Abstract
Focusing on the chemical basis of dietary selection while investigating the nutritional ecology of animals helps understand their feeding biology. It is also important to consider food abundance/biomass while studying the mechanism of animal food selection. We studied leaf selection in two Bornean folivorous primates in relation to plant chemistry and abundance: proboscis monkeys inhabiting a secondary riverine forest and red leaf monkeys inhabiting a primary forest. Both species tended to prefer leaves containing higher protein levels, although more abundant plant species were chosen within the preferred species, probably to maximise energy gain per unit time. However, the two species showed clear differences in their detailed feeding strategy. Red leaf monkeys strictly chose to consume young leaves to adapt to the poor nutritional environment of the primary forest, whereas proboscis monkeys were not highly selective because of the better quality of its common food in the riverine forest.
Similar content being viewed by others
Introduction
All free-living animals must make choices regarding the foods to be included in their daily diet and these choices influence their nutritional state and ultimately their health and fitness. In investigations of the nutritional ecology of animals, focusing on the chemical basis of dietary selection promotes understanding of their feeding biology1. Such mineral and phytochemical studies have revealed a universal trend in food choice in herbivorous primates, particularly colobine monkeys: they prefer foods with more protein2,3,4,5,6. On the basis of this finding, it has been proposed that the protein–fibre ratio of leaves positively correlates with the abundance of colobine monkeys5,7,8.
It is equally important to investigate differences in the chemical basis of diet choice between closely related species with similar anatomy and, presumably, physiology because body size and subtle physiological differences in different colobines9 may influence their tolerance of unfavourable environments such as a lower protein–fibre ratio of leaves; higher tolerance would enable the animals to be widely distributed among various types of forests. However, although many comparative studies of feeding ecology considering more than a single colobine species have been conducted10,11,12,13, such studies have rarely investigated variation in feeding strategy in terms of food plant chemistry. Nonetheless, large variation in the chemical basis of diet selection, particularly leaf selection, between two Asian colobines (Presbytis rubicunda and P. melalophos) has been found, which may be explained by the difference in their habitats, i.e. peninsular Malaysia and the island of Borneo2. However, there is too little relevant information available to give an account of the mechanism of such variation.
In addition to plant chemistry, food abundance/biomass is an important factor for understanding animal food selection6,14,15,16. This is because the time required to find and handle food is a significant cost for animals in some cases and it is important to consider the classic optimal foraging strategy model, i.e. animals maximise energy gain per unit time (travelling + foraging time)17. A multivariate approach is therefore essential to reveal interactions between chemical content and distribution or processing time. Although such studies are rare, some authors have investigated dietary selection on the basis of complex, integrated, multiple criteria, including both classic optimal foraging models and chemical content14,16.
We studied leaf selection in two Bornean folivorous primates: proboscis monkeys (Nasalis larvatus) inhabiting a secondary riverine forest in the lower Kinabatangan, which spend more than 65% of their feeding time eating leaves18 and red leaf monkeys (P. rubicunda) inhabiting a primary forest in the Danum Valley, which spend 46% of their feeding time eating leaves6. Both species are members of the subfamily Colobinae, which includes at least 30 species that can be grouped into four to nine genera distributed in Asia and Africa19. The different species of colobine monkeys have similar stomach anatomy and physiology, with an enlarged, sacculated forestomach9 and are endemic to the island of Borneo. N. larvatus is a large, sexually dimorphic species; adult males have the largest body size among colobine species (approximately 20 kg), whereas adult females are about half the weight of males20. P. rubicunda is small-bodied and gracile and is less sexually dimorphic than N. larvatus; adults weigh approximately 6.0 kg21. N. larvatus is restricted to lowlands and is typically associated with coastal forest, including mangroves and riverine, peat swamp and freshwater swamp forests, whereas P. rubicunda is found in almost all habitat types of Borneo, excluding mangroves and freshwater swamp forests22.
In a study of N. larvatus inhabiting peat swamp forests, Yeager et al. (1997)23 compared consumed and nonconsumed leaves and provided comprehensive data on the roles of mineral and phytochemical constituents in leaf selection. Leaves of species used as food sources were significantly higher in protein and lower in fibre than those of species not used as food sources. Food sources of N. larvatus had higher concentrations of phosphorus and potassium, whereas nonfood sources had higher concentrations of calcium and manganese. However, the behavioural data of that study were mostly derived from boat-based observations when the monkeys were close to riverside trees because the terrain was frequently swampy and impassable on foot24; therefore, the data must be regarded as potentially biased, particularly in relation to the animals' feeding behaviour. Furthermore, there are no data on the effects of both plant chemistry and food abundance on food selection in N. larvatus. Thus, to gain a more complete picture of leaf selection in N. larvatus, continuous observations in both riverbank and inland forest habitats, including data on food abundance, are crucial. Food selection by P. rubicunda has been better studied than that of N. larvatus in terms of plant chemistry and food abundance. Studies at two different study sites in Borneo showed that P. rubicunda preferred not only plants with higher protein content but also plants that were more abundant2,6.
In the light of new data on leaf selection by N. larvatus observed throughout the day for 13 months and published data on P. rubicunda6, we examined and compared leaf selection with a focus on plant chemistry and abundance. In particular, we sought to (1) compare the chemical properties of leaves in common plant species between the two habitat types (primary and secondary forests) of the two colobines; (2) describe leaf selection in detail, focusing on plant chemistry and abundance of N. larvatus in the secondary forest, which is comparable with that of P. rubicunda6 and (3) compare the chemical properties of consumed leaves between N. larvatus and P. rubicunda. Lastly, we discuss the reason for the variation in diet choice among these two closely related species with similar anatomy and physiology and the significance of their different feeding strategies with their different distribution patterns on the island of Borneo.
Results
Comparison of chemical properties of young leaves of common plant species between primary and secondary forests
As shown in Table 1A, the difference between the primary and secondary forests was not significant for NDF, crude protein and crude lipid but significant for crude ash. The proportion of condensed tannin-bound species did not differ between the study sites (χ2 = 1.10, P = 0.295). Furthermore, the protein–fibre ratio was significantly different between the two forests, indicating that the quality of young leaves of common plant species for the colobines in the secondary forest was higher than that of young leaves in the primary forest.
Young leaf selection by N. larvatus
Selection between preferred species and common species
GLM for predicting whether N. larvatus monkeys decide the preferred young leaf species using their chemical properties and abundance indicated that abundance and crude protein were the significant factors (Table 2). The negative linear term for abundance indicated that N. larvatus selectively fed on young leaves in the forests. On the other hand, the positive linear term for crude protein indicated that they chose young leaves containing more crude protein.
Selection within the preferred plant species
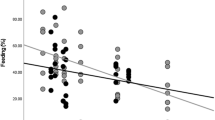
The best-fit model predicting the percentages of the time eating young leaves of the preferred species in N. larvatus included only the abundance of the plant species, with a negative effect (Table 3). The negative linear term, however, indicated that the monkeys spent more time eating young leaves of more abundant plants within the consumed plant species (Fig. 1, see Methods), though this tendency seems to be driven by the two “super-abundant” plant species, i.e. Mallotus muticus (Euphorbiaceae) and Lophopyxis maingayi (Lophopyxidaceae).
Comparison of chemical properties of consumed young leaf between P. rubicunda and N. larvatus
Young leaves of only four (20%) of the common plant species in the primary forest were consumed by P. rubicunda, although N. larvatus consumed young leaves of all common plant species (100%). In addition, in P. rubicunda, young leaves of S. macropterus (Leguminosae), the most consumed plant species, accounted for 62.3% of the total young leaves consumed and 27.9% of the total feeding time6, whereas in N. larvatus, the most consumed plant species (young leaves: M. muticus; Euphorbiaceae) accounted for only 9.9% and 6.6%, respectively. Furthermore, the dietary diversity index of N. larvatus (H′ = 0.98) was clearly higher than that of P. rubicunda (H′ = 0.52), indicating that P. rubicunda is a more selective eater of young leaves than N. larvatus.
As shown in Table 1B, the differences between P. rubicunda and N. larvatus in the chemical properties of the top seven plant species consumed were not significant. In addition, the proportion of condensed tannin-bound species among the consumed species did not differ between the two colobines (χ2 = 1.70, P = 0.193).
Discussion
The results of this study revealed significant differences in some chemical properties of the leaves in the common plant species between the secondary riverine and the primary forests and showed that leaf quality, i.e. protein–fibre ratio, was higher in the secondary riverine forest. This may be explained by differences in soil quality between the study sites. The leaves of trees growing on poor soils apparently contain lower levels of protein and higher levels of fibre than those of trees growing on better soils42,43. As discussed by Waterman et al. (1988)44, many common soils of Borneo are rather poor compared with those of Peninsular Malaysia. However, even in Borneo, certain areas exposed to seasonal flooding, such as forests along rivers, may be supplied with fertile soil by the flooded water. Plants inhabiting such forests along rivers may be expected to exhibit a higher protein–fibre ratio than those in inland forests. Indeed, seasonal flooding of the secondary riverine forest at our study site has been recorded and the results showed that water covers a wide range of the secondary riverine forest45. In addition to the better soil hypothesis, it is also important to consider the plant life history. Compared with the primary forest, in the secondary riverine forest there are potentially fewer shade-tolerant plants and more plants that are gap specialists which generally contain more water and protein compared to shade-tolerant plant species46. Therefore, to test this possibility, further research on nutritional quality of leaves in riverine forest in relation to the distance from rivers is needed to better understand leaf quality in Bornean forests.
Echoing the results on leaf selection between preferred and common plant species by N. larvatus in this study, Yeager et al. (1997)23 noted that N. larvatus preferred leaves with higher protein levels. Preference for leaves containing higher protein levels is a general trend not only in N. larvatus but also in other colobine monkeys, including P. rubicunda2,3,4,5, except for some colobine species inhabiting nonprotein-limited forests47. Compared with primary forests, secondary riverine forests, with higher protein–fibre ratios of the leaves, would be more suitable habitats for colobines48. Nonetheless, forests along rivers in Borneo may be considered as a protein-limited habitat for N. larvatus because this animal may need more protein owing to a larger body size (approximately 20 kg for adult males) compared with colobines with a smaller body size, including P. rubicunda (approximately 6 kg for adults). Thus, although N. larvatus inhabited forests along the river, with high-quality leaves, the general trend found for colobines was detected in this study.
This is the first study to demonstrate leaf selection by N. larvatus within its preferred plant species. N. larvatus consumed the more abundant of its preferred plant species, though this tendency appeared to arise from the two preferred super-abundant plant species, i.e. there may not be any clear tendency if these plant species are excluded. In the latter case, the lack of a clear relationship may be explained by the better quality of common foods in the secondary riverine forest compared to that in the primary forest; it may not be necessary for monkeys inhabiting such forests to strictly rely on abundant foods. Nonetheless, the fact that N. larvatus consumed the more abundant of its preferred plant species may reflect maximisation of their energy gain per unit time, i.e. the classic optimal foraging strategy model17 appears to be applicable to this colobine species. Hanya and Bernard (2012)6 also showed that food abundance is more important than other chemical properties in leaf selection by P. rubicunda in primary forests. These are good examples showing that the travelling cost saved by selecting common plants exceeds the benefit gained by selecting higher-quality foods, i.e. leaves with higher protein levels. Thus, this study provides the novel finding that the classical optimal foraging strategy model can be applied to herbivores that spend most of their feeding time for ubiquitous food sources such as leaves.
Comparing the feeding strategies of different animals living in different habitats in relation to plant chemistry and abundance is a considerable challenge because it is usually difficult to determine whether the difference(s) seen is(are) attributable to the characteristics of the animal species or of the habitat types. Nonetheless, in this study, we were able to reveal the different feeding strategies of two Bornean colobines living in different habitat types. Although P. rubicunda inhabits primary forests, which produce lower-quality leaves than the secondary riverine forests inhabited by N. larvatus, to gain a similar level of chemical properties, P. rubicunda was highly selective in choosing which young leaves to consume as a means of adapting to the poor nutritional environment of the primary forest in Borneo. On the other hand, the feeding strategy of N. larvatus was more opportunistic; it chose to consume leaves with higher protein levels, although it was not highly selective because of the better quality of its common food. Although we performed a comparative analysis of only the major foods eaten by the two colobines (top seven plant species), our results may still adequately reflect their general similarities and differences in feeding strategies; if we include other rare food plants eaten, the tendencies observed might be obscured as the analysis applied did not consider the relative amounts of each plant species eaten. A tendency similar to that found to this study has been reported by Davies et al. (1988)2: banded leaf monkeys (Presbytis melalophos) inhabiting low-lying vegetation associated with a river terrace with rather fertile land soil in Peninsular Malaysia consumed foliage of many common plant species, although P. rubicunda inhabiting primary forests in Borneo relies on rare plant species. These studies indicate the flexible leaf-eating strategy of colobines, depending on the nutritional environment in their habitat.
The difference in feeding strategy between the two colobines in this study provides insights into the distribution patterns of Bornean colobine monkeys. Although N. larvatus is closely associated with waterways, travelling inland to forage (generally up to 1 km) and returning to its sleeping sites along the river edge almost every evening45,49, other smaller-bodied colobine species, including P. rubicunda, are frequently found even in the inland primary forests of Borneo22. To expand their distribution to poor nutritional environments, i.e. inland forests of Borneo dominated by the family Dipterocarpaceae, colobine species may need to reduce their body size to acquire sufficient nutrition from patchily distributed high-quality leaves, such as those of the vine S. macropterus eaten by P. rubicunda in this study.
In addition to the benefit of effective predation avoidance proposed previously45,50,51, N. larvatus may be forced to range in flooded forests where it can easily obtain higher-quality leaves rich in protein (with a low fibre content) and minerals (crude ash) to maintain its large body. A previous explanation for riverine refuging by N. larvatus mainly focused on an antipredator strategy: the monkeys may guard more effectively against attack from feline predators while in trees at the riverbank because predators can approach the monkeys only from the landward side45,50,51. However, the strategy of riverine refuging may provide not only N. larvatus but also other sympatric primates with the common benefit of an effective means of predation avoidance. Thus, to clarify the reasons why riverine refuging is more common in N. larvatus than in sympatric primates, it is important to consider the additional benefit of riverine refuging by N. larvatus51. Because a large body size generally permits a low-energy diet with a low nutrient density52, the large body of N. larvatus—the largest among the colobines—may allow more fibrous foods to be consumed. Nonetheless, it may be impossible to obtain adequate nutrition only from those limited high-quality leaves in inland forests to maintain the large body. Furthermore, the possibility of other nutritional problems in N. larvatus resulting from a large body size may be an important consideration48. For example, the large herbivorous mammals with forestomach such as sambar deer (Cervus unicolor) and banteng (Bos javanicus) inhabiting Bornean primary forests dominated by the family Dipterocarpaceae frequently use salt licks to obtain rich minerals such as calcium, magnesium, potassium and sodium53, suggesting that they acquire sufficient nutrition not only from plants but also from other sources.
The effect of the social system of N. larvatus on its specific distribution pattern along rivers may be worth considering. N. larvatus is one of the various species reported to exhibit a multilevel social system, in which lower levels of social organisation aggregate into larger units54,55 in Borneo. Therefore, the biomass of N. larvatus in the riverine habitat is estimated to be rather higher than that of sympatric primate species24 and the local density of individuals becomes high depending on temporal changes in food availability56. To organise their specific social system, N. larvatus may be forced to live in flooded forests with abundant food sources of better quality.
This study presents novel findings on leaf selection by two Bornean colobine monkeys living in the different habitat types. On the other hand, recent Asian colobine studies provide evidence of the dietary flexibility of these species: although most of the feeding time in Asian colobines, including N. larvatus and P. rubicunda, is devoted to the exploitation of young leaves, more than half of their monthly feeding time is accounted for by fruits (mostly unripe) and/or seeds at certain times of the year6,18,57,58,59. However, there are fewer studies on the chemical basis of diet selection focusing on consumed fruits/seeds in Asian colobines compared with African colobines60,61. Future research should therefore consider not only leaves but also fruits/seeds in Asian colobines to provide a general understanding of colobine feeding strategies and better knowledge of the nutritional ecology of animals.
Methods
Study sites and collection of behavioural data
N. larvatus in secondary riverine forest
I. M. and two trained assistants conducted observations in the riverine forest along the Menanggul River, a tributary of the Kinabatangan River in Sabah, Borneo, Malaysia (118° 30′ E, 5° 30′ N), from May 2005 to May 2006. The southern area of the Menanggul River is extensively covered by secondary forest, whereas the northern area has been deforested for oil palm plantations, except for a protected zone along the river. Mean minimum and maximum temperatures were approximately 24 and 32°C, respectively and the total annual precipitation at the site was 2510 mm (June 2005–May 2006)25. During the observation period, the river level varied by approximately 1 m daily, the average level rising by more than 3 m during seasonal flooding in February and March 2006.
The one-male multifemale group of N. larvatus (BE group: one adult male, six adult females, nine immatures) was habituated and all monkeys in the group were identified. Observational data were collected using focal animal sampling26 on each observation day from around 06:00 h until 18:30 h on 11–17 days/month. The observers were separated into two groups to simultaneously follow one adult male and one arbitrarily selected female; note that this observation protocol may have introduced some degree of pseudoreplication into the data set, though we believe that its effect on the analysis performed was small. While following focal individuals, the time (s) spent in feeding behaviour was recorded. These continuous observations facilitated the calculation of time budgets of focal individuals, including the proportion of the day spent feeding and time spent feeding on individual food items18. The total observation time was 3506 h.
P. rubicunda in primary forest
G. H. and six trained assistants conducted observations in a primary forest around the Danum Valley Field Centre (117° 48′ E, 4° 57′ N) within the Danum Valley Conservation Area (438 km2) located in Sabah, Borneo, Malaysia, from December 2006 to 2008. During 2007 and 2008, mean minimum and maximum temperatures were approximately 22.5 and 31.4°C, respectively and the total annual precipitation at the site was 3115 mm6. The forest is predominantly composed of lowland dipterocarp trees27.
One habituated group of P. rubicunda with 8–12 individuals was observed from around 06:00 h until 16:00 h on 5–10 days/month. Behaviour was recorded by scan sampling every 10 min. Feeding behaviour was recorded for all visible individuals, except for dependent infants. During feeding, the food category was recorded and the samples were collected for later identification6. The total observation time was 1141 h. Although the behavioural data collection method used for P. rubicunda and N. larvatus differed, this is unlikely to have affected our results because different methodologies, i.e. focal and scan sampling, have previously been demonstrated to produce similar tendencies concerning colobine activity budgets (e.g. Colobus vellerosus28,29).
Vegetation survey
Secondary riverine forest
Along the trails (TR1–TR16; 200–500-m long) in the study site, we labelled trees ≥10 cm in diameter at breast height (DBH) and vines ≥5 cm in diameter located 1.5 m from the trail; hence, the labelled width was 3 m and the vegetation survey area covered 2.15 ha (3 m × 7150 m). All the labelled trees and vines were taxonomically identified with the help of the Forest Research Center, Sandakan, Sabah.
Primary forest
Data on the botanical composition of the forest were obtained from Lingenfelder (2005)30. We used data for a 4-ha plot in the study site, for which all trees ≥10 cm DBH were identified to species and their DBH was measured. Because these data did not include lianas, data on Spatholobus macropterus (Leguminosae), an important food source for P. rubicunda at this site, were added6. A transect was set along a circular trail that covered most of the home range (approximately 16 ha), of 4 m × 1.2 km and the presence/absence of this liana for all trees along the transect was recorded.
Leaf sampling
N. larvatus in secondary riverine forest
To compare the chemical properties of young leaves that were consumed and not consumed by N. larvatus, from October to December 2008, samples were collected of young leaves of the top 25 species that accounted for 49.7% of the total feeding time (leaves of all species that accounted for ≥0.5 of the feeding time) and the 25 most abundant species (number of plant species) in the study area of the vegetation survey. However, because of the difficulty in leaf sampling, of the top 25 species in feeding time and abundance, only 18 and 19 plant species, respectively, were sampled for chemical analysis. For each tree species consumed, young leaf samples were collected from at least four individual trees in the vegetation survey area. At least 20 leaves were sampled for each species. The leaves were kept in plastic bags and taken to FRC, where they were dried at 60°C for 60 h in an oven. The leaves were then stored in plastic bags and taken to the laboratory at the Primate Research Institute, Kyoto University, where they were again dried at 60°C for 48 h using a vacuum incubator. After weighing, the leaves were milled, placed in a plastic tube and kept in a desiccator.
P. rubicunda in primary forest
The chemical properties of young leaves consumed and not consumed by P. rubicunda were compared. Young leaves of all species that accounted for ≥1% of the feeding time (31.4% of the total feeding time) and the 20 most abundant species (basal area) were sampled in the study area of the vegetation survey30,31. Seven consumed species and 16 nonconsumed species were included in the analyses. All other processes for chemical analysis were as described for N. larvatus6.
Chemical analysis
Crude protein was determined using the Kjeldahl procedure for total nitrogen and multiplying the result by 6.25. Since fibre-bound nitrogen was not taken into account, metabolizable protein may have been lower32. Crude lipid was measured as diethyl ether extract using the Soxhlet method33. Crude ash was determined by ashing at 550°C. Neutral detergent fibre (NDF) was estimated using the method of van Soest et al. (1991)34. Condensed tannin was extracted with 50% methanol and its presence/absence was determined using the butanol–HCl method35. Presence/absence was used because it is difficult to detect subtle differences in leaf tannin content using this method32,36. The ratio of weight of the constituent to the total dry weight was used for analysis of crude protein, crude lipid, NDF and crude ash.
Data analysis
Comparison of chemical properties of young leaves in common plant species between primary and secondary forests
The collected plant species (19 species) within the top 25 abundant species in the secondary forest (Sukau) and the top 20 abundant species in the primary forest (Danum Valley) were defined as the common plant species at each study site. Using the Mann–Whitney U test, the mean values of chemical properties (NDF, crude protein, crude ash and crude lipid) in the common plant species were compared between the study sites. Because the protein–fibre ratio of leaves has been suggested to indicate leaf quality for colobines4, the mean protein–fibre ratio in the common plant species was also compared between the study sites using the Mann–Whitney U test. Because the data on condensed tannin were binominal (presence/absence), the χ2 test was used to determine whether the ratio of the common plant species containing the condensed tannin differed between the study sites.
Young leaf selection by N. larvatus: comparison between preferred and common species
The top 25 plant species (18 species were available for analysis) consumed by N. larvatus in terms of the feeding time were defined as the preferred species. To examine whether N. larvatus decided on the preferred species using chemical properties (NDF, crude protein, crude ash, crude lipid and condensed tannin) and abundance (number of plant species), generalised linear model (GLM) analysis was performed, applying the binomial family to calculate the Akaike's information criterion for small samples (AICc37; dispersion parameter, 1.09). The categorisation of preferred/common species (excluding species listed as preferred young leaf species) was the dependent variable and chemical properties and abundance were independent variables. To select the best-fit model, models were assessed with the AICc using the AICc function of MuMIn Version 1.7.738. The variance inflation factors (VIFs) were 1.18 for crude NDF, 1.17 for crude protein, 1.19 for crude ash content, 1.20 for crude lipid, 1.14 for condensed tannin and 1.14 for plant abundance, which were less than the cutoff value (10)39; therefore, collinearity among independent factors did not affect the results. The model with the smallest AICc among all possible combinations of independent factors, including the null model, was chosen. Likelihood ratio tests were used to evaluate the linear model and the independent variables affecting categorisation.
Young leaf selection by N. larvatus within the preferred plant species
Using GLM, the effects of chemical properties and abundance of the preferred plant species on the percentage of feeding time were assessed. The gamma family (link function = inverse, i.e. the calculated coefficient value reflects the inverse effect) was used to calculate the AICc (dispersion parameter, 0.65). VIFs were 1.09 for crude NDF, 1.47 for crude protein, 1.48 for crude ash content, 1.60 for crude lipid, 1.23 for condensed tannin and 1.53 for plant abundance, which were less than the cutoff value (10)39; therefore, collinearity among independent factors did not affect the results. The model with the smallest AICc among all possible combinations of independent factors, including the null model, was chosen.
Comparison of chemical properties of consumed young leaves between P. rubicunda and N. larvatus
To compare P. rubicunda with N. larvatus in terms of food selectivity and the chemical properties of young leaves consumed, the top seven plant species that accounted for approximately 30% of the total feeding time in both the colobines (P. rubicunda, 31.4%; N. larvatus, 31.8%) were chosen. Using the time spent eating young leaves of the top seven plant species in the two colobines, the Shannon–Wiener index of diversity (H′)40 was calculated to evaluate dietary selectivity for the top seven plant species; less dietary diversity means higher food selectivity. The mean chemical properties and the protein–fibre ratio of the top seven young leaves were also compared between the two colobines using the Mann–Whitney U test. The χ2 test was used to determine whether the ratio of consumed plant species containing condensed tannins differed between the two species of colobines. R 2.14.141 was used for all statistical analyses.
References
Lambert, J. E. Primate nutritional ecology: feeding biology and diet at ecological and evolutionary scales. In: Campbell, C., Fuentes, A.,MacKinnon, K. C., Panger, M. & Bearder, S. (eds) Primates in Perspective 2nd Edition, Oxford University Press. Oxford, pp 512–522 (2010).
Davies, A. G., Bennett, E. L. & Waterman, P. G. Food selection by two South-east Asian colobine monkeys (Presbytis rubicunda and Presbytis melalophos) in relation to plant chemistry. Biol. J. Linn. Soc. 34, 33–56 (1988).
Kar-Gupta, K. & Kumar, A. Leaf chemistry and food selection by common langurs (Presbytis entellus) in Rajaji National Park, Uttar Pradesh, India. Int. J. Primatol. 15, 75–93 (1994).
Chapman, C. A. & Chapman, L. J. Foraging challenges of red colobus monkeys: Influence of nutrients and secondary compounds. Comp. Biochem. Physiol. A. 133, 861–875 (2002).
Wasserman, M. D. & Chapman, C. A. Determinants of colobine monkey abundance: the importance of food energy, protein and fibre content. J. Anim. Ecol. 72, 650–659 (2003).
Hanya, G. & Bernard, H. Fallback foods of red leaf monkey (Presbytis rubicunda) in Danum Valley, Borneo. Int. J. Primatol. 33, 322–337 (2012).
Chapman, C. A., Chapman, L. J., Bjorndal, K. A. & Onderdonk, D. A. Application of protein to fiber ratios to predict colobine abundance on different spatial scales. Int. J. Primatol. 23, 283–310 (2002).
Fashing, P. J., Dierenfeld, E. S. & Mowry, C. B. Influence of plant and soil chemistry on food selection, ranging patterns and biomass of Colobus guereza in Kakamega Forest, Kenya. Int. J. Primatol. 28, 673–703 (2007).
Chivers, D. Functional anatomy of the gastrointestinal tract. In: Davies, A. G. & Oates, J. F. (eds.) Colobine Monkeys: Their Ecology, Behaviour and Evolution. Cambridge University Press, Cambridge, pp 205–227 (1994).
Clutton-Brock, T. H. Feeding behavior of red colobus and black and white colobus in East Africa. Fol. Primatol. 23, 165–207 (1975).
Struhsaker, T. T. & Oates, J. F. Comparison of the behavior and ecology of red colobus and black-and-white colobus in Uganda: A summary. In Tuttle, R. H. (ed.), Socioecology and Psychology of Primates, Mouton, The Hague, pp. 103–123 (1975).
Hadi, S., Ziegler, T., Waltert, M., Mühlenberg, M. & Hodges, J. K. Habitat use and trophic niche overlap of two sympatric colobines, Presbytis potenziani and Simias concolor, on Siberut Island, Indonesia. Int. J. Primatol. 33, 218–232 (2012).
Tombak, K. J. et al. Patch depletion behavior differs between sympatric folivorous primates. Primates 53, 57–64 (2012).
Leighton, M. Modeling dietary selectivity by Bornean orangutans: evidence for integration of multiple criteria in fruit selection. Int. J. Primatol. 14, 257–313 (1993).
Barton, R. A. & Whiten, A. Reducing complex diets to simple rules: food selection by olive baboons. Behav. Ecol Sociobiol. 35, 283–293 (1994).
Hanya, G., Kiyono, M., Takafumi, H., Tsujino, R. & Agetsuma, N. Mature leaf selection of Japanese macaques: Effects of availability and chemical content. J. Zool. 273, 140–147 (2007).
Krebs, J. R. & Davies, N. B. An introduction to behavioural ecology, 3rd edn. Oxford: Blackwell (1993).
Matsuda, I., Tuuga, A. & Higashi, S. The feeding ecology and activity budget of proboscis monkeys. Am. J. Primatol. 71, 478–492 (2009a).
Oates, J. F. & Davies, A. G. What are the colobines? In: Davies, A. G. & Oates, J. F. (eds.) Colobine Monkeys: Their Ecology, Behaviour and Evolution. Cambridge University Press, Cambridge, pp 1–9 (1994).
Schultz, A. H. Growth and development of the proboscis monkey. Harvard Bull. Mus. Comp. Zool. 89, 279–314 (1942).
Davis, D. D. Mammals of the lowland rainforests of North Borneo. Bull. Natl. Museum Singapore 31, 1–129 (1962).
Meijaard, E. & Nijiman, V. Primate hotspots on Borneo: predictive value for general biodiversity and the effects of taconomy. Cons. Biol. 17, 725–732 (2003).
Yeager, C. P., Silver, S. C. & Dierenfeld, E. S. Mineral and phytochemical influences on foliage selection by the proboscis monkey (Nasalis larvatus). Am. J. Primatol. 41, 117–128 (1997).
Yeager, C. P. Feeding ecology of the proboscis monkey (Nasalis larvatus). Int. J. Primatol. 10, 497–530 (1989).
Matsuda, I., Tuuga, A. & Higashi, S. Ranging behaviour of proboscis monkeys in a riverine forest with special reference to ranging in inland forest. Int. J. Primatol. 30, 313–325 (2009b).
Altmann, J. Observational study of behavior: sampling methods. Behaviour 69, 227–267 (1974).
Newbery, D. M., Kennedy, D. N., Petol, G. H., Madani, L. & Ridsdale, C. E. Primary forest dynamics in lowland dipterocarp forest at Danum Valley, Sabah, Malaysia and the role of the understorey. Philos Trans R Soc Lond B Biol Sci 354, 1763–1782 (1999).
Teichroeb, J. A., Saj, T. L., Paterson, J. D. & Sicotte, P. Effect of group size on activity budgets of Colobus vellerosus in Ghana. Int. J. Primatol. 24, 743–758 (2003).
Teichroeb, J. A. & Sicotte, P. Test of the ecological constraints model on ursine colobus monkeys (Colobus vellerosus) in Ghana. Am. J. Primatol. 71, 49–59 (2009).
Lingenfelder, M. Rain forest dynamics and response to drought in a Bornean primary lowland dipterocarp forest. PhD dissertation, University of Bern, Bern (2005).
Newbery, D. M., Campbell, E. J. F., Proctor, J. & Still, M. J. Primary lowland dipterocarp forest at Danum Valley, Sabah, Malaysia. Species composition and patterns in the understorey. Vegetatio 122, 193–220 (1996).
Rothman, J. M., Chapman, C. A. & Pell, A. N. Fiber-bound nitrogen in gorilla diets: Implications for estimating dietary protein intake of primates. Am. J. Primatol. 70, 690–694 (2008).
Soxhlet, F. Die gewichtsanalytische Bestimmung des Milchfettes. Polytechnisches Journal 232, 461–465 (1879).
van Soest, P. J., Robertson, J. B. & Lewis, B. A. Methods for dietary fiber, neutral detergent fiber and nonstarch polysaccharides in relation to animal nutrition. J. Dairy Science 74, 3583–3597 (1991).
Porter, L. J. Tannins. In: Dey, P. M. & Harborne, J. B. (eds.) Methods in plant biochemistry, vol 1, Plant Phenolics, London: Academic, pp 389–419 (1989).
Rautio, P., Bergvall, U. A., Karonen, M. & Salminen, J. P. Bitter problems in ecological feeding experiments: Commercial tannin preparations and common methods for tannin quantifications. Biochem. Syst. Ecol. 35, 257–262 (1989).
Burnham, K. P. & Anderson, D. R. Model selection and multimodel inference: A practical information-theoretic approach (2nd ed.), Springer-Verlag (2002).
Bartoń, K. MuMIn: Multi-model inference (R package version 1.7.7). http://cran.r-project.org/web/packages/MuMIn/MuMIn.pdf (accessed 2012 May 14).
Quinn, G. P. & Keough, M. J. Experimental design and data analysis for biologists. Cambridge University Press, Cambridge (2002).
Pielou, E. C. Shannon's formula as a measure of specific diversity: its use and misuses. Am. Nat. 104, 463–465 (1966).
R Development Core Team. R: A language and environment for statistical computing. R foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0, URL http://www.R-project.org/ (accessed 2011 November 3).
Janzen, D. H. Tropical blackwater rivers, animals and mast fruiting by the Dipterocarpaceae. Biotropica 6, 69–103 (1974).
McKey, D. B., Gartlan, J. S., Waterman, P. G. & Choo, G. M. Food selection by black colobus monkeys (Coiobus satanas) in relation to plant chemistry. Biol. J. Linn. Soc. 16, 115–146 (1981).
Waterman, P. G., Ross, J. A. M., Bennett, E. L. & Davies, A. G. A comparison of the floristics and leaf chemistry of the tree flora in two Malaysian rain forests and the influence of leaf chemistry on populations of colobine monkeys in the Old World. Biol. J. Linn. Soc. 34, 1–32 (1988).
Matsuda, I., Tuuga, A. & Higashi, S. Effects of water level on sleeping-site selection and inter-group association in proboscis monkeys: why do they sleep alone inland on flooded days? Ecol. Res. 25, 475–482 (2010a).
Coley, P. D. Interspecific variation in plant anti-herbivore properties: The role of habitat quality and rate of disturbance. New Phytologist 106, 251–263 (1987).
Kool, K. M. Food selection by the silver leaf monkey, Trachypithecus auratus sondaicus, in relation to plant chemistry. Oecologia 90, 527–533 (1992).
Bennett, E. L. & Sebastian, A. C. Social organization and ecology of proboscis monkeys (Nasalis larvatus) in mixed coastal forest in Sarawak. Int. J. Primatol. 9, 233–256 (1988).
Sha, J. C. M., Bernard, H. & Nathan, S. Status and conservation of proboscis monkeys (Nasalis larvatus) in Sabah, East Malaysia. Prim. Cons. 23, 107–120 (2008).
Matsuda, I., Tuuga, A., Akiyama, Y. & Higashi, S. Selection of river crossing location and sleeping site by proboscis monkeys (Nasalis larvatus) in Sabah, Malaysia. Am. J. Primatol. 70, 1097–1101 (2008).
Matsuda, I., Tuuga, A. & Bernard, H. Riverine refuging by proboscis monkeys (Nasalis larvatus) and sympatric primates: implications for adaptive benefits of the riverine habitat. Mam. Biol. 76, 165–171 (2011).
Clauss, M. et al. The influence of natural diet composition, food intake level and body size on ingesta passage in primates. Comp. Biochem. Physiol. A. 150, 274–281 (2008).
Matsubayashi, H. et al. Importance of natural licks for the mammals in Bornean isaland tropical rain forests. Ecol. Res. 22, 742–748 (2007).
Yeager, C. P. Proboscis monkey (Nasalis larvatus) social organization: intergroup patterns of association. Am. J. Primatol. 23, 73–86 (1991).
Grueter, C. C., Matsuda, I., Zhang, P. & Zinner, D. Multilevel societies in primates and other mammals: introduction to the special issue. Int. J. Primatol. 33, 993–1001 (2012).
Matsuda, I., Kubo, T., Tuuga, A. & Higashi, S. A Bayesian analysis of the temporal change of local density of proboscis monkeys: implications for environmental effects on a fission-fusion society. Am. J. Phys. Anthropol. 142, 235–245 (2010b).
Davies, G. Seed-eating by red leaf monkeys (Presbytis rubicunda) in dipterocarp forest of northern Borneo. Int. J. Primatol. 12, 119–144 (1991).
Kool, K. M. The diet and feeding behavior of the silver leaf monkey (Trachypithecus auratus sondaicus) in Indonesia. Int. J. Primatol. 14, 667–700 (1993).
Dela, J. D. S. Western purple-faced langurs (Semnopithecus vetulus nestor) feed on ripe and ripening frutis in human-modified environments in Sri Lanka. Int. J. Primatol. 33, 40–72 (2012).
Maisels, F., Gautier-Hion, A. & Gautier, J. P. Diets of two sympatric colobines in Zaire: more evidence on seed-eating in forests on poor soils. Int. J. Primatol. 15, 681–701 (1994).
Davies, A. G., Oates, J. F. & Dasilva, G. L. Patterns of frugivory in three West African colobine monkeys. Int. J. Primatol. 20, 327–357 (1999).
Acknowledgements
I.M. and G.H. thank the Economic Planning Unit of the Malaysian Government, the Sabah Wildlife Department, the Sabah Forestry Department, the Kinabatangan Orangutan Conservation Project, the Danum Valley Field Centre and our research assistants for support. We would like to thank Enago (www.enago.jp) for the English language review. I.M. truly appreciates Y. Matsuda for supporting my field works. Finally, we are grateful to anonymous reviewers for their fruitful comments that improved this manuscript. The Economy Planning Unit of Malaysia and the State of Sabah and the Danum Valley Management Committee of Yayasan Sabah permitted our study. This study was partly financed by HOPE and Human Evolution Project of KUPRI, Grant-in-Aid for challenging Exploratory Research (24657170 to IM) and Sasakawa Scientific Research Grant from the Japan Science Societyand (to I.M.) and was conducted in compliance with animal care regulations and applicable Malaysian laws.
Author information
Authors and Affiliations
Contributions
I.M., A.T., H.B., J.S. and G.H. were involved in planning, designing and realizing the research. Data collection conducted by I.M. and G.H. I.M. analysed the data and wrote the manuscript. All authors discussed the results and provided comments on the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/3.0/
About this article
Cite this article
Matsuda, I., Tuuga, A., Bernard, H. et al. Leaf Selection by Two Bornean Colobine Monkeys in Relation to Plant Chemistry and Abundance. Sci Rep 3, 1873 (2013). https://doi.org/10.1038/srep01873
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep01873
This article is cited by
-
Modifying the diets of captive proboscis monkeys in a temperate zoo to reduce weight loss and renal disease
Primates (2023)
-
Deconstructing Protein in the Diet and Biomass of Colobine Primates
International Journal of Primatology (2021)
-
Factors influencing riverine utilization patterns in two sympatric macaques
Scientific Reports (2020)
-
Patterns and Constraints of Craniofacial Variation in Colobine Monkeys: Disentangling the Effects of Phylogeny, Allometry and Diet
Evolutionary Biology (2019)
-
Functional decline of sweet taste sensitivity of colobine monkeys
Primates (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.