Abstract
As tendon stem/progenitor cells were reported to be rare in tendon tissues, tendons as vulnerable targets of sports injury possess poor self-repair capability. Human ESCs (hESCs) represent a promising approach to tendon regeneration. But their teno-lineage differentiation strategy has yet to be defined. Here, we report that force combined with the tendon-specific transcription factor scleraxis synergistically promoted commitment of hESCs to tenocyte for functional tissue regeneration. Force and scleraxis can independently induce tendon differentiation. However, force alone concomitantly activated osteogenesis, while scleraxis alone was not sufficient to commit, but augment tendon differentiation. Scleraxis synergistically augmented the efficacy of force on teno-lineage differentiation and inhibited the osteo-lineage differentiation by antagonized BMP signaling cascade. The findings not only demonstrated a novel strategy of directing hESC differentiation to tenocyte for functional tendon regeneration, but also offered insights into understanding the network of force, scleraxis and bmp2 controlling tendon-lineage differentiation.
Similar content being viewed by others
Introduction
Tendons are frequently injured during sports and other rigorous activities1. Unfortunately, tendon injuries often lead to suboptimal repair associated with significant dysfunction and disability due to very limited self-repair capability and scar formation1,2,3,4. Therefore, it necessitates the development of therapeutic strategies to functionally repair injured tendons. At present, stem cells and tissue engineering techniques offer great potential for tendon regeneration. We and others have shown that direct implantation of somatic mesenchymal stem cells (MSCs) functionally improves tendon defects. However, it fails to regenerate functional tendons due to their inability to differentiate into mature tenocytes and potential ectopic ossification5,6,7. This indicates that additional cell source with greater regenerative potential and controlled tenogenesis differentiation is needed for tendon repair Fetal tendon has been shown to possess high intrinsic regenerative capacity8, suggesting that tenocyte progenitor at early developmental stage is a superior candidate for tendon regeneration. In this setting, human embryonic stem cells (hESCs) offer great promise as lineage committed progenitors during early development can be derived in vitro indefinitely. We have previously derived MSCs from hESCs by stepwise induction and utilized them for tendon repair7. Although implantation of hESC-MSCs improved the function of injured tendon, the lack of full regeneration in the repaired tendon suggests that hESC-derived MSCs need to further differentiate into a tenocyte lineage for tendon regeneration. To date, researchers have not reported strategies to specifically drive the differentiation of hESCs into a tenocyte fate. The signaling pathways involved in tendon development may potentially be harnessed to direct differentiation of hESCs into this tendon lineage.
Tenocyte development progresses at least through two stages: (i) formation of progenitor cells expressing Scleraxis (SCX), the only direct molecular regulator of tenocyte differentiation identified to date; (ii) tenocyte maturation and tendon formation9,10,11,12. Despite the early expression of SCX in tendon progenitors, ectopic expression of SCX in vivo and overexpression in MSCs in vitro are insufficient to induce tendon formation and maturation10,13,14. It suggests that other factors are required to work synergistically with SCX to drive tendon differentiation. SCX gene knockout has been shown to result in severe force-transmitting tendon defects12, indicating that mechanical stress and SCX may play a synergic role in the tendon development and maturation. Indeed, mechanical stress enhances teno-lineage differentiation of bone marrow derived MSCs into the tenocyte lineage15,16,17,18. However, mechanical stress also causes bone differentiation of tendon stem cells by increasing BMP2 signaling3,19. BMP signaling is transduced by BMP type I receptor which directly phosphorylates and activates receptor regulated Smads (R-Smad) Smad1, Smad5 and Smad8. Activated Smads1/5/8 associate with common mediator Smad4 and the Smad complexes then translocate to the nucleus to activate target gene expression such as Runx220,21. Despite a line of evidence showing that overexpression of a biologically active Smad8 variant promotes tendon differentiation of murine MSCs22, BMP signaling inhibits tendon development by decreasing the formation of progenitor cells in embryonic tissue10,20. During development, BMP signaling has been shown to restrict SCX expression and antagonizing endogenous BMP activity induces ectopic SCX expression10,20, suggesting that BMP signaling is inhibitory to SCX expression. However, it remains unknown how overexpression of SCX may regulate BMP signaling to control the cell fate decision.
In this study, we investigated how combination of SCX and mechanical stress modulates the BMP signaling cascade in hESCs-derived MSCs to induce teno-lineage commitment. We provide the first evidence to demonstrate that SCX overexpression combined with mechanical stimulation synergistically drives differentiation of hESCs to the teno-lineage for tendon tissue engineering. This synergy was achieved by the function of SCX to selectively inhibit osteogenic differentiation of hESCs-MSCs activated by mechanical force through the antagonizing BMP signaling pathway.
Results
Overexpression of SCX augments differentiation of hESCs into tenocyte
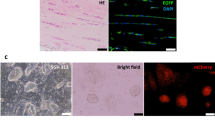
To explore the effect of SCX overexpression on differentiation of hESCs into tenocytes, we firstly generated hESC-MSCs as previously described7 (Fig. 1a, Phase 1). SCX was then overexpressed in hESC-MSCs by infection with lentiviruses carrying SCX or control genes (Fig. 1a, Phase 2). Overexpression of SCX, confirmed by qPCR, GFP expression and western blotting analysis (see Supplementary Fig. 1a–c), induced more hESC-MSCs to develop an elongated morphology with a higher degree of alignment of actin fibers compared to the control (Fig. 1b). Immunostaining further confirmed that both cells express collagen I and tenascin C, the markers of tenocyte20 . A stronger collagen I expression was observed in cells overexpressing SCX compared to the control while tenascin C showed no different (Fig. 1b, c). SCX overexpression augmented collagen deposition and scaffold-free engineered tendon formation (Fig. 1d, n = 22). Moreover, in the engineered tendon, SCX reduced the expression of osteogenesis gene ALP, the transcription factor SOX9 which associated with chondrogenesis23 and mesodermal-related gene markers Snail1 and Snail224 (Fig. 1e). These results suggest that SCX overexpression, consistent with its role in somatic MSCs14 promotes differentiation of hESC-MSCs into tenocyte.
Effect of SCX overexpression on tenocytic differentiation.
(a) Flow chart illustrating experimental design for stepwise induction of hESC into tenocytes through SCX overexpression and mechanical stress. (b) Morphology and immunofluorescence analysis of actin, collagen I and Tenascin-C expression in control and SCX overexpressing cell monolayer at 3 days. Cells were co-stained with DAPI (blue) and images were captured at the same parameters. Scale bar, 100 um (left), 50 um (middle) and 20 um (right). (c) Collagen I and Tenascin-C gene expression of control and SCX overexpressing cell sheet at 3, 7, 11 and 16 days after reaching confluence. Transcripts were normalized to GAPDH, fold over control group at day 0. (d) Percentage of scaffold free engineered tendon formation within 24 wells. (e) SCX reduce bone markers ALP, cartilage markers SOX9 and mesodermal markers snail1 and snail2 expression in engineered tendon (day3 after scaffold free engineered tendon formation). Transcripts were normalized to GAPDH. (**, p<0.01; **, p<0.05).
Combination of SCX overexpression and mechanical stress synergistically promotes tendon maturation in vitro
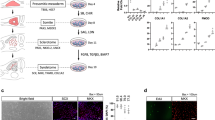
Tenocyte maturation is the second stage of tendon differentiation7,25,26, which can be examined using the in vitro scaffold-free engineered tendon formation model6,7,27. Mechanical stress has been previously shown to increase differentiation of bone marrow derived MSCs into tenocytic lineage15,16,17,18,28,29. This prompted us to examine the synergistic effect of SCX overexpression combined with mechanical stress on tendon maturation of hESC-MSCs in vitro. Mechanical stress alone induced the formation of a compact and sling-like tissue structure from hESC-MSCs (see Supplementary Fig. 2a). Histological staining revealed highly aligned fibrils representative of mature tendon formation (Fig. 2a), which was further confirmed by the histological score (Fig. 2b). Mechanic stress also increased the mean diameters of collagen fiber larger than 22 nm (Fig. 2c), which indicative of mechanical properties and maturation rate of the tendon30. Combination of SCX overexpression and mechanical stress had a synergistic effect on inducing more mature collagen fibril arrangement as shown by polarized light microscopy (Fig. 2a) and increasing collagen fibril diameter by 18% (25.04±1.76 nm vs. 21.2±1.7 nm, p<0.05)compared to the mechanical stress alone (Fig. 2c, 2d). Moreover, SCX together with mechanical stress induced significantly higher frequency of samples to develop more mature collagen fibers (larger collagen diameters and more bands of collagen fibers with crimp pattern25,26,30,31,32) than the control (percentage of mean collagen diameters larger than 22 nm30: 83%:10/12 vs 17%:2/12) after 2 to 3 weeks of mechanical stress application (Fig. 2c and Supplementary Fig. 2b). Furthermore, it caused the formation of more mature collagen fibers with arrangement in the engineered tendon as shown by electron microscopy (Fig. 2c, longitudinal section). Collectively, these results indicated that mechanical stress alone augments hESC-MSC derived tendon maturation. SCX overexpression together with mechanical stress had a synergistic effect on promoting hESC-MSC derived tendon maturation in vitro.
Combination of SCX overexpression with mechanical stress enhanced tendon maturation in vitro.
(a) Histology of the engineered tendons after 1 week of mechanical stimulus. Scale bar, 50 um. (b) Histological score of engineered tendon. (c) Quantitative analysis of collagen fiber diameters of engineered tendons at 3 to 21 days (N = 2 to 5 each group) (d) Ultrastructure and histogram of collagen fiber diameters of engineered tendons at 3 days. SCX and mechanical interaction, *p<0.05; mechanical stress effects #, p<0.05, ##, p<0.01.
SCX overexpression boosts tenocyte maturation activated by mechanical stress through antagonizing BMP signaling mediated osteogenic differentiation
We next investigated the molecular mechanisms underlying the synergistic effect of SCX overexpression and mechanical stress on tendon maturation in engineered tendon. Gene expression analyses were performed on various extracellular matrixes (collagen Ia1, collagen Ia2 and collagen XIV33 , transcription factors Eya2 and surface marker tenomodulin)34,35 that are associated with tendon differentiation. While SCX overexpression alone had little effect on tenocyte gene expression at this stage of tendon differentiation, mechanical stress upregulated the expression of a few genes (Collagen 1a 2 and Eya 2), if not all (Fig. 3a). However, combination of SCX overexpression and mechanical stress synergistically increased the expression of almost all genes examined (Fig. 3a). Immunostaining results further confirmed this observation (Fig. 3b). These results suggest that SCX overexpression may potentiate tenocyte differentiation that is suboptimally activated by mechanical stress to achieve the synergistic effect. To further look into the underlying mechanism, we examined the role of SCX overexpression and mechanical stress in modulating BMP signaling which has been shown to promote osteogenesis and inhibit tenocyte differentiation during development. Mechanical stress upregulated the expression of BMP2, Smad8 and osteogenesis marker osteocalcin (OCN) and this effect was abolished by SCX overexpression9,10,11,12. (Fig. 3c). These results suggested that mechanical stress may activated BMP signaling pathway to promote osteogenesis and SCX overexpression is able to antagonize BMP signaling to inhibit osteogenesis of hESC-MSCs.
Combination of SCX with mechanical stress synergistically promotes tendon differentiation in engineered tendon.
(a) qPCR analysis of the gene expression levels of tenogenic markers Collagen Ia1, Ia2, XIV, Eya2, Epha4 and tenomodulin in engineered tendons under mechanical stress after day3 and day 7 (fold over control group without SCX nor mechanical stress). (b) Immunofluorescence analysis of Eya2, collagen XIV and tenomodulin expression in scaffold-free tendon subjected to mechanical stress for 7 days. Cells are co-stained with DAPI (blue) and images were captured at the same parameters. Scale bar, 50 um. (c) Gene expression of osteocalcin(OCN), Smad8 and BMP2 that are associated with osteogenesis, after day3 and day 7. Transcripts were normalized to GAPDH (fold over control group without SCX nor mechanical stress). (n = 2 to 3 for each group from two independent experiments). SCX and mechanical interaction, *p<0.05, **p<0.01; mechanical effects, #, p<0.05, ##, p<0.01; SCX effects, @, p<0.05, @@, p<0.01.
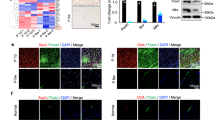
To test this, we treated the cells with recombinant BMP2 and BMP antagonist noggin. While activation of BMP signaling pathway by BMP2 downregulated endogenous SCX expression, noggin induced SCX expression and antagonized the effect of BMP2 (Fig. 4a). Moreover, blocking BMP signaling by noggin induced spindle-shaped morphology of hESC-MSCs (Fig. 4b) and had trend to induce expression of tendon related ECM genes including collagen Ia1, collagen Ia2 and collagen III and transcription factor Six1 (Fig. 4c). These results suggest that activation of BMP signaling is inhibitory to tenocyte differentiation and antagonizing BMP signaling is able to promote differentiation of hESC-MSCs into teno-lineage.
Combination of SCX overexpression and mechanical stress synergistically induces tendon differentiation through differential regulation of BMP.
(a) Endogenous SCX expression of cells treated with BMP2 or Noggin after 12 h to 3 days ( n = 3, *, P<0.05 noggin effects). (b) The morphology of the cells treated with or without noggin. (c) Tenocyte gene expression of engineered tendon with or without noggin treatment after day 3 and day 7 (n = 2, fold over control group without noggin). (d) hESC-MSC, Hela and C3H10T1/2 cells were transiently transfected with BMP2 promoter reporter vector alone, or with increasing amounts of expression vectors encoding scleraxis after 24 h. Luciferase expression levels were normalized to co-transfected rentilia vector. (n> = 3). *, P<0.05 compared to control. (e) Western blot analysis of phosphorylation of Smad1/5/8 on control and SCX overexpressing cells treated with BMPs 1 hour. Beta-actin and Smad1-Smad5-Smad8 served as loading control. Representative results from 3 independent experiments. (f) Immunostaining of Runx2 in control, SCX overexpressing and noggin treated cells after 3 days. Arrows are pointing to cells which are shown at high magnification as inserts at the bottom left corners. (g) Immunostaining of Eya2 in control, SCX overexpressing and noggin treated cells after 3 days. Arrows are pointing to cells which are shown at high magnification as inserts at the bottom left corners. Scale bar, 50 um (f, g).
To determine whether SCX overexpression antagonize BMP signaling, we transiently co-transfected hESC-MSC, C3H10T1/2 and HeLa cell lines with SCX-expressing vector together with a construct that contained BMP2 promoter region fused to luciferase cDNA (pGL3 BMP2). BMP2 promoter activity was downregulated in the presence of SCX in the co-transfected cells (Fig. 4d). Western blot analysis also showed that SCX overexpression was able to attenuate phosphorylated Smad1, Smad5 and Smad8 levels activated by BMP2 and BMP4 treatments (Fig. 4e, Supplementary Fig. 3). Moreover, similar to noggin, SCX overexpression inhibited the nuclear translocation of osteogenesis transcription factor Runx2, the downstream factor of BMP2-Smad signaling, in monolayer cells (Fig. 4f). SCX overexpression increased nuclear translocation of Eya2 and noggin enhanced Eya2 translocation induced by SCX after 3 days treatment (Fig. 4g). Taken together, these data suggested that SCX ovexpression may facilitate tendon differentiation and inhibit osteogenesis through repressing BMP-Smad-Runx2 signaling activated by mechanical stress.
Combination of SCX with mechanical stress enhances ectopic tendon regeneration in nude mice
The effect of SCX overexpression combined with mechanical stress in vitro motivated us to look into ectopic tendon regeneration in vivo. The engineered tendons were implanted subcutaneously into SCID mice for ectopic tendon regeneration. Cell tracking showed that engineered tendons were detected subcutaneously within the mice at least 4 weeks after implantation (see Supplementary Fig. 4a and 4b). The HE and Masson trichrome staining results demonstrated that mechanical stress in situ induced the formation of a dense tissue mass consisting of bands of collagen fibers with small crimp patterns and spindle-shaped tenocytes (Fig. 5). Additionally, mechanical stress in situ led to more collagen maturation within the engineered tendons compared to the control as shown by polarized light microscopy results and longitudinal section of TEM (Fig. 5). More mature collagen fibers were formed in SCX+mech engineered tendon (Fig. 5 polarized light) that has 20% larger collagen fibers (Fig. 5 and Supplementary Fig. 4c, p<0.05). This data thus indicated that SCX expression together with mechanical stress had a synergistic effect on promoting tendon maturation in vivo.
Combination of SCX with mechanical stress enhances ectopic tendon regeneration in nude mice.
(Upper panel) Hematoxylin and Eosin staining of engineered tendons after 4 weeks of mechanical stress. (Second panel) Masson-staining. (Third panel) Polarized light microscopy. (Bottom panels) TEM and histogram of collagen fiber diameters showing the collagen fibers within in vivo engineered tendons in cross and longitudinal sections. Scale bars, 50 um (HE), 200 nm (cross), 500 nm (longitudinal).
Discussion
This study demonstrated the sequential induction of hESC-MSC differentiation into teno-lineage for tendon regeneration by the combination of SCX overexpression and mechanical stress. The induction strategy for mature tendon differentiation was sub-divided into three stages: stage I: Differentiation of hESCs into MSCs; stage II: Initiation of tendon differentiation by SCX overexpression; and stage III: Induction of tendon maturation with mechanical stress together with SCX overexpression. In particular, mechanical stress and SCX overexpression have synergistic effects on tendon maturation. This synergy is achieved by the ability of SCX overexpression to boost tendon differentiation by selectively inhibiting ostoegenensis concurrently activated by mechanical stress through antagonizing BMP2-smad-Runx2 signaling pathway.
During embryonic development, expression of SCX, the earliest known marker of tendon progenitor cells and tenocytes, is highly specific10,11,36,37. However, little is known about the role of SCX in regulating tendon differentiation and maturation. Although a recent study has reported that ectopitc expression of SCX promotes human BM-MSCs into tendon progenitors38, it remains unknown how SCX may regulate tendon differentiation and maturation. Using hESC-MSCs and in vitro engineered tendon as the system to address this question, we revealed that SCX is able to drive the initiation of tendon differentiation from hESC-MSCs as it augmented the synthesis of tendon specific ECM and increased collagen deposition. However, SCX overexpression alone had little effect on the tendon maturation in the in vitro engineered tendon. These results suggest that SCX overexpression drives the initiation of tendon differentiation from hESC-MSCs and it may need combine with secondary signal for tendon maturation. Interestingly, SCX has also been shown to regulate genes associated with bone and cartilage differentiation and development3,9,39,40,41. It suggests that SCX may influence cell fate decision beyond tendon lineage. How SCX may control lineage differentiation other than tendon needs future investigation.
Our results showed that mechanical stress, a well-known inducer for tendon differentiation, promotes tendon maturation of in vitro engineered tendon as shown by collagen fibers with crimp patterns and spindle-shaped cells aligned along the axis of tensile load. On the other hand, previous studies have also demonstrated that dynamic mechanical stress induces osteogenesis by activating BMP signaling3,19. Confirming this observation, we also found that dynamic mechanical stress increased gene expression of BMP signaling components BMP2 and Smad842 and osteogenesis maturation stage gene OCN42 in our engineered tendon model. It may implicated that SCX alone can only suppress early osteogenesis, rather than the late osteogenesis. But SCX can inhibit the end osteo-differentiation effect of mechanical stimulus. It suggests that mechanical stress activated both tenocyte maturation and BMP mediated osteogenesis program. However, activation of BMP signaling is known to inhibit mesenchymal cell developing into tendon progenitor cells during tendon development, whereas blocking BMP signaling by antagonist noggin induces ectopic SCX expression10. Moreover, BMP signaling also inhibits SCX expression and increases cartilage gene expression in the precursor cells of heart valve which has similar structure of the tendon43. Furthermore, activation of BMP signaling causes tendon progenitor to differentiate into osteocytes and results in ectopic bone formation in the tendon10,20. Collectively, these observations suggest that the activation of the BMP signaling induce osteogenesis, which is inhibitory to tendon differentiation. Our results confirmed this observation as activation of BMP signaling downregulated endogenous SCX expression, whereas blockage with noggin promotes tendon differentiation. SCX overexpression, although it is insufficient for inducing tendon maturation by itself, robustly boosted tendon maturation-inducing effect of mechanical stress. Our results demonstrated that, similar to noggin, overexpression of SCX was able to antagonize BMP signaling cascade mediated osteogenesis. Therefore, we revealed a mutual inhibitory interaction between SCX overexpression and BMP signaling. SCX overexpression selectively inhibits osteogenic differentiation of hESC-MSCs activated by mechanical force through antagonizing BMP signaling pathway.
Our results showed that SCX overexpression combined with mechanical stress also synergistically improved tendon regeneration in nude mouse model. Because the size of construct for ectopic tendon regeneration in nude mice is relatively small, it was not possible to do mechanical test in this study. Although this in vivo effect is encouraging, further investigation will be needed to evaluate the synergistic effect on long-term regeneration using additional animal model such as immunodepressed rat model. Despite that it is still preliminary to apply this strategy for functional regeneration of tendon injuries in the clinic, our study provides a proof of concept to establish a novel approach by combining genetic and physical methods to drive lineage specification of hESC-MSCs and has broad applications beyond tendon differentiation and regeneration.
Our results collectively demonstrated that SCX overexpression and mechanical stress are able to synergistically drive tendon lineage differentiation and ectopic tendon regeneration. This synergy is achieved by selective inhibition of SCX overexpression to osteo-lineage differentiation of hESC-MSC induced by mechanical force through antagonizing BMP signaling pathway. Our findings not only established a novel strategy of coming both genetic and physical approaches for directing tenogenic differentiation of hESCs, but also offer new insights into developing novel therapeutics to prevent tendon pathological ossification.
Methods
Cell culture
An undifferentiated NIH-registered human ESC H9 cell line was cultured on mitotically inactivated mouse embryonic fibroblasts and maintained under growth conditions and passaging techniques as described previously44. Mesenchymal differentiation was performed as described in our previous study7. Briefly, a confluent 6-well plate of hESCs was trypsinized for 5 minutes at 37°C, neutralized, centrifuged and resuspended in knockout Dulbecco's modified Eagle's medium (DMEM) (Gibco, Grand Island, NY, http://www.invitrogen.com)45, supplemented with 10% serum replacement medium (Gibco) and 5 ng/ml FGF2 (Gibco) on a gelatinized 10-cm2 plate. Most of the cells in culture appeared fibroblast-like after two passages and the cells were then seeded at very low density (10 cells/cm2) to form colonies. The hESC-derived colonies forming fibroblast-like (CFU-F) cells were designated as hESC-derived mesenchymal stem cells (hESC-MSCs).
Lentiviral production and infection
Human scleraxis gene (NM_001080514.1) was synthetized by Jin-Si-Te co. (Nanjing, Jiangsu, China) and then subcloned into the pleti-Gateway vector for constructing lentivirus (Invitrogen Inc., Carlsbad, CA, USA, http://www.invitrogen.com) and in packaging cell line Hek293T. Lentiviruses carrying SCX-GFP, SCX and their corresponding GFP and lacZ controls were generated in packaging cell line HEK293T to infect hESC-MSCs. After infection, the cells were then selected with blasticin (Invitrogen) at the concentration of 2 ug/ml for 2 weeks. The selection pressure was maintained to generate cells stably expressing SCX/SCX-GFP and control vectors. All the subsequent experiments were performed on cells infected with SCX-GFP and GFP control except that cells transduced with SCX and lacZ were used for immunostaining experiments to avoid confounding issue of GFP with FITC fluorochrome. For simplicity, SCX and control were used in the text unless it was specifically stated. Transduced cells at passage 3 and 5 after selection were utilized for functional studies.
Fabrication of hESC-MSC-engineered tendon in vitro under dynamic mechanical stress
As previously described, hESC-MSCs upon reaching confluence were cultured in high glucose DMEM supplemented with 10% (v/v) FBS and 50 μg/ml ascorbic acid. A multi-layered hESC-MSC cell sheet that formed within 2 to 3 weeks in a 70-cm2 dish was detached from the substratum by applying a small roll-up force. The tissue-engineered constructs were then cultured under uniaxial dynamic tension as previously described46. To apply mechanical stress, the ends of the engineered tendons (40 mm) were anchored at 10% elongation to 44 mm in length. Culture medium was then added, followed by application of dynamic stress (1 HZ, 10% for 2 h/day) with medium change being carried out every other day. Engineered tendons were harvested 3, 7, 14 and 21 days after assembly for TEM, RNA isolation and histological staining.
TEM observation
Tissue specimens were fixed by standard procedures for TEM to assess collagen fibril diameter and alignment47. The samples were then mounted and sputter-coated with gold for viewing under TEM (Quanta 10 FEI). About 250 collagen fibers in vitro and 500 collagen fibrils were measured for each sample in vivo to obtain a true representation of the fibril diameter distribution.
Histological examination
Specimens were immediately fixed in 10% neutral buffered formalin, dehydrated through an alcohol gradient, cleared and embedded in paraffin blocks. Histological sections (7 μm) were prepared using a microtome and subsequently stained with hematoxylin and eosin and Masson trichrome. General histological scoring was performed using hematoxylin and eosin staining. Three parameters (fiber structure, fiber arrangement, rounding of nuclei) were semi-quantitatively assessed of in vitro engineered tendon. The scoring system is a modification of that utilized in a previous study48. These parameters were semi-quantitatively graded into a four-point scale (0–III), with 0 being normal and 3 being maximally abnormal. Therefore, a normal tendon would score 0 and would maximally score 9.
Signaling pathways analysis
Effect of BMP-2 ,4 and noggin on the Differentiation of hESC-MSCs
hESC-MSCs were plated at 4×103 cells/cm2 in a 6-well plate and cultured in complete culture medium until the cells reached confluence. Then the cells were used for signaling pathway analysis. For endogenous SCX gene expression assay, they were incubated in complete culture medium with 30 ng/ml BMP2 and/or 1000 ng/ml noggin for 12 h, 24 h, 72 h49,50, at 37°C, 5% CO2. To determine the phosphorylation of Smad1/5/8, the cells were treated with 30 ng/ml BMP2, BMP4 for 1 h. The immunofluorescence of Runx2 and Eya2 were detected after treated with noggin for 3 days.
Luciferase assays
hESC-MSC, hela and C3H10T1/2 cell lines were seeded in 96-well plates at densities of 3000 and 10000 cells per well. Then cells were co-transfected with the human scleraxis or mouse scleraxis (gift from Olson EN) expressing constructs or the corresponding empty vectors (4 ng to 20 ng ) along with, bmp2-pGL3-Luc (0.1 ug, gift of Rogers MB, New Jersey Medical School) and pRL-Renilla (20 ng, Promega) for 12 h using Lipofectamine (0.3 ul/well). After further culture for 24 h, the cells were finally harvested for dual luciferase activity assays (Promega).
In vivo remodeling of in vitro engineered tendons
In order to determine the potential for ectopic tendon formation by the tissue-engineered tendon under in vivo mechanical loading, a nude mouse model was utilized as previously described7. The experimental protocol was approved by the Zhejiang University Institutional Animal Care and Use Committee (ZJU2010102003). To provide in vivo mechanical loading, the engineered tendon (3 cm) was sutured to the fascia nuchae cranially at the level of the tenth thoracic vertebra and to the fascia of the musculi sacrospinalis at the level of the second sacral vertebra. Thereafter, natural movement of the mouse back provided cyclic mechanical loading on the engineered tendon.
Statistical analysis
All data are expressed as mean ± SD, unless otherwise indicated. Statistical comparison between groups was analyzed by the Student's t-test. The interactive effect of mechanical stress and SCX on tendon differentiation was analyzed using factorial design analysis-of-variance (ANOVA). Values of p<0.05 were deemed to be statistically significant.
References
Butler, D. L. et al. Functional tissue engineering for tendon repair: A multidisciplinary strategy using mesenchymal stem cells, bioscaffolds and mechanical stimulation. J. Orthop. Res. 26, 1–9 (2008).
Lui, P. P., Chan, L. S., Cheuk, Y. C., Lee, Y. W. & Chan, K. M. Expression of bone morphogenetic protein-2 in the chondrogenic and ossifying sites of calcific tendinopathy and traumatic tendon injury rat models. J Orthop Surg Res. 4, 27 (2009).
Rui, Y. F. et al. Mechanical loading increased BMP-2 expression which promoted osteogenic differentiation of tendon-derived stem cells. J. Orthop. Res. 29, 390–396 (2011).
Gulotta, L. V., Chaudhury, S. & Wiznia, D. Stem cells for augmenting tendon repair. Stem Cells Int. 2012, 291431 (2012).
Ouyang, H. W., Goh, J. C., Thambyah, A., Teoh, S. H. & Lee, E. H. Knitted poly-lactide-co-glycolide scaffold loaded with bone marrow stromal cells in repair and regeneration of rabbit Achilles tendon. Tissue Eng. 9, 431–439 (2003).
Ouyang, H. W. et al. Mesenchymal stem cell sheets revitalize nonviable dense grafts: implications for repair of large-bone and tendon defects. Transplantation 82, 170–174 (2006).
Chen, X. et al. Stepwise Differentiation of Human Embryonic Stem Cells Promotes Tendon Regeneration by Secreting Fetal Tendon Matrix and Differentiation Factors. Stem Cells 27, 1276–1287 (2009).
Favata, M. et al. Regenerative properties of fetal sheep tendon are not adversely affected by transplantation into an adult environment. J. Orthop. Res. 24, 2124–2132 (2006).
Brown, D., Wagner, D., Li, X., Richardson, J. A. & Olson, E. N. Dual role of the basic helix-loop-helix transcription factor scleraxis in mesoderm formation and chondrogenesis during mouse embryogenesis. Development 126, 4317–4329 (1999).
Schweitzer, R. et al. Analysis of the tendon cell fate using Scleraxis, a specific marker for tendons and ligaments. Development 128, 3855–3866 (2001).
Brent, A. E., Schweitzer, R. & Tabin, C. J. A somitic compartment of tendon progenitors. Cell 113, 235–248 (2003).
Murchison, N. D. et al. Regulation of tendon differentiation by scleraxis distinguishes force-transmitting tendons from muscle-anchoring tendons. Development 134, 2697–2708 (2007).
Shukunami, C., Takimoto, A., Oro, M. & Hiraki, Y. Scleraxis positively regulates the expression of tenomodulin, a differentiation marker of tenocytes. Dev. Biol. 298, 234–247 (2006).
Alberton, P. et al. Conversion of human bone marrow-derived mesenchymal stem cells into tendon progenitor cells by ectopic expression of scleraxis. Stem Cells Dev. 21, 846–858 (2012).
Altman, G. H. et al. Cell differentiation by mechanical stress. FASEB J. 16, 270–272 (2002).
Butler, D. L. et al. Using functional tissue engineering and bioreactors to mechanically stimulate tissue-engineered constructs. Tissue Eng Part A 15, 741–749 (2009).
Mendias, C. L., Gumucio, J. P., Bakhurin, K. I., Lynch, E. B. & Brooks, S. V. Physiological loading of tendons induces scleraxis expression in epitenon fibroblasts. J. Orthop. Res. 30, 606–612 (2012).
Mendias, C. L., Gumucio, J. P. & Lynch, E. B. Mechanical loading and TGF-beta change the expression of multiple miRNAs in tendon fibroblasts. J. Appl. Physiol. 113, 56–62 (2012).
Eliasson, P., Fahlgren, A. & Aspenberg, P. Mechanical load and BMP signaling during tendon repair: a role for follistatin. Clin. Orthop. Relat. Res. 466, 1592–1597 (2008).
Bi, Y. et al. Identification of tendon stem/progenitor cells and the role of the extracellular matrix in their niche. Nat. Med. 13, 1219–1227 (2007).
Lo, Y. C., Chang, Y. H., Wei, B. L., Huang, Y. L. & Chiou, W. F. Betulinic acid stimulates the differentiation and mineralization of osteoblastic MC3T3-E1 cells: involvement of BMP/Runx2 and beta-catenin signals. J. Agric. Food Chem. 58, 6643–6649 (2010).
Hoffmann, A. et al. Neotendon formation induced by manipulation of the Smad8 signalling pathway in mesenchymal stem cells. J. Clin. Invest. 116, 940–952 (2006).
Yang, L. et al. Rab23 regulates differentiation of ATDC5 chondroprogenitor cells. J. Biol. Chem. 283, 10649–10657 (2008).
Levay, A. K. et al. Scleraxis is required for cell lineage differentiation and extracellular matrix remodeling during murine heart valve formation in vivo. Circ. Res. 103, 948–956 (2008).
Chen, X. et al. Ligament regeneration using a knitted silk scaffold combined with collagen matrix. Biomaterials 29, 3683–3692 (2008).
Frank, C., McDonald, D. & Shrive, N. Collagen fibril diameters in the rabbit medial collateral ligament scar: a longer term assessment. Connect. Tissue Res. 36, 261–269 (1997).
Calve, S. et al. Engineering of functional tendon. Tissue Eng. 10, 755–761 (2004).
Chen, Y. J. et al. Effects of cyclic mechanical stretching on the mRNA expression of tendon/ligament-related and osteoblast-specific genes in human mesenchymal stem cells. Connect. Tissue Res. 49, 7–14 (2008).
Farng, E., Urdaneta, A. R., Barba, D., Esmende, S. & McAllister, D. R. The effects of GDF-5 and uniaxial strain on mesenchymal stem cells in 3-D culture. Clin. Orthop. Relat. Res. 466, 1930–1937 (2008).
Kadler, K. E., Holmes, D. F., Trotter, J. A. & Chapman, J. A. Collagen fibril formation. Biochem. J. 316 (Pt 1), 1–11 (1996).
Redaelli, A. et al. Possible role of decorin glycosaminoglycans in fibril to fibril force transfer in relative mature tendons--a computational study from molecular to microstructural level. J. Biomech. 36, 1555–1569 (2003).
Oryan, A. & Shoushtari, A. H. Histology and ultrastructure of the developing superficial digital flexor tendon in rabbits. Anat. Histol. Embryol. 37, 134–140 (2008).
Young, B. B., Gordon, M. K. & Birk, D. E. Expression of type XIV collagen in developing chicken tendons: association with assembly and growth of collagen fibrils. Dev. Dyn. 217, 430–439 (2000).
Docheva, D., Hunziker, E. B., Fassler, R. & Brandau, O. Tenomodulin is necessary for tenocyte proliferation and tendon maturation. Mol. Cell. Biol. 25, 699–705 (2005).
Qi, J. et al. Differential Expression and Cellular Localization of Novel Isoforms of the Tendon Biomarker Tenomodulin. J. Appl. Physiol. (2012).
Brent, A. E. & Tabin, C. J. FGF acts directly on the somitic tendon progenitors through the Ets transcription factors Pea3 and Erm to regulate scleraxis expression. Development 131, 3885–3896 (2004).
Pryce, B. A. et al. Recruitment and maintenance of tendon progenitors by TGF{beta} signaling are essential for tendon formation. Development 136, 1351–1361 (2009).
Alberton, P. et al. Conversion of Human Bone Marrow-Derived Mesenchymal Stem Cells into Tendon Progenitor Cells by Ectopic Expression of Scleraxis. Stem Cells Dev. (2011).
Blitz, E. et al. Bone ridge patterning during musculoskeletal assembly is mediated through SCX regulation of Bmp4 at the tendon-skeleton junction. Dev. Cell 17, 861–873 (2009).
Espira, L. et al. The basic helix-loop-helix transcription factor scleraxis regulates fibroblast collagen synthesis. J. Mol. Cell. Cardiol. 47, 188–195 (2009).
Furumatsu, T., Shukunami, C., Amemiya-Kudo, M., Shimano, H. & Ozaki, T. Scleraxis and E47 cooperatively regulate the Sox9-dependent transcription. Int. J. Biochem. Cell Biol. 42, 148–156 (2010).
Engler, A. J., Sen, S., Sweeney, H. L. & Discher, D. E. Matrix elasticity directs stem cell lineage specification. Cell 126, 677–689 (2006).
Lincoln, J., Alfieri, C. M. & Yutzey, K. E. BMP and FGF regulatory pathways control cell lineage diversification of heart valve precursor cells. Dev. Biol. 292, 292–302 (2006).
Zhang, S. C., Wernig, M., Duncan, I. D., Brustle, O. & Thomson, J. A. In vitro differentiation of transplantable neural precursors from human embryonic stem cells. Nat. Biotechnol. 19, 1129–1133 (2001).
Barberi, T., Willis, L. M., Socci, N. D. & Studer, L. Derivation of multipotent mesenchymal precursors from human embryonic stem cells. PLoS Med. 2, e161 (2005).
Chen, J. L. et al. Efficacy of hESC-MSCs in knitted silk-collagen scaffold for tendon tissue engineering and their roles. Biomaterials 31, 9438–9451 (2010).
Schormann, W. et al. Tracking of human cells in mice. Histochem. Cell Biol. 130, 329–338 (2008).
Chen, J. M., Willers, C., Xu, J., Wang, A. & Zheng, M. H. Autologous tenocyte therapy using porcine-derived bioscaffolds for massive rotator cuff defect in rabbits. Tissue Eng. 13, 1479–1491 (2007).
Li, J. et al. BMP-2 and CDMP-2: stimulation of chondrocyte production of proteoglycan. J. Orthop. Sci. 8, 829–835 (2003).
Chen, X. D., Fisher, L. W., Robey, P. G. & Young, M. F. The small leucine-rich proteoglycan biglycan modulates BMP-4-induced osteoblast differentiation. FASEB J. 18, 948–958 (2004).
Acknowledgements
This work was supported by NSFC grants (81101355, 81071461, 81125014, 31000440, J0830833, J1103603), The National Key Scientific Program (2012CB966600). China Postdoctoral Science Foundation (20100481417, 201104723), Zhejiang Province Grants (Z2100086), Zhejiang Provincial Program for the Cultivation of High-level Innovative Health talents, the Foundation of Zhejiang Provincial Key Medical Discipline (Medical Tissue Engineering), Zhejiang Innovation Program for Graduates(YK2010013), Zhejiang Provincial Key Laboratory of Tissue Engineering and Regenerative Medicine, Fundamental Research Funds for the Central Universities, International Science & Technology Cooperation Program of China (Grant No. 2011DFA32190 and Scholarship Award for Excellent Doctoral Student granted by Ministry of Education. The authors thank Boon Chin Heng for the manuscript preparation, Wang Li for the TEM imaging and Xiao Guifeng for the confocal imaging.
Author information
Authors and Affiliations
Contributions
CX and YZ design the experiments, collection and/or assembly of data, data analysis and interpretation, prepared all figures and wrote the main manuscript text. CJL, SWL, LHH, TQM and FZ: Collection and/or assembly of data, animal maintenance, assistance with in vivo experiments and collection of tissue samples, paraffin-embedded tissue sections. LLL: data analysis and interpretation. JJF and OYHW: Conception and design of experiments, financial support, data analysis and interpretation and revise the main manuscript text. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Supplementary Information
Supplementary
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareALike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Chen, X., Yin, Z., Chen, Jl. et al. Force and scleraxis synergistically promote the commitment of human ES cells derived MSCs to tenocytes. Sci Rep 2, 977 (2012). https://doi.org/10.1038/srep00977
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep00977
This article is cited by
-
LSD1-mediated demethylation of OCT4 safeguards pluripotent stem cells by maintaining the transcription of PORE-motif-containing genes
Scientific Reports (2021)
-
Grafting of iPS cell-derived tenocytes promotes motor function recovery after Achilles tendon rupture
Nature Communications (2021)
-
Cyclically stretched ACL fibroblasts emigrating from spheroids adapt their cytoskeleton and ligament-related expression profile
Cell and Tissue Research (2021)
-
Using Tools in Mechanobiology to Repair Tendons
Current Tissue Microenvironment Reports (2020)
-
The chemokines CXCL12 and CXCL14 differentially regulate connective tissue markers during limb development
Scientific Reports (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.