separate the two surfaces apart. If the viscosity is not high enough to form a full EHD film, there would appear some asperities in the contact region with direct contact (dry friction), which can cause a high friction. In addition, the proportion of direct contact would become higher with viscosity decreasing due to the hydrodynamic effect becoming weaker, which would lead to friction increasing. It is the reason why the friction coefficient is higher if the viscosity of added C2H6O2 solution is lower. After C2H6O2 solution was added in the contact region, the water in the solution would evaporate out with time, which would lead to the viscosity increasing. In this case, the proportion of direct contact would become lower and thereby the friction coefficient would reduce with time. If the concentration of C2H6O2 solution is lower, it would need a longer time for water evaporation to reach the same viscosity, which is the reason why the running-in period becomes longer with concentration reducing.
Second, the speed was changed from 5 rpm to 900 rpm when the lowest friction coefficient (μ = 0.004) appeared, as shown in Fig. 5. It is observed that the friction coefficient almost keeps constant (μ = 0.004) when the rotation speed is greater than 120 rpm (a sliding speed of 0.05 m/s). However, when the rotation speed decreases from 120 rpm to 5 rpm (a sliding speed of 0.002 m/s), the friction coefficient has an increase from 0.005 to 0.074. It suggests that the change of friction coefficient with speed is in accordance with the stribeck curve21. For comparison, the relationship between friction coefficient of C2H6O2 solution (no acid) and speed was also investigated (Fig. 5). It can be seen that the change of friction coefficient of C2H6O2 solution with speed is also in accordance with the stribeck curve. Moreover, the lowest friction coefficient of C2H6O2 solution is 0.026, which indicates that the superlubricity cannot be achieved for C2H6O2 solution no matter how to change the sliding speed.
According to H-D theory20, the film thickness between two sliding surfaces as a function of speed can be estimated, as shown in Fig. 5. When the speed is less than 90 rpm, the film thickness is less than 14 nm due to the weak hydrodynamic effect. It indicates that the lubrication regime is in mixed lubrication, in comparison with the roughness of friction surfaces (Ra = 12 nm for the track and Ra = 10 nm for the worn region of ball, measured by white light interferometer). In this case, it is difficult to form a full EHD film and instead there would appear some asperities in the contact region with direct contact (dry friction), leading to friction increasing. In contrast, when the speed is greater than 180 rpm, the film thickness is more than 22 nm, which can lead to the formation of a full EHD film. In this case, the two sliding surfaces are separated by C2H6O2 solution completely. As for mixture of C2H6O2 and H2SO4, the EHD film is formed between two sliding surfaces with attached hydrogen ions, leading to an ultra-low friction. As for C2H6O2 solution, the EHD film is formed between two original surfaces without hydrogen ions, leading to a high friction.
Finally, to remove the other factors such as chemical reaction and molecule structure of C2H6O2 influencing superlubricity, other four kinds of aqueous solution (80%, v/v), including glycerol (C3H8O3), dimethyl sulfoxide (C2H6OS), 1,4-butanediol (C4H10O2) and 1,5-pentanediol (C5H12O2), were introduced in the contact region instead of C2H6O2 solution after testing H2SO4 (pH = 1) for about 250 s. The final friction results are shown in Table 3. It can be found that all of these solutions can achieve a friction coefficient less than 0.005 if there are hydrogen ions attached on the friction surfaces, which indicates that the superlubricity has no significant correlation with C2H6O2 solution. In other words, the chemical reaction and molecule structure of C2H6O2 are not the main factors for superlubricity. From this result, it can be inferred that the superlubricity would appear as long as there is an EHD film formed between two sliding surfaces with attached hydrogen ions, regardless of the chemical content of aqueous solution.
Discussion
These friction results indicate that there are indeed an EHD film in the contact region when superlubricity (μ = 0.004) appears. Moreover, the superlubricity is independent on the chemical content of EHD film. However, as for common EHD lubrication, just like C2H6O2 or C3H8O3 (not mixed with acid), the friction coefficient is usually in the range of 0.01–0.1. Why adding hydrogen ions in the solution can lead to the friction coefficient of EHD lubrication reducing by an order of magnitude? As shown above, the EHD film is formed between two sliding surfaces with attached hydrogen ions (by tribochemical reaction), which is the key factor for the reduction of friction stress. Here, a possible lubrication model of mixture of C2H6O2 and H2SO4 was proposed according to these results and analyses, as illustrated in Fig. 6.
It is thought that there are two kinds of EHD films in the contact region. The first one is the common EHD film formed between two original surfaces (Si3N4 and SiO2) where there are no hydrogen ions attached on the friction surfaces. The second one is the case that there are hydrogen ions attached on the friction surfaces. In this case, the counter-ions (HSO4−) in the solution would be adsorbed by hydrogen ions on the two sliding surfaces to form the hydration layer (Fig. 6 inside) and thereby the EHD film would be formed on the hydration layer. Assuming that the proportion of the first case is k (0 ≤ k < 1), the friction coefficient for the first case is μ1 and the friction coefficient for the second case is μ2, the final friction coefficient μ can be obtained as follows:

As for μ1, the value is equal to the friction coefficient of common EHD lubrication approximately (μ1 ≈ 0.05 for C2H6O2 solution). As for μ2, it is in proportion to the friction stress of hydration layer approximately because the shear mainly occurs in the hydration layer due to the excellent fluidity of hydration layer22. Thus, the value of μ2 (about 0.0002 in Ref. 22) is much lower than μ1 because of the extremely low friction stress of hydration layer23. According to this, the Equation (1) can be rewritten as follows:

Based on the value of μ (μ = 0.004 in Fig. 1) in our test, we can get the value of k (k = 0.08). It is indicates that most of the EHD film in the contact region is formed on the hydration layer when superlubricity appears. Therefore, the ultra-low friction stress of C2H6O2 solution is mainly attributed to the extremely low shear strength of hydration layer produced by attached hydrogen ions on the friction surfaces.
This lubrication model indicates that the hydrogen ions attaching on the friction surfaces and the C2H6O2 solution forming an EHD film in the contact region are two key factors for superlubricity. It therefore can be inferred the superlubricity is independent on the properties of surfaces and the types of solutions as long as the two factors above are satisfied for superlubricity. From the data in Table 3, it can be confirmed that the superlubricity is independent on the types of solutions. To confirm whether the superlubricity is independent on the properties of surfaces, other three kinds of friction pairs (Si3N4/Si3N4, Si3N4/α-Al2O3 and α-Al2O3/α-Al2O3) were chosen instead of Si3N4/SiO2 and the friction coefficient of mixture of C2H6O2 (20%) and H2SO4 (pH = 1) was measured between these friction pairs. The results show that all of these friction pairs can lead to the friction coefficient of mixture reducing to about 0.003–0.005. Because all of these friction pairs can adsorb hydrogen ions by tribochemical reaction during the running-in period, it can be confirmed that the superlubricity is also independent on the properties of surfaces if there are hydrogen ions adsorbed on the surfaces.
In our previous work, the superlubricity is attributed to the formation of hydrogen bond network on the stern layer (induced by hydrogen ions) after the running-in period24. However, the hydrodynamic effect of hydrogen bond network is not considered before. The present work proves that the hydrodynamic effect also plays an important role on superlubricity. Moreover, from the lubrication model above, it can be found that the role of hydrogen ions is being attached on the friction surfaces to form the hydration layer and the role of C2H6O2 solution is forming an EHD film in the contact region. Both of them are very important for the reduction of friction stress. If there is only hydrogen ions adsorbed on the friction surfaces, the friction coefficient is higher than 0.02, such as various kinds of acid solutions19. If there is only an EHD film of C2H6O2 solution in the contact region, the friction coefficient is higher than 0.04 (Fig. 1). Based on the roles of hydrogen ions and C2H6O2 solution, we can propose a new method to reduce friction coefficient of aqueous solution if it can meet the three conditions below. First, the friction surfaces should have SiOH or AlOH bonds, which can adsorb hydrogen ions by triochemical reaction. Second, there should be sufficient hydrogen ions attached on the friction surfaces, which can form the hydration layer. Finally, the viscosity of aqueous solution should be high enough to form an EHD film in the contact region, which can separate the two friction surfaces apart. If three of them are met simultaneously, the friction coefficient of aqueous solution would decrease by an order of magnitude to achieve superlubricity, just like C2H6O2 solution. According to this method, we believe, most aqueous solutions used as lubricants would save energy effectively in sliding system.
In conclusion, we showed that the friction stress of C2H6O2 solution can decrease by an order of magnitude to achieve superlubricity if it is mixed with one acid solution. Experimental result indicates that the interaction between hydrogen ions and friction surfaces plays the key role on the reduction of friction stress. The function of hydrogen ions is being attached on the friction surfaces by tribochemical reaction to form the hydration layer and the function of C2H6O2 solution is forming an EHD film on the hydration layer to separate the two sliding surfaces apart. The ultra-low friction stress is mainly attributed to the low shear strength of hydration layer on the friction surfaces. According to these results, a new method to reduce friction stress of aqueous solution is proposed that adding hydrogen ions in the solution or attaching hydrogen ions on the friction surface. This work seems to have an important engineering value to the traditional mechanical lubrication systems and nano-mechanical systems.
Methods
Sample preparation
Five kinds of aqueous solution, including ethylene glycol (C2H6O2), glycerol (C3H8O3), dimethyl sulfoxide (C2H6OS), 1,4-butanediol (C4H10O2) and 1,5-pentanediol (C5H12O2) and four kinds of acids, including sulfuric acid (H2SO4), hydrochloric acid (HCl), oxalic acid (H2C2O4) and aminosulfonic acid (HNO3S), were all commercial products (J&K) with a purity of greater than 99%. Before test, the pure ethylene glycol was diluted by de-ionized water (resistivity > 18 MΩ·cm) with a concentration of 20%, 50%, 70%, 90% (v/v) and the four kinds of acids were diluted or dissolved by de-ionized water to a pH of 1.0. In addition, the sodium sulfate (Na2SO4) solution with a concentration of 5% (pH = 7.0) and the sodium hydroxide (NaOH) solution with a pH of 13 were also prepared. The mixtures of C2H6O2 and acid were obtained by mixing C2H6O2 solution (20%) with acid solutions (pH = 1) in a volume ratio of 10:1 (the pH value of the mixtures is approximately equal to 2). The mixture of C2H6O2 and Na2SO4 and the mixture of C2H6O2 and NaOH were prepared by mixing C2H6O2 solution (20%) with Na2SO4 solution (5%) and NaOH solution (pH = 13) in a volume ratio of 10:1, respectively.
Friction measurements
Before test, the mixtures above were introduced between a ball and a disk with a volume of 20 μL and then the friction force between the ball and disk were measured on a Universal Micro-Tribotester (UMT-3, Bruker, America) with a rotation mode. The ball is made of silicon nitride (Si3N4) or sapphire (α-Al2O3) with a diameter of 4 mm. There are six kinds of materials used as disks, which are glass (SiO2), silicon nitride (Si3N4), sapphire (α-Al2O3), polytetrafluoroethylene (PTFE), polished steel (AISI 52100) and diamond-like carbon coating (DLC, ta-C) on Si substrate. Both the balls and the disks were cleaned in an ultrasonic bath with acetone and ethanol each for 10 min and then they were washed by deionized water and dried by compressed air for test. The load applied on the disk was 3 N and the rotation speed of the disk was changed from 5 rpm to 900 rpm with a track radius of 4 mm, corresponding to a sliding speed from 0.002 m/s to 0.375 m/s. All the tests were performed at the temperature of 26°C and the relative humidity of 20–30%.
References
Perry, S. S. & Tysoe, W. T. Frontiers of fundamental tribological research. Tribol. Lett. 19, 151–161 (2005).
Martin, J. M., Donnet, C., Lemogne, T. & Epicier, T. Superlubricity of Molybdenum-Disulfide. Phys. Rev. B 48, 10583–10586 (1993).
Erdemir, A., Eryilmaz, O. L. & Fenske, G. Synthesis of diamondlike carbon films with superlow friction and wear properties. J. Vac. Sci. Technol. A 18, 1987–1992 (2000).
Dienwiebel, M. et al. Superlubricity of graphite. Phys. Rev. Lett. 92, 126101 (2004).
Jin, Z. & Duncan, D. Bio-friction. Friction 1, 100–113 (2013).
Damiens, B., Venner, C. H., Cann, P. M. E. & Lubrecht, A. A. Starved lubrication of elliptical EHD contacts. J. Tribol.-Trans. ASME 126, 105–111 (2004).
Klein, J. Hydration lubrication. Friction 1, 1–23 (2013).
Hirano, M., Shinjo, K., Kaneko, R. & Murata, Y. Observation of superlubricity by scanning tunneling microscopy. Phys. Rev. Lett. 78, 1448–1451 (1997).
Erdemir, A. & Martin, J.-M. Superlubricity. (Elsevier Academic Press, 2007).
Li, J. J. & Luo, J. B. Advancements in superlubricity. Sci. China-Technol. Sci. 56, 2877–2887 (2013).
Xu, J. G. & Kato, K. Formation of tribochemical layer of ceramics sliding in water and its role for low friction. Wear 245, 61–75 (2000).
Chen, M., Kato, K. J. & Adachi, K. The comparisons of sliding speed and normal load effect on friction coefficients of self-mated Si3N4 and SiC under water lubrication. Tribol. Int. 35, 129–135 (2002).
Zhou, F., Adachi, K. & Kato, K. Sliding friction and wear property of a-C and a-CNx coatings against SiC balls in water. Thin Solid Films 514, 231–239 (2006).
Matta, C., Joly-Pottuz, L., Bouchet, M. I. D. & Martin, J. M. Superlubricity and tribochemistry of polyhydric alcohols. Phys. Rev. B 78, 085436 (2008).
Drobek, T. & Spencer, N. D. Nanotribology of surface-grafted PEG layers in an aqueous environment. Langmuir 24, 1484–1488 (2008).
Li, J. J., Zhang, C. H. & Luo, J. B. Superlubricity Behavior with Phosphoric Acid-Water Network Induced by Rubbing. Langmuir 27, 9413–9417 (2011).
Li, J. J., Zhang, C. H. & Luo, J. B. Superlubricity achieved with mixtures of polyhydroxy alcohols and acids. Langmuir 29, 5239–5245 (2013).
Li, J. J., Zhang, C. H., Ma, L. R., Liu, Y. H. & Luo, J. B. Superlubricity Achieved with Mixtures of Acids and Glycerol. Langmuir 29, 271–275 (2013).
Li, J. J., Zhang, C. H., Sun, L., Lu, X. C. & Luo, J. B. Tribochemistry and superlubricity induced by hydrogen ions. Langmuir 28, 15816–15823 (2012).
Dowson, D. & Higginson, G. R. Elasto-Hydrodynamic Lubrication. (Pergamon Press, 1977).
Lu, X., Khonsari, M. M. & Gelinck, E. R. M. The Stribeck curve: Experimental results and theoretical prediction. J. Tribol.-Trans. ASME 128, 789–794 (2006).
Raviv, U. & Klein, J. Fluidity of bound hydration layers. Science 297, 1540–1543 (2002).
Briscoe, W. H. et al. Boundary lubrication under water. Nature 444, 191–194 (2006).
Li, J. J. et al. Investigations on the mechanism of superlubricity achieved with phosphoric acid solution by direct observation. J. Appl. Phys. 114, 114901 (2013).
Acknowledgements
The work is financially supported by National Key Basic Research Program of China (2013CB934200), NSFC of China (51405256, 51222507, 51335005) and China Postdoctoral Science Foundation funded project (2014M550056).
Author information
Authors and Affiliations
Contributions
J.J.L., C.H.Z., M.M.D. and J.B.L. designed the experiments. Most of the experiments were carried out by J.J.L. with contributions from all authors. All authors contributed to the analysis and discussion of the data and the writing of the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License. The images or other third party material in this article are included in the article's Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder in order to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/
About this article
Cite this article
Li, J., Zhang, C., Deng, M. et al. Reduction of friction stress of ethylene glycol by attached hydrogen ions. Sci Rep 4, 7226 (2014). https://doi.org/10.1038/srep07226
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep07226
This article is cited by
-
Achieving ultrafast superlubricity with layered double hydroxides
Nano Research (2023)
-
Macroscale superlubricity achieved via hydroxylated hexagonal boron nitride nanosheets with ionic liquid at steel/steel interface
Friction (2022)
-
Insight into macroscale superlubricity of polyol aqueous solution induced by protic ionic liquid
Friction (2022)
-
Low friction in self-mated silicon carbide tribosystem using nanodiamond as lubricating additive in water
Friction (2021)
-
Superlubricity achieved with two-dimensional nano-additives to liquid lubricants
Friction (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.