Abstract
Many animals can detect the taste of calcium but it is unclear how or whether humans have this ability. We show here that calcium activates hTAS1R3-transfected HEK293 cells and that this response is attenuated by lactisole, an inhibitor of hT1R3. Moreover, trained volunteers report that lactisole reduces the calcium intensity of calcium lactate. Thus, humans can detect calcium by taste, T1R3 is a receptor responsible for this and lactisole can reduce the taste perception of calcium by acting on T1R3.
Similar content being viewed by others
Introduction
Many animals can satisfy their physiological need for calcium by locating and consuming calcium salts1,2. This “calcium appetite” is controlled by taste: Concentrated calcium salts are rejected by nutritionally replete animals but avidly ingested by calcium-deprived ones3,4. The mechanism involves changes in oral sensitivity to calcium3,4,5,6 but how calcium is detected is unclear. There is evidence, albeit incomplete, for calcium taste receptors in amphibians and rodents2,7,8,9,10,11 but it is unknown whether humans can detect calcium per se rather than as a combination of, for example, bitter and sour taste components12.
One receptor implicated in the detection of calcium taste by mice is T1R313. Polymorphisms in Tas1r3, the gene encoding T1R3, are associated with the calcium preferences of 40 inbred mouse strains and Tas1r3 knockout mice are indifferent to concentrations of CaCl2 and calcium lactate that wild-type littermates avoid13. Moreover, the electrophysiological response of the chorda tympani nerve elicited by oral calcium is less pronounced in Tas1r3 knockout mice than wild-type littermate controls13. But despite this evidence, T1R3 seems an unlikely candidate to be a calcium taste receptor because it has well-established roles as a detector of sweet and umami (monosodium glutamate) tastes14,15. Critically, the taste intensities of most sweeteners and monosodium glutamate are inhibited by lactisole [sodium 2-(4-methoxyphenoxy)propanoate], which interacts with the transmembrane domain of human T1R3 (hT1R3)15,16,17. This raises the possibility of using lactisole to test whether T1R3 influences calcium taste. To accomplish this, we first determined whether hT1R3 was sensitive to calcium and lactisole in vitro. We then asked human volunteers to rate the calcium intensity of calcium lactate and other taste solutions, with or without added lactisole.
Results
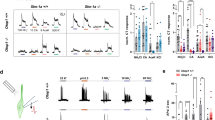
HEK293 cells transiently transfected with hT1R3 and a chimeric G-protein, Gα16-gust44, responded to calcium (but not magnesium) in a dose-dependent manner (EC50 = 53 mM; Fig. 1a). Cells transfected with Gα16-gust44 but not T1R3 were completely unresponsive to either mineral (not shown). The response to calcium was inhibited by lactisole (EC50 = 3 mM; Fig. 1b). This was not simply a nonspecific inhibitory effect on cellular responses because the response to carbachol was unaffected by lactisole (Fig. 1c).
Intracellular calcium responses of hT1R3-transfected HEK293 cells.
a. Cells transfected with hT1R3 respond to extracellular CaCl2 in a dose-dependent manner (EC50 = 53 mM) but not to MgCl2. Cells transfected with Gα16-gust44 alone are unresponsive to either mineral (not shown). The responses to CaCl2 are markedly reduced in the presence of 8 mM lactisole (EC50 = 25 mM). b. Lactisole produces a dose-dependent inhibition of the response of T1R3-transfected cells to 75 mM calcium (EC50 = 3 mM). c. Lactisole does not affect the response of T1R3-transfected cells to the muscarinic agonist, carbachol. d. Calcium activates cells transfected with hT1R3 (EC50 = 53 mM) and both hT1R2+hT1R3 (EC50 = 67 mM) but not hT1R2 alone.
T1R3 is a shared subunit of both the sweet taste receptor (T1R2+T1R3) and umami taste receptor (T1R1+T1R3). Consistent with earlier characterizations of the T1R3 receptor18,19,20, HEK293 cells expressing hT1R3 alone did not respond to sweeteners or to monosodium glutamate (data not shown). However, hT1R3 could account entirely for the response of dual-transfected cells to calcium (Fig 1a,b and d): Transfection with hT1R3 alone or both hT1R3 and hT1R2 supported the response whereas transfection with hT1R2 alone did not (Fig. 1d).
Human subjects were trained to recognize calcium by taste and then were asked to rate various taste solutions. They rated calcium lactate solution as having a predominant calcium taste with minor bitter and sour intensities. Lactisole decreased the intensity of the calcium taste (Fig. 2). It did this despite simultaneously increasing calcium lactate's sourness, bitterness, saltiness and overall intensity (Supplementary Table 1). As expected, lactisole significantly reduced the overall intensity and sweetness intensity of sucrose14,15 and decreased the sweetness intensity of urea. It also caused slight but significant increases in the bitter, umami and calcium intensities of sucrose. Consistent with earlier work, lactisole did not influence the taste perception of citric acid, NaCl, quinine hydrochloride or MSG+IMP14 (Supplementary Table 2).
Influence of 8 mM lactisole on the calcium intensity of various taste solutions, including three concentrations of calcium lactate (CaLa).
*p<0.05 relative to same taste compound without lactisole. “Calcium intensity” is the judged intensity of the calcium component of the taste solutions, measured using a general linear magnitude scale (see text). Ratings of the intensity of other components (sweet, sour, salty, bitter and umami) of each taste solution are provided in Supplementary Tables 1 and 2.
Discussion
These results, together with evidence that T1R3 is a calcium taste receptor in mice13, suggest that humans can detect calcium by taste, that T1R3 is a receptor responsible for this and that lactisole can reduce the taste perception of calcium by acting on T1R3. The results are consistent with what little is known about the molecular mechanisms of calcium detection. A calcium-binding pocket has been delineated in the Venus fly trap region of the calcium-sensing receptor, CaSR and the same residues are conserved in T1R321. Lactisole interacts with the transmembrane domain of T1R3, restricting the molecule to its ground state conformation and thus inhibiting sweet and umami taste15,22. It appears reasonable that the same mechanism is responsible for lactisole's effects on calcium taste.
T1R3 is unlikely to be the only receptor involved in the detection of calcium salts by humans. Our subjects were trained to recognize calcium specifically but, even so, they rated calcium lactate as having bitter and sour components. Although lactisole reduced the calcium intensity of calcium lactate it also increased its sourness, saltiness, bitterness and overall intensity. This probably reflects disinhibition of the lactate anion from mixture suppression, allowing it to exert a greater effect on intensity than when accompanied by the calcium cation23. The concentration of lactisole required to effectively inhibit calcium (and also umami taste) was higher than that required to inhibit sweetness14,15. Perhaps this is because sweet taste detection depends entirely on T1R3 (dimerized with T1R2) whereas calcium and umami taste compounds recruit additional receptors that are insensitive to lactisole. The existence of additional receptors can also explain why lactisole's effect on calcium taste intensity was largest with the highest concentration of calcium lactate we tested and the effect was a reduction but not an elimination of calcium taste intensity. One possibility is that lactisole blocks the action of T1R3 but leaves active CaSR, the calcium-sensing receptor, which may function as a taste receptor7,8,10,23.
T1R3 is a detector of sweet and umami tastes so the present findings raise the question of why calcium is not perceived as being sweet or umami-like. There is no confusion between calcium taste and sweet or umami taste (Fig. 2) and no evidence for an interaction between calcium and the sweet or umami taste qualities (Supplementary Table 2). Perhaps there is a subpopulation of taste cells that are armed with T1R3 and wired specifically as calcium-sensitive. We have speculated that T1R3 in these cells dimerizes with CaSR or another receptor13 although the finding made here that T1R3 can detect calcium by itself (Fig. 1) suggests that a T1R3 homodimer is responsible.
Human T1R3-transfected HEK293 cells did not respond to magnesium, which contrasts with results we have found previously with mice13: Unlike wild-type controls, Tas1r3 knockout mice do not avoid MgCl2. We have also found that (a) lactisole does not affect intensity ratings of magnesium chloride solutions made by humans (unpublished results) and (b) HEK293 cells transfected with mouse (as opposed to human) T1R3 respond readily to MgCl2 (see supplementary material, Figure 1). Thus, we suspect that there is a species difference in the response of T1R3 to magnesium. Of course, species differences in the response of T1R3 are not without precedent; there are well-known differences in the response of the human and rodent forms of T1R3 to lactisole and to artificial sweeteners (e.g.15,24).
Our focus here is on T1R3 as a taste receptor but T1R3 has also been identified in the gastrointestinal tract25 so it may signal the presence of high concentrations of calcium in the stomach or intestines. T1R3 is also found in human pancreas and liver26 but it is unlikely to act as a postabsorptive calcium sensor because, at least under our experimental conditions, the lowest calcium concentrations to activate T1R3-mediated responses were well above the 1–2 mM concentrations found in blood.
We are often asked whether calcium is a basic taste, akin to sweet, sour, salty, bitter and umami. Our demonstration of a receptor in the oral cavity fulfills a universally accepted criterion for a basic taste but there is little consensus about what other criteria must be met. For example, is a specific taste quality or a central representation required? The “basic taste” concept is under fire27,28. It cannot deal well with observations that the tastes of complex carbohydrates are bland to humans but avidly preferred by many other species27, that dozens of receptors all produce a unitary sensation of bitterness28,29, or that there is apparently no region encoding sour taste in primary taste cortex30. Without a better definition we cannot determine whether calcium is a basic taste. Nevertheless, our results showing that calcium and lactisole interact with T1R3 establish T1R3 as a calcium taste receptor and consequently provide the first evidence for calcium taste transduction in the human oral cavity.
Methods
Heterologous expression and assay of hT1R3
HEK293 (peakRapid) cells were obtained from ATCC (CRL-2828) and cultured at 37°C in Dulbecco's modified Eagle's medium supplemented with 10% fetal bovine serum. Human T1R2, T1R3 and Gα16-gust44 clones were as described previously15. For transient transfection, cells were seeded onto 96-well plates coated with poly-lysine at 40,000 cells per well in optiMEM (Invitrogen no. 51985) supplemented with 5% dialyzed fetal bovine serum. Cells were transfected with T1R2 and T1R3 either singly or in combination, along with Gα16-gust44. Equal amounts of each cDNA were transfected, totaling 0.2 µg per well. After 24 h, media were replaced with fresh OPTIMEM supplemented with 5% FBS. After another 24 h, the cells were washed with Hank's balanced salt solution (HBSS) and loaded with 50 µl of 3 µm Fluo-4 in HBSS and incubated for 2 h. Then the cells were washed three times with HBSS and left in HBSS. The dye-loaded transfected cells in plates were placed into a Molecular Devices FlexStation III system to monitor the change in fluorescence (excitation, 488 nm; emission, 525 nm; cutoff, 515 nm) after the addition of 50 μl Dulbecco's phosphate-buffered saline (DPBS) mixed with 50 µl taste solution at twice the required concentration. For each trace, a taste solution (with or without lactisole) was added 30 s after the start of the scan, scanning continued for an additional 150 s and data were collected every 2 s.
Intracellular calcium mobilization was quantified as the percentage of change (peak fluorescence - baseline fluorescence level, denoted as ΔF) from its own baseline fluorescence level (denoted as F). Peak fluorescence intensity occurred about 20–30 s after the addition of taste solutions. As controls, buffer alone or compounds that do not taste sweet evoked no change of fluorescence (ΔF/F ≈ 0, S.E. is about 1%). The data were expressed as the mean ± S.E. of the ΔF/F value of three independent samples. Curve-fitting was carried out using Graph-Pad Prism 5.0 (GraphPad Software, Inc.).
Taste Tests
The procedures were modeled on those used by Galindo-Cuspinera and Breslin14 to study the effect of lactisole on umami taste. We tested 8 male and 8 female subjects, aged 25 – 65 yr. Informed consent was obtained from each subject and the protocol was approved by Institutional Review Board 4 of the University of Pennsylvania (Protocol no. 812064). Each subject attended five ∼1 h sessions, held on different days. In the first session, subjects were trained to use 96-mm general linear magnitude scales (gLMSs) that were anchored with the term “strongest imaginable sensation of any kind” at the top and “no sensation” at the bottom. Initially, subjects provided ratings of ten imagined situations (e.g., “The pain from biting your tongue”) to familiarize them with the range and use of the multimodal gLMS. They then repeatedly rated samples of 300 mM sucrose, 3 mM citric acid, 150 mM NaCl, 0.05 mM quinine hydrochloride (QHCl), 300 mM urea, 100 mM monosodium glutamate + 50 mM inosine-5′ monophosphate (MSG + IMP) and 100 mM calcium lactate on seven gLMSs, provided on a single 11 x 8.5 inch page, with the scales titled “Intensity”, “Sweet”, “Salty”, “Sour”, “Bitter”, “Umami” and “Calcium”. Subjects were considered to be trained when ratings of the taste compounds were consistently made in the weak-to-moderate range of the pertinent scale.
In the following four sessions, the subjects were asked to rate each of the seven solutions they received during training, plus water and two additional solutions: 56 and 177 mM calcium lactate. Each taste compound was presented once alone and once in a mixture with 8 mM lactisole, so there were a total of 20 samples tested per session. Subjects rinsed with water four times before starting. They tasted a 10-mL sample for 5 s (timed by a handheld timer), expectorated, made all seven gLMS ratings and then rinsed four times during a timed 75-s interstimulus interval. For two of the sessions, the subjects wore nose clips; for the other two, they did not. We counterbalanced the order of sample presentation within tests and the use of nose clips between tests. We used calcium lactate in preference to CaCl2 or other calcium salts because it is soluble and has been used historically in animal studies of calcium taste.
Separate statistical analyses were conducted on the intensity ratings of each taste type. The ratings were analyzed by within-subject (dependent) t-tests, or for the three concentrations of CaLa by within-subject two-way ANOVA with factors of Concentration and Lactisole. Differences between pairs of means were ascertained using post hoc LSD tests. Preliminary analyses found no differences in ratings made by male and female participants so gender was not included as a factor in analyses. There were no concerted differences among the four test sessions, including no difference in ratings made with or without nose clips, so the analyses presented here are based on average scores. The criterion for significance of all statistical tests was p< 0.05. Values given in the text and shown in the figures are means ± standard errors of the mean.
References
Schulkin, J. Calcium hunger (Cambridge University Press, Cambridge, 2001).
Tordoff, M. G. Calcium: taste, intake and appetite. Physiol. Rev. 81, 1567–1597 (2001).
Inoue, M. & Tordoff, M. G. Calcium deficiency alters chorda tympani nerve responses to oral calcium chloride. Physiol. Behav. 63, 297–303 (1998).
McCaughey, S. A., Forestell, C. A. & Tordoff, M. G. Calcium deprivation increases the palatability of calcium solution in rats. Physiol. Behav. 84, 335–342 (2005).
McCaughey, S. A. & Tordoff, M. G. Calcium-deprived rats sham-drink CaCl2 and NaCl. Appetite 34, 305–311 (2000).
McCaughey, S. A., Forestell, C. A. & Tordoff, M. G. Calcium deprivation increases the palatability of calcium solutions in rats. Physiol. Behav. 84, 335–342 (2005).
Okada, Y. et al. A calcium-receptor agonist induces gustatory neural responses in bullfrogs. Cell. Molec. Neurobio. 27, 771–781 (2007).
San Gabriel, A., Uneyama, H., Maekawa, T. & Torii, K. The calcium-sensing receptor in taste tissue. Biochem. Biophys. Res. Comm. 378, 414–418 (2009).
Tordoff, M. G., Reed, D. R. & Shao, H. Calcium taste preferences: Genetic analysis and genome screen of C57BL/6J x PWK/PhJ hybrid mice. Genes Brain Behav. 7, 618–628 (2008).
Bystrova, M. F., Romanov, R. A., Rogachevskaja, O. A., Churbanov, G. D. & Kolesnikov, S. S. Functional expression of the extracellular-Ca2+-sensing receptor in mouse taste cells. J. Cell Sci. 123, 972–982 (2010).
Ohsu, T. et al. Involvement of the calcium-sensing receptor in human taste perception. J. Biol. Chem. 285, 1016–1022 (2009).
Henning, H. Die qualitatenreihe des geschmaks. Z. Psychol. 74, 203–219 (1916).
Tordoff, M. G. et al. Involvement of T1R3 in calcium-magnesium taste. Physiol. Genomics 34, 338–348 (2008).
Galindo-Cuspinera, V. & Breslin, P. A. The liaison of sweet and savory. Chem. Senses 31, 221–225 (2006).
Jiang, P. et al. Lactisole interacts with the transmembrane domains of human T1R3 to inhibit sweet taste. J. Biol. Chem. 280, 15238–15246 (2005).
Lindley, M. G. Method of inhibiting sweetness (ed. United States Patent) (1986).
Schiffman, S. S., Booth, B. J., Sattely-Miller, E. A., Graham, B. G. & Gibes, K. M. Selective inhibition of sweetness by the sodium salt of +/-2-(4-methoxyphenoxy)propanoic acid. Chem. Senses 24, 439–447 (1999).
Li, X. et al. Human receptors for sweet and umami taste. Proc. Natl. Acad. Sci. USA 99, 4692–4696 (2002).
Nelson, G. et al. Mammalian sweet taste receptors. Cell 106, 381–390 (2001).
Li, X. T1R receptors mediate mammalian sweet and umami taste. Am J Clin Nutr 90, 733S–737S (2009).
Silve, C. et al. Delineating a Ca2+ binding pocket within the venus flytrap module of the human calcium-sensing receptor. J. Biol. Chem. 280, 37917–37923 (2005).
Xu, H. et al. Different functional roles of T1R subunits in the heteromeric taste receptors. Proc. Natl. Acad. Sci. USA 101, 14258–14263 (2004).
Keast, R. S. J. & Breslin, P. A. S. An overview of binary taste-taste interactions. Food. Qual. Pref. 14, 111–124 (2003).
Jiang, P. et al. Identification of the cyclamate interaction site within the transmembrane domain of the human sweet taste receptor subunit T1R3. J. Biol. Chem. 280, 34296–34305 (2005).
Margolskee, R. F. et al. T1R3 and gustducin in gut sense sugars to regulate expression of Na+-glucose cotransporter 1. Proc. Natl. Acad. Sci. USA (2007).
Taniguchi, K. Expression of the sweet receptor protein, T1R3, in the human liver and pancreas. J Vet Med Sci 66, 1311–1314 (2004).
Sclafani, A. The sixth taste? Appetite 43, 1–3 (2004).
Cxhandrashekar, J. et al. T2Rs function as bitter taste receptors. Cell 100, 703–711 (2000).
Meyerhof, W. et al. The molecular receptive ranges of human TAS2R bitter taste receptors. Chem Senses 35, 157–170 (2010).
Chen, X., Gabitto, M., Peng, Y., Ryba, N. J. & Zuker, C. S. A gustotopic map of taste qualities in the mammalian brain. Science 333, 1262–1266 (2011).
Acknowledgements
The authors thank Drs. R. Margolskee and D. Reed for insightful comments on earlier drafts of the manuscript. This work was supported by the National Institute of Diabetes and Digestive and Kidney Diseases at the National Institutes of Health [grant number DK-46791]. Cell culture was assisted by the Monell Histology and Cellular Localization Core, which is supported, in part, by funding from the NIH-NIDCD Grant P30 DC-11735. The funders had no role in study design, data collection or analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
MGT and PJ conceived and designed the experiments. SV and PJ performed the cell assay experiments. LA performed the human tests and preliminary data analyses. MGT conducted statistical analyses and wrote the paper.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Supplementary Information
Supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareALike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Tordoff, M., Alarcón, L., Valmeki, S. et al. T1R3: A human calcium taste receptor. Sci Rep 2, 496 (2012). https://doi.org/10.1038/srep00496
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep00496
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.