Abstract
Rice blast caused by Magnaporthe oryzae is a devastating disease of rice. Mechanisms of rice resistance to blast have been studied extensively and the rice–M. oryzae pathosystem has become a model for plant–microbe interaction studies. However, the mechanisms of non-host resistance (NHR) to rice blast in other plants remain poorly understood. We found that penetration resistance to M. oryzae in multiple mutants, including pen2 NahG pmr5 agb1 and pen2 NahG pmr5 mlo2 plants, was severely compromised and that fungal growth was permitted in penetrated epidermal cells. Furthermore, rice Pi21 enhanced movement of infection hyphae from penetrated Arabidopsis epidermal cells to adjacent mesophyll cells. These results indicate that PEN2, PMR5, AGB1 and MLO2 function in both penetration and post-penetration resistance to M. oryzae in Arabidopsis, and suggest that the absence of rice Pi21 contributed to Arabidopsis NHR to M. oryzae.
Similar content being viewed by others
Introduction
Rice is a staple crop of economic importance in many countries. One of the most serious and widespread rice diseases is blast, which is caused by the ascomycete fungus Magnaporthe oryzae. The mechanisms underlying rice resistance to blast have been studied extensively and the rice–M. oryzae pathosystem has become a model for the study of plant–microbe interactions because both whole genome sequences and functional genomic approaches are available1,2,3,4,5. The infection of rice by M. oryzae follows a developmental process that occurs in many foliar fungal pathogens. A germ tube produced from the conidium differentiates into a specialized infectious structure called the appressorium, which adheres tightly to the plant surface with mucilage. The fungus generates massive turgor pressure inside the melanized appressorium, forcing a narrow penetration peg through the host surface and allowing the entry of the fungus into a leaf epidermal cell6. After penetration, the peg differentiates into bulbous and lobed infectious hyphae that grow intra- and intercellularly.
Most plants are immune to the majority of potential pathogens and are susceptible to only a few adapted microbes. Consequently, disease is the exception rather than the rule. Disease resistance in all members of a plant species to all genetic variants of a non-adapted pathogen species is the most common form of plant immunity, which is termed non-host resistance (NHR)7,8. Although NHR represents the most common and durable form of plant resistance in nature, it is poorly understood at the molecular level.
Arabidopsis mutants with altered non-host interactions following Blumeria graminis hordei (Bgh) infection were described recently and three genes were identified: PENETRATION 1 (PEN1), PEN2 and PEN39,10,11,12. PEN1 encodes a plasma membrane–anchored syntaxin with a soluble N-ethylmaleimide-sensitive factor attachment protein receptor (SNARE) domain9. PEN2 encodes an atypical myrosinase involved in glucosinolate metabolism in defense responses10,13,14. PEN3 encodes a pleiotropic drug resistance (PDR) ATP-binding cassette (ABC) transporter11,12. Collectively, these studies demonstrate that Arabidopsis NHR to non-adapted biotrophic powdery mildews has two successive and multicomponent defense layers: pre- and post-invasion resistance. Notably, PEN2 and PEN3 contribute to both stages of resistance10,11. Moreover, enhanced disease susceptibility 1 (EDS1), phytoalexin-deficient 4 (PAD4) and senescence-associated gene 101 (SAG101) are factors in post-invasion resistance10.
The Arabidopsis pen2 mutant also shows a significantly elevated M. oryzae penetration ratio15,16,17. Furthermore, PMR5 and AGB1 are positive regulator factors for penetration resistance in the Arabidopsis–M. oryzae interaction15. Broad-spectrum resistance to adapted powdery mildews is conferred by loss-of-function mutant alleles of MILDEW RESISTANCE LOCUS O (MLO) genes in barley and Arabidopsis. MLO encodes a plant-specific family of integral membrane proteins. Barley plants carrying a mutation in the MLO locus, which confers a durable resistance to powdery mildew, are hypersusceptible to the rice blast fungus Magnaporthe oryzae18 . Rice pi21 is a recessive gene conferring durable resistance to blast disease; it encodes a loss-of-function mutation in a cytoplasmic proline-rich protein consisting of a putative heavy metal–binding domain and putative protein–protein interaction motifs. The rate of hyphae penetration from penetrated cells into adjacent cells, which is an indicator of hyphal growth, is significantly lower in pi21 plants than in Pi21 plants, suggesting that the susceptible Pi21 allele negatively regulates resistance19. Despite the identification of these factors, their roles in NHR to M. oryzae are unknown.
Here, we report the NHR genetic interactions revealed by our examination of the mechanisms operating in Arabidopsis. We found PEN2, PMR5, AGB1 and MLO2 to be involved in several steps of NHR to M. oryzae. Moreover, we demonstrated that rice Pi21 enhanced movement of M. oryzae infection hyphae from penetrated Arabidopsis epidermal cells to adjacent mesophyll cells.
Results
The roles of PMR5 and AGB1 in Arabidopsis NHR to M. oryzae
We recently demonstrated that PMR5 and AGB1 contributed to Arabidopsis resistance to penetration by M. oryzae, indicating that a genetic network regulated this resistance15. In the present study of the genetic network, we generated pen2 NahG pmr5, pen2 NahG agb1, pen2 pmr5 agb1 and pen2 NahG pmr5 agb1 mutants. We performed an experiment to compare M. oryzae entry rates among different Arabidopsis mutants. Around 24 h post-inoculation (hpi), M. oryzae penetrated Arabidopsis epidermal cells; we harvested leaves of infected plants at 26 and 48 hpi and examined them microscopically. Consistent with our previous observations, entry rates into pen2 NahG, pen2 pmr5 and pen2 agb1 double mutants were higher than the rate into pen2 mutants (Fig. 1a)15. The triple mutants (pen2 NahG pmr5, pen2 NahG agb1 and pen2 pmr5 agb1) had higher rice blast entry rates than the pen2 double mutants (Fig. 1a). Moreover, the entry rate was significantly increased (P < 0.05) into the pen2 NahG pmr5 agb1 mutant as compared with the other mutants (Fig. 1a). Entry rates into these mutants at 48 hpi were usually higher than those at 26 hpi, indicating that penetration continued up to 48 hpi (Fig. 1a and Supplementary Fig. S1a). We estimated the penetration ratio of multiple mutants by summing the frequencies of individual mutants and showed that NahG, pmr5 and agb1 may have additive effects on Arabidopsis penetration resistance to M. oryzae.
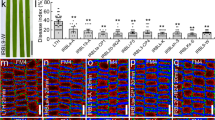
Quantitative analysis of Arabidopsis mutant non-host resistance to M. oryzae.
(a) Mean frequency of M. oryzae penetration into Arabidopsis mutants 48 h post-inoculation (hpi) expressed as a percentage of total infection sites. (b) Mean length of infection hyphae was measured at 48 hpi. (c) The penetration process was divided into four events (I–IV): I, successful penetration without infection hyphae; II, successful penetration with short (<10 μm) infection hyphae; III, successful penetration with long (>10 μm) infection hyphae; and IV, successful penetration with branched hyphae. The epidermal cells penetrated accumulated autofluorescent compounds (pale yellow). ap, appressorium, ih, infection hypha. (d–g) Mean frequencies of infection site types on Arabidopsis mutants at 48 hpi, expressed as percentages of penetrated cells. (d) Type I, (e) type II, (f) type III, (g) type IV. Values are means ± standard errors (n = 3). Bars sharing the same lowercase letters are not significantly different (Tukey's highly significant difference test; P ≥ 0.05; n = 3).
Previously, we reported that M. oryzae penetrated epidermal cells, but showed no further intra- or intercellular growth in Arabidopsis pen2 NahG, pen2 pmr5 and pen2 agb1 mutants15; we concluded that PMR5 and AGB1 contributed only to penetration resistance. However, during microscopic examination of pen2 NahG pmr5 agb1 mutants in the present study, we noticed that infection hyphae in the plants were longer (≥30 μm) than in the pen2 mutant (<ca. 10 μm) and some of them had branched by 48 hpi (Fig. 1b–g). Accordingly, we measured the lengths of the longest infection hyphae in these multiple mutants at 26 and 48 hpi. The longest infection hyphae in pen2 NahG, pen2 pmr5 and pen2 agb1 double mutants were longer than those in the pen2 mutant at 26 and 48 hpi (Fig. 1b and Supplementary Fig. S1b). Infection hypha in the triple mutants (pen2 NahG pmr5, pen2 NahG agb1 and pen2 pmr5 agb1) were longer yet than those in pen2 double mutants at 26 and 48 hpi (Fig. 1b and Supplementary Fig. S1b). Furthermore, infection hypha length in the pen2 NahG pmr5 agb1 mutant was significantly (P < 0.05) greater than lengths in pen2 double mutants at 26 and 48 hpi (Fig. 1b and Supplementary Fig. S1b). Infection hypha lengths in these multiple mutants increased between 26 and 48 hpi, except in pen2 and pen2 NahG plants, indicating that elongation of infection hyphae continued through 48 hpi in most of the mutants (Fig. 1b and Supplementary Fig. S1b). We found that infection hypha lengths in these multiple mutants could be roughly estimated by summing the lengths in individual mutants. Hence, NahG, pmr5 and agb1 may have additive effects on fungal elongation in Arabidopsis tissues.
We evaluated M. oryzae penetration success through the occurrence of autofluorescence at infection sites; the autofluorescence we observed resulted from hypersensitive responses resembling cell death that were triggered by penetration. Hence, the penetration process included not only the breaching of epidermal cell walls, but also hyphal elongation in penetrated cells that induced cell death. Therefore, we examined the penetration process by using higher-magnification microscopy to classify Arabidopsis defense responses. We were readily able to divide the process into four events (I–IV): (I) cell wall penetration, (II) establishment of infection hyphae, (III) elongation of infection hyphae and (IV) branch formation on infection hyphae (Fig. 1c). No visible infection hyphae (Fig. 1d) were detected in about 30% of penetrated pen2 epidermal cells, short (<10 μm) infection hyphae (Fig. 1e) developed in about 40% of penetrated cells, long (>10 μm) infection hyphae (Fig. 1f) developed in about 25% and branched hyphae (Fig. 1g) developed in <5% of penetrated cells. Thus, the PEN2-mediated pathway likely controlled not only cell wall penetration, but also the establishment of M. oryzae infection hyphae in penetrated epidermal cells.
We subsequently examined the penetration process in pen2 double mutants (pen2 NahG, pen2 pmr5 and pen2 agb1) under high magnification. Among these mutants, no visible infection developed in ∼10% of penetrated cells; this proportion differed significantly from that in pen2 plants (P < 0.05; Fig. 1d). These double mutants produced short (<10 μm) infection hyphae in approximately 25% of penetrated cells (Fig. 1e). The pen2 pmr5 and pen2 agb1 mutants had significantly higher proportions of long (>10 μm) infection hypha development (P < 0.05) as compared with pen2 mutants (Fig. 1f). Notably, long infection hyphae developed in >50% of penetrated cells in pen2 pmr5 plants (Fig. 1f). Branched hyphae developed in only about 10% of penetrated cells in pen2 pmr5 plants; the proportions of penetrated cells (∼20%) in pen2 NahG and pen2 agb1 mutants that developed branched hyphae were significantly higher than that in the pen2 mutant (P < 0.05; Fig. 1g).
We further examined the penetration process in pen2 NahG pmr5, pen2 NahG agb1, pen2 pmr5 agb1 and pen2 NahG pmr5 agb1 mutants. Among these pen2 multiple mutants, visible infection hyphae developed in >90% of penetrated cells; this proportion differed significantly from that in pen2 plants (P < 0.05; Fig. 1d). Short (<10 μm) infection hyphae were produced in these multiple mutants (with the exception of pen2 pmr5 agb1 plants) much less frequently than in the pen2 double mutants (Fig. 1e). The pen2 NahG pmr5 agb1 plants had especially reduced proportions of this fungal phenotype (P < 0.05; Fig. 1e). The proportions of long (>10 μm) infection hyphae in these multiple mutants did not differ markedly from those in pen2 double mutants (Fig. 1f) after penetration. The proportion of branched hyphae formed in pen2 NahG agb1 plants equaled the sum of the proportions in pen2 NahG and pen2 agb1 plants, indicating that the effects of NahG and agb1 on branch formation were additive (Fig. 1g). Although we detected no effect of pmr5 in pen2 NahG pmr5 and pen2 pmr5 agb1 plants in our comparison with pen2 NahG and pen2 agb1 plants, pen2 NahG pmr5 agb1 plants had significantly higher proportions of branched hyphae than any other genotype (P < 0.05; Fig. 1g).
The role of MLO2 in Arabidopsis NHR to M. oryzae
Previously, we showed that MLO2 had no NHR function in Arabidopsis–M. oryzae interactions15. However, barley plants carrying a mutation in the MLO locus, which confers durable resistance to powdery mildew, are hypersusceptible to the rice blast fungus Magnaporthe oryzae18 . Therefore, we continued analysis of the mlo mutation in different genetic backgrounds to determine the NHR MLO function in Arabidopsis–M. oryzae interactions. To identify the role of MLO2 in NHR, we generated pen2 NahG mlo2, pen2 pmr5 mlo2 and pen2 NahG pmr5 mlo2 plants and performed an experiment to compare M. oryzae entry rates into these mutants. We harvested leaves of infected plants at 26 and 48 hpi and examined them microscopically. Consistent with our previous observations, entry rates into pen2 mlo2 plants did not differ from that into the pen2 mutant (Fig. 2a)15. The entry rate into pen2 NahG mlo2 plants was higher than the rate into pen2 double mutants and equal to the sum of entry rates into pen2 NahG and pen2 mlo2 plants (Fig. 2a). Thus, mlo2 had an additive effect on penetration resistance in the pen2 NahG genetic background. Moreover, pen2 pmr5 mlo2 plants had significantly higher penetration rates than pen2 double mutants (P < 0.05; Fig. 2a); pen2 NahG pmr5 mlo2 plants also had significantly higher penetration rates than other mutants (P < 0.05; Fig. 2a), indicating that mlo2 likely had synergistic effects on penetration resistance in the pen2 pmr5 genetic background. Based on these data, we suggest that mlo2 and NahG acted independently and that mlo2 and pmr5 acted synergistically in their effects on Arabidopsis penetration resistance against M. oryzae.
Quantitative analysis of mlo2 multiple mutant Arabidopsis non-host resistance to M. oryzae.
(a) Mean frequencies of M. oryzae penetration in Arabidopsis mutants at 48 h post-inoculation (hpi) expressed as percentages of total infection sites. (b) Mean lengths of infection hypha measured at 48 hpi. (c–f) Mean frequencies of infection site types in Arabidopsis mutants at 48 hpi expressed as percentages of penetrated cells. (c) Type I, (d) type II, (e) type III, (f) type IV. Values are means ± standard errors (n = 3). Bars sharing the same lowercase letters are not significantly different (Tukey's highly significant differences test; P ≥ 0.05, n = 3).
We further investigated the role of MLO2 in post-penetration resistance by measuring lengths of the longest infection hyphae in mlo mutants at 26 and 48 hpi (Fig. 2b and Supplementary Fig. S2b). The lengths of infection hyphae in the pen2 mlo2 double mutant and pen2 mutant did not differ (Fig. 2b). The lengths of infection hyphae in pen2 NahG mlo2 plants and pen2 NahG double mutants also did not differ (Fig. 2b). However, the infection hyphae in pen2 pmr5 mlo2 and pen2 NahG pmr5 mlo2 plants were longer than those in control plants (Fig. 2b). Thus, the effect of mlo2 was likely expressed only in the pen2 pmr5 genetic background and MLO2 likely played a role in post-penetration resistance.
Subsequently, we examined the penetration process in the pen2 mlo2 double mutant under high magnification. The distribution of cellular reaction types in the pen2 mlo2 and pen2 mutants did not differ (Fig. 2c–f). We also examined hyphal penetration in pen2 NahG mlo2, pen2 pmr5 mlo2 and pen2 NahG pmr5 mlo2 plants. Among these, visible infection hyphae developed in more than ∼90% of penetrated cells, a proportion that differed significantly from that in pen2 mlo2 plants (P < 0.05; Fig. 2c). Short (<10 μm) infection hyphae were produced much less frequently in these multiple mutants (except for pen2 NahG mlo2 plants) than in pen2 mlo2 plants (Fig. 2d). Proportions of penetrated cells that developed long (>10 μm) infection hyphae did not differ between pen2 pmr5 mlo2 and pen2 pmr5 plants (Fig. 2e). The proportion of penetrated cells that developed branched hyphae in pen2 NahG mlo2 was intermediate between proportions in pen2 mlo2 and pen2 NahG plants. The branched hyphal development proportion in pen2 pmr5 mlo2 plants was significantly (P < 0.05) higher than those in pen2 mlo2 and pen2 pmr5 plants (Fig. 2f); the proportion in pen2 NahG pmr5 mlo2 plants was significantly higher than that in pen2 NahG pmr5 plants (P < 0.05; Fig. 2f). These data indicate that mlo2 likely had a synergistic effect on the formation of branched hyphae in the pen2 pmr5 genetic background.
Rice Pi21 enhances hyphal growth of M. oryzae in Arabidopsis
The pen2 NahG pmr5 agb1 and pen2 NahG pmr5 mlo2 Arabidopsis plants had the highest rates of hyphal penetration (Figs. 1a, 2a and Supplementary Figs. S1a, S2a), longest hyphae (Figs. 1b, 2b and Supplementary Figs. S1b, S2b), highest proportions of branched hypha formation (Figs. 1g and 2f) and highest proportions of hyphal growth from penetrated cells to adjacent cells (Supplementary Fig. S3); however, they prevented M. oryzae from completing its life cycle to form asexual conidia. Hence, unidentified genes in Arabidopsis probably control post-penetration resistance to M. oryzae. Alternatively, essential factors needed to establish the rice blast infection may be absent in Arabidopsis, in which case, these factors should be present in rice. Rice blast susceptibility factor Pi21 may function in this role.
To test whether rice Pi21 affected Arabidopsis NHR to M. oryzae, we generated transgenic lines in pen2, pen2 NahG pmr5 agb1, and pen2 NahG pmr5 mlo2 genetic backgrounds, expressing rice Pi21 under the control of a 35S promoter. Although we were able to generate sufficient numbers of transgenic Pi21 lines in pen2 and pen2 NahG pmr5 mlo2 backgrounds (Fig. 3a and Supplementary Fig. S4), we were not able to produce sufficient numbers in the pen2 NahG pmr5 agb1 background to permit further study. Our analysis was therefore restricted to two genetic backgrounds.
Characterization of transgenic Arabidopsis Pi21 lines.
(a) Reverse transcription polymerase chain reaction (RT-PCR) analysis of Pi21 in transgenic Arabidopsis Pi21 lines (pen2 NahG pmr5 mlo2 background). Col (g), genomic DNA of Arabidopsis accession Col-0 served as a control template for distinguishing cDNA and genomic DNA–derived PCR products. (b) Mean frequencies of M. oryzae penetration in transgenic Arabidopsis Pi21 lines (pen2 NahG pmr5 mlo2 background) at 48 h post-inoculation (hpi) expressed as percentages of total infection sites. (c) Mean lengths of infection hyphae measured at 48 hpi. (d–g) Mean frequencies of infection site types in transgenic Arabidopsis Pi21 lines (pen2 NahG pmr5 mlo2 background) at 48 hpi expressed as percentages of penetrated cells. (d) Type I, (e) type II, (f) type III, (g) type IV. Cont., control pen2 NahG pmr5 mlo2 plants; 4-1 and 5-1, two independent T3 lines (pen2 NahG pmr5 mlo2 background). Values are means ± standard errors (n = 3). Bars sharing the same lowercase letters are not significantly different (Tukey's highly significant differences test; P ≥ 0.05, n = 3).
We first examined M. oryzae entry rates in these transgenic Pi21 lines. Leaves of infected plants were harvested at 48 hpi and inspected microscopically. Entry rates into transgenic Pi21 lines did not differ among pen2 and pen2 NahG pmr5 agb1 backgrounds and control plants (Fig. 3b and Supplementary Fig. S5a). Subsequently, we measured the lengths of the longest infection hyphae in these transgenic lines at 48 hpi. The lengths of the infection hyphae did not differ between transgenic lines in the pen2 background and pen2 plants (Supplementary Fig. S5b); however, the lengths of infection hyphae in transgenic lines in the pen2 NahG pmr5 mlo2 background were significantly greater than those in control pen2 NahG pmr5 mlo2 plants (P < 0.05; Fig. 3c).
We further examined the penetration process in transgenic Pi21 lines by high-magnification microscopy. The distribution of cellular reaction types did not differ between transgenic lines in pen2 and pen2 NahG pmr5 mlo2 backgrounds and control plants (Fig. 3d–g and Supplementary Fig. S6).
We also examined the movement of fungal hyphae in transgenic lines from penetrated epidermal cells to adjacent epidermal or mesophyll cells. The movement phenotypes were grouped as follows: (A) movement to adjacent epidermal cell (Fig. 4a, b), (B) movement to adjacent mesophyll cell (Fig. 4c, d) and (C) movement to adjacent epidermal and mesophyll cells. Significantly higher rates of hyphal movement from penetrated cells into adjacent mesophyll cells were observed in transgenic Pi21 lines in the pen2 NahG pmr5 mlo2 background (P < 0.05); however, this was not the case for movement into adjacent epidermal cells (Fig. 4e–g). This Arabidopsis phenotype resembled transgenic rice plants expressing Pi2119. However, no hyphal movement was observed in Arabidopsis transgenic lines expressing Pi21 in the pen2 background (Supplementary Fig. S7), indicating that NahG, pmr5, and mlo2 mutations are likely necessary factors allowing Pi21 to function in the pen2 Arabidopsis mutant.
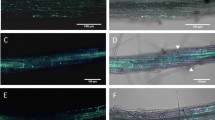
Movement of infection hyphae from penetrated epidermal cells to adjacent cells in transgenic Arabidopsis Pi21 lines.
Microscopic view of infection sites in transgenic Arabidopsis Pi21 lines (pen2 NahG pmr5 mlo2 background). (a) Movement of infection hyphae from a penetrated epidermal cell to an adjacent epidermal cell in transgenic Arabidopsis Pi21 lines (pen2 NahG pmr5 mlo2 background). The infection site was photographed at 48 h post-inoculation (hpi). The first invaded epidermal cell was associated with slight browning. ap, appressorium; ih, infection hyphae in adjacent epidermal cell. Bars = 50 μm. (b) Fluorescence microscopic view of the infection site of (a). ih, infection hyphae in adjacent epidermal cell. Bars = 50 μm. (c) Movement of infection hyphae from a penetrated epidermal cell to adjacent mesophyll cells in transgenic Arabidopsis Pi21 lines (pen2 NahG pmr5 mlo2 background). The infection site was photographed with the mesophyll cells in focus (48 hpi). ap, appressorium; ih, infection hyphae in adjacent mesophyll cell. Bars = 50 μm. (d) Fluorescence microscopic view of the infection site in (c). m, penetrated adjacent mesophyll cell. Bars = 50 μm. (e–g) Cell–cell movements of infection hyphae were grouped into categories (A–C): A, hyphal movement from penetrated epidermal cell to adjacent epidermal cell; B, hyphal movement from penetrated epidermal cell to adjacent mesophyll cell; C, hyphal movement from penetrated epidermal cell to adjacent epidermal and mesophyll cells. Mean frequencies of various types of infection site in transgenic Arabidopsis Pi21 lines (pen2 NahG pmr5 mlo2 background) at 48 hpi expressed as percentages of penetrated cells. (e) Type A, (f) type B, (g) type C. Cont., control pen2 NahG pmr5 mlo2 plants; 4-1 and 5-1, two independent T3 lines (pen2 NahG pmr5 mlo2 background). Values are means ± standard errors, n = 3 independent experiments. Bars sharing the same lowercase letters are not significantly different (Tukey's highly significant differences test; P ≥ 0.05, n = 3).
Discussion
Our systematic analyses of multiple mutant combinations revealed the genetic network of penetration and post-penetration resistance to M. oryzae in Arabidopsis. In multiple mutants, including pen2 NahG pmr5 agb1 and pen2 NahG pmr5 mlo2 plants, penetration resistance to M. oryzae was severely compromised and fungal growth in penetrated epidermal cells was also greater than in the pen2 mutant (Figs. 1, 2 and Supplementary Figs. S1, S2). Thus, PEN2, PMR5, AGB1 and MLO2 were likely involved in Arabidopsis penetration and post-penetration resistance to M. oryzae. Moreover, Arabidopsis transgenic lines expressing rice Pi21 in the pen2 NahG pmr5 mlo2 background permitted fungal cell–cell movement (Fig. 4), suggesting that the absence of rice susceptibility to the M. oryzae gene Pi21 may contribute to Arabidopsis NHR.
Transgenic NahG plants and the pmr5, agb1 and mlo2 single mutants did not support fungal penetration success and hyphal elongation in M. oryzae–Arabidopsis interactions15; however, the entry rates into pen2 NahG, pen2 pmr5 and pen2 agb1 plants exceeded those into the pen2 mutant15. The penetration ratio of pen2 pmr5 agb1 plants was obtained by summing the frequencies for individual mutants (Figs. 1a and 2a), suggesting that pmr5 and agb1 have additive effects on the rates in the pen2 background. Hence, PMR5 and AGB1 probably acted independently in Arabidopsis penetration resistance to M. oryzae. In our previous study, the pen2 and pen2 mlo2 mutants did not differ in penetration resistance to rice blast15. However, in the present work, mlo2 had both an additive effect on hyphal penetration rates in the pen2 NahG background (Fig. 2a) and a synergistic effect on these rates in the pen2 pmr5 background (Fig. 2a). Hence, MLO2 and NahG probably acted independently and MLO2 and PMR5 likely acted synergistically, in Arabidopsis rice blast penetration resistance.
Because we detected no difference in post-penetration resistance among pen2, pen2 NahG, pen2 pmr5 and pen2 agb1 plants in our previous study, we concluded that PMR5 and AGB1 were not involved in post-penetration resistance15. However, in the present investigation, we found that frequencies of long (>10 μm) infection hyphae in pen2 pmr5 and pen2 agb1 plants were significantly (P < 0.05) higher than those in pen2 plants (Fig. 1f). Further, rates of infection hypha branch formation were significantly higher in pen2 NahG and pen2 agb1 plants than in pen2 plants (Fig. 1g). Hence, PMR5 functioned mainly in the inhibition of hyphal elongation following fungal penetration in the pen2 genetic background. NahG preferentially stimulated hyphal branching and AGB1 restricted both hyphal elongation and branching. Although detected no effect of pmr5 on post-penetration resistance in pen2 NahG pmr5 and pen2 pmr5 agb1 plants (Fig. 1d–g), pen2 NahG pmr5 agb1 plants had significantly higher frequencies of branched hypha establishment than other mutants (Fig. 1g). Because PMR5 inhibited hyphal elongation and AGB1 restricted hyphal elongation and branching, agb1 likely had synergistic effects on hyphal branch formation in the pen2 NahG pmr5 background. The mlo2 mutation enhanced hyphal length and significantly increased the rate of infection hypha branch formation in the pen2 pmr5 background (Fig. 2b, f), which suggests that MLO2 restricted both hyphal elongation and branching in this background.
Rice pi21 is a recessive gene conferring durable resistance to blast disease. Wild-type Pi21 appears to slow host defense responses, suggesting that Pi21 may promote fungal growth in plants19. We determined whether rice Pi21 would support fungal growth in non-host Arabidopsis. In transgenic Pi21 lines in pen2 and pen2 NahG pmr5 mlo2 backgrounds, rice Pi21 did not affect Arabidopsis penetration resistance (Fig. 3b and Supplementary Fig. S5a). However, although rice Pi21 significantly enhanced hyphal elongation and hyphal movement from penetrated epidermal cells to adjacent mesophyll cells in the pen2 NahG pmr5 mlo2 background (Figs. 3c and 4f), this was not the case in the pen2 background (Supplementary Figs. S5b and S7). Hence, rice Pi21 regulated hyphal elongation and movement, but not penetration defense, in Arabidopsis. Factors other than Pi21 may have affected hyphal movement from penetrated epidermal cells to adjacent epidermal cells. Rice Pi21 contains putative protein–protein interaction motifs19, which suggests that Pi21 interacting factors occur in Arabidopsis. Rice Pi21 also contains a heavy metal–transport/detoxification protein domain. Yeast copper chaperone ATX1 represents the proteins containing this domain20. Although homologs of yeast ATX1 are present in Arabidopsis, rice Pi21 (Os04g0401000) belongs to a group of genes that are distinct from these homologs19 , suggesting that Pi21, which is a gene for susceptibility to M. oryzae, occurs in rice but not in Arabidopsis. Hence, Pi21 may predispose rice to blast disease.
PEN2, PMR5, AGB1 and MLO2 functioned in both penetration and post-penetration resistance. Moreover, epistasis occurred among these genes at both levels of resistance. Elevated fungal entry rates were related to fungal growth rates in penetrated cells (Figs. 1, 2 and Supplementary Figs. S1, S2). Therefore, penetration and post-penetration resistance in Arabidopsis likely share a common mechanism. Indeed, PEN2 and PEN3 function in both penetration and post-penetration resistance in Arabidopsis–powdery mildew interactions10,11. PEN2 converts a nontoxic substrate into a toxic product, which is then exported either directly or following further modification to the apoplast by PEN3; the toxic product poisons the fungal penetration peg as it attempts to breach the cell wall11. Post-penetration roles of PEN2 and PEN3 may therefore have involved PEN3 toxin export to the extrahaustorial matrix, where the haustorium was poisoned; this would have limited the initiation and growth of secondary fungal hyphae. Therefore, PEN2 in Arabidopsis likely operated by poisoning fungal penetration (i) as penetration pegs passed through the cell wall and (ii) when infection hyphae invaded the plasma membrane.
Silencing of the arbuscular mycorrhizal (AM) symbiosis-induced gene Vapyrin impairs epidermal penetration by AM fungi and prevents arbuscule formation in Medicago truncatula21 . The phenotypes of Vapyrin-silencing plants indicate that a common cellular mechanism may be required to enable hyphal growth through epidermal cells and arbuscule development in cortical cells21. Vapyrin may play a role in cellular remodeling processes that support entry, possibly those that facilitate membrane invagination21. In compatible rice–M. oryzae interactions, primary hyphae and then bulbous invasive hyphae penetrate living rice cells while separating host cytoplasm from host extracellular space with a plant-derived extra-invasive hyphal membrane (EIHM)2. Moreover, infection hyphae are established in host-adapted and non-adapted Colletotrichum species through invagination of the host plasma membrane22. Considering these diverse lines of evidence, we suggest that establishment of infection hyphae through invagination of the host plasma membrane is a common mechanism of fungal penetration into plant cells. We also suggest that PMR5, AGB1 and MLO2 are involved in the invagination process. These factors function in plant cell membranes; PMR5 likely targets the endoplasmic reticulum (ER)23, AGB1 is localized in both the plasma membrane and the ER24 and MLO is localized in the plasma membrane25.
The fungus never sporulated in any of the multiple mutants we tested (including Pi21 transgenic lines), indicating that post-penetration resistance to M. oryzae was effective in Arabidopsis. Therefore, additional and currently unidentified genes probably had roles in Arabidopsis post-penetration resistance to M. oryzae. Alternatively, essential requirements needed to establish the pathogen's biotrophic stage may be absent in Arabidopsis. NHR may now be considered as the consequence of ineffective microbial effectors with no suppression of pathogen-associated molecular pattern (PAMP)-triggered immunity (PTI) and/or effector-triggered immunity (ETI)26. Alternatively, (individual) effectors may not have been selected to evade recognition; they would thus be recognized in non-host plants, resulting in ETI. Adapted pathogens might conceivably use effector molecule transfer to interfere with both penetration and post-penetration defense mechanisms and to establish basic compatibility. Studies examining the functions of blast effectors in NHR of Arabidopsis are certainly warranted.
Here, we have presented the interaction between Arabidopsis and M. oryzae as a model system for dissecting NHR mechanisms. Correlated genetic analysis and cytological investigation allowed us to characterize genes involved in several steps of Arabidopsis NHR to M. oryzae. This work will contribute to improved and durable disease resistance in important crops.
Methods
Plant material
Arabidopsis plants were grown under short-day conditions (9:15 L:D) at 22°C in a growth room. The Arabidopsis accession code was Col-0. We used the following mutants and transgenic plants: pen2-110 , pmr5-127 , agb1-228 , Atmlo2-7 (SALK_079850) and NahG (all with the Col-0 background)29 . These mutants were used for crosses. Respective multiple mutants were identified in F2 progeny by polymerase chain reaction (PCR) using suitable CAPS or derived CAPS (dCAPS) markers; genotypes were verified in the subsequent (F3) generation.
Transgenic Arabidopsis lines overexpressing rice Pi21 cDNA
A cDNA containing the susceptible Pi21 allele from Nipponbare (AK070581) was digested with SfiI and cloned into the SfiI sites of the binary vector pBIG2113SF30. Arabidopsis pen2, pen2 NahG pmr5 agb1 and pen2 NahG pmr5 mlo2 plants were transformed with the resulting construct by floral dipping31 and primary transformants (T1 generation) were selected on hygromycin in tissue culture. Progeny of T2 lines (T3 generation) were used for M. oryzae inoculations. RNA samples were prepared from the T3 lines. Reverse transcription (RT)-PCR was performed for Pi21 and actin using the following oligonucleotide primers: Pi21-F, CGGCAAATTTGACAGATGGGTAT; Pi21-R, CTTCTCCGGGTCGAACTTC; Actin-F, GTTGGGATGAACCAGAAGGA; and Actin-R, GACCACCGATCCAGACACT. Genomic DNA of Arabidopsis accession Col-0 served as the control template for distinguishing cDNA and genomic DNA–derived PCR products.
Fungal material
M. oryzae isolate Kita 1 (race 007) was incubated on oatmeal agar media in Petri dishes at 25°C; the inoculum was prepared as previously described32. To inoculate M. oryzae, 15-μl droplets (104 spores/ml) were applied to leaves of 4–5-week-old plants, which were then kept in conditions of saturating humidity until harvested.
Cytology and quantification of fungal growth
Infected leaves were harvested at the time point indicated. After cutting, the leaves were submerged directly in ethanolic lactophenol solution and were heated to 90°C for 3 min. The leaf samples were cooled to room temperature for 10 min and were subsequently incubated in saturated chloral hydrate (2.5 g/ml) for 2 days. To quantify cell entry and fungal growth, we examined the germinated fungal sporelings that developed appressoria on six leaves from six independent plants per experiment and genotype (minimum of 100 appressoria/leaf evaluated). Penetration success of M. oryzae was detected by the occurrence of autofluorescence or hyphal elongation at infection sites using fluorescence and bright-field microscopy (BX51; OLYMPUS). Fluorescence was examined under a microscope with a mirror unit (U-MWIB3; OLYMPUS; excitation, 460–495 nm; dichroic, 505 nm; emission, 510 nm). The images were recorded with a digital camera (DP72; OLYMPUS). Length and branch formation of infection hyphae at infection sites were also examined by bright-field microscopy. Cell entry and fungal growth on each plant genotype were quantified in at least three independent experiments.
Data collection and analysis
Data were collected from six leaves from six independent plants per line. A minimum of 100 infection sites were inspected per leaf. Data were compared using Tukey's highly significant difference (HSD) tests. Calculations were performed on three data sets (n = 3) and P < 0.05 indicated statistically significant effects.
References
Koga, H. in Major Fungal Diseases of Rice Recent Advances 87–110 (Kluwer Academic Publishers, 2001).
Kankanala, P., Czymmek, K. & Valent, B. Roles for rice membrane dynamics and plasmodesmata during biotrophic invasion by the blast fungus. Plant Cell 19, 706–724 (2007).
Wilson, R. A. & Talbot, N. J. Under pressure: investigating the biology of plant infection by Magnaporthe oryzae . Nat Rev Microbiol 7, 185–195 (2009).
Dean, R. A. et al. The genome sequence of the rice blast fungus Magnaporthe grisea . Nature 434, 980–986 (2005).
Ebbole, D. J. Magnaporthe as a model for understanding host-pathogen interactions. .Annu Rev Phytopathol 45, 437–456 (2007).
Howard, R. J., Ferrari, M. A., Roach, D. H. & Money, N. P. Penetration of hard substrates by a fungus employing enormous turgor pressures. Proc Natl Acad Sci USA 88, 11281–11284 (1991).
Lipka, U., Fuchs, R. & Lipka, V. Arabidopsis non-host resistance to powdery mildews. .Curr Opin Plant Biol 11, 404–411 (2008).
Heath, M. C. Nonhost resistance and nonspecific plant defenses. Curr Opin Plant Biol 3, 315–319 (2000).
Collins, N. C. et al. SNARE-protein-mediated disease resistance at the plant cell wall. Nature 425, 973–977 (2003).
Lipka, V. et al. Pre- and postinvasion defenses both contribute to nonhost resistance in Arabidopsis . Science 310, 1180–1183 (2005).
Stein, M. et al. Arabidopsis PEN3/PDR8, an ATP binding cassette transporter, contributes to nonhost resistance to inappropriate pathogens that enter by direct penetration. .Plant Cell 18, 731–746 (2006).
Kobae, Y. et al. Loss of AtPDR8, a plasma membrane ABC transporter of Arabidopsis thaliana, causes hypersensitive cell death upon pathogen infection. Plant Cell Physiol 47, 309–318 (2006).
Bednarek, P. et al. A glucosinolate metabolism pathway in living plant cells mediates broad-spectrum antifungal defense. Science 323, 101–106 (2009).
Clay, N. K., Adio, A. M., Denoux, C., Jander, G. & Ausubel, F. M. Glucosinolate metabolites required for an Arabidopsis innate immune response. Science 323, 95–101 (2009).
Maeda, K. et al. AGB1 and PMR5 contribute to PEN2-mediated preinvasion resistance to Magnaporthe oryzae in Arabidopsis thaliana . Mol Plant-Microbe Interact 22, 1331–1340 (2009).
Maeda, K. et al. Nonhost resistance to Magnaporthe oryzae in Arabidopsis thaliana . Plant Signal Behav 5, 755–756 (2010).
Schreiber, C., Slusarenko, A. J. & Schaffrath, U. Organ identity and environmental conditions determine the effectiveness of nonhost resistance in the interaction between Arabidopsis thaliana and Magnaporthe oryzae . Mol Plant Pathol 12, 397–402 (2011).
Jarosch, B., Jansen, M. & Schaffrath, U. The ambivalence of the barley Mlo locus: Mutations conferring resistance against powdery mildew (Blumeria graminis f. sp. hordei) enhance susceptibility to the rice blast fungus Magnaporthe grisea . Molecular Plant-Microbe Interactions 6, 508–514 (1999).
Fukuoka, S. et al. Loss of function of a proline-containing protein confers durable disease resistance in rice. Science 325, 998–1001 (2009).
Lin, S. J. & Culotta, V. C. The ATX1 gene of Saccharomyces cerevisiae encodes a small metal homeostasis factor that protects cells against reactive oxygen toxicity. Proc Natl Acad Sci USA 92, 3784–3788 (1995).
Pumplin, N. et al. Medicago truncatula Vapyrin is a novel protein required for arbuscular mycorrhizal symbiosis. .Plant J 61, 482–494 (2010).
Hiruma, K. et al. Entry mode-dependent function of an indole glucosinolate pathway in Arabidopsis for nonhost resistance against anthracnose pathogens. Plant Cell 22, 2429–2443 (2010).
Vogel, J. P., Raab, T. K., Somerville, C. R. & Somerville, S. C. Mutations in PMR5 result in powdery mildew resistance and altered cell wall composition. Plant J 40, 968–978 (2004).
Wang, S., Narendra, S. & Fedoroff, N. Heterotrimeric G protein signaling in the Arabidopsis unfolded protein response. Proc Natl Acad Sci USA 104, 3817–3822 (2007).
Bhat, R. A., Miklis, M., Schmelzer, E., Schulze-Lefert, P. & Panstruga, R. Recruitment and interaction dynamics of plant penetration resistance components in a plasma membrane microdomain. Proc Natl Acad Sci USA 102, 3135–3140 (2005).
Jones, J. D. & Dangl, J. L. The plant immune system. Nature 444, 323–329 (2006).
Vogel, J. & Somerville, S. Isolation and characterization of powdery mildew-resistant Arabidopsis mutants. Proc Natl Acad Sci USA 97, 1897–1902 (2000).
Chen, J. G., Gao, Y. & Jones, A. M. Differential roles of Arabidopsis heterotrimeric G-protein subunits in modulating cell division in roots. Plant Physiol 141, 887–897 (2006).
Lawton, K. et al. Systemic acquired resistance in Arabidopsis requires salicylic acid but not ethylene. Mol Plant-Microbe Interact 8, 863–870 (1995).
Ichikawa, T. et al. The FOX hunting system: an alternative gain-of-function gene hunting technique. Plant J 48, 974–985 (2006).
Clough, S. J. & Bent, A. F. Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana . Plant J 16, 735–743 (1998).
Koga, H. & Nakayachi, O. Morphological studies on attachment of spores of Magnaporthe grisea to the leaf surface of rice. J Gen Plant Pathol 70, 11–15 (2004).
Acknowledgements
We would like to acknowledge ARBC for providing seeds of Col-0, agb1-2 and Atmlo2-7 and the Rice Genome Resource Center (National Institute of Agrobiological Sciences, Japan) for providing Pi21 cDNA. We thank H. Koga (Ishikawa Prefectural University) for providing the M. oryzae isolate, P. Schulze-Lefert (Max Planck Institute for Plant Breeding Research) for seeds of pen2-1, Syngenta for NahG, J. Vogel (United States Department of Agriculture) for pmr5-1 and Y. Kondou (RIKEN) for the binary vector. This work was supported by a grant-in-aid for scientific research at Fukui Prefectural University to A.I. from Fukui Prefecture, Japan.
Author information
Authors and Affiliations
Contributions
Author contricutions M.N., R.N., K.K. and R.I. performed experiments. A.I. designed experiments, interpreted results and wrote the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Supplementary Information
Article File
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareALike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Nakao, M., Nakamura, R., Kita, K. et al. Non-host resistance to penetration and hyphal growth of Magnaporthe oryzae in Arabidopsis. Sci Rep 1, 171 (2011). https://doi.org/10.1038/srep00171
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep00171
This article is cited by
-
Functional analysis of a susceptibility gene (HIPP27) in the Arabidopsis thaliana-Meloidogyne incognita pathosystem by using a genome editing strategy
BMC Plant Biology (2023)
-
Transcriptomic and metabolomic analyses identify a role for chlorophyll catabolism and phytoalexin during Medicago nonhost resistance against Asian soybean rust
Scientific Reports (2015)
-
Different non-host resistance responses of two rice subspecies, japonica and indica, to Puccinia striiformis f. sp. tritici
Plant Cell Reports (2014)
-
Plant innate immunity: An updated insight into defense mechanism
Journal of Biosciences (2013)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.