Abstract
Ozone is a strong oxidant that is globally used as disinfection agent for many purposes including indoor building air cleaning, during food preparation procedures and for control and killing of bacteria such as E. coli and S. aureus. However, it has been shown that effective ozone concentrations for controlling e.g., microbial growth need to be higher than 5 ppm, thereby exceeding the recommended U.S. EPA threshold more than 10 times. Consequently, real-time monitoring of such ozone concentration levels is essential. Here, we describe the first online gas sensing system combining a compact Fourier transform infrared (FTIR) spectrometer with a new generation of gas cells, a so-called substrate-integrated hollow waveguide (iHWG). The sensor was calibrated using an UV lamp for the controlled generation of ozone in synthetic air. A calibration function was established in the concentration range of 0.3–5.4 mmol m−3 enabling a calculated limit of detection (LOD) at 0.14 mmol m−3 (3.5 ppm) of ozone. Given the adaptability of the developed IR sensing device toward a series of relevant air pollutants and considering the potential for miniaturization e.g., in combination with tunable quantum cascade lasers in lieu of the FTIR spectrometer, a wide range of sensing and monitoring applications of beyond ozone analysis are anticipated.
Similar content being viewed by others
Introduction
During recent decades, ozone has evolved into the main disinfection agent at a global scale1. Due to its substantial oxidation potential (+ 2.07 V vs. standard hydrogen electrode) and facile generation, ozone is nowadays used in a wide variety of disinfection scenarios such eliminating indoor odors2, food preservation3, residential air cleaning4, treatment of carious lesions5 and for microbial control6.
The effect of ozone on the human health was initially reported as an irritant gas7. However, long-term exposure has been proven to potentially result in decrease of lung function and respiratory diseases such as asthma8,9. The Environmental Protection Agency of the United States (U.S. E.P.A) has therefore established an ozone threshold of 0.08 ppm for an exposure period of 8 hours and 0.09 ppm for an exposure period of 1 hour. Complementarily, the World Health Organization (WHO) has limited the ozone threshold at 0.06 ppm for an exposure period of 8 hours10.
However, the apparent disinfection power of ozone is limited at concentrations yielding such exposure limits7,11. Moreover, most commercial ozone generators produce substantially higher levels of ozone by nature of the generation process, i.e., via electrical discharge (corona) or by photoreaction with ultraviolet (UV) radiation12,13. Consequently, a series of studies have reported that useful ozone levels for disinfection readily exceed the regulated ozone concentrations by a factor of ten or more14,15.
As ozone is a highly reactive compound, indirect chemical methods are frequently used for its determination. However, secondary reactions and other oxidizing species may affect the accuracy of the obtained results16. Iodometry, based on the reaction between ozone and iodide forming the colored ion triiodide (I3−) is to date the standard method for the - indirect - determination of high levels of ozone in air17. Among other practical limitations for in-field usage, this method is not suitable for real-time analysis. As ozone is an excellent UV absorber, UV spectroscopy based and chemiluminscent analyzers have been considered the most sensitive techniques for online monitoring to date18,19. Nevertheless, other absorbing constituents in the UV wavelength range may cause false positive signals20; also, considerable costs and the rather bulky dimensions are major arguments against widespread field usage.
Even thought ozone has distinct absorption features at mid-infrared frequencies, the use of mid-infrared (MIR) sensing techniques for quantification in indoor and outdoor air has not been reported in literature. This may be attributed to the fact that until recently the bulkiness and cost of instrumentation along with the moderate sensitivity rendered this wavelength regime less suitable for environmental and especially for atmospheric analysis, which demands analyzing ozone concentrations at approximately 5–100 ppb. At these trace levels, to date only chemiluminesce, high-resolution FTIR and UV spectroscopy based methods are applicable21,22,23.
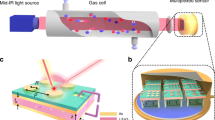
However, as mentioned above a series of applications requires the detection of ozone at elevated concentration levels. Serra et al. have evaluated the use of ozone to reduce molds in a cheese ripening room via the generation of 8 g h−1 15. 5 ppm of ozone was used during 60 minutes in order to disinfect vegetables3. For building disinfections purposes, 24 materials were exposed for 16 h to ozone concentrations of 1000 – 1200 ppm14. Particularly for these types of applications, real-time monitoring of ozone is essential. The approach presented herein takes advantage if the inherent molecular selectivity provided by mid-infrared vibrational spectroscopy using particularly compact and portable FTIR spectrometers combined with a new generation of miniaturized gas cells based on hollow waveguide technology.
A hollow waveguide (HWG) may generally be described as a light pipe made from dielectric materials or metals with a coaxial hollow core enabling radiation propagation by reflection at the inside wall24. If gaseous samples are injected into the hollow core, the HWG may simultaneously act as a miniaturized gas cell with a well-defined optical absorption path length, thereby enabling quantitative optical analysis25. Recent studies have already demonstrated the utility of HWGs coupled to FTIR spectroscopy operating in the MIR band (3–20 μm) as gas sensing system26,27,28,29,30,31. Very recently, a fundamentally new generation of hollow waveguides has been developed, so-called substrate-integrated hollow waveguides (iHWG). The most remarkable features are the compact dimensions of the waveguide substrate (75 × 50 × 12 mm; made from aluminum), the adaptable optical path length via integration of a meandered hollow waveguide channel at virtually any desired geometry into an otherwise planar substrate, the small volume of gas sample required for analysis and the short transient time of such small volumes (few hundred microliters) through the active transducer region facilitating real-time monitoring32.
As MIR sensing has not been explored as an analytical tool for the quantification and monitoring of ozone in gaseous samples, the present study demonstrate for the first time the capability of this new generation of iHWG-based sensing devices providing a compact, simply assembled and robust system as a versatile alternative to conventional analytical techniques.
Results
Reproducibility of ozone generation
A critical parameter in chemical analysis is the reliability of standards used for device calibration. As commercial ozone cylinders providing certified standards are not available, any ozone generation must be tested for reproducibility. Here, the precision was evaluated using the peak area at 1055 cm−1 during 25 independent measurements of 4.10 mmol m−3 of ozone. The achieved intra-day relative standard deviation (RSD) was 1.41%. Inter-day repeatability was determined within three consecutive days and was better than 2.26%.
Analytical figures-of-merit
For quantitative purposes, a calibration function was established based on the evaluation of the peak area at 1055 cm−1 versus the ozone concentration. For each concentration, the mean value of five replicate measurements was calculated. The goodness of the fit was determined at r2 > 0.99 in the concentration range of 0.36 to 5.40 mmol m−3 of ozone following A = 0.234 [O3]. A represents the peak area at 1055 cm−1 and [O3] is the ozone concentration in mmol m−3 (Figure 1a) The calculated limit of detection (LOD) was considered to be three times the standard deviation of the blank signal and was determined at 0.14 mmol m−3 (3.49 ppm). The validation results are summarized in Table 1.
(a) Calibration curve of ozone determination, (b) Continuous online monitoring of ozone concentrations in real-time and (c) contour plot (top view) of the ozone plume migrating through the iHWG via 2D IR-spectra of the spectral region comprising the relevant ozone IR signatures; the color bar encodes the absorption intensity (i.e., the ozone concentration) from low (blue) to high (red).
Real-time monitoring of ozone
The capabilities of the developed iHWG-FTIR sensor for online monitoring was evaluated by continuously monitoring the sample flow provided from the ozone generator. For this purpose, the UV lamp was switched off at intervals of 1.1 min, in order to instantaneously reduce the ozone concentration and switched back on after 1.1 min, again producing ozone. The maximum signal value was obtained once the dead volume of the device was completely filled with ozone, which occurred within 55 seconds. The obtained results are shown in Figure 1b and 1c. Excellent reproducibility of the peak height and of the transient signal for different ozone concentrations was obtained.
Discussion
Within the mid-infrared band, the most pronounced band of ozone is located at 1055 cm−1 and is associated with the ν3 asymmetric stretching vibration33. Furthermore, a Fermi resonance band at 2100 cm−1, associated with the ν1 + ν3 combination band, is evident, yet though with at least 4-times less intensity. Hence, the peak at 1055 cm−1 is most suitable for quantitative data evaluation. Figure 2 shows an exemplary IR spectrum obtained for 5.40 mmol m−3 of ozone using the gold-coated iHWG coupled with the FTIR spectrometer. A typical signal-to-noise ratio (SNR) for such measurements was determined at 159.
An online real-time gas sensing system for the quantitative determination of high levels of ozone in air was developed using for the first time substrate-integrated hollow waveguides (iHWG) coupled to an FTIR spectrometer operating in the mid-infrared spectral range. Next to a rapid dynamic response toward concentration changes of 55 s, the sensing device provides an LOD of 0.14 mmol m−3 for ozone, with a significant dynamic range suitable for threshold sensing at U.S. EPA and WHO demanded levels. In future, a significant size reduction of the sensing device is anticipated by replacing the FTIR spectrometer with a suitable tunable quantum cascade laser (QCL)24,34,35,36. Moreover, the sensitivity may be further improved by extending the optical path length via variation of the meandered waveguide channel geometry integrated into the iHWG, as previously demonstrated32.
Methods
Ozone generation
Synthetic air and nitrogen were provided by MTI Industriegase AG (Neu-Ulm, Germany). Ozone was generated by exposing synthetic air to ultraviolet radiation (UV Technik, Germany) using a home-made quartz tube flow device. This device facilitates the interaction between the sample gas and UV radiation at a wavelength of 185 nm, as illustrated in Figure 3.
Ozone calibration
The obtained ozone concentration was verified using the conventional iodometric method prior to IR analysis17. The method is based on the reaction between ozone and potassium iodide in buffered solution (pH 6.8; phosphate buffer). The absorbance of the formed triiodide ion was analyzed at 357 nm using an UV-VIS spectrophotometer (SPECORD S600, Analytik Jena, Jena, Germany). A custom-made gas mixing system prototype based on mass flow controllers was developed by the Institute of Analytical and Bioanalytical Chemistry at University of Ulm and Lawrence Livermore National Laboratory and was used for preparing and delivering synthetic air samples at a flow rate of 20 mL min−1. The obtained concentration of ozone was 5.40 ± 0.80 mmol m−3. Dilutions of ozone-containing samples were prepared by mixing different ratios of nitrogen and synthetic air, which were then flown through the quartz tube coiled around the UV lamp at a constant flow rate at 20 mL min−1 for all experiments.
FTIR spectrometer
All measurements reported herein were performed using a compact FTIR spectrometer (IR cube, Bruker Optics Inc, Ettlingen, Germany) equipped with a liquid nitrogen cooled mercury-cadmium-telluride (MCT) detector (Infrared Associates, Stuart/FL, USA). A gold-coated iHWG with an absorption path length of 23 cm and dimensions of 75 × 50 × 12 mm (length × width × depth) was coupled to the spectrometer using off-axis parabolic mirrors with a focal length of 2″ (Thorlabs, Germany). The ozone standards were injected into the iHWG at a constant flow rate of 20 mL min−1. The IR spectra were recorded in the range of 4000-650 cm−1 at a spectral resolution of 4 cm−1; 100 spectra were averaged per measurement. The OPUS 6.5 software package (Bruker Optics Inc, Ettlingen, Germany) was used for data acquisition.
References
Piazza, T., Lee, R. H. & Hayes, J. Survey of the use of ozone-generation air cleaners by the California Public. Final Report, California Air Resources Board, (2006). Prepared for: California Air Resources Board; Available at: http://www.arb.ca.gov/research/apr/past/05-301rev.pdf; Date of access: September 25, 2013.
Nazaroff, W. W. et al. Indoor air chemistry: Cleaning agents, ozone and toxic air contaminants. Final Report: Contract No. 01-336 (2006). Prepared for: California Air Resources Board; Available at: http://www.arb.ca.gov/research/apr/past/01-336.pdf; Date of Access: September 25, 2013.
Bermúdez-Aguirre, D. & Barbosa-Cánovas, G. V. Disinfection of selected vegetables under nonthermal treatments: Chlorine, acid citric, ultraviolet light and ozone. Food Control 29, 82–90 (2013).
Tung, T. C. W., Niu, J. L., Burnett, J. & Hung, K. Determination of ozone emission from a domestic air cleaner and decay parameters using environmental chamber tests. Indoor Built Environ. 12, 29–37 (2005).
Baysan, A. & Beighton, D. Assessment of the ozone-mediated killing of bacteria in infected dentine associated with non-cavitated occlusal carious lesions. Carie Res. 41, 337–341 (2007).
Barkhudarov, E. M. et al. Killing bacteria present on surfaces in films or in droplets using microwave UV lamps. World J. Microbiol. Biotech. 24, 761–769 (2008).
Shaughnessy, R. J., Levetin, E., Blocker, J. & Sublette, K. L. Effectiveness of portable indoor air cleaners: Sensory testing results. Indoor Air 4, 179–188 (1994).
Britigan, N., Alshawa, A. & Nizkorodov, S. A. Quantification of ozone levels in indoor environments generated by ionization and ozonolysis Air Purifiers. J. Air Waste Manag. Assoc. 565, 601–610 (2006).
Wolkoff, P., Clausen, P. A., Wilkins, C. K. & Nielsen, G. D. Formation of strong airway irritants in terpene/ozone mixtures. Indoor Air 10, 82–91 (2000).
Weschler, C. J. Ozone in indoor environments: Concentration and chemistry. Indoor Air 10, 269–288 (2000).
Huth, K. C. et al. Effectiveness of ozone against periodontal pathogenic microorganisms. Eur. J. Oral Sci. 119, 204–210 (2011).
Ohkubo, T., Hamasaki, S., Nomoto, Y., Chang, J.-S. & Adachi, T. The effect of corona wire heating on the downstream ozone concentration profiles in an air-cleaning wire-duct electrostatic precipitator. IEEE Trans. Ind. App. 26, 542–549 (1990).
Philips, T. & Jakober, C. Evaluation of ozone emissions from portable indoor air cleaners that intentionally emit ozone. Staff technical report to the California air resources board 1 (2006). Prepared for: California Air Resources Board; Available at: http://www.arb.ca.gov/research/indoor/o3g-rpt.pdf; Date of Access: September 25, 2013.
Poppendieck, D., Hubbard, H., Ward, M., Weschler, C. & Corsi, R. L. Ozone reactions with indoor materials during building disinfection. Atmos. Environ. 41, 3166–3176 (2007).
Serra, R., Abrunhosa, L., Kozakiewicz, Z., Venancio, A. & Lima, N. Use of ozone to reduce molds in a cheese ripening room. J. Food Protec. 66, 2355–2358 (2003).
Felix, E. P., de Souza, K. A. D., Dias, C. M. & Cardoso, A. A. Measurement of ambient ozone using indigo blue-coated filters. J. AOAC Internat. 89, 480–485 (2006).
Stern, A. C. Air Pollution: Measuring, Monitoring and Surveillance of Air Pollution. (Academic Press, San Diego, 1976).
Proffitt, M. H. & McLaughlin, R. J. Fast-response dual- beam UV-absorption ozone photometer suitable for use on stratospheric balloons. Rev. Sci. Instru. 54, 1719–1728 (1983).
Ridley, B. A., Grahek, F. E. & Walega, J. G. A small, high- sensitivity, medium- response ozone detector suitable for measurements from light aircraft. J. Atmos. Oceanic Techn. 9, 142–148 (1992).
Eipel, C. A., Jeroschewski, P. A. & Steinke, I. Determination of ozone in ambient air with a chemiluminescence reagent film detector. Anal. Chim. Acta 491, 145–153 (2003).
Schurath, U., Speuser, W. & Schmidt, R. Principle and application of a fast sensor for atmospheric ozone. Fresenius J. Anal. Chem. 340, 544–547 (1991).
Ischer, H. Remote sensing of atmospheric trace constituents using Fourier transform spectrometry. Berichte der Bunsengesellschaft für physikalische Chemie 96, 306–314 (1992).
Grosjean, D. & Harrison, J. Response of chemiluminescence NOx analyzers and ultraviolet ozone analyzers to organic air pollutants. Environ. Sci. Techn. 19, 862–865 (1985).
Charlton, C., de Melas, F., Inberg, A., Croitoru, N. & Mizaikoff, B. Hollow-waveguide gas sensing with room-temperature quantum cascade lasers. IEE Proc.-Optoelectron. 150, 306–309 (2003).
Frey, C. M., Luxenburguer, F., Droege, S., Mackoviak, V. & Mizaikoff, B. Near-Infrared hollow waveguide gas sensors. Appl. Spectrosc. 65, 1269–1974 (2011).
Young, C., Hartwig, S., Lambrecht, A., Kim, ., S. S. & Mizaikoff, B. Optimizing gas sensors based on quantum cascade lasers and photonic bandgap hollow waveguides. IEEE Sensors 1345–1348 (2007).
Kim, S.-S. et al. Mid-Infrared trace gas analysis with single pass Fourier Transform Infrared hollow waveguides gas sensor. Appl. Spectrosc. 63, 331–337 (2009).
Young, C. et al. Infrared hollow waveguide sensor for simultaneous gas phase detection of benzene, toluene and xylenes in field environments. Anal. Chem. 83, 6141–6147 (2011).
Kozodoy, R. L., Micheels, R. H. & Harrington, J. A. Small-Bore hollow waveguide infrared absorption cells for gas sensing. Appl. Spectrosc. 50, 415–417 (1996).
Saggese, S. J., Harrington, J. A. & Sigel Jr, G. H. Attenuation of incoherent infrared radiation in hollow sapphire and silica waveguides., Opt. Lett. 16, 27–29 (1991)
Matsuura, Y., Abel, T. & Harrington, J. A. Optical properties of small-bore hollow glass waveguides. Appl. Opt. 34, 6842–6647 (1995).
Wilk, A. et al. Substrate-integrated hollow waveguides: A new level of integration in mid-infrared gas sensing. Anal. Chem., 10.1021/ac402391m (2013).
Ennis, C. P., Bennett, C. J. & Kaiser, R. On the formation of ozone in oxygen-rich solar system ices via ionizing radiation. Phys. Chem. Chem. Phys. 13, 9469–9482 (2011).
Hvozdara, L., Pennington, N., Kraft, M., Karlowatz, M. & Mizaikoff, B. Quantum cascade lasers for mid-infrared spectroscopy. Vib. Spectrosc. 30, 53–58 (2002).
Young, C. et al. External cavity widely tunable quantum cascade laser based hollow waveguide gas sensors for multianalyte detection. Sens. Actuators B Chem. 140, 24–28 (2009).
Woerle, K. et al. Breath analysis with broadly tunable quantum cascade lasers. Anal. Chem. 85, 2697–2702 (2013).
Acknowledgements
J.F.S.P.; P.R.F.; I.M.R.Jr and A.A.C. thank the Brazilian agencies CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior), CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico) and FAPESP-BEPE (2012/05573-6) for fellowships and financial support. This work was performed in part under the auspices of the U.S. Department of Energy by Lawrence Livermore National Laboratory (LLNL) under Contract DE-AC52-07NA27344. This project was in part funded under LLNL sub-contract Nos. B598643 and B603018.
Author information
Authors and Affiliations
Contributions
J.F.S.P., V.K., P.R.F., I.M.R.Jr., A.A.C. and B.M. have designed and discussed the experiments. J.F.S.P and V.K. have performed the experiments. A.W. and B.M. have designed the substrate integrated hollow waveguides. J.F.S.P., A.A.C. and B.M. have written and edited the manuscript. J.F.S.P., V.K. and B.M. contributed to the discussions during data analysis. All authors contributed to discussion of the obtained results and to assembly of the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareALike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
da Silveira Petruci, J., Fortes, P., Kokoric, V. et al. Real-time monitoring of ozone in air using substrate-integrated hollow waveguide mid-infrared sensors. Sci Rep 3, 3174 (2013). https://doi.org/10.1038/srep03174
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep03174
This article is cited by
-
Efficient facemask decontamination via forced ozone convection
Scientific Reports (2021)
-
Molecular Species Generated by Surface Dielectric Barrier Discharge Micro-plasma in Small Chambers Enclosing Atmospheric Air and Water Samples
Plasma Chemistry and Plasma Processing (2021)
-
A Hyphenated Preconcentrator-Infrared-Hollow-Waveguide Sensor System for N2O Sensing
Scientific Reports (2018)
-
A facile synthesis of hybrid nanocomposites of reduced graphene oxide/ZnO and its surface modification characteristics for ozone sensing
Journal of Materials Science: Materials in Electronics (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.