Abstract
Using a Drosophila model, we previously demonstrated truncated life span and neurotoxicity with exposure to 1-octen-3-ol, the volatile organic compound (VOC) responsible for much of the musty odor found in mold-contaminated indoor spaces. In this report, using biochemical and immunological assays, we show that exposure to 0.5 ppm 1-octen-3-ol induces a nitric oxide (NO) mediated inflammatory response in hemocytes, Drosophila innate immune cells. Moreover, exposed Drosophila brains show increased peroxynitrite expression. An increase in nitrite levels is observed with toluene and 1-octen-3-ol but not with 1-butanol. Pharmacological inhibitors of nitric oxide synthase (NOS) namely, L-NAME, D-NAME and minocycline and NOS mutants show improvements of life span among 1-octen-3-ol exposed flies. Exposure to 1-octen-3-ol also induces NOS expression in larval tracheal tissues and remodels tracheal epithelial lining. These findings suggest a possible mechanistic basis for some of the reported adverse health effects attributed to mold exposure and demonstrates the utility of this in vivo Drosophila model to complement existing model systems for understanding the role of inflammation in VOC-mediated toxicity.
Similar content being viewed by others
Introduction
Inflammation via NO plays a crucial role in the development and progression of neurodegenerative diseases and asthma1,2. Macrophages and their mediators, including NO, are implemented in xenobiotic induced tissue injury and toxicity, where activation of macrophages protects the host from toxins and pathogens and serves essential functions for the survival of organisms. Conversely, excessive and unregulated activation of macrophages including microglia, the resident immune cells of brain, act as agents of destruction and thus promote tissue injury and disease3. Indoor air-VOCs and other chemical exposures are associated with neurogenic inflammation4. Similarly, NO induction is detected in lung linings of rodent models after exposure to VOC5,6. Peroxynitrite, derived from the combination of reactive oxygen species, especially superoxide and NO, interacts with cellular components, lipids, DNA and proteins and triggers cellular responses that range from harmless cell signaling to overwhelming oxidative injury leading to necrosis or apoptosis2.
Invertebrate hemocytes have been used as a model to study and measure the impact of chemicals on the immune system, including pesticides and heavy materials7. Drosophila hemocytes act as a surveillance system and respond to foreign agents like bacteria and parasites via stimulation of nitric oxide synthase (NOS), an enzyme for NO production8,9. These hemocytes are present in both larval and adult Drosophila, and are considered morphologically and functionally similar to mammalian microglia10.
Exposure to fungal contamination in water damaged buildings is correlated with adverse human health effects11,12. Fungal VOCs associated with moldy odors are hypothesized to contribute to such health effects13. Several of these compounds are toxic to mammalian models and cell cultures14. One of the major components of mold VOC mixtures is 1-octen-3-ol, a compound known to be ubiquitously produced by fungi, which is commonly detected reported in moldy, water damaged office buildings, residences and classrooms15,16,17.
We have pioneered a Drosophila model to characterize the toxicity profile of mixtures of VOCs emitted from living fungal cultures18. Exposure to 1-octen-3-ol leads to neurotoxicity, stimulated apoptotic signaling pathways in flies19,20,21 and cytotoxicity in human embryonic stem cells22. Furthermore, human volunteers exposed to 1.9 ppm of 1-octen-3-ol for 2 hours showed an increase in inflammatory markers in nasal secretions23.
Induction of the Drosophila innate immune response has been demonstrated against parasites and bacteria8,9 There has been no study with respect to a possible toxic chemical mediated induction of such innate immune response in flies that is comparable to microglial activation in mammals. In this report, we demonstrate that exposure to low concentrations (0.5 ppm) of 1-octen-3-ol vapors results in induction of NOS in Drosophila adult brain and larval tracheal linings. The exposure to 1-octen-3-ol appears to activate Drosophila innate immune cells. Taken together, these data demonstrate that a common fungal VOC leads to excessive stimulation of the inflammatory response and subsequent toxicity, thereby providing a possible mechanistic basis for some of the reported adverse health effects attributed to mold exposure.
Results
To evaluate if 1-octen-3-ol exposure leads to activation of the inflammatory marker, NO; we quantified the level of nitrites, the product of NO breakdown, after exposure to 0.5 ppm of 1-octen-3-ol, using a modified Griess reagent24. An increase in nitrite levels was found in extracts of 0.5 ppm 1-octen-3-ol-exposed head, body and whole flies but not in unexposed flies (Fig. 1a). The increased nitrite levels from exposed whole flies were further verified by performing qRT-PCR (Fig. 1b). After 6 hr, a more intense purple discoloration (indicative of elevated diaphorase activity) was detected in the anterior lobe and mushroom body regions of adult brains from exposed brains than in unexposed brains (Fig. 1c, d). The increase in the nitrite levels and diaphorase activity in the exposed flies suggests the activation of NO in response to 1-octen-3-ol.
1-octen-3-ol activates NOS in flies.
(a) Elevated nitrite levels were detected in the head, body and whole flies extracts after 6 hr exposure. (n = 250-300 heads and 80-120 bodies) ** = P < 0.01 and represent the significant difference between control (unexposed) and 1-octen-3-ol exposed groups (b) qRT-PCR showed approximately 10-fold upregulation of NOS in the whole flies extract of exposed flies compared to control flies after 6 hr of exposure. (n = 48); ** = P < 0.01 and shows a significant difference between control and 1-octen-3-ol groups. (c,d) 1-octen-3-ol exposed adult brain exhibited increased purple discoloration in the regions of anterior lobe (arrows) and mushroom bodies (arrowheads) as compared to control brain. (n = 10–12) (e,f) 1-octen-3-ol exposure caused expression of peroxynitrite in the adult brain exposed to 0.5 ppm for 6 hr but not in the unexposed control adult brains. (n = 8–10). (g,h) Insets showing the magnified regions shown in e and f. Arrows indicate the peroxynitrite positive signal in the exposed brain in inset (h).
Since we have previously showed that exposure to 1-octen-3-ol induces reactive oxygen species in Drosophila head extracts19, we hypothesized that peroxynitrite is also involved in 1-octen-3-ol mediated toxicity. We performed immunostaining using anti-nitrotyrosine antibody and found increased expression in the exposed adult brains as compared to non-exposed control brains (Fig 1e, f, g, h).
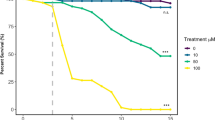
Furthermore, we also previously showed that exposure to 1-octen-3-ol is associated with the shortening of the fly survival span18,19,20,21. In order to assess the role of NO/peroxynitrite in 1-octen-3-ol mediated truncation of life span, we exposed 0.5 ppm 1-octen-3-ol to adult flies in the presence of the NO inhibitors, D-NAME, L-NAME and minocycline at a concentration of 2 mM. The feeding of L-NAME and minocycline led to an improvement in survival span by 6 and 8 days, respectively, indicating that truncation of life span with 1-octen-3-ol exposure is at least partially due to NO/peroxynitrite-induced damage and that prevention of 1-octen-3-ol-mediated NOS induction could be neuroprotective (Fig. 2a). Furthermore, the heterozygous mutant flies for NOS, NOS24283 exposed to 0.5 ppm of 1-octen-3-ol had a 3 day improvement in survival duration compared to exposed wild type flies (Fig 2b). To determine if the induction of NO/peroxynitrite is specific to 1-octen-3-ol, we tested toluene, a well-known industrial solvent and 1-butanol another fungal VOC. Previously, we have found that exposure of 2.8 ppm of toluene, but not 1-butanol, led to significant truncation of survival span of flies18. Here we assessed the level of nitrites in the extracts of head, body and whole flies exposed to 2.8 ppm of toluene and 1-butanol. There was a significant increase in the nitrite levels in the extracts of flies exposed to toluene as reported in mammalian model25 but not in those exposed to 1-butanol when compared with the control extracts (Fig. 2c). This suggests that the phenomenon of induction of NO/peroxynitrite is common to 1-octen-3-ol and toluene but not to 1-butanol.
Pharmacological inhibitors and NOS mutants show improvement in the survival span.
(a) 48 hr old post eclosion flies were exposed to 0.5 ppm of 1-octen-3-ol alone or with 2 mM each of L-NAME, D-NAME and minocycline continuously until the death of all flies and the average survival duration was calculated. The feeding of L-NAME and minocycline led to improvement in survival span by 6 and 8 days, respectively compared to flies exposed to 1-octen-3-ol alone. (n = 100–120). NS = non-significant. ** and *** = P < 0.01 and P < 0.001 and represent a significant difference between 1-octen-3-ol and other groups. (b) Upon exposure of 0.5 ppm of 1-octen-3-ol to NOS mutant and wild type flies, the heterozygous NOS mutants survived about 3 days longer than the wild type flies. * = P < 0.05 represent the significant difference between the mutant and wild type flies. (c) Elevated nitrite levels were detected in the head, body and whole flies extracts after 6 hr exposure to 2.8 ppm toluene but not with 1-butanol (n = 250–300 heads and 80–120 bodies) ** = P < 0.01 and represent the significant difference between control (unexposed) and exposed groups.
The activation of NO and peroxynitrite in response to 1-octen-3-ol exposure led us to seek for the source of the Drosophila NO. In Drosophila, glial cells are known to perform immune-like functions during development and remodeling of the nervous system26 and in a genetic model of neurodegeneration27. Therefore, we used glial reporter, Repo-GAL4; UAS-GFP transgenic lines to determine if glial cells are the source of NOS positive signal in response to 1-octen-3-ol exposure. Although 1-octen-3-ol exposure intensified the GFP signal of the repo-positive glial cells in the adult brain but no co-localization between NOS signal and GFP labelled glial cells was observed (Fig. 3a,b). Since Drosophila responded to infection by gram negative bacteria and parasites via stimulation of NO in hemocytes8,9, we then sought to investigate if hemocytes were involved in our observed activation of NO. We extracted hemocytes from control and exposed 3rd instar larvae of He-GAL4; UAS-eGFP and performed co-immunostaining for NOS and larval hemocytes using universal anti-NOS and anti-H2 antibodies. We detected co-immunostaining of NOS and H2 with GFP-labeled larval hemocytes, thus confirming that larval hemocytes were the source of NOS (Fig. 3 c,d,e,f). There was also increase in the expression of NOS in the exposed hemocytes (Fig. 3g).
1-octen-3-ol triggers increased induction of NOS in the exposed hemocytes but not in glial cells.
(a,b) The GFP labelled repo-positive glial cells (repo-GAL4; UAS-eGFP) failed to show any co-localization with NOS positive cells although exposure of 1-octen-3-ol intensified the GFP signal of repo-positive glial cells (arrowheads). (c,d) The hemocytes from the 3rd instar stage larvae of He-GAL4; UAS-eGFP upon exposure to 1-octen-3-ol for 1 hr demonstrated stimulation of NO by hemocytes which co-localize with H2 specific antibodies where the insets demonstrate the morphology and size of the co-localized cells in control and exposed brain (e,f). (g) The mean fluorescence pixel intensity (MFPI) of NOS was elevated in hemocytes exposed to 1-octen-3-ol. (n = 100–150 hemocytes;** = P < 0.01) and represents a significant difference between control and octen-3-ol exposed larvae.
Our earlier studies showed that 1-octen-3-ol exposure causes neurotoxicity in adult Drosophila brain19,21. In order to determine if the activation of NOS was via adult hemocytes in response to 1-octen-3-ol induced toxicity, we performed co-immunostaining to detect the expression of NOS and anti-P1 (plasmatocyte-specific) antibodies in adult Drosophila brain in control (unexposed) and exposed (0.5 ppm for 6 hr) adult flies. Unexposed control adult brain showed few discrete NOS positive cells, while exposed adult brain showed an increased number of NOS positive cells that exhibited rounded to amoeboid cell shape (ranging from 0.5 μm to 2 μm in size) that co-localized with anti-P1 positive cells (Fig. 4 a,b,c,d). Since fly-hemocytes are considered morphologically and functionally similar to mammalian microglia, the co-localization of expression of NOS and P1 positive plasmatocytes in adult brain, along with an increase in the expression of NOS and P1 positive cells in the exposed brains, indicate stimulation of a microglial-like response against 1-octen-3-ol in adult brain (Fig. 4 e,f).
Induction of NOS in adult brain by 1-octen-3-ol.
(a) Few NOS positive cells were seen in control adult brains (b) Increased NOS positive cells expressed in adult brain exposed to 1-octen-3-ol for 6 hr which co-localized with adult P1 positive cells. The insets demonstrate the morphology and size of the colocalized cells (c,d). (e) The increase in MFPI for NOS and P1 labeled hemocytes in the exposed adult brains and was expressed as a percentage of that of control adult brains. (f) The Pearson co-relation coefficient for control and exposed adult brains for NOS and adult hemocytes showing the degree of co-localization between NOS positive cells and adult hemocytes (n = 10–12).
In rats, exposure of volatile industrial compounds is associated with up regulation of NO and other inflammatory markers in airway linings5. Upon exposure to 0.5 ppm of fungal VOC, 1-octen-3-ol for 6 hr, induction of NOS was seen in primary and secondary branches of larval tracheal linings where the source of NOS was confirmed as larval hemocytes (Fig 5 a,b,c,d). Furthermore, increased numbers of nuclei were detected in the distal branches of tracheal linings of exposed larvae indicating that exposure to 1-octen-3-ol possibly led to remodeling of the epithelial lining (Fig 5 e,f). Similar changes in the Drosophila tracheal lining have been observed in flies challenged with gram negative bacteria28 and such remodeling is a common pathological feature of asthma29.
1-octen-3-ol induces NOS expression in larval tracheal tissues and remodels tracheal epithelial linings.
(a,b) The increased expression of NOS was seen in the epithelial linings of exposed tracheal primary and secondary branches as compared to that of control larvae. (c) The increase in NOS intensity as assessed with mean fluorescence intensity for NOS was detected in 1-octen-3-ol exposed larval tracheal linings (n = 20–25) (d,e) The btl>GFP stained with anti-H2 (hemocytes) and anti-NOS antibodies display co-localization (shown with arrows) between hemocytes, NOS and tracheal epithelial lining in exposed larvae but tracheal linings of control larvae fail to show any measurable co-localization. (n = 10–14) (f,g) The increased numbers of DAPI-positive nuclei (shown with arrows) were detected in the distal branches of tracheal linings of exposed larvae but not in control tracheal linings (n = 8–10).
Discussion
Exposure to environmental agents is a major risk factor for various pathological conditions30. Although industrial chemicals have received the most intense research focus, epidemiological studies have correlated the presence of fungi and their metabolic products with adverse health consequences in indoor environments11,12. Most of the research on mold has focused on mycotoxins31 while fungal volatile organic compounds (VOCs) have received less attention13,14. These VOCs are low-molecular-weight compounds found in the gaseous state under normal atmospheric temperature and pressure, that are emitted by growing molds as a mixture of aldehydes, alcohols, esters, ethers, terpenoids and other compounds14. The C-8 compound, 1-octen-3-ol, commonly called “mushroom alcohol”, is a major VOC produced by fungi and is also one of the major fungal VOCs emitted by molds commonly found in moldy and water damaged buildings15,16,17. Our previous reports have demonstrated that exposure of flies to 1-octen-3-ol causes neurotoxicity by means of a selective loss of dopaminergic neurons19,21 and stimulates the caspase-3 dependent apoptotic signaling pathway20. Furthermore, 1-octen-3-ol and its enantiomers display cytotoxicity to human embryonic stem cellss22.
Nitric oxide (NO), a fundamental signaling agent, regulates various cellular functions and serves as a potent mediator of inflammation and cellular damage/cytotoxicity1,2. In a Scandinavian study, using human volunteers, 2 hr exposure to 1.9 ppm of 1-octen-3-ol induced activation of the inflammatory markers; eosinophil cationic protein, myeloperoxidase, lysozyme and albumin in nasal secretions23. The NO signaling pathway is well-conserved among different species, including Drosophila. The enzyme nitric oxide synthase (NOS) mediates the oxidation of L-arginine into citrulline and NO in the presence of NADPH and other cofactors32. Drosophila possesses a single NOS gene (dNOS) that shares 47% sequence similarity to mammalian neuronal NOS33,34. dNOS activity is reported in fly brains, larval tracheal linings and other tissues35,36. Like mammalian NOS, dNOS regulates diverse biological processes including host immune response37,38,39 Up-regulation of other components of the Drosophila immune response has been reported in fly models of neurodegenerative diseases27. Moreover, in a fashion similar to mammals, Drosophila airway linings express NOS, one of the known asthma susceptibility genes40.
In this report, we used Drosophila to investigate if exposure of fungal VOC, 1-octen-3-ol induces an inflammatory response via activation nitric oxide. Foley and O'Farrell8 and Carton et al.9 found that injections of NOS inhibitor, L-NAME into the Drosophila body cavity prior to infection with gram-negative bacteria and parasites decreased survival of flies, thereby suggesting that NO functions as an important component of the host immune response that counteract the cytotoxicity associated with infectious agents. Thus, NO seems to be a protective mechanism in responding to microbial infections. However, inflammation is a Janus-faced mechanism. It is well known that many of the volatile industrial solvents, environmental chemicals/pollutants and xenobiotics also induce NOS-mediated inflammatory response in different organ systems including brain and airway linings1,4,5,41. In our study, exposure to the industrial solvent, toluene and the fungal VOC, 1-octen-3-ol, led to an increase in the nitrite levels in the head, body and whole fly extracts while another fungal VOC, 1-butanol, failed to alter nitrite levels. Exposure to toluene is known to induce activation of NO/cGMP in mammalian models and in rat synaptosomes and bronchoalveolar lavage42,43,44. In our study, the pharmacological inactivation of NOS via inhibitors of NOS, L-NAME and minocycline also lead to improvement in 1-octen-3-ol mediated truncation of survival span. In addition, upon exposure to 1-octen-3-ol, the heterozygous mutant strain for NOS survived 3 days longer than the wild type flies. The heterozygous NOS mutant flies survival was comparable to that of wild type flies under unexposed conditions (data not shown). The immunostaining of adult brains with anti-peroxynitrite antibodies showed the increase in peroxynitrite levels in the 1-octen-3-ol exposed brains indicating that 1-octen-3-ol mediated truncation of survival span is due to production of peroxynitrite as a result of possible interaction of NO with reactive oxygen species generated upon exposure of 1-octen-3-ol19. In summary, our data collectively suggest that 1-octen-3-ol mediated toxicity is at least partly mediated via activation of NOS and peroxynitrite in Drosophila.
In mammals, glial cells are non-neuronal cells constituting about 50% of the volume of the CNS and play a key role in support and nutrition to CNS neurons, formation of myelin sheaths and in signal transmission in the CNS. The Drosophila nervous system possesses counterparts for mammalian glia. Further, the glial cell specific transcription factor, repo (reverse polarity), is expressed in nearly all fly glial cells26. Recently, glial cells were implicated in the induction of immune response and subsequent neurodegeneration in the Drosophila model of the human disease, Ataxia-Telangiectasia27,45. In contrast, in our study, failure of co-localization between the 1-octen-3-ol induced NOS positive cells and GFP labelled repo-positive glial cells demonstrates that glial cells are not the source of the NOS signal we detected.
Drosophila hemocytes are immune surveillance cells and form crucial components of the cellular immune response46. There are mainly three different types of Drosophila hemocytes: plasmatocyte, lamellocytes and crystal cells. The plasmatocytes comprise 90–95% of all mature hemocytes which appear as small rounded cells capable of altering their sizes in response to foreign agents and resemble the mammalian monocyte/macrophage lineage10,46. The plasmatocytes are the only class of hemocytes known to be present in adult Drosophila47,48 where hemese-specific (H2) and plasmatocyte-specific (P1) antibodies have been reported49,50. Infection with gram-negative bacteria and parasites induce elevated expression of NOS in hemocytes of Drosophila and other invertebrates8,9,51. In mammalian models for neurodegeneration and xenobiotic toxicity, there is an up-regulation of NOS in macrophages/microglia in response to MPTP, paraquat and other xenobiotics41,52,53.
Using 3rd instar larvae of transgenic line for hemese, a blood-cell-specific transmembrane protein, He-GAL4; UAS-GFP54, we performed co-immunostaining with anti-hemese monoclonal (H2)50 and anti-NOS antibodies8 and confirmed that larval hemocytes indeed express NOS. The quantification of mean fluorescent intensity of NOS confirmed the elevated expression level NOS in exposed hemocytes as compared to the control hemocytes. Similarly, we detected NOS positive microglia-like cell population in adult Drosophila brain by performing the co-immunostaining with anti-NOS8 and anti-plasmatocytes (P1)50 antibodies in the control and 1-octen-3-ol (0.5 ppm for 6 hr) exposed adult brain. Interestingly, few discrete NOS positive plasmatocytes were detected in the control brains while exposed adult brain showed an increased number of NOS positive cells that exhibited rounded to amoeboid cell shape, ranging from 0.5 μm to 2 μm in size that co-localized with plasmatocytes. The quantification of mean fluorescent intensity of NOS and plasmatocytes confirmed the elevated expression level of NOS and hemese in exposed hemocytes as compared to the control hemocytes. Increased expression of NOS positive plasmatocytes in the 1-octen-3-ol exposed adult brain is a provocative finding and reports for the first time presence of increased number of microglial cell-like population in the adult Drosophila capable of expressing NOS and acting possibly as cytotoxic agent due to dysregulated expression of plasmatocytes in response to environmental fungal derived chemical, 1-octen-3-ol.
Several mammalian studies have shown that the overactivation and dysregulation of microglia results in amplification of the neuronal damage induced by foreign stimuli/toxins, thereby enhancing neurotoxicity and neurodegeneration55. Our study suggests that exposure to a low dose of 1-octen-3-ol induces similar neuroinflammatory responses in adult Drosophila brain and may provide a mechanistic basis for the reported neurological symptoms associated with exposure to mold56,57,58.
Multiple epidemiological studies have found a positive association of mold and dampness with multiple allergic and respiratory effects11,12,59. Few groups have advocated Drosophila models for understanding the genetic components of asthma and other inflammatory types of lung disorders37,40. The Drosophila larval airway system shows striking similarities with that of mammals in terms of its physiology, architecture and reaction towards foreign pathogens along with expression of NOS in larval airway linings28. In our study, we detected the up-regulation of NOS in the airway linings of the transgenic strains of 3rd instar btl-GAL4::UAS-GFP larvae exposed to 0.5 ppm of 1-octen-3-ol for 6 hr. Moreover, increased numbers of nuclei were detected in the distal branches of tracheal linings of exposed larvae indicating that exposure to 1-octen-3-ol possibly led to remodeling of the epithelial lining. Wagner et al.60 also reported the similar changes in the Drosophila tracheal lining challenged with gram negative bacteria. Our study shows that exposure of 1-octen-3-ol induces increase expression of NOS as well as airway remodeling, both of which are the common pathological features of asthma and inflammatory lung diseases29 and may provide cues for the reported positive association between the mold exposure and respiratory symptoms16,61.
In summary, our study demonstrates that a common fungal VOC associated with mold-contaminated damp indoor spaces stimulates a NO mediated inflammatory response in Drosophila nervous and respiratory tissues. This work opens a new avenue for gaining a mechanistic understanding of the human health effects by mold-emitted metabolites and also demonstrates the utility of this in vivo Drosophila model to complement existing cell culture systems for studying VOC toxicity.
Methods
Drosophila strains
Unless otherwise stated, all experiments were performed using 48 hr post-eclosion, wild type y1, w1118, a yellow body and white-eyed strain. The heterozygous mutant strain for NOS, NOS24283 and y w; repo-GAL4 and UAS-eGFP transgenic lines were obtained from Bloomington Stock Center. All fly stocks were reared on Ward's Instant Drosophila medium (blue) and all experiments were performed at room temperature. The transgenic lines for hemese, hemocyte-specific transmembrane protein, y w; He-Gal-4; UAS-eGFP (chromosome III), y w, btl-Gal4; UAS-btl::GFP which expresses a Btl::GFP fusion protein in btl-expressing tissues, including trachea were gifts from Dr. Janis O'Donnell (University of Alabama) and have been described previously62,63.
Chemicals and exposure conditions
1-octen-3-ol (99%), NG-Nitro-D-arginine-methyl ester hydrochloride (D-NAME), NG-Nitro-L-arginine-methyl ester hydrochloride (L-NAME), Minocycline (MC), toluene (≥99.9%), 1-butanol (99%) and Modified Griess reagent were purchased from Sigma, USA while NADPH and Nitroblue tetrazolium were supplied by Calbiochem and Alfa Aesar, USA respectively. The exposure protocol for 1-octen-3-ol was performed as described in Inamdar et al20 using 0.5 ppm (volume: volume) of undiluted liquid 1-octen-3-ol for all exposure experiments while toluene and 1-butanol were exposed at the concentration of 2.8 ppm (volume: volume) as described in Inamdar et al18. The NO synthase inhibitors NG-nitro-L-arginine methyl ester hydrochloride (L-NAME), D-NAME and Minocycline were added at 2 mM concentration in agar food (2% agar and 5% sucrose).
Viability study
The effect of 1-octen-3-ol on survival was assessed by exposing five replicates of 10 adult flies at 0.5 ppm with or without the simultaneous presence of NOS inhibitors, D-NAME, L-NAME or minocycline. Survival was monitored by counting the number of the dead flies every day until all flies were dead; the percent of total dead flies at each time period was calculated for each replicate.
Quantitative reverse transcriptase PCR (qRT-PCR)
qRT-PCR analysis for NOS was performed following the protocol of Brown et al64 using the primers described therein. Briefly, upon 6 hr exposure of adult flies to 1-octen-3-ol, total RNA was extracted from 8 adults using TRizol (Invitrogen). The cDNAs were obtained using a high capacity cDNA reverse transcriptase kit (Applied Biosystems). The qRT-PCR was performed using STBR Green 1 (Invitrogen) on StepOnePlus™ Real-Time PCR System (Applied Biosystems) using the cycle conditioned described earlier55. The primers for NOS and internal controls, Rp 49 were NOS: F: 5′-AGCAACAGAAGGCACAGACA-3′and R: 5′-AGGCGATGCTGTGGAGATAC-3′; Rp49:- F: 5′-TCCTACCAGCTTCAAGATGAC-3′and R: CACGTTGTGCACCAGGAACT-3′. The data were recorded from three replicates from two independent experiments. The cycle threshold (Ct) value was determined and the expression levels of NOS in control and exposed extracts was calculated relative to Rp49 using the ΔΔCt method. The relative gene expression for NOS in the exposed flies was presented as a fold upregulation in the NOS expression of control flies.
Nitric oxide synthase (Nitrite) assay
Male adult flies at 48 hr post-eclosion were exposed for 6 hr to 0.5 ppm 1-octen-3-ol, while 2.8 ppm toluene or 1-butanol. Head, body and whole fly-extracts for control and each exposure group were prepared by homogenizing in buffer (0.1 M phosphate buffer at pH 7.4, 0.015 M potassium chloride) followed by centrifugation for 10 min at 10,000 g at 4°C. The supernatants were mixed in a 1:1 proportion with freshly prepared Modified Griess reagent (Sigma) and incubated for 15 min. Nitrite levels were measured spectrophotometrically at 595 nm, with concentrations of nitrite calculated against a silver nitrite-derived standard curve. Data were presented as a concentration of nitrite in mM for 50 fly-heads, 10 fly-bodies or 8 whole-flies.
Nitric oxide synthase histochemical assay
The induction of NOS in the adult brain was detected with the NADPH diaphorase assay described earlier8. The control and experimental (exposed to 1-octen-3-ol for 6 hr) adult brains were dissected from adult flies in PBS and kept in a fixing buffer (4% paraformaldehyde, 100 mM PIPES at pH 7.4, 2 mM MgSO4, 1 mM EGTA) for 30 min at room temperature. The tissues were then washed in PBS and incubated in staining solution (1 mM NADPH, 0.2 mM nitrobluetetrazolium, 100 mM Tris at pH 7.2, 0.2% Triton X-100) for 30 min at room temperature. The tissues were mounted in hydromount and imaged using an Olympus light microscope, BH-2 attached with camera.
Immunohistochemistry for hemocytes and larval tracheal linings
The protocol for staining hemocytes was adapted from previous reports8,50,62. After exposure of the larvae of a transgenic line for hemese (y w; He-Gal-4; UAS-eGFP) to 0.5 ppm 1-octen-3-ol for 6 hr, the control and exposed hemocytes were expressed from larvae on Superfrost Plus Gold microscope slides (Fisher Scientific, USA). The control and exposed tracheal linings from 3rd instar larvae that had been exposed to 1-octen-3-ol for 6 hr were dissected from transgenic line for breathless (y w; btl-Gal4; UAS-btl::GFP). The hemocytes were fixed with 2% paraformaldehyde (PF) and 0.01% phenylthiourea (PTU) while tracheal tissues were fixed with only 2% PF. The tissues were washed with PBS thrice and then with 0.1% (v/v) Triton X- 100, 0.2% (w/v) bovine serum albumin in PBS (PBT). The blocking agent (5% Fetal bovine serum [FBS] in PBT) treatment for 30 min was followed with overnight incubation at 4°C in the following primary antibodies: anti-rabbit universal NOS (1:100) (Abcam, Cat. No. ab3142); Chicken anti-GFP (1:1000); and anti-mouse H2, hemese antigen specific monoclonal antibodies (1:50). The secondary antibodies were Cy-3-conjugated goat anti-rabbit IgG, FITC-conjugated rabbit anti-chicken and Mouse IgG Atto 633. DAPI staining (Santa Cruz) was used at 1:1000 dilutions in PBS and added to the final wash step before mounting the hemocytes with SlowFade Gold antifade (Invitrogen). For the experiments with only DAPI staining, tissues were fixed with PF and then stained with DAPI.
Immunostaining of adult brains with anti-NOS and anti-plasmatocyte antibodies
The staining was performed using the protocol published by Kurucz et al50 with slight modifications. Upon exposure to 0.5 ppm 1-octen-3-ol to wild type flies, the exposed and control (un-exposed) adult brains were dissected in PBS and fixed in acetone for 3 min. The brains were rehydrated with PBST and then blocked with 5% FBS in PBT for 30 min. The overnight incubation at 4°C with primary antibodies: anti-rabbit universal NOS (1:100) (Abcam, Cat. No. ab3142) and anti-mouse P1 antibodies, plasmatocyte specific monoclonal antibodies against NimC1 antigen expressed in plasmatocytes of adult flies (1:50) was followed by incubation with secondary antibodies as Cy-3-conjugated goat anti-rabbit IgG (1:1000) and mouse IgG Atto 633(1:500). Similar protocol was used for repo-GAL4; UAS-eGFP flies to determine the co-localization between glial and NOS-positive cells using anti-GFP (Abcam, Cat. No. ab291) and anti-NOS antibodies in the dissected adult brains from unexposed and exposed groups.
Immunostaining of adult brains with anti-peroxynitrite antibodies
Upon exposure to 0.5 ppm 1-octen-3-ol to wild type flies, the exposed and control (un-exposed) adult brains were dissected in PBS and fixed in 2% PF for 30 min. The dissected brains were then blocked with 10% FBS in PBT overnight at 4°C. The overnight incubation at 4°C with primary antibodies: anti-rabbit nitro-tyrosine (1:100) (Life Technologies, Cat. No. A21285) was followed by incubation with secondary antibodies as Cy-3-conjugated goat anti-rabbit IgG before performing confocal microscopy.
Confocal microscopy
The images were captured with a Zeiss LSM 710 confocal microscope. Appropriate control experiments were performed to avoid co-localization due to auto-fluorescence or bleed-through of signals. The acquisition parameters were kept similar for control and experimental samples for each tissue. Using the frame mode, each tissue was scanned sequentially to obtain the final image from 12-15 Z-sections. The co-localization and mean intensity were evaluated using ZEN 2010 image processing software. The quantification of mean pixel intensity and co-localization was performed using intensity and co-localization modules in Zen 2010 image processing software where Pearson coefficient was used to evaluate the degree of co-localization by taking into consideration the weighted mean intensity.
Statistical analysis
Statistical significance was determined by Student's one-tailed t-test or one way ANOVA with Dunnett's/Bonferroni post-test using GraphPad Prism software (CA, USA). The relevant details of the statistical analysis are described in the Figure legends.
References
Coleman, J. W. Nitric oxide in immunity and inflammation. Int. Immunopharmacol. 1, 1397–1406 (2001).
Pacher, P., Beckman, J. S. & Liaudet, L. Nitric oxide and peroxynitrite in health and disease. Physiol. Rev. 87, 315–424 (2007).
Laskin, D. L., Sunil, V. R., Gardner, C. R. & Laskin, J. D. Macrophages and tissue injury: agents of defense or destruction? Annu. Rev. Pharmacol. Toxicol. 51, 267–288 (2011).
Meggs, W. J. Neurogenic inflammation and sensitivity to environmental chemicals. Environ. Health Perspect. 101, 234–238 (1993).
Wang, F., Li, C., Liu, W. & Jin, Y. Effect of exposure to volatile organic compounds (VOCs) on airway inflammatory response in mice. J. Toxicol. Sci. 37, 739–48 (2012).
Wang, F., Li, C., Liu, W., Jin, Y. & Guo, L. Effects of subchronic exposure to low-dose volatile organic compounds on lung inflammation in mice. Environ. Toxicol. 10.1002/tox.21844 (2013).
Galloway, T. S. & Depledge, M. H. Immunotoxicity in invertebrates: measurement and ecotoxicological relevance. Ecotoxicology 10, 5–23 (2001).
Foley, E. & O'Farrell, P. H. Nitric oxide contributes to induction of innate immune responses to gram-negative bacteria in Drosophila. Genes Dev. 17, 115–125 (2003).
Carton, Y., Frey, F. & Nappi, A. J. Parasite-induced changes in nitric oxide levels in Drosophilaparamelanica. J. Parasitol. 95, 1134–1141 (2009).
Lemaitre, B. & Hoffman, J. The host defense of Drosophila melanogaster. Annu. Rev. Immuno. 25, 697–743 (2007).
IOM. Institute of Medicine of the National Academies. Damp indoor spaces and health. Washington, DC: Institute of Medicine of the National Academies, The National Academies Press (2004).
WHO. World Health Organization (WHO). Guidelines for indoor air quality: dampness and mould. Germany: Druckartner Moser (2009).
Mølhave, L. [Volatile organic compounds and the sick building syndrome]. Environmental toxicants: human exposures and their health effects. [Lipmann, M. (ed.)] [241–56] (Wiley-Interscience, New York, 2009).
Korpi, A., Jarnberg, J. & Pasanen, A. L. Microbial volatile organic compounds. Crit. Rev. Toxicol. 39, 139–193 (2009).
Strom, G., West, J., Wessen, B. & Palmgren, U. [Quantitative analysis of microbial volatiles in damp Swedish houses]. Health implications of fungi in indoor environments. Air quality monographs. [Samson, R. A., Flannigan, B., Flannigan, M. E., Verhoeff, A. P., Adan, O. C. G. & Hoekstra, E. S. (ed.)] [291–305] (Elsevier Science, Amsterdam, 1994).
Kim, J. L. et al. Indoor molds, bacteria, microbial volatile organic compounds and plasticizers in schools–associations with asthma and respiratory symptoms in pupils. Indoor Air 17, 153–163 (2007).
Schleibinger, H., Laussmann, D., Bornehag, C. G., Eis, D. & Rueden, H. Microbial volatile organic compounds in the air of moldy and mold-free indoor environments. Indoor Air 18, 113–124 (2008).
Inamdar, A. A., Zaman, T., Morath, S. U., Pu, D. C. & Bennett, J. W. Drosophila melanogaster as a model to characterize fungal volatile organic compounds. Environ Toxicol. 10.1002/tox.21825 (2012).
Inamdar, A. A. Masurekar, P. & Bennett, J. W. Neurotoxicity of fungal volatile organic compounds in Drosophilamelanogaster. Toxicol Sci. 117, 418–426 (2010).
Inamdar, A. A., Masurekar, P., Hossain, M., Richardson, J. R. & Bennett, J. W. Signaling Pathways Involved in 1-Octen-3-ol-Mediated Neurotoxicity in Drosophilamelanogaster: Implication in Parkinson's Disease. Neurotox. Res. 10.1007/s12640-013-9418-z (2013).
Inamdar, A. A. et al. Fungal-derived semiochemical 1-octen-3-ol disrupts dopamine packaging and causes neurodegeneration. Proc Natl Acad Sci U S A. 110, 19561–19566 (2013).
Inamdar, A. A., Moore, J. C., Cohen, R. I. & Bennett, J. W. A model to evaluate the cytotoxicity of the fungal volatile organic compound 1-octen-3-ol in human embryonic stem cells. Mycopathologia 173, 13–20 (2012).
Wålinder, R., Ernstgård, L., Norbäck, D., Wieslander, G. & Johanson, G. Acute effects of 1-octen-3-ol, a microbial volatile organic compound (MVOC)-an experimental study. Toxicol Lett. 181, 141–147 (2008).
Green, L. C. et al. Analysis of nitrate, nitrite and [15N] nitrate in biological fluids. Anal. Biochem. 126, 131–138 (1982).
Persecă, O., Orasăn, R., Cătoi, C., Taulescu, M. A. & Pârvu, A. E. Nitro-oxidative stress in amice model of toluene diisocyanate -induced asthma. Bulletin UASMV, Vet. Med. 69, 153–161 (2012).
Freeman, M. R. & Doherty, J. Glial cell biology in Drosophila and vertebrates. Trends. Neurosci. 29, 82–90 (2006).
Petersen, A. J. & Wassarman, D. A. Drosophila innate immune response pathways moonlight in neurodegeneration. Fly (Austin) 6, 169–172 (2012).
Wagner, C., Isermann, K. & Roeder, T. Infection induces a survival program and local remodeling in the airway epithelium of the fly. FASEB J. 23, 2045–2054 (2009).
Holgate, S. T. Pathogenesis of asthma. Clin. Exp. Allergy 38, 872–897 (2008).
Norman, R. E., Carpenter, D. O., Scott, J., Brune, M. N. & Sly, P. D. Environmental exposures: an underrecognized contribution to noncommunicable diseases. Rev. Environ. Health 28, 59–65 (2008).
Robbins, C. A., Swenson, L. J., Nealley, M. L., Kelman, B. J. & Gots, R. E. Health effects of mycotoxins in indoor air: a critical review. Appl. Occup. Environ. Hyg. 15, 773–784 (2000).
Mayer, B. Regulation of nitric oxide synthase and soluble guanylyl cyclase. Cell Biochem. Funct. 12, 167–177 (1994).
Regulski, M. & Tully, T. Molecular and biochemical characterization of dNOS: a Drosophila Ca2+/calmodulin-dependent nitric oxide synthase. Proc Natl Acad Sci U S A 92, 9072–9076 (1995).
Stasiv, Y., Regulski, M., Kuzin, B., Tully, T. & Enikolopov, G. The Drosophila nitric-oxide synthase gene (dNOS) encodes a family of proteins that can modulate NOS activity by acting as dominant negative regulators. J. Biol. Chem. 276, 42241–42251 (2001).
Müller, U. Ca2+/calmodulin-dependent nitric oxide synthase in Apismellifera and Drosophilamelanogaster. Eur. J. Neurosci. 6, 1362–1370 (1994).
Davies, S. Nitric oxide signalling in insects. Insect Biochem. Mol. Biol. 30, 1123–1138 (2000).
Roeder, T., Isermann, K. & Kabesch, M. Drosophila in asthma research. Am. J. Respir. Crit. Care Med. 179, 979–983 (2009).
Mukherjee, T., Kim, W. S., Mandal, L. & Banerjee, U. Interaction between Notch and Hif-alpha in development and survival of Drosophila blood cells. Science 332, 1210–1213 (2011).
Nappi, A. J., Vass, E., Frey, F. & Carton, Y. Nitric oxide involvement in Drosophila immunity. Nitric oxide 4, 423–430 (2000).
Roeder, T., Isermann, K., Kallsen, K., Uliczka, K. & Wagner, C. A. Drosophila asthma model - what the fly tells us about inflammatory diseases of the lung. Adv. Exp. Med. Bio. 710, 37–47 (2012).
Laskin, D. L. Macrophages and inflammatory mediators in chemical toxicity: a battle of forces. Chem. Res. Toxicol. 22, 1376–1385 (2009).
Chan, M. H., Chien, T. H., Lee, P. Y. & Chen, H. H. Involvement of NO/cGMP pathway in toluene-induced locomotor hyperactivity in female rats. Psychopharmacology (Berl.) 176, 435–439 (2004).
Myhre, O. & Fonnum, F. The effect of aliphatic, naphthenic and aromatic hydrocarbons on production of reactive oxygen species and reactive nitrogen species in rat brain synaptosome fraction: the involvement of calcium, nitric oxide synthase, mitochondria and phospholipase A. Biochem. Pharmacol. 62, 119–128 (2001).
Huffman, L. J. et al. Inhalation of toluene diisocyanate is associated with increased production of nitric oxide by rat bronchoalveolar lavage cells. Toxicol. Appl. Pharmacol. 145, 61–70 (1997).
Petersen, A. J., Rimkus, S. A. & Wassarman, D. A. ATM kinase inhibition in glial cells activates the innate immune response and causes neurodegeneration in Drosophila. Proc Natl Acad Sci U S A 109, E656–664 (2012).
Williams, M. J. Drosophila Hemopoiesis and Cellular Immunity. J. Immunol. 178, 4711–4716 (2007).
Lanot, R., Zachary, D., Holder, F. & Meister, M. Postembryonic hematopoiesis in Drosophila. Dev. Biol. 230, 243–257 (2001).
Holz, A., Bossinger, B., Strasser, T., Janning, W. & Klapper, R. The two origins of hemocytes in Drosophila. Development 130, 4955–4962 (2003).
Asha, H. et al. Analysis of ras-induced overproliferation in Drosophila hemocytes. Genetics 163, 203–215 (2003).
Kurucz, E. et al. Definition of Drosophila hemocyte subsets by cell-type specific antigens. Acta. Biol. Hung. 58, 95–111 (2007).
Krishnan, N., Hyrsl, P. & Simek, V. Nitric oxide production by hemocytes of larva and pharate prepupa of Galleria mellonella in response to bacterial lipopolysaccharide: Cytoprotective or cytotoxic? Comp. Biochem. Physiol. C. Toxicol. Pharmacol. 142, 103–110 (2006).
Day, B. J., Patel, M., Calavetta, L., Chang, L. Y. & Stamler, J. S. A mechanism of paraquat toxicity involving nitric oxide synthase. Proc Natl Acad Sci USA 96, 12760–12765 (1999).
Dehmer, T., Lindenau, J., Haid, S., Dichgans, J. & Schulz, J. B. Deficiency of inducible nitric oxide synthase protects against MPTP toxicity in vivo. J. Neurochem. 74, 2213–2216 (2000).
Kurucz, E. et al. Hemese, a hemocyte-specific transmembrane protein affects the cellular immune response in Drosophila. Proc Natl Acad Sci USA 100, 2622–2627 (2001).
Block, M. L., Zecca, L. & Hong, J. S. Microglia-mediated neurotoxicity: uncovering the molecular mechanisms. Nat. Rev, Neurosci. 8, 57–69 (2007).
Kilburn, K. H. Role of molds and mycotoxins in being sick in buildings: neurobehavioral and pulmonary impairment. Adv. Appl. Microbiol. 55, 339–359 (2004).
Kilburn, K. H. Neurobehavioral and pulmonary impairment in 105 adults with indoor exposure to molds compared to 100 exposed to chemicals. Toxicol. Ind. Health 25, 681–692 (2009).
Empting, L. D. Neurologic and neuropsychiatric syndrome features of mold and mycotoxin exposure. Toxicol. Ind. Health 25, 577–581 (2009).
Mendell, M. J., Mirer, A. G., Cheung, K., Tong, M. & Douwes, J. Respiratory and Allergic Health Effects of Dampness, Mold and Dampness-Related Agents: A Review of the Epidemiologic Evidence. Environ Health Perspect. 119, 748–756 (2011).
Wagner, C., Isermann, K., Fehrenbach, H. & Roeder, T. Molecular architecture of the fruit fly's airway epithelial immune system. BMC Genomics 9, 446–458 (2008).
Jaakkola, M. S. & Jaakkola, J. J. Indoor molds and asthma in adults. Adv. Appl. Microbiol. 55, 309–338 (2004).
Zettervall, C. J. et al. A directed screen for genes involved in Drosophila blood cell activation. Proc Natl Acad Sci U S A 10, 114192–114197 (2004).
Hsouna, A., Lawal, H. O., Izevbaye, I., Hsu, T. & O'Donnell, J. M. Drosophila dopamine synthesis pathway genes regulate tracheal morphogenesis. Dev. Biol. 308, 30–43 (2007).
Brown, A. E., Baumbach, J., Cook, P. E. & Ligoxygakis, P. Short-term starvation of immune deficient Drosophila improves survival to gram-negative bacterial infections. PLoS One 4, e4490 (2009).
Acknowledgements
This work was supported by the Rutgers University Reserve Funding to JWB. We are thankful to Richard Hung, Samantha Lee, Shannon Morath, Prakash Masurekar and Sally Padhi for helpful discussions concerning fungal volatile organic compounds and to Priya Sudheer for help with maintaining the Drosophila stocks. Special thanks to Dr. Ando Istvan (Biological Research Centre of Hungarian Academy of Sciences, Szeged) for providing hemocytes specific H2 and P1 antibodies.
Author information
Authors and Affiliations
Contributions
A.A.I. initiated and conducted the experiments. J.W.B. supervised the project. Both authors participated in the writing and editing of the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/3.0/
About this article
Cite this article
Inamdar, A., Bennett, J. A common fungal volatile organic compound induces a nitric oxide mediated inflammatory response in Drosophila melanogaster. Sci Rep 4, 3833 (2014). https://doi.org/10.1038/srep03833
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep03833
This article is cited by
-
Discovery, total syntheses and potent anti-inflammatory activity of pyrrolinone-fused benzoazepine alkaloids Asperazepanones A and B from Aspergillus candidus
Communications Chemistry (2022)
-
Toxicant-induced loss of tolerance for chemicals, foods, and drugs: assessing patterns of exposure behind a global phenomenon
Environmental Sciences Europe (2021)
-
Fungal volatile organic compounds and their role in ecosystems
Applied Microbiology and Biotechnology (2015)
-
Immune responses to airborne fungi and non-invasive airway diseases
Seminars in Immunopathology (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.