Abstract
Interactions between individuals of different cetacean species are often observed in the wild. Killer whales (Orcinus orca) can be potential predators of many other cetaceans and the interception of their vocalizations by unintended cetacean receivers may trigger anti-predator behavior that could mediate predator-prey interactions. We explored the anti-predator behaviour of five typically-solitary male sperm whales (Physeter macrocephalus) in the Norwegian Sea by playing sounds of mammal-feeding killer whales and monitoring behavioural responses using multi-sensor tags. Our results suggest that, rather than taking advantage of their large aerobic capacities to dive away from the perceived predator, sperm whales responded to killer whale playbacks by interrupting their foraging or resting dives and returning to the surface, changing their vocal production and initiating a surprising degree of social behaviour in these mostly solitary animals. Thus, the interception of predator vocalizations by male sperm whales disrupted functional behaviours and mediated previously unrecognized anti-predator responses.
Similar content being viewed by others
Introduction
Interspecific interactions across multiple scales (species, population, individual) remain a central question in understanding the organization of animal communities1,2,3. In predator-prey interactions, the importance of predation over evolutionary time (e.g. regulating prey population) has long been clear, but the influence of predation on phenotype (e.g. body size) and behaviour of prey has seen a growing recognition in the last decade. In fact, prey can be affected by predators in other ways than direct (lethal) predation, in the form of stress4, changes in behaviour and anti-predator strategies5. Prey must search for food and reproduce while maintaining the capability to detect and avoid predators. There is thus a constant trade-off for prey between avoiding predation and continuing fitness-enhancing activities such as foraging or mating5,6.
Cetaceans spend most of their time underwater and thus the effect of predator-prey interactions on the animals' behaviour is difficult to observe and rarely studied, especially in comparison to terrestrial animals7. However, the advanced multi-sensor tag developed by Johnson and Tyack (2003) contributed to a major advance in cetacean behavioural studies, enabling reliable monitoring and quantification of the animals' behaviour, even below the sea surface8. Using such technology, it becomes possible to study the effects of predation risk on cetaceans' behaviour.
The two main potential predators of cetaceans are killer whales and sharks9. Both of these predator types are widespread in the world's oceans and have been observed to feed on a large number of prey items including fishes, cephalopods, seabirds, seals and cetaceans. In general, small cetaceans such as dolphins and porpoises are likely more vulnerable to predation than larger cetaceans. Sharks seem not to pose a significant risk to large whales, though small calves of some species may be vulnerable to attacks occasionally9. Killer whales, however, clearly prey on large cetacean species including mysticetes and some odontocetes such as adult female and juvenile sperm whales10,11,12,13. In the last decade, there has been a growing interest in the role of killer whale predation in shaping marine ecosystems and regulating prey population14,15. However, the effect of predation pressure on behaviour and social organization of prey and especially of cetacean prey, remains to be experimentally tested16. Although various observations have reported interspecific predatory or non-predatory encounters between killer whale and other cetaceans, there is a lack of consistency in these observations10,13 and thus no prediction on how cetaceans' behaviour is altered by killer whale presence and how these interactions are mediated.
The sperm whale is the largest toothed whale. A considerable number of observations of lethal and non lethal attacks by killer whale on calves and adult female sperm whales have been reported10,11. Male and female sperm whales are geographically segregated outside of the breeding grounds. Females stay in tropical and subtropical areas in warm surface waters where they live in cohesive groups including adult females and immature males and females. Males leave their family unit at approximately 10 years of age and gradually move to higher latitudes into colder surface waters where they are usually seen solitary17. In their late twenties, males start to return periodically to the breeding grounds inhabited by females to mate18. In female sperm whales, grouping behaviour to defend themselves and their calves has been observed in response to killer whale attacks10,15 and may be the primary function of their sociality11,17,18.
Killer whales are a highly vocal species and their vocalizations can be heard underwater over long distances19. Specific avoidance responses to killer whale sounds have been demonstrated experimentally in fish20,21, pinnipeds22 and few cetacean species23,24,25. As sperm whales hear well at the frequencies of killer whale vocalizations26, the unintended interception of killer whale sounds may inform the eavesdroppers about the presence of predators, enabling them to make adjustments of their behavior27,28.
To our knowledge, no reports exist on how high-latitude male adult sperm whales might respond to predation risk, suggesting that male sperm whales are rarely attacked by killer whales. It's possible that anti-predator strategies exhibited by male sperm whales would be sufficiently powerful to actually discourage killer whales to attack such low benefit prey.
In natural conditions, sperm whales spend 80% of their time foraging and 20% of their time resting29,30,31. To meet their energy requirements, they predate mainly upon cephalopods and have developed a sophisticated and efficient foraging strategy, performing almost continuously long deep foraging dives and producing loud echolocation clicks to localize their prey29,30. If killer whales are perceived as a potential predation risk for male sperm whales, we expect that the detection of predator presence should induce significant changes in the foraging or resting behaviour of sperm whales. Alteration of foraging should be reflected in a change of diving behaviour along with a change in the production of sounds associated with foraging such as clicks and buzzes. Alteration of resting behaviour should be reflected in a change in the stereotypical resting dives pattern31. The solitary nature of adult males leads to the prediction that social defences would not be part of the response repertoire, while their large aerobic diving capacity would enable a diving escape strategy15,16,17. Grouping of male sperm whales is indeed rarely observed in natural conditions15, but a male sperm whale could alert other males in the area to enrol them in a social defence. Such social behaviour should be indicated by the grouping of several whales and the emission of social sounds called ‘codas’32,33.
Our objectives were therefore to investigate whether the detection of mammal-feeding killer whale sounds 1) alters the behaviour (i.e. foraging or resting) of male sperm whales and whether they respond by avoiding the sound source and diving deeper and longer and 2) induces an initiation of social response as an anti-predator strategy. To test what anti-predator strategies are employed by adult male sperm whales, we conducted playback of killer whale vocalizations to five male sperm whales off Norway and monitored the behavioural responses of the animals using a high resolution depth-and-sound-recording tag8.
Results
Overall, the five tested whales changed their behaviour in response to killer whale sounds playbacks (abbreviated as KW) (see Table 1).
Change in foraging/resting behaviour
Change in the dive profile
Four animals of the five tested whales (sw09_141a, sw09_160a, sw10_149a and sw10_140a) were conducting deep foraging dives before the start of KW sounds playback and 1 whale (sw09_142a) was in a resting mode, performing stereotypical resting dives (Table 1).
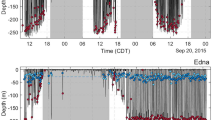
Playback of KW sounds resulted in significantly shorter and shallower dives compared to the baseline (Fig. 1a, b) and none of the whales extended the duration of their dives. For all whales except one (sw09_160a), KW sounds elicited a clear interruption in the descent phase of the dive and a return to the sea surface (Fig. 2a, b, d and e). Two of the four whales that were foraging were also exposed to a broadband noise as negative control (abbreviated as CTRL) (sw10_149a and sw10_150a) and performed apparent similar foraging dives during CTRL playback compared to the baseline (similar duration and max depth) (Fig. 1a, b and Fig. 2d, e). The foraging whale that did not interrupt its dive during KW playback (sw09_160a) performed an unusual foraging dive pattern showing multiple vertical wiggles (i.e. short ascents) during the descent phase of the dive (Fig. 2c). The tagged whale sw09_142a that was in a resting mode preceding the KW sounds exposure stopped resting in response to KW playback (Fig. 2b) and performed a spyhop (raising of the whale's head above the sea surface) just after coming up to the surface.
Time-depth profile of tagged whales sw09_141a (a), sw09_142a (b), sw09_160a (c), sw10_149a (d) and sw10_150a (e) during baseline, CTRL and KW playbacks.
The diving periods highlighted in red correspond to clicking activity, the blue circles represent emission of buzzes and the yellow arrows indicate production of codas. For each surfacing phase, the range of group size is shown between brackets.
Change in regular click production
During baseline periods, the foraging sperm whales spent most of their dive clicking (mean 77 ± 9.4% s.e.m., range: 54.7–94.5%, n = 4). Buzzes were produced for all baseline foraging dives (Fig. 2a, c, d, e). For the whale that was resting during baseline, as expected, the clicking activity was close to zero and there was no buzz emitted during the dives (Fig. 2b).
Clicking activity was significantly lower during KW playback compared to baseline (Fig. 1c) (P = 0.043, Z = 2.022, n = 5) but appeared similar during CTRL playback compared to baseline. Buzz production was not significantly different between KW playback and baseline (Fig. 1d) although for two whales of four that interrupted their foraging dive to return to the surface in response to the KW playback, no buzz was produced during the dive (Fig. 2d, e).
Change in social behaviour (whale grouping and production of codas)
Sperm whales were solitary during the baseline period preceding the playback experiments. During KW playbacks, we found evidence of a social response in three of the five tested whales (Fig. 2a, d, e and Table 1). Two tested whales appeared in association with other individuals at the first surfacing phase following KW playback (Fig. 2a, e). In addition, one of these 2 whales and another tested whale produced codas (Fig. 2a, d). We provide below a descriptive analysis of these behaviours along with Fig. 2 and Table 1.
For sperm whale sw09_141a, the tagged whale was observed solitary at the surface for the 3 h preceding the start of KW playback (group size: 1) (see Fig. 2a). During KW playback, the tagged animal interrupted its foraging dive and returned to the surface (group size: 1). At the first surfacing phase following the end of KW playback, three other whales appeared within 50 m apart from the tagged animal (group size: 1–4 whales). Simultaneously, twelve codas were produced by the group of individuals at the sea surface. At the next surfacing phase of the tagged whale, the animal was observed alone indicating that the group had spread out.
Sperm whale sw10_149a was solitary during the entire period preceding playback experiments. This whale produced one coda at the end of one single baseline dive, just before surfacing (3 h 30 before the start of KW playback) but did not produce any codas during the next baseline dives or during CTRL playback. Then, during KW playback, two codas were produced, one at the max depth of the dive and one when the animal returned to the surface (Fig. 2d). The animal was not observed with other whales and no more than one whale was acoustically detected by the passive acoustics equipment deployed in the water.
The tagged whale sw10_150a was observed as a single animal during baseline and CTRL playback (group size: 1) but then appeared with three more individuals at the first surfacing phase following the end of KW playbacks (group size: 3–4; distance of whales to the focal animal: 0–100 m) (see Fig. 2e). Then, at the next surfacing, the tagged whale appeared solitary again. No coda was recorded during baseline or during sound exposure.
Change in the direction of horizontal movement (avoidance or attraction)
The visual tracks of the surfacing tagged whales indicated that four of the five tested whales (all except sw09_160a) changed their direction of horizontal movement in response to the KW playback. In two cases (sw09_141a and sw10_150a), the whales were originally moving towards the KW sound source but made a clear turn away from the source after they surfaced (avoidance). Sw09_160a did not show any apparent changes in direction of horizontal movement during the KW playback. This subject was moving towards the sound source before the playback started and kept this course during and after the KW playback. The two subjects that were exposed to CTRL playback (sw10_149a and sw10_150a) showed an avoidance response. Sw10_149a was moving towards the sound source before CTRL playback and made a clear turn away from the sound source during the playback. Sw10_150a slightly avoided the CTRL sound source during the playback and went back to its original course after the end of playback.
Responses to CTRL playbacks
The two individuals tested with CTRL playbacks (sw10_149a and sw10_150a) did not show any changes of behaviour except in the direction of horizontal movement (avoidance response). All the other behavioural parameters taken into account in our analyses had apparent similar values between CTRL playback and baseline period (Fig. 1 and Fig. 2d). Dive duration and maximum depth values during CTRL playback were essentially the same as those measured during baseline dives (Fig. 1a, b). Moreover, the rate of click production and number of buzzes indicating foraging activity appeared similar between CTRL playback and baseline (Fig. 1c, d). No indication of social behaviour, i.e., production of codas or association with other whales, was recorded during CTRL playback (Fig. 2d, e).
Discussion
Playbacks that simulated killer whale presence strongly changed the behaviour of five tested male sperm whales. This indicates that sperm whales have perceived the KW sounds in spite of the fact that the main frequencies of KW vocalizations are between 1–2 kHz which is outside the best hearing sensitivity (5–20 kHz) of sperm whales. Moreover, the fact that the two animals exposed to CTRL playback changed their direction of horizontal movement away from the sound source indicates that the whales also perceived the CTRL stimulus.
Animals that were initially foraging during the baseline exhibited significantly shorter and shallower dives compared to the baseline dives and produced less echolocation clicks and buzzes. In general, prey are predicted to alter their behaviour in response to the presence of predators by avoiding them or by reducing risk activities like feeding5,6. The best way for prey to avoid killer whales may be to keep quiet and hide from them by travelling to where the predator cannot follow. In offshore waters, deep diving species such as beaked whales or sperm whales may escape killer whales by going to great depths where killer whales cannot reach them10. Solitary male sperm whales off Norway are not constrained by diving limitations of calves in their group so they could potentially use this strategy of escaping at depth. In contrast to this prediction, our results showed that during KW sounds playbacks, sperm whales interrupted their foraging or resting dive and returned to the surface. The animal's decision to reduce or stop foraging in response to KW playback represents a high energy cost and could be explained by the detection of a potential danger. A possible explanation for the behavioural response of the animals returning to the sea surface may be therefore that animals replenish their oxygen stores in preparation for a potential coming flight or deep avoidance dive if the predator approaches. At the surface, they might also search for additional cues of predator presence34, a possible function of the spyhop observed by one of the tested whales.
In our study, all of the four sperm whales performing deep foraging dives at the start of exposure reduced vocal activity or silenced completely in response to the KW playback. In addition, three of these four whales and the initially resting whale interrupted their dive and returned to the surface, resulting in shorter and shallower dives compared to baseline. The two control experiments with the playback of a broad band noise (CTRL) showing the same frequency range than KW stimulus, verified that animals responded specifically to KW sounds and not to any unspecific acoustic stimuli.
On one hand, our findings are consistent with responses of six right whales (Eubalaena glacialis) exposed to experimental synthetic signals resembling to KW signals that interrupted their foraging dives and returned to the surface35. On the other hand, these results contrast with the typical straight avoidance responses reported in grey whales (Eschrichtius robustus) and belugas (Delphinapterus leucas) in response to KW playbacks23,24. Moreover, a Blainville's beaked whale (Mesoplodon densirostris), which is a deep diver like sperm whales, silenced and broke off the deep dive when exposed to a playback of KW sounds but returned to the sea surface only after an unusually slow ascent from depth25. Therefore, variations across cetacean species in their pattern of anti-predator responses to the detection of killer whale presence do not appear to be solely driven by physiological factors like diving capability.
We showed that in the feeding grounds off Norway, male sperm whales spend most of their time solitary and seldom produce codas, which is in agreement with the literature15,36,37,38. Indeed, male sperm whales were rarely observed socializing with other whales and when it happened, it was usually in cluster of two animals and for no more than few hours15. Moreover, codas, which play a major role in communication between conspecifics, have only rarely been recorded in the Norwegian Sea36,37,38. In contrast to females that live in cohesive groups, solitary males would not have the benefit of communal defence against predators. It has been suggested that as males are bigger than females, they would be less vulnerable to killer whale predation than females. Indeed, all documented predatory interactions between killer whales and sperm whales referred to attacks on group of female sperm whales with calves or wounded animals. However, in the present study, in three of five tested male sperm whales, KW playback induced a social response (clustering of animals at the sea surface and/or production of social/alerting sounds), indicating the initiation of anti-predator social strategy. This suggests that typically solitary males may also exhibit a social defence strategy in response to KW presence.
There have been observations of non-predatory interactions between killer whales and sperm whales or between killer whales and other species, where sometimes the prey ignores the predator10,18. A possible explanation for this absence of cetaceans' reaction facing their predator could be that animals can discriminate between vocalizations of different killer whale ecotypes and perceive as a potential predation risk only mammal-eating KW sounds. Thus, by intercepting KW sounds, prey would be able to assess food preference of killer whales and thus to evaluate the risk of predation. Here, we used unfamiliar mammal-eating KW sounds associated with foraging behaviour that may represent a higher threat for sperm whales compared to other sound types such as local fish-eating KW sounds that would likely pose no threat to them. Playback experiments using different KW sounds are needed to elucidate these questions, as has been explored in seals22.
In summary, we found that the playback of unfamiliar mammal-eating KW sounds simulating potential predator presence modified the behaviour of five tested adult male sperm whale. Although, at the ecological scale, killer whale predation probably has a small role in regulating sperm whale populations15, we show experimental evidence here that it must have at the individual scale a large impact on the behaviour and sociality of animals. Even if the predation risk might be lower for male sperm whales than for females with calves, it still induced a dramatic change in males' behaviour including the interruption of foraging or resting dives. Rather than taking advantage of their large aerobic capacities to dive away from the perceived predator, sperm whales returned quickly to the surface and initiated social responses, suggesting that high-latitude male sperm whales may have previously-unrecognized social interactions.
Methods
General protocol
Field work was conducted aboard a research vessel in the Northern Norwegian Sea during May/June 2009–201039,40. The vessel had a dedicated observation platform from which visual data collection was conducted. A dedicated small motor boat was launched from the vessel for tagging operations and playback experiments. The protocol consisted of the following phases: 1) tagging, 2) start of visual tracking of the tagged animal, 3) collection of baseline behavioural data (before sound exposure), 4) collection of behavioural data during sound exposures, 5) end of visual tracking when the tag released, 6) tag recovery and data download for analysis.
Tagging and visual data collection
To monitor the behavioural response of sperm whales exposed to the playback sounds, we used an advanced miniature high-resolution movement-and-sound-recording tag called D-tag8 (see Supplementary Material). We used the depth sensor and hydrophones on the tag to investigate potential changes in dive pattern and vocal production, respectively, in response to the playbacks. Using the VHF radio beacon on this tag, we were able to visually track the position of the animal at each surfacing event and to simultaneously assess whether the tagged whale was associated to other whales. At each surfacing phase, whale positions were determined at regular 2 min intervals (range: 3–8 recorded positions per surfacing phase) (see Supplementary Material).
Acoustic stimuli
Sperm whales were presented with playback of ‘unfamiliar’ mammal-eating KW sounds previously recorded in the North Pacific using D-tags41 and corresponding to calls and clicks produced during eating activity. We expected these sounds to simulate a high risk of potential predation22. These KW sounds show a frequency range of 0.5–120 kHz with most energy distributed between 1 and 2 kHz which correspond to the fundamental frequency of the majority of the calls42. KW stimuli were transmitted through a speaker designed to broadcast in the 0.20–20 kHz frequency band. A large number of odontocetes are most sensitive in the 30–120 kHz43,44 range whereas sperm whales show best hearing sensitivity between 5 kHz and 20 kHz26 which matches with the frequency range covered by the speaker. Therefore, even if the frequencies of KW sounds above 20 kHz were transmitted by the speaker, sperm whales would probably hardly hear them. Our system of playback was thus appropriate to our species model. To control for sperm whales' reaction to any unspecific acoustic stimulus, two subjects were also exposed to a broad band noise (0.5–15 kHz) as a negative control (CTRL). CTRL stimuli were created from non-calling periods during the recordings from which KW stimuli were taken, amplified to get an average root mean square power equal to the KW stimuli. These CTRL stimuli had most energy between 1 and 2 kHz, like KW stimuli42. All stimuli had 15 min duration and were re-sampled at 44.1 kHz before being used for playback experiments. The average duration of the KW sounds within each 15 min KW stimulus was 7 min 21 sec ± 6 sec s.d. (n = 3). For each stimulus type (KW and CTRL), three different stimulus versions, i.e. collected from different acoustic recordings, were used among the tested whales to avoid pseudoreplication45.
Playback experiments
We tested five male sperm whales encountered off Andenes, Norway. In 2009, three whales were tested with KW stimulus and in 2010, 2 whales were tested with both KW and CTRL stimuli. The baseline period was defined as the period preceding the start of the KW/CTRL playback experiments and had a duration of 5 h 12 min ± 1 h 49 min s.e.m. (n = 5). The three whales tested only with KW stimulus were previously exposed to sonar sounds as part of a parallel project39. For these whales, the baseline period was thus the period between the end of sonar exposure and the start of KW/CTRL playback experiments. Stimuli were transmitted using a M-Audio Microtrack II recorder and amplified by a Cadence Z8000 amplifier connected to a Lubell LL9642T underwater loudspeaker (frequency response: 0.2–20 kHz) submerged at a depth of 8 m46. To later measure the sound level of the source and to ensure that sounds were faithfully played back by the system without distortion, playback stimuli were monitored using a calibrated hydrophone (Bruel & Kjaer 8105 amplified by a Bruel & Kjaer 2635 charge amplifier) placed 1 m from the source and recorded using a M-Audio Microtrack II recorder. The sound level of the KW stimuli ranged from 145 to 151 dBrms re 1 μPa (149 ± 1 dBrms re 1 μPa s.d., n = 3 stimuli) which corresponds to the source level of killer whale vocalizations observed in natural conditions19. The sound level of CTRL stimuli ranged from 145 to 150 dBrms re 1 μPa (147 ± 2 dBrms re 1 μPa s.d., n = 3 stimuli). After the baseline period during which we collected data on normal behavioural patterns, we waited until the tagged whale came up at the surface to then place the motor boat with the playback source at an approximate distance of 800 m from the tagged whale (713 ± 73 m s.e.m., n = 7 playback trials). At the end of a surfacing phase, the whale raises its flukes prior to diving29,30. We started to broadcast a sound stimulus at the end of a surfacing phase, just after fluking. For the two subjects that were first exposed to CTRL stimulus, the KW stimulus was played back after fluking of the next surfacing phase.
Measure of the behavioural response
Quantification of the change in dive behaviour
Vertical movements of the tagged animal were analysed using Matlab software (version 7.8.0). During the pre-exposure period (baseline), four sperm whales were conducting deep foraging dives interspersed with short surface periods and few shallow dives close to the surface29. For the analysis of the foraging dives, we excluded the shallow dives, i.e. dives with max depth <100 m. One whale was in a resting mode during baseline, performing characteristic resting dives (shallow dives <100 m, 20–30 min duration, no clicking31). For all dives performed during baseline and sounds exposure, we measured the dive duration (time from the fluke-up to the following surfacing) and the maximum depth. As an indication of what is a normal dive profile during foraging (n = 4 whales) or resting (n = 1 whale), we calculated for each whale the average of dive duration and max depth for all the dives performed during baseline (9 ± 3 s.d. baseline dives analysed/tagged whale, n = 5). Then, we compared the average baseline to the parameters measured for the dive performed during sound exposure (either KW or CTRL playback).
Quantification of the change in vocal production
Sound files recorded on the D-tags were viewed as spectrograms using Adobe Audition software (Blackman-Harris window; FFT length: 4096; time resolution: 21.3 ms). We identified on spectrograms the production of regular clicks and buzzes (associated with foraging) and codas (associated with socializing behaviour or alarm signalling). For each dive, we summed the duration of regular clicking periods to calculate the proportion of time clicking compared to the total duration of the dive, we scored the number of buzzes and we monitored whether codas were produced.
Social behaviour
Simultaneously with visual recording of the tagged whales' positions, we scored the group size defined as the number of individuals within 200 m of the focal animal and the group spacing defined as the distance between individuals in the group47. Group size and group spacing were compared between the baseline surfacing phases and the surfacing phase occurring right after the end of a stimulus playback (KW or CTRL).
Change in direction of horizontal movement
Potential changes of direction of horizontal movement were investigated using the visual tracks of the tagged whale recorded before, during and after the playbacks.
Statistical analyses
Statistical analyses were performed using Statistica software (version 6.0; StatSoft 2001). To test for differences in dive duration, max depth, proportion of clicking activity during the dive and number of buzzes between baseline (pre exposure) and KW playback exposure period, we used the non-parametric Wilcoxon matched-pair test suitable for small sample size. The sample size for CTRL playbacks was too small though (n = 2) to conduct the test comparison to baseline. A descriptive analysis was performed to assess production of codas and potential changes in direction of horizontal movement, group size and group spacing.
Ethical statement
All experiments comply with the current laws of the country where they were performed. Animal experiments were carried out with permission from the Norwegian Animal Research Authority (Permit No. S-2007/61201). Protocols were approved by the Animal Welfare and Ethics Committee of the University of St Andrews (AWEC, UK) and the Institutional Animal Care and Use Committee (IACUC) of the Woods Hole Oceanographic Institute (WHOI, USA).
References
Agrawal, A. A. et al. Filling key gaps in population and community ecology. Front. Ecol. Environ. 5, 147–152 (2007).
Ings, T. C. et al. Review: Ecological networks - beyond food webs. J. Anim. Ecol. 78, 253–269 (2009).
Golubski, A. J. & Abrams, P. A. Modifying modifiers: what happens when interspecific interactions interact? J. Anim. Ecol. 80, 1097–1108 (2011).
Remage-Healey, L., Nowacek, D. P. & Bass, A. H. Dolphin foraging sounds suppress calling and elevate stress hormone levels in a prey species, the Gulf toadfish. J. Exp. Biol. 209, 4444–4451 (2006).
Lima, S. L. & Dill, L. M. Behavioural decisions made under the risk of predation: a review and prospectus. Can. J. Zool. 68, 619–640 (1990).
Frid, A. & Dill, L. M. Human-caused disturbance stimuli as a form of predation risk. Cons. Ecol. 6 (1), 11 (2002).
Wirsing, A. J., Heithaus, M. R., Frid, A. & Dill, L. M. Seascapes of fear: Evaluating sublethal predator effects experienced and generated by marine mammals. Mar. Mamm. Sci. 24, 1–15 (2008).
Johnson, M. P. & Tyack, P. L. A digital acoustic recording tag for measuring the response of wild marine mammals to sound. IEEE J. Ocean. Eng. 28 (1), 3–12 (2003).
Weller, D. W. Predation on marine mammals. In The Encyclopedia of Marine Mammals (eds. Perrin, W. F., Würsig B. & Thewissen, J. G. M.) 985–994 (Academic Press, San Diego, 2002).
Jefferson, T. A., Stacey, P. J. & Baird, R. W. A review of killer whale interactions with other marine mammals: predation to co-existence. Mamm. Rev. 21, 151–180 (1991).
Pitman, R. L., Ballance, L. T., Mesnick, S. I. & Chivers, S. J. Killer whale predation on sperm whales: observations and implications. Mar. Mamm. Sci. 17, 494–507 (2001).
Ford, J. K. B. et al. Killer whale attacks on minke whales: prey capture and anti-predator tactics. Mar. Mamm. Sci. 21, 603–618 (2005).
Ford, J. K. B. & Reeves, R. R. Fight or flight: anti-predator strategies of baleen whales. Mamm. Rev. 38 (1), 50–86 (2008).
Barrett-Lennard, L. G. & Heise, K. A. The natural history and ecology of killer whales. In Whales, Whaling and Ocean Ecosystems. (eds. Estes, J. A., Demaster, D. P., Doak, D. F., Williams, T. M. & Brownell, R. I.) 163–173 (University of California Press, Berkeley, 2006).
Whitehead, H. Sperm whales in ocean ecosystems. In Whales, Whaling and Ocean Ecosystems. (eds. Estes, J. A., Demaster, D. P., Doak, D. F., Williams, T. M. & Brownell, R. I.) 324–334 (University of California Press, Berkeley, 2006).
Reeves, R. R., Berger, J. & Clapham, P. J. Killer whales as predators of large baleen whales and sperm whales. In Whaling, Oceans and Food Webs. (ed. Estes, J.) 172–186 (University of California Press, Berkeley, 2007).
Whitehead, H. In Sperm Whales: Social Evolution in the Ocean. (ed. Whitehead, University of Chicago Press, Chicago, 2003).
Whitehead, H. et al. Multilevel societies of female sperm whales (Physeter macrocephalus) in the Atlantic and Pacific: why are they so different? Int. J. Primatol. 33 (5), 993–1001 (2012).
Miller, P. J. O. Diversity in sound pressure levels and estimated active space of resident killer whale vocalizations. J. Comp. Physiol. A. 192, 449–459 (2006).
Doksæter, L. et al. Behavioral responses of herring (Clupea harengus) to 1–2 kHz sonar signals and killer whale feeding sounds. J. Acoust. Soc. Am. 125, 554–564 (2009).
Sivle, L. D. et al. Impact of naval sonar signals on herring (Clupea harengus) during summer feeding. ICES. J. Mar. Sci. 69 (6), 1–8 (2012).
Deecke, V. B., Slater, P. J. B. & Ford, J. K. B. Selective habituation shapes acoustic predator recognition in harbour seals. Nature 420, 171–173 (2002).
Cummings, W. C. & Thompson, P. O. Gray whales, Eschrichtius robustus, avoid the underwater sounds of killer whales, Orcinus orca. Fish. Bull. 69, 525–530 (1971).
Fish, J. F. & Vania, J. S. Killer whale, Orcinus orca, sounds repel white whales, Delphinapterus leucas. Fish. Bull. 69, 531–535 (1971).
Tyack, P. L. et al. Beaked Whales Respond to Simulated and Actual Navy Sonar. PLoS ONE 6, e17009 (2011).
Ridgway, S. H. & Carder, D. Assessing hearing and sound production in cetacean species not available for behavioral audiograms: experience with Physeter, Kogia and Eschrichtius. Aquat. Mamm. 27, 267–276 (2001).
Peake, T. Eavesdropping in communication network. In Animal Communication Networks (ed. McGregor, P. K.) 13–37 (Cambridge University Press, New York, 2005).
Blanchet, S., Clobert, J. & Danchin, E. The role of public information in ecology and conservation: an emphasis on inadvertent social information. Ann. NY. Acad. Sci. 1195, 149–168 (2010).
Watwood, S. L., Miller, P. J. O., Johnson, M., Madsen, P. T. & Tyack, P. L. Deep-diving foraging behaviour of sperm whales (Physeter macrocephalus). J. Anim. Ecol. 75, 814–825 (2006).
Miller, P. J. O., Johnson, M. P. & Tyack, P. L. Sperm whale behaviour indicates the use of of echolocation click buzzes ‘creaks’ in prey capture. Proc. R. Soc. B. 271, 2239–2247 (2004).
Miller, P. J. O., Aoki, K., Rendell, L. E. & Amano, M. Stereotypical resting behavior of the sperm whale. Curr. Biol. 18 (1), R21–R23 (2008).
Madsen, P. T. et al. Sperm whale sound production studied with ultrasound time/depth-recording tags. J. Exp. Biol. 205, 1899–1906 (2002).
Frantzis, A. & Alexiadou, P. Male sperm whale (Physeter macrocephalus) coda production and coda-type usage depend on the presence of conspecifics and the behavioural context. Can. J. Zool. 86, 62–75 (2008).
Cresswell, W., Butler, S., Whittingham, M. J. & Quinn, J. L. Very short delays prior to escape from potential predators may function efficiently as adaptive risk-assessment periods. Behaviour 146, 795–813 (2009).
Nowacek, D. P., Johnson, M. P. & Tyack, P. L. Right whales ignore ships but respond to alarm stimuli. Proc. R. Soc. B. 271, 227–231 (2003).
Madsen, P. T. & Mohl, B. Sperm whales (Physeter catodon) do not react to sounds from detonators. J. Acoust. Soc. Am. 107, 668–671 (2000).
Madsen, P. T., Wahlberg, M. & Møhl, B. Male sperm whale (Physeter macrocephalus) acoustics in a high-latitude habitat: implications for echolocation and communication. Behav. Ecol. Sociobiol. 53, 31–41 (2002).
Wahlberg, M. The acoustic behaviour of diving sperm whales observed with a hydrophone array. J. Exp. Mar. Biol. Ecol. 281, 53–62 (2002).
Kvadsheim, P. et al. Cetaceans and naval sonar – the 3S-2009 cruise report. FFI report 2009/01140 (2009). Available: http://rapporter.ffi.no/rapporter/2009/01140.pdf.
Miller, P. J. O. et al. 3S-2010 Baseline Cruise Report: baseline behavior of long-finned pilot whales and sperm whales. (2010).
Miller, P. J. O., Shapiro, A. D. & Deecke, V. B. The diving behaviour of mammal eating killer whales (Orcinus orca L., 1758): variations with ecological not physiological factors. Can. J. Zool. 88, 1103–1112 (2010).
Foote, A. D. & Nystuen, J. A. Variation in call pitch among killer whale ecotypes. J. Acoust. Soc. Am. 123, 1747–1752 (2008).
Au, W. W. L. In The Sonar of Dolphins. ( Springer Verlag, New York, 1993).
Au, W. W. L. Nachtigall, P. E. & Pawloski, J. L. Acoustic effects of the ATOC signal (75 Hz, 195 dB) on dolphins and whales. J. Acoust. Soc. Am. 101, 2973–2977 (1997).
McGregor, P. K. et al. Design of playback experiments. In Playback and Studies of Animal Communication. (ed. McGregor, P. K.) 1–9 (Plenum Press, New York, 1992).
Curé, C. et al. Pilot whales attracted to killer whales sounds: acoustically-mediated interspecific interactions in cetaceans. PLoS ONE 7 (12), e52201 (2012).
Visser, F., Miller, P. J. O., Lam, F. P., Kvadsheim, P. & Tyack, P. Group behaviour sampling protocol for behavioural response studies. In Kvadsheim, P. et al. Behavioural Response Studies of Cetaceans to Naval Sonar Signals in Norwegian Waters - 3S-2011 Cruise Report. FFI-rapport 2011/01289. Available: http://rapporter.ffi.no/rapporter/2011/01289.pdf (2011).
Acknowledgements
Research funding was provided by the US Office of Naval Research, the Royal Norwegian Navy and Ministries of Defence of Norway and the Netherlands. RA was supported by WWF-Norway. CC was supported by Fondation Total and Fondation Bleustein-Blanchet. The authors are grateful to the many researchers and vessel crew members who helped to collect the data. The authors are also grateful to Volker Deecke for providing the D-tag acoustic recordings of killer whale sounds and for his thoughtful comments that improved the manuscript. The authors thank Marie Chalandre for offering her art skills on the whale drawing used in Figure 2.
Author information
Authors and Affiliations
Contributions
All authors conducted the experiments and contributed extensively to the collection of the data. P.H.K. and P.J.O.M. are scientific project leaders. C.C. and P.J.O.M. were responsible for the protocol design of the playback experiments. F.V. elaborated the methodology for data collection on group size and group spacing. C.C. prepared the acoustic stimuli, performed the data analyzes and wrote the manuscript.
Ethics declarations
Competing interests
This study was mainly funded by three naval organizations: the US Office of Naval Research, the Norwegian Ministry of Defence and the Netherlands Ministry of Defence. In addition, WWF-Norway, Fondation TOTAL and the Fondation Bleustein-Blanchet also contributed financially. The funders had no role in study design, data analysis, or preparation of the manuscript. Authors are employed by government (Norwegian Defence Research Establishment), independent no-profit (Netherlands Organization for Applied Scientific Research and Kelp Marine Research), or academic (University of St. Andrews) research organizations. No authors are employed by naval organizations.
Electronic supplementary material
Supplementary Information
Supplementary Material
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/3.0/
About this article
Cite this article
Curé, C., Antunes, R., Alves, A. et al. Responses of male sperm whales (Physeter macrocephalus) to killer whale sounds: implications for anti-predator strategies. Sci Rep 3, 1579 (2013). https://doi.org/10.1038/srep01579
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep01579
This article is cited by
-
On a quasilinear fully parabolic predator–prey model with indirect pursuit-evasion interaction
Journal of Evolution Equations (2023)
-
Friend or foe: Risso’s dolphins eavesdrop on conspecific sounds to induce or avoid intra-specific interaction
Animal Cognition (2022)
-
Global solvability of prey–predator models with indirect predator-taxis
Zeitschrift für angewandte Mathematik und Physik (2021)
-
Fear of Killer Whales Drives Extreme Synchrony in Deep Diving Beaked Whales
Scientific Reports (2020)
-
The effect of underwater sounds on shark behaviour
Scientific Reports (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.