Abstract
An inadequate selenium level is supposed to be a risk factor for cardiovascular diseases. However little is known about variation of the genes encoding selenium-containing proteins that would confirm the causality in these diseases. The aim of this study was to analyze the relationships between two functional variants of selenoprotein P gene (SEPP1 rs3877899G>A, rs7579G>A) and the occurrence of abdominal aortic aneurysm (AAA) and aortoiliac occlusive disease (AIOD), as well as their metabolic risk factors. In AAA, the rs3877899A allele was associated with higher systolic blood (P < .003) and pulse pressure (P < .003) values (recessive model) and with coexistence of peripheral arterial disease (PAD; carriers: P = .033). The other SEPP1 variants were associated with BMI values and influenced the risk of aortic diseases, depending on body weight. The strongest associations in the case-control analysis was found between the presence of the rs3877899G-rs7579G haplotype and development of AAA in overweight and obese subjects (OR = 1.80, 95%CI = 1.16–2.79, P = .008). The higher BMI values were correlated with lower age of AAA patients and larger size of aneurysm. Our results suggests the potential role of the selenoprotein P in pathogenesis of AAA. Future studies should consider the role of the rs3877899G-rs7579G haplotype as a risk factor for aggressive-growing AAAs.
Similar content being viewed by others
Introduction
Abdominal aortic aneurysm (AAA) is a progressive dilatation of the abdominal aorta with a diameter of at least 3.0 cm1. AAAs are often asymptomatic until they rupture. The disease affects ~9% of adults older than 65 years, causing almost 2% of male deaths in industrialized countries. AAA has a complex pathophysiology, in which both environmental and genetic factors play important roles2. The mechanisms of AAA initiation and rupture are still not fully elucidated, partially because of the coexistence of atherosclerotic occlusive disease in the majority of patients, which has hindered the designation of risk factors specific to aneurysm formation.
Increasing evidence indicates that oxidative stress in the aorta may contribute to aortic wall weakening in AAA3,4 whereas factors with antioxidant capacity, such as resveratrol5, fruit consumption6 and increased levels of high-density lipoproteins7, could prevent its development or decrease the risk of rupture. The role of the antioxidant trace element selenium deficiency in AAA pathogenesis could also be postulated in countries with a relatively low dietary selenium intake8. European soil, particularly in Central Europe, is relatively low in this micronutrient, which results in low selenium content in food products. A decreased plasma selenium levels has been associated in European populations with development of different cardiovascular diseases8, including AAA in the Polish population9. In addition to nutritional selenium deficiency, cigarette smoking, the most important risk factor for AAA10, may substantially contribute to the reduction of selenium levels in patients11. What is more, it was found that the levels of thioredoxin, a component of the selenium-dependent redox signaling system, correlate with the size and expansion rate of AAAs12.
Selenium exerts its biological role as a selenocysteine residue in a family of 25 selenoproteins. The tissue distribution and availability of selenium depend on the status of selenoprotein P, a major selenoprotein in plasma with both transport and antioxidant activity. This protein might also act as a heavy-metal chelator13 and an anti-angiogenic agent14.
Selenoprotein P has two major domains: N-terminal, which contain 1 selenocysteine residue in a redox motif and several potential heparin-binding and C-terminal with up to 9 selenocysteine residues. The heparin-binding domain facilitate binding of protein to proteoglycans on the vascular endothelium and this may contribute to the protection against oxidants15. In humans, the full length protein (60-kDa) contain 10 selenocysteine residues, while the truncated isoform (50-kDa) only one16.
The liver produces most plasma selenoprotein P, although this protein is also expressed in other tissues and is presumably secreted by them17. The expression of SEPP1 gene encoding selenoprotein P is upregulated in abdominal aorta of AAA patients (the data available in the NCBI's GEO database http://www.ncbi.nlm.nih.gov/geo/)18, most probably to protect the tissue from the effects of oxidative stress and inflammation19. Upregulation of this gene was also observed in a subclass of anti-inflammatory macrophages present in atherosclerotic plaques20 and in vessels stimulated to remodel invardly by reduced blood flow, the process that mimics the effect of arterial hypertension on small arteries21. On the contrary, in dissected regions of ascending aorta a downregulation of SEPP1 gene was observed22, which demonstrates a difference in the pathobiology between dissected thoracoabdominal aortic aneurysms and AAA.
The two studied SEPP1 single nucleotide polymorphisms (SNPs) have been reported to have a functional effect: rs3877899G>A (Ala234Thr) in exon 5 and rs7579G>A in the 3′ untranslated region23,24. Both of these variants influence the proportion of the two protein isoforms; the rs3877899G and rs7579A alleles favor the production of the 60-kDa isoform, whereas the rs3877899A and rs7579G alleles - the production of the 50-kDa isoform25. These SNPs affect selenium's bioavailability for the synthesis of all other selenoproteins (e.g. proteins that are involved in regulation of the extra- and intracellular redox environment and protection against endoplasmic reticulum stress) by influencing the body's selenium status23, effectiveness of supplementation23 and selenium supply to target tissues26. In consequence, they may influence cardiovascular outcomes independently of dietary selenium intake. Determination of the frequency of the SEPP1 variants in AAA could be crucial for further studies of serum biomarkers that improve clinical prediction in this disease, because markers of inflammation and endothelium injury were previously found to be useful for indicating the development of life-threatening complications of repair surgery for AAA27.
In search for the associations specific for the development of abdominal aortic aneurysms, the present study analyzed the SEPP1rs3877899 and rs7579 polymorphisms in three groups: patients with AAA, patients suffering from aortoiliac occlusive disease (AIOD), an atherosclerotic disease partially sharing its location with AAAs and controls. There are no previous reports on the distribution of SEPP1 SNPs in these aortic diseases.
Results
Traditional risk factors distribution
Smoking was the most prevalent and the most important risk factor for AAA (80.0%, P < .0001) and AIOD (91.3%, P < .0001, Table 1). The other risk factors for both aortic diseases were: arterial hypertension and low high-density lipoprotein cholesterol level. Advanced age, male sex, overweight and obesity constituted risk factors that were more common in AAA. In AIOD, an increased proportion of underweight subjects (7.8%), as compared to both controls (1.8%) and AAA patients (1.8%, P < .0001) were observed. The AAA patients with concomitant PAD, compared with those without, had a significantly higher frequency of smokers (88.2% vs 77.3%; P = .02) and smaller aortic diameters (median [interquartile range], 57.5 [48, 69] mm vs 62.5 [55, 72] mm; P < .01).
Genotype distribution
The frequency of genotypes did not deviate significantly from the Hardy-Weinberg equilibrium in either the patients or the controls (Tables 2 and 3). Because of the small distance between the two SEPP1 variants (460 bp), resulting in the complete linkage disequilibrium between them (D′ = 1.0), only the three common rs3877899-rs7579 haplotypes: G-G, G-A and A–G were observed in the our population. As a consequence, the effects of the rs3877899A and rs7579A alleles correspond to the impacts of the rs3877899A-rs7579G and rs3877899G-rs7579A haplotypes, respectively. The estimated pairwise r2 value for studied SNPs (.129) was similar to that observed in other populations of European ancestry (range between .122 and .221, according to the Ensembl database).
The distribution of polymorphisms differed between the subgroups of AAA patients stratified by PAD coexistence. There were fewer carriers of the SEPP1rs3877899A (rs3877899A-rs7579G) allele among patients without concomitant PAD than among those with PAD; respectively: 37.1% and 44.1% (OR = 0.61, 95%CI: 0.38–0.96, P = .033, Table 2 and 4). A 40% reduction in the risk of AAA without coexisting PAD was observed for the carriers of this allele (OR = 0.60 95%CI: 0.39–0.91, P = .017). These effects remained significant after adjusting for age and sex (Table 4). The studied SEPP1 polymorphisms were not significantly correlated with the aortic diameter of AAA patients.
Effects of the polymorphisms on the metabolic risk factors for cardiovascular disease
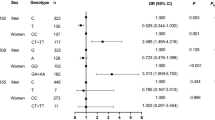
The relations between SEPP1rs3877899-rs7579 haplotypes and age, sex, smoking status, blood pressure parameters, arterial hypertension, glucose levels, diabetes, BMI values, overweight, obesity, blood lipid and lipoproteins level in AAA and AIOD were investigated. The associations of the specific SEPP1 haplotypes with blood pressure parameters, BMI values and occurrence of overweight and obesity (BMI > 25) were found (Figure 1, Tables 3 and 4). In AAA, carriers of two copies of the A-G haplotype had significantly higher systolic blood pressure (SBP) and pulse pressure (PP) values, as compared to subjects with one copy of this haplotype and to non-carriers (these effects remained also significant after adjusting for other potential confounders: age, sex, hypertension and PAD). The G-G haplotype was associated with higher, while G–A – with lower BMI values in this patient group (Figure 1). Carriers of the G-G haplotype had more frequently BMI values > 25 in the whole studied population (two copies: OR = 1.51, 95%CI = 1.11–2.06, P = .007) and display a significant 1.80-fold increase in risk for the development of AAA in overweight and obese subjects (dominant model, 95%CI: 1.16–2.79, P = .008). In patients group (AAA plus AIOD), carriers of the G-A haplotype had less frequently BMI values > 25 (OR = 0.69, 95% CI = 0.50–0.93, P = 0.016) and display a 1.54-fold increase in the risk of studied aortic diseases in non-overweight subjects (95%CI: 1.01–2.351, P = .045). These relationships remained significant after adjusting for age and sex. The associations of the A–G haplotype with aortic diseases in overweight and obese subjects were significant only in univariate analysis.
BMI values and other risk factors for cardiovascular disease and aortic diameter
In both patients group, BMI values were positively correlated with TG levels (AAA: r = .231, P < .0001, AIOD: r = .338, P < .0001) and negatively with the HDLC concentration (AAA: r = −.183, P = .001, AIOD: r = −.286, P = .001). Positive associations between increased BMI values and hypertension (AAA: P = .004, AIOD: P = .007) or diabetes (AAA and AIOD: P < .0001) were also noted. In AAA, BMI values were also positively correlated with the aortic diameter (r = .130, P = .023), while negatively with the age of patients (r = −.156, P = .005). All these associations were significant after adjusting for sex or sex and age.
Discussion
The results of our study provide preliminary evidence of the role of SEPP1 polymorphisms in AAA and AIOD pathogenesis and their association with the metabolic risk factors of these diseases and the coexistence of PAD in AAA. The main outcomes were: 1) the association of the SEPP1rs3877899A allele (the rs3877899A-rs7579G haplotype), that favors the production of the low-selenium isoform of selenoprotein P, with coexistence of PAD in AAA as well as with increased systolic blood pressure and pulse pressure parameters in these patients and 2) the association of the SEPP1rs3877899G-rs7579G haplotype with increased BMI values in the studied population and AAA formation in overweight and obese subjects. These associations were novel in the context of cardiovascular disease development.
The SEPP1 rs3877899A allele, related to a more complex phenotype of AAA, was previously associated with reduced risk of gender–dependent cancers: breast28 and prostate29, while the opposite allele of this SNP has been shown to enhance the risk of prostate cancer in carriers of the variant of the mitochondrial superoxide dismutase gene30. Furthermore, the risk of colorectal cancer was influenced by the two-loci interactions between the SEPP1rs3877899 alleles and polymorphisms in other selenoprotein encoding genes31. However, this relation, while observed in Europe, was not replicated in populations of United States32 and Korea33, most probably because of population differences in selenium supplementation8 and allele frequencies. The analysis of data from the International HapMap Project, the 1000 Genomes Project, the NHLBI GO Exome Sequencing Project and other previously published23,24,28,29,30,31,32,33,34,35,36 demonstrates significant population diversity in the distribution of the SEPP1 SNPs (Supplementary Table), which may influence the impact of alleles on cardiovascular morbidity. The frequency of the rs3877899A allele ranged from 0.001 to 0.412 (from 0.213 to 0.360 in subjects with European ancestry), while the frequency of the rs7579A allele was more similar in ethnically different populations and it ranged from 0.120 to 0.325 (from 0.248 to 0.337 in subjects with European ancestry).
Polymorphisms in several selenoprotein encoding genes have been previously found to significantly influence cardiovascular health. For example, a variant allele of glutathione peroxidase 1 was found to protect against thoracic aortic aneurysm development in hypertensive patients, while increasing the risk of coronary artery disease, PAD and stroke37. Other examples include a polymorphism of the glutathione peroxidase 3, which has been associated with the occurrence of stroke and cerebral venous thrombosis38 and the selenoprotein S gene variants which influence the risk of stroke and coronary artery disease39. The genome-wide association study approach for AAAs has so far failed to find any statistically significant associations with SEPP12. However, atherosclerosis may be a potential confounder in these studies, since the polymorphisms identified in these reports have been shown to be also associated with other cardiovascular diseases. Similarly, shared disease pathways between AAA and atherosclerosis may contribute to failure of studies, that focus on AAA-specific biomarkers40.
The link between the SEPP1rs3877899A allele and the presence of PAD in AAAs corroborates the reports of the protective role of selenoprotein P and selenium against atherosclerosis development8. Studies show that the initiation and acceleration of atherogenesis in AAA may be a consequence of a pro-inflammatory response induced by cigarette smoking41. In accordance with this observation, smoking in our study was more common in patients with concomitant PAD, than in those without. This environmental factor has been found to exert an adverse effect on both the severity of inflammation42 and the levels of selenium9 in AAA tissue. SEPP1 gene expression is induced by interleukin-1019, thus the preferential expression of this cytokine in AAA tissue, as compared to occlusive atherosclerotic lesions41, may partially explain the observed lack of effects of this gene variant on AIOD development. The relative decrease in the prevalence of the rs3877899A allele in AAA without concomitant PAD, as compared to controls seems to be a consequence of subgroup selection. Nevertheless, this association confirm the heterogeneity of AAA pathogenesis, which may result in different mechanisms of disease initiation in unrelated cases.
Our data indicate that the specific SEPP1 variants may affect the risk of AAA by enhancing the adverse effects of other risk factors: isolated systolic hypertension (the rs3877899A allele) and high BMI values (the rs3877899G-rs7579G haplotype). This type of hypertension is characterized by increased systolic and normal diastolic blood pressure values, resulting in an increase in pulse pressure values. It is primarily a consequence of a reduction in the elasticity of the large arteries and progresses with age. Clinical studies show increased prevalence of AAA in systolic hypertension and an independent association between high pulse pressure values and the occurrence of the disease43. Thus, genetically determined reduced activity of selenoprotein P resulting in the promotion of elastin degradation and endothelial dysfunction44 under oxidative-stress conditions and may be the common mechanisms for both AAA and systolic hypertension development. Experiments involving the induction of aortic aneurysms by angiotensin II infusions in hyperlipidemic mouse strains1 confirmed the important role of blood pressure in AAA pathogenesis, whereas recent functional studies in spontaneously hypertensive rats demonstrated the link between hypertension, low selenium diet, endothelial injury and extensive degeneration of the aortic wall45.
The presence of lower extremity arterial occlusion in AIOD could alter the effects of genotype, since PAD itself was found to be an independent predictor of isolated systolic hypertension in the elderly46. Accordingly, we observed slightly higher prevalence of this type of hypertension in AIOD than in AAA (22% vs 18%, P = .080, data not presented). Isolated systolic hypertension is a strong predictor of cardiovascular mortality, whereas diet modification, smoking cessation and supplementation with antioxidants may serve as a therapeutic strategy for the improvement of endothelial dysfunction; therefore, further studies on selenoprotein variants in hypertension seem advisable.
In clinical studies, the two functional SEPP1 polymorphisms, together with the values of BMI, have been shown to predict baseline plasma selenium content in the general population23, but there are no previous reports on the role of these variants in human obesity. Notwithstanding, this function may be postulated from studies conducted on mice without the selenoprotein P gene, as these animals lost weight when fed with low-selenium diet and did not gain it significantly when fed with a high-fat, high-sucrose diet8. Moreover, studies in diabetics show correlation between the levels of selenoprotein P and adiponectin47, an adipose tissue hormone with protective function in cardiovascular diseases. The associations between the SEPP1 polymorphisms and several metabolic phenotypes related to diabetes have also been documented; for example, the rs7579 variant was previously associated with insulin sensitivity, fasting insulin and fibrinogen levels34. Because BMI values in AAA were associated with the aorta diameter and the age of patients, evaluation of the clinical utility of SEPP1 polymorphisms for stratification of AAA risk and growth rate should be considered in future studies.
There are several limitations to our study; the statistical power of study is restricted by the sample size, then a future replication study should be drawn to confirm the findings. However, the low P-values for most analyzes indicate that the results are not random. The second limitation is the absence of the ankle-brachial index (ABI) measurements for PAD diagnosis. We had to use a more precise method, because ABIs could not be reliably obtained in many cases. The use of BMI for defining the risk of cardiovascular diseases has also numerous limitations; abdominal adiposity, which correlates with inflammation and systemic oxidative stress, would be a better predictor for cardiovascular risk. However, the correlation of BMI values with the size of aneurysm, age of patients and modifiable risk factors confirm the usefulness of this parameter for estimating the AAA risk. On the other hand, the symptomatic and chronic course of AIOD may weaken the strength of the relationship between the SEPP1 polymorphisms and BMI values. As the severe symptoms of lower extremity ischemia lead to physical exhaustion in a high number of patients, an increased frequency of underweight individuals was observed among examined AIOD cases. The fourth limitation is that this was only a clinical association study and thus selenium content and other confounding factors that might impair the selenoprotein P production: deficiency of other nutrients, exposition to heavy-metals and food additive carrageenan, insulin resistance, visceral obesity, nonalcoholic fatty liver disease, were not examined; therefore, potential mechanisms were not fully elucidated. Also the influence of cigarette smoking could not be reliably evaluated, because of underrepresentation of nonsmokers in studied diseases (statistical analyzes indicated no effect of smoking; data not presented). Nevertheless, it should be stressed that genetic research is highly useful in delineating causal disease pathways, because it represents lifelong exposure and is not affected by reverse causality.
In conclusion, this study provide preliminary genetic evidence that the SEPP1 functional variants contribute to the AAA risk. The results suggests that the rs3877899A allele may increase the susceptibility or promote AAA progression through influencing the risk of peripheral atherosclerosis and isolated systolic hypertension. The SEPP1rs3877899G-rs7579G haplotype seems to be a factor that increases predisposition to AAA in overweight and obese subjects and a potential marker of aggressive-growing AAAs. Further work is required to confirm the findings in other populations and to study the functional role of these variants in AAA.
Methods
Study population
The study enrolled 335 AAA patients and 333 AIOD patients scheduled for surgery at the Department of General and Vascular Surgery of the Poznan University of Medical Sciences in the years 1999–2011. The control group, consisting of 336 subjects matched for age (±5 years) and sex to the AAA patients, was selected during the same time from the Poznan district. The patients and controls included in this study were previously investigated for polymorphisms involved in homocysteine metabolism48 and the hypoxia signaling pathway49. Detailed definitions and clinical characteristics were presented in previous publications48,49. In short, the exclusion criteria for the controls included known aneurysms and peripheral arterial disease (PAD), whereas for AIOD – the coexistence of AAA. The diagnosis of AAA was evaluated by computed tomography angiography or magnetic resonance angiography, while the diagnosis of AIOD – by computed tomography angiography alone. Based on physical examination supplemented with ultrasound duplex color scanning, the coexistence of PAD was recognized in 60.3% of the AAA patients. All patients were treated pharmacologically with statins, antiplatelet drugs and other drugs (antihypertensive or antidiabetic), depending on their clinical condition.
Questionnaires were used to obtain information concerning cigarette smoking history and standardized methods were employed for the evaluation of the remaining known risk factors for cardiovascular diseases. Hypertension was defined as systolic blood pressure (SBP) ≥ 140 mmHg and/or diastolic blood pressure (DBP) ≥ 90 mmHg, as well as in the case of antihypertensive therapy. Diabetes was determined by fasting plasma glucose levels (>6.8 mmol/L) and antidiabetic medication status. Obesity was recognized at a body mass index (BMI) ≥ 30.0 kg/m2 overweight at BMI = 25.0–29.9 kg/m2, whereas underweight at BMI < 18.5 kg/m2. The study was conducted in accordance with the Declaration of Helsinki. The study protocol was approved by the Bioethical Committee of the Poznan University of Medical Sciences, Poland (decision no 628/10) and all subjects provided their informed and written consent to participate in this study.
Genotyping
Genomic DNA was extracted from circulating blood lymphocytes by a chemical method (using the acid guanidinium thiocyanate and phenol/chloroform extraction). Polymorphisms were ascertained with the use of the TaqMan Pre-Designed SNP Genotyping Assays (Life Technologies) employing the ABI 7900HT Fast Real-Time PCR System (Life Technologies).
Data analysis
Genotype frequencies were tested for Hardy-Weinberg equilibrium using the χ2 test (http://ihg.gsf.de/cgi-bin/hw/hwa1.pl). Haploview was used for SEPP1 haplotype analysis and QUANTO software (http://hydra.usc.edu/GxE/) was employed for sample size calculations. For genotypes, alleles and haplotypes, the values of odds ratio (OR) and 95% confidence intervals (95% CI) were calculated. The post-hoc analysis revealed that the statistical power of our study for analyses of the differences in genotype distribution between subgroups of patients and controls ranged between 50%–70%. In univariate analyses, the Mann-Whitney U test, the Kruskal-Wallis or ANOVA tests were performed for quantitative variables and the χ2 test was employed for qualitative variables. Logistic and conditional regression analysis or MANOVA tests were used to adjust the effects of modifiable risk factors of AAA and AIOD and genotype for age, sex and other potential confounders. In parametric analyses, continuous variables which deviate in distribution from the normal curve were analyzed after logarithmic transformation. Pearson's coefficient of correlation, were calculated (r) to assess the strength of the association between BMI values and other risk factors (when appropriate). These analyses were performed using STATISTICA version 10.0 software. The observed differences were considered significant at P < .05, with the exception of the effects of the rs3877899G-rs7579G haplotype, which were considered significant at P < .025 (the Bonferroni correction for multiple comparisons was applied).
References
Kuivaniemi, H. & Elmore, J. R. Opportunities in abdominal aortic aneurysm research: epidemiology, genetics and pathophysiology. Ann Vasc Surg 26, 862–70 (2012).
Harrison, S. C., Holmes, M. V., Agu, O. & Humphries, S. E. Genome wide association studies of abdominal aortic aneurysms–Biological insights and potential translation applications. Atherosclerosis 217, 47–56 (2011).
Miller, F. J., Jr et al. Oxidative stress in human abdominal aortic aneurysms: a potential mediator of aneurysmal remodeling. Arterioscler Thromb Vasc Biol 22, 560–5 (2002).
Cafueri, G. et al. Endothelial and smooth muscle cells from abdominal aortic aneurysm have increased oxidative stress and telomere attrition. PLoS One 7, e35312 (2012).
Kaneko, H. et al. Resveratrol prevents the development of abdominal aortic aneurysm through attenuation of inflammation, oxidative stress and neovascularization. Atherosclerosis 217, 350–357 (2011).
Stackelberg, O., Bjorck, M., Larsson, S. C., Orsini, N. & Wolk, A. Fruit and vegetable consumption with risk of abdominal aortic aneurysm. Circulation 128, 795–802 (2013).
Torsney, E. et al. Elevation of plasma high-density lipoproteins inhibits development of experimental abdominal aortic aneurysms. Arterioscler Thromb Vasc Biol 32, 2678–2686 (2012).
Joseph, J. & Loscalzo, J. Selenistasis: epistatic effects of selenium on cardiovascular phenotype. Nutrients 5, 340–58 (2013).
Socha, K., Borawska, M. H., Gacko, M. & Guzowski, A. Diet and the content of selenium and lead in patients with abdominal aortic aneurysm. Vasa 40, 381–9 (2011).
Wilmink, T. B. M., Quick, C. R. G. & Day, N. E. The association between cigarette smoking and abdominal aortic aneurysms. J Vasc Surg 30, 1099–1105 (1999).
Ellingsen, D. G., Thomassen, Y., Rustad, P., Molander, P. & Aaseth, J. The time-trend and the relation between smoking and circulating selenium concentrations in Norway. J Trace Elem Med Biol 23, 107–115 (2009).
Martinez-Pinna, R. et al. Increased levels of thioredoxin in patients with abdominal aortic aneurysms (AAAs). A potential link of oxidative stress with AAA evolution. Atherosclerosis 212, 333–8 (2010).
Sasakura, C. & Suzuki, K. T. Biological interaction between transition metals (Ag, Cd and Hg), selenide/sulfide and selenoprotein P. J Inorg Biochem. 71, 159–62 (1998).
Ishikura, K. et al. Selenoprotein P as a diabetes-associated hepatokine that impairs angiogenesis by inducing VEGF resistance in vascular endothelial cells. Diabetologia 57, 1968–76 (2014).
Arteel, G. E., Franken, S., Kappler, J. & Sies, H. Binding of selenoprotein P to heparin: characterization with surface plasmon resonance. Biol Chem 381, 265–8 (2000).
Mostert, V., Lombeck, I. & Abel, J. A Novel Method for the Purification of Selenoprotein P from Human Plasma. Arch Biochem Biophys 357, 326–330 (1998).
Burk, R. F. & Hill, K. E. Selenoprotein P-expression, functions and roles in mammals. Biochim Biophys Acta 1790, 1441–7 (2009).
Lenk, G. M. et al. Whole genome expression profiling reveals a significant role for immune function in human abdominal aortic aneurysms. BMC Genomics 8, 237 (2007).
Bosschaerts, T. et al. Alternatively activated myeloid cells limit pathogenicity associated with african Trypanosomiasis through the IL-10 inducible gene selenoprotein P. J Immunol 180, 6168–6175 (2008).
Brochériou, I. et al. Antagonistic regulation of macrophage phenotype by M-CSF and GM-CSF: Implication in atherosclerosis. Atherosclerosis 214, 316–324 (2011).
van den Akker, J. et al. The redox state of transglutaminase 2 controls arterial remodeling. PLoS One 6, e23067 (2011).
Müller, B. T. et al. Gene expression profiles in the acutely dissected human aorta. Eur J Vasc Endovasc Surg 24, 356–364 (2002).
Méplan, C. et al. Genetic polymorphisms in the human selenoprotein P gene determine the response of selenoprotein markers to selenium supplementation in a gender-specific manner (the SELGEN study). FASEB J 21, 3063–3074 (2007).
Karunasinghe, N. et al. Serum selenium and single-nucleotide polymorphisms in genes for selenoproteins: relationship to markers of oxidative stress in men from Auckland, New Zealand. Genes Nutr 7, 179–90 (2012).
Meplan, C. et al. Relative abundance of selenoprotein P isoforms in human plasma depends on genotype, se intake and cancer status. Antioxid Redox Signal 11, 2631–40 (2009).
Kurokawa, S., Bellinger, F. P., Hill, K. E., Burk, R. F. & Berry, M. J. Isoform-specific Binding of Selenoprotein P to the β-Propeller Domain of Apolipoprotein E Receptor 2 Mediates Selenium Supply. J Biol Chem 289, 9195–9207 (2014).
Kokot, M. et al. Endothelium injury and inflammatory state during abdominal aortic aneurysm surgery: scrutinizing the very early and minute injurious effects using endothelial markers - a pilot study. Arch Med Sci 9, 479–86 (2013).
Meplan, C. et al. Association between polymorphisms in glutathione peroxidase and selenoprotein P genes, glutathione peroxidase activity, HRT use and breast cancer risk. PLoS One 8, e73316 (2013).
Steinbrecher, A. et al. Effects of selenium status and polymorphisms in selenoprotein genes on prostate cancer risk in a prospective study of European men. Cancer Epidemiol Biomarkers Prev 19, 2958–68 (2010).
Cooper, M. L. et al. Interaction between single nucleotide polymorphisms in selenoprotein P and mitochondrial superoxide dismutase determines prostate cancer risk. Cancer Res 68, 10171–10177 (2008).
Meplan, C. et al. Genetic variants in selenoprotein genes increase risk of colorectal cancer. Carcinogenesis 31, 1074–9 (2010).
Peters, U. et al. Variation in the selenoenzyme genes and risk of advanced distal colorectal adenoma. Cancer Epidemiol Biomarkers Prev 17, 1144–54 (2008).
Sutherland, A., Kim, D.-H., Relton, C., Ahn, Y.-O. & Hesketh, J. Polymorphisms in the selenoprotein S and 15-kDa selenoprotein genes are associated with altered susceptibility to colorectal cancer. Genes Nutr 5, 215–223 (2010).
Hellwege, J. N. et al. Genetic variants in selenoprotein P plasma 1 gene (SEPP1) are associated with fasting insulin and first phase insulin response in Hispanics. Gene 534, 33–39 (2014).
Sun, W. et al. Selenoprotein P gene r25191g/a polymorphism and quantification of selenoprotein P mRNA level in patients with Kashin-Beck disease. Br J Nutr 104, 1283–7 (2010).
Calvo, A. et al. Alterations in gene expression profiles during prostate cancer progression: functional correlations to tumorigenicity and down-regulation of selenoprotein-P in mouse and human tumors. Cancer Res 62, 5325–35 (2002).
Kato, K. et al. Assessment of Genetic Risk Factors for Thoracic Aortic Aneurysm in Hypertensive Patients. Am J Hypertens 21, 1023–1027 (2008).
Leopold, J. A. & Loscalzo, J. Oxidative risk for atherothrombotic cardiovascular disease. Free Radic Res 47, 1673–1706 (2009).
Alanne, M. et al. Variation in the selenoprotein S gene locus is associated with coronary heart disease and ischemic stroke in two independent Finnish cohorts. Hum Genet 122, 355–365 (2007).
Skora, J. et al. Prognostic value of tissue factor in patients with abdominal aortic and iliac arterial aneurysms - preliminary study. Arch Med Sci 9, 1071–7 (2013).
Shimizu, K., Mitchell, R. N. & Libby, P. Inflammation and cellular immune responses in abdominal aortic aneurysms. Arterioscler Thromb Vasc Biol 26, 987–994 (2006).
Arapoglou, V. et al. The influence of total plasma homocysteine and traditional atherosclerotic risk factors on degree of abdominal aortic aneurysm tissue inflammation. Eur J Vasc Endovasc Surg 43, 473–479 (2009).
Naydeck, B. L., Sutton-Tyrrell, K., Schiller, K. D., Newman, A. B. & Kuller, L. H. Prevalence and risk factors for abdominal aortic aneurysms in older adults with and without isolated systolic hypertension. Am J Cardiol 83, 759–764 (1999).
Steinbrenner, H., Bilgic, E., Alili, L., Sies, H. & Brenneisen, P. Selenoprotein P protects endothelial cells from oxidative damage by stimulation of glutathione peroxidase expression and activity. Free Radic Res 40, 936–943 (2006).
Ruseva, B., Atanasova, M., Georgieva, M., Shumkov, N. & Laleva, P. Effects of selenium on the vessel walls and anti-elastin antibodies in spontaneously hypertensive rats. Exp Biol Med 237, 160–166 (2012).
Safar, M. E. et al. Peripheral arterial disease and isolated systolic hypertension: The ATTEST Study. J Hum Hypertens 23, 182–187 (2008).
Misu, H. et al. Inverse correlation between serum levels of selenoprotein P and adiponectin in patients with type 2 diabetes. PLoS One 7, e34952 (2012).
Strauss, E., Waliszewski, K. & Pawlak, A. L. [The different genotypes of MTHFR 1298A>C and PON1 -108C>T polymorphisms confer the increased risk of the abdominal aortic aneurysm in the smoking and nonsmoking persons]. Przegl Lek 62, 1023–30 (2005).
Strauss, E., Waliszewski, K., Oszkinis, G. & Staniszewski, R. Polymorphisms of genes involved in the hypoxia signaling pathway and the development of abdominal aortic aneurysms or large-artery atherosclerosis. J Vasc Surg (2014), 10.1016/j.jvs.2014.02.007 [Epub ahead of print].
Acknowledgements
The work has been supported by the National Science Centre in Poland under grant No. NN403250440.
Author information
Authors and Affiliations
Contributions
E.S. designed the study, analyzed and interpreted the data, performed genotyping and statistical analysis, drafted the manuscript and prepared figure. E.S. and G.O. participated in data acquisition. G.O. and R.S. took part in patients selection and examination and performed critical revision of the article. E.S., R.S. and G.O. participated in obtaining funding. All authors read and approved the final manuscript. Overall responsibility of E.S.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Supplementary Information
Supplementary Table
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License. The images or other third party material in this article are included in the article's Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder in order to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/
About this article
Cite this article
Strauss, E., Oszkinis, G. & Staniszewski, R. SEPP1 gene variants and abdominal aortic aneurysm: gene association in relation to metabolic risk factors and peripheral arterial disease coexistence. Sci Rep 4, 7061 (2014). https://doi.org/10.1038/srep07061
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep07061
This article is cited by
-
Estimation of Selected Minerals in Aortic Aneurysms—Impaired Ratio of Zinc to Lead May Predispose?
Biological Trace Element Research (2021)
-
Systems Biology of Selenium and Complex Disease
Biological Trace Element Research (2019)
-
Genetic polymorphism in selenoprotein P modifies the response to selenium-rich foods on blood levels of selenium and selenoprotein P in a randomized dietary intervention study in Danes
Genes & Nutrition (2018)
-
A case–control study of selenoprotein genes polymorphisms and autoimmune thyroid diseases in a Chinese population
BMC Medical Genetics (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.