Key Points
-
Describes new drugs that have already been implicated in osteonecrosis of the jaw.
-
Highlights the difficulty in managing patients at risk of jaw necrosis.
-
Discusses the likelihood of the increasing pattern of jaw necrosis incidence.
-
Provides realistic solutions to an important issue the dental fraternity will be facing.
Abstract
Osteonecrosis of the jaw (ONJ) has a number of causes, the most familiar being radiation or bisphosphonate induced. Various other novel anti-neoplastic and bone-targeting therapies that can also cause jaw necrosis have recently become available. This has led to the suggested acronym MRONJ for medication-related osteonecrosis of the jaw. This article summarises the available information on these drugs and their implications for the dental surgeon.
Similar content being viewed by others
Introduction
The jaw bones have a predisposition to developing osteonecrosis. There is a historical precedent with 'phossy jaw' which was due to phosphorous ingestion by those employed in the match making industry.1,2,3
The mandible is more predisposed to necrosis, when compared to the remaining skeleton, because of its high bone metabolism and ready exposure to bacteria through breaches in the thin oral mucosa or the dentition. The two well-known predisposing risk factors are radiation and bisphosphonate medication.
At present mandibular necrosis is a relatively uncommon condition but its relevance lies in the fact that the condition has no guaranteed cure. Consequently, the 'at risk' patient requires a blanket of dental care over their lifetime. The inadvertent induction of osteonecrosis through unguarded dental treatment has important repercussions. Herein lies the relevance of this topic to the dental profession. The current status quo is about to change. A number of new cancer drugs are reported to induce osteonecrosis. As treatment outcomes improve the pool of 'at risk' patients in the population will increase and with it the prospect of developing necrosis of the jaw.
This article reports on a new class of cancer drugs that demonstrate the ability to induce jaw necrosis. The potential implications for the dental professionals are discussed.
Definitions and pathogenesis
Osteonecrosis
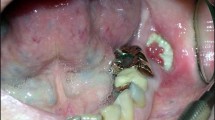
Osteonecrosis in its simplest form, consists of an area of exposed avascular dead bone. It has a number of causes with different historical time lines.
Radiation-induced osteonecrosis of the jaw was first reported by Regaud in 1922.4 Since then both the pathophysiology and management have been continually re-defined.5,6 Recently, Delanian7 has proposed that an important inductive factor is radiation-induced fibrosis which leads to a poor blood supply and vulnerability to infection. The suggested management is pentoxifylline and vitamin E (PVe) with anecdotal evidence showing promise for early lesions.
In comparison bisphosphonate-related osteonecrosis of the jaw (BRONJ) is a relatively new entity described by Marx8 in 2003. The American Association of Oral and Maxillofacial Surgeons (AAOMS) has proposed the acronym MRONJ (medication-related osteonecrosis of the jaw)9 to describe the condition which includes bisphosphonates as well as a number of new medications implicated in jaw necrosis.
Definition
MRONJ9 has the following three characteristics:
-
Current or previous treatment with anti-resorptive or anti-angiogenic agents
-
Exposed bone or bone that can be probed through an intra-oral or extra-oral fistula(e) in the maxillofacial region that has persisted for more than eight weeks
-
No history of radiation therapy to the jaws or obvious metastatic disease to the jaws.
In contrast, the criteria for osteoradionecrosis (ORN) have not been universally agreed. Exposed jaw bone in the absence of tumour recurrence following head and neck radiation is a prerequisite. However, the controversy relates to the duration of bone exposure needed for it to be described as ORN with the suggested definitions ranging from 2–6 months.10,11,12,13,14
Bone modulating drug therapy
Bone modulating therapy (BMT) is undertaken with drugs including bisphosphonates (BPs), RANKL inhibitors (RANKL is an acronym of 'receptor activator of nuclear factor kappa b ligand') as well as strontium ranelate, calcitonin and parathyroid hormone, but only BPs and RANKL inhibitors have been reported to cause ONJ.
BMT is used in both primary bone tumours (such as multiple myeloma) as well as cancers that metastasise to bone, such as breast, lung and prostate cancer. They are also used to control hypercalcaemia and in non-neoplastic conditions including osteoporosis, Paget's disease and osteogenesis imperfecta. There are a number of other miscellaneous uses including treatment of fibrous dysplasia and giant cell granulomas.
Denosumab
Denosumab (DB) is a human monoclonal IgG2 antibody15,16,17 and prevents the activation of receptor activator of nuclear factor kappa b ligand (RANKL) which drives the proliferation and function of osteoclasts. Inhibition leads to decreased osteoclastic activity18 and thereby reduces bone resorption.
This drug is slowly replacing BPs as a drug of choice for modulating bone metabolism. It is licenced by NICE19,20 with restrictions for its use (see Table 1); however, there is already positive evidence for its use in prostate cancer with bony metastases.21
Denosumab and BPs have different actions, although the end point is generally the same in achieving decreased bone turnover by inhibiting effective osteoclast function. BPs are internalised within the cell to induce their effect whereas denusomab acts on the cell membrane.22 This difference is significant as cessation of denosumab will usually allow osteoclastic reactivation once the drug is eliminated, whereas BPs cause osteoclastic cell death (apoptosis) and bind to the skeleton with an estimated half-life of approximately 10 years. Both drugs have fast onset when delivered parenterally but denosumab does not become incorporated into bone and bone resorption markers return to baseline about six months after cessation of taking the drug.23 Thus, its action may be considered to be intentionally reversible after some time. Unfortunately, cessation may not be a realistic option in all cancer cases but the reversibility is relevant in patients treated for osteoporosis.
Estimation of an individual dental patient's risk of developing ONJ is extremely difficult as they may be switched between the different anti-resorptive drugs and their immune status, previous or concomitant steroid therapy and the magnitude of dental treatment all complicate the situation. The risk of necrosis seems similar for both denusomab and zolendronic acid and can be compared to other drugs in Table 2.
Anti-angiogenic drug therapy
Tumours induce their own vascular supply through angiogenic growth factors, including members of the vascular endothelial growth factor (VEGF) family31 which are overexpressed in most solid cancers.32,33,34 Inhibition of VEGF suppresses tumour growth in animal models32,33,34 and this therapeutic approach is now likely to be used more widely in the management of tumours in humans.35 It is predominantly through VEGF inhibition that the drugs bevacizumab, sunitinib and cabozantinib act. However, VEGF is also essential for regulation of osteoclastic function, promotion of osteoclast differentiation and survival.36,37,38 These vascular endothelial growth factors (VEGF) and platelet derived growth factors (PDGF) also play an important role in wound healing and PDGF products are already commercially available and used for these properties.
Both bevacizumab39 and sunitinib40,41 have been associated with ONJ when used alone or in conjunction with BPs.42,43,44,45,46,47,48,49 The manufacturer50 of bevacizumab (Avastin®) as well as the Medicines and Healthcare Products Regulatory Agency (MHRA) have issued a drug safety warning on these products regarding the risk of ONJ.51
Bevacizumab
Bevacizumab (Avastin®) is a humanised monoclonal antibody that recognises and blocks VEGF. It is administered as a slow (60–90 minute) IV infusion and is licensed for the treatment of colorectal cancer, breast cancer, renal cancer, non-small cell lung cancer and ovarian/fallopian tube cancer.52
Recently, two large analyses (3,560 patients53 and 2,120 patients54) provided some evidence of ONJ risk. Bevacizumab used in isolation had an ONJ incidence of 0.2%;53 however, when combined with BPs this increases to approximately 2.4%53 but significantly higher rates have been reported.42,43,44,45,46,47,48,55 The combination of BPs with anti-angiogenics roughly halves the time to necrosis from 23 months with BPs alone to 12.4 months.49 In such circumstances 70%49 of the dual therapy patients developed ONJ spontaneously, which is a very significant concern, as preventative dental treatment would have no effect on this.
Sunitinib
Sunitinib (Sutent®) is a tyrosine kinase inhibitor that is orally administered, in contrast to conventional chemotherapy, which is delivered in pulses. In contrast to conventional chemotherapy, which is delivered in pulses. Sunitinib is usually a background medication taken over many years.56
Sunitinib targets an array of factors such as VEGF receptors, PDGF receptors, c-Kit, FLT3 and RET kinases.57,58 It is used for advanced or metastatic renal cell carcinoma, metastatic malignant gastro-intestinal stromal tumours and metastatic pancreatic neuroendocrine tumours.
Multiple case reports of sunitinb producing ONJ have appeared in the recent literature.40,41,59 The risk of ONJ appears to increase when dual therapy with BPs is used, mirroring the results with other combination therapies. In a small series of 21 patients with metastatic renal cell carcinoma treated concomitantly with zolendronic acid and sunitinib, five patients (24%) developed ONJ after a mean duration of exposure of 18.5 months of zolendronic acid and 5.4 months of sunitinib.60
Beuselinck et al.43 reported an incidence of ONJ in 10% of patients on tyrosine kinase inhibitors and concomitant BPs. Brunello et al.45 described an interesting case where a patient previously on BPs was recommenced on BPs and sunitinib due to progression of disease. The patient soon developed spontaneous ONJ, which improved on cessation of sunitinib but recurred as soon as it was restarted. Eventually sunitinib was withdrawn and the ONJ resolved. With further disease progression, sunitinib was tried tentatively again and a new area of necrosis occurred.
Cabozantinib
Cabozantinib (Cometriq®) is an oral bioavailable tyrosine kinase inhibitor with activity against VEGF61 and is a new therapy for treatment of progressive, unresectable, locally advanced or metastatic medullary thyroid carcinoma.
In a phase III trial ONJ was reported as 1.4%62 and been listed as a side-effect in the drug's patient information leaflet advising appropriate dental assessment before commencing. Recently a case report63 also reported cabozantinib-related ONJ.
Radiation drug therapy
Radium-223
Radium-223 dichloride (Xofigo®) is given as an intravenous infusion as an internal form of radiotherapy. Radium is a calcium-mimetic and is therefore taken up by the active tumour and normal bone cells. It selectively binds to areas of increased bone turnover such as bone metastases and emits high-energy alpha particles of short range (<100 μm).64 The high-energy, alpha-particle radiation induces mainly double-stranded DNA breaks that result in a potent and highly localised cytotoxic effect in the target areas.65,66,67,68 In a recent Phase III study, radium-223 significantly prolonged overall survival in patients who had castration-resistant prostate cancer and bone metastases, with a 30% reduction in the risk of death, as compared with placebo.69
In this group of patients there is a theoretical risk of ONJ. The high bone turnover in the jaws will attract the drug. The incidence of ONJ in a Phase III trial was 0.67% patients (4/600). All patients who developed ONJ had been previously exposed to BPs (for example, zolendronic acid).69 However,it was not reported if any had oral surgical procedures.
Strontium-89
Strontium-89 shares a number of similarities to that of radium-223. It has recently been deployed in a clinical trial (Trapeze)70 and like radium-223 it is a form of internal radiotherapy. Currently, there are no reported cases of ONJ. The trial's investigative arms included both strontium-89 as well as dual therapy with BPs and it remains to be seen whether ONJ occurred in either or both arms.
Other bone modulating theraputic agents
Odanacatib
Ondanacatib is classified as a selective cathepsin K inhibitor. Cathepsins are homeostatic enzymes which are involved in the proteolytic processing of specific substrates and importantly contribute to the physiological process of collagen turnover in bone and cartilage.71 Of the eleven known cathepsins, cathepsin K is the most important with respect to bone remodeling.72
It inhibits osteoclast function but preserves osteoclast viability. No cases of ondanacatib-associated ONJ have been reported. As with many of the therapies mentioned in this article the actual risk of ONJ will only be exposed after extensive use of the drug in clinical practice where it will be prescribed in combination with, or after, other anti-resorptive/anti-angiogenic medication and when they have oral surgical procedures.
Implication on dental health and dental services
The population characteristics in the UK are changing with an increasing number of elderly people in the community. Patients within this group often have complex medical and drug histories as well as existing or potentially complex dental problems. At the same time a reservoir of patients at risk of ONJ is increasing as cancer is being turned into a chronic disease. The effect of increased cancer survival has huge, and as yet largely unrecognised, implications for the dental profession. There are serious issues of risk recognition, risk assessment and risk management, which previously did not really exist as a problem.
Many dental patients in the 'heavy metal generation' and those with 'semi-preserved dentitions' are likely to need significant expertise to avoid ONJ. How they access this expertise and meet the costs of care is likely to become an increasingly important issue. The problems posed by this group of patients frequently require a novel approach to tooth care that may not follow traditional norms; for example, endodontic therapy with no intention to restore the tooth thereafter but rather just covering the roots with radiopaque conventional glass ionomer cement. Very little, if any, thought seems to have been given to having appropriately trained and experienced dental clinicians available for this patient group.
Insight to the potential magnitude of the problem can be gained from the 2009 Adult Dental Health Survey (ADHS),73 which describes the dental health of the population, by age group (Fig 1). The middle and late age group now retain their teeth for longer, but in various states of disrepair with chronic periodontal disease and structural and pulpal problems and many with sub-optimal endodontics. The very simplistic view that 'retention of teeth' means that there will be no long-term problems in this group is misconceived and short sighted. However, using that oversimplified 'retention of teeth' as a surrogate measure for improving dental health in older patients means that little resource is allocated to dealing with the increasingly more technically demanding and time consuming treatments to avoid ONJ. In many ways this is a problem that will erupt and worsen over time and it is being largely ignored by senior healthcare professionals responsible for allocating national budgets.
Having three or more indicators would generally suggest either a fair degree of current need for dental care or the probability of significant future maintenance need or both. Graph taken from 4: Complexity and maintenance – a report from the Adult Dental Health Survey 200977
Aggressive prevention of MRONJ
'Aggressive prevention' of potential problems is currently being approached on a pragmatic basis. Patients need to be strongly encouraged to use interspace and interdental brushing twice a day, every day with high strength (5000 ppm) fluoride toothpaste. This meticulous daily removal of plaque should be coupled with using customised mouthguards, with reservoirs in the cervical regions (Figs 2 and 3), in the style of bleaching trays into which very high strength toothpaste eg Duraphat 5000 ppm fluoride toothpaste (Colgate-Palmolive Ltd, Guildford, Surrey GU2 8JZ) (Fig. 4) can be placed for overnight bathing of vulnerable teeth, with the therapeutic fluoride gel not being washed out readily by saliva. A suggested pragmatic approach using this combination overnight or for a couple of hours at least three times a week to help prevent recurring decay seems sensible. This approach has been used for many years in patients who have had radiotherapy for cancer of the head and neck with variable results. The benefits in preventing new decay seem to depend on sugar avoidance, compliance with mouthguards and fluoride gel combination and avoiding smoking. There is a lack of high level research from a meta-analysis of randomised controlled clinical trials to support this approach. However, from first principles such an approach in high risk patients is unlikely to do harm and potentially could help avoid ONJ.
Solution
It is proposed a national surveillance unit be set up to monitor prescriptions of these drugs. The unit could then be responsible for automatically sending information packs to GMPs and GDPs, as well as to the patients. Warning cards with the drug details and on how to access a website for up-to-date practical advice should be developed and circulated. This would help provide much better information to the patient and the health professionals to form a united and informed care team. The patients should also be encouraged to self-report a possible diagnosis of necrosis to the surveillance unit. The disease is sporadic in nature and this presents an almost insurmountable task of understanding the disease, its pathophysiology, and incidence. This is essential information if one is to develop effective management of the condition. Treatment protocols need to be fashioned to guide dentists in the care of these patients.
The NHS will inevitably be faced with a rise in both MRONJ and ORN. This has significant resource implications, especially if the patient is relatively young and has a long lifespan ahead of them. Prevention seems an optimum approach but progress will depend largely on a much better understanding of the real risks involved, a better organised approach and appropriate resource allocation.
References
Chevalier M A, Pirier A . J Chim Med 1858; 4: 139.
Smith J . Case report. St Bart Hosp Rep 1865; 1: 101.
Marx R E . Uncovering the cause of “phossy jaw” circa 1858 to 1906: oral and maxillofacial surgery closed case files-case closed. J Oral Maxillofac Surg 2008; 66: 2356–2363.
Regaud C . Sur la necrose des os attente - par un processus cancereux et traites par les radiaions. Compt Rend Soc Biol 1922; 87: 629. Quoted in Oral Surg 1951; 4.
Meyer I . Infectious diseases of the jaws. J Oral Surg 1970; 28: 17–26.
Marx R . Osteoradionecrosis: a new concept of its pathophysiology. J Oral Maxillofac Surg 1983; 41: 283–288.
Delanian S, Lefaix J L . The radiation-induced fibroatrophic process: therapeutic perspective via the antioxidant pathway. Radiother Oncol 2004; 73: 119–131.
Marx R E . Pamidronate (Aredia) and zoledronate (Zometa) induced avascular necrosis of the jaws: a growing epidemic. J Oral Maxillofac Surg 2003; 61: 1115–1117.
http://www.aaoms.org/images/uploads/pdfs/mronj_position_paper.pdf.
Beumer J, Silverman S, Benak S B . Hard and soft tissue necroses following radiation therapy for oral cancer. J Prosthet Dent 1972; 27: 640–644.
Marx R E . A new concept in the treatment of osteoradionecrosis. J Oral Maxillofac Surg 1983; 41: 351–357.
Hutchinson I L . Complications of radiotherapy in head and neck: an orofacial surgeon's view. In Tobias J S, Thomas P R M (eds) Current radiation oncology. pp 144–177. London: Arnold, 1996
Epstein J B, Wong F L, Dickens A, Szasz I, Lepawsky M . Bone and gallium scans in postradiotherapy osteonecrosis of the jaw. Head Neck 1992; 14: 288–292.
Harris M . The conservative management of osteoradionecrosis of the mandible with ultrasound therapy. Br J Oral Maxillofac Surg 1992; 30: 313–318.
Anastasilakis A D, Toulis K A, Polyzos S A, Terpos E . RANKL inhibition for the management of patients with benign metabolicbone disorders. Expert Opin Investig Drugs 2009; 18: 1085–1102.
Kyrgidis A, Triaridis S, Vahtsevanos K, Antoniades K . Osteonecrosis of the jaw and bisphosphonate use in breast cancer patients. Expert Rev Anticancer Ther 2009; 9: 1125–1134.
Kyrgidis A, Toulis K A . Denosumab-related osteonecrosis of the jaws. Osteoporos Int 2011; 22: 369–370.
Tat S K, Padrines M, Theoleyre S et al. OPG/membranous-RANKL complex is internalized via the clathrin pathway before a lysosomal and a proteasomal degradation. Bone 2006; 39: 706–715.
NICE. Denosumab for the prevention of osteoporotic fractures in postmenopausal women. 2010. Available online at https://www.nice.org.uk/guidance/ta204 (accessed August 2015).
NICE. Denosumab for the prevention of skeletal-related events in adults with bone metastases from solid tumours. Available online at http://www.nice.org.uk/guidance/ta265 (accessed August 2015).
Smith M R, Coleman R E, Klotz L et al. Denosumab for the Prevention of Skeletal Complications in Metastatic Castration-Resistant Prostate Cancer: Comparison of Skeletal-Related Events and Symptomatic Skeletal Events. Ann Oncol 2015; 26: 368–374.
Baron R, Ferrari S, Russel R G . Denosumab and bisphosphonates: different mechanisms of action and effects. Bone 2011; 48: 677–692.
Miller P D, Bolognese M A, Lewiecki E M et al. Effect of denosumab on bone density and turnover in postmenopausal women with low bone mass after long-term continued, discontinued, and restarting of therapy: a randomized blinded phase 2 clinical trial. Bone 2008; 43: 222–229.
Stopeck A T, Lipton A, Body J J et al. Denosumab compared with zoledronic acid for the treatment of bone metastases in patients with advanced breast cancer: A randomized, double-blind study. J Clin Oncol 2010; 28: 5132–5139.
Henry D, Von Moos R, Vadhan-Raj S et al. A double-blind, randomized study of denosumab versus zoledronic acid for the treatment of bone metastases in patients with advanced cancer (excluding breast and prostate cancer) or multiple myeloma. Presented at the European Cancer Organization 15 European Society for Medical Oncology 34 European Multidisciplinary Congress, Berlin, Germany; September 20–24, 2009.
Fizazi K, Carducci M, Smith M et al. Denosumab versus zoledronic acid for treatment of bone metastases in men with castration-resistant prostate cancer: a randomised, double-blind study. Lancet 2011; 377: 813–822.
Mozzati M, Arata V, Gallesio G . Tooth extraction in patients on zoledronic acid therapy. Oral Oncol 2012; 48: 817.
Malden N, Lopes V . An epidemiological study of alendronate-re-lated osteonecrosis of the jaws. A case series from the south-east of Scotland with attention given to case definition and prevalence. J Bone Miner Metab 2012; 30: 171.
FGDP. National study on avascular necrosis of the jaws including bisphosphonate-related necrosis. 2012. Available online at http://www.fgdp.org.uk/_assets/pdf/research/final%20report-27.11.12.pdf (accessed August 2015).
Kunchur R, Need A, Hughes T et al. Clinical investigation of C-terminal crosslinking telopeptide test in prevention and man-agement of bisphosphonate-associated osteonecrosis of the jaws. J Oral Maxillofac Surg 2009; 67: 1167.
Vasudev N S, Reynolds A R . Anti-angiogenic therapy for cancer: current progress, unresolved questions and future directions. Angiogenesis 2014; 17: 471–494.
Carmeliet P, Jain R K . Molecular mechanisms and clinical applications of angiogenesis. Nature 2011 473: 298–307.
Leite de Oliveira R, Hamm A, Mazzone M . Growing tumour vessels: more than one way to skin a cat—implications for angiogenesis targeted cancer therapies. Mol Aspects Med 2011; 32: 71–87.
Ellis L M, Hicklin D J . VEGF-targeted therapy: mecha-nisms of anti-tumour activity. Nat Rev Cancer 2008; 8: 579–591.
Folkman J . Tumour angiogenesis: therapeutic implications. N Engl J Med 1971; 285: 1182–1186.
Cher M L, Towler D A, Rafii S et al. Cancer interaction with the bone microenvironment. Am J Pathol 2006; 168: 1405–1412.
Nakagawa M, Kaneda T, Arakawa T et al. Vascular endothelial growth factor (VEGF) directly enhances osteoclastic bone resorption and survival of mature osteoclasts. FEBS Lett 2000; 473: 161–164.
Aldridge S E, Lennard T W, Williams J R, Birch M A . Vascular endothelial growth factor receptors in osteoclast differentiation and function. Biochem Biophys Res Commun 2005; 335: 793–798.
Estilo C L, Fornier M, Farooki A, Carlson D, Bohle G 3rd, Huryn J M . Osteonecrosis of the jaw related to bevacizumab. J Clin Oncol 2008; 26: 4037–4038.
Koch F P, Walter C, Hansen T et al. Osteonecrosis of the jaw related to sunitinib. J Oral Maxillofac Surg 2011; 15: 63.
Fleissig Y, Regev E, Lehman H : Sunitinib related osteonecrosisof jaw: A case report. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2012; 113: e1.
Christodoulou C, Pervena A, Klouvas G et al. Combination of bisphosphonates and anti-angiogenic factors induces osteonecrosis of the jaw more frequently than bisphosphonates alone. Oncology 2009; 76: 209.
Beuselinck B, Wolter P, Karadimou A et al. Concomitant oral tyrosine kinase inhibitors and bisphosphonates in advanced renal cell carcinoma with bone metastases. Br J Cancer 2012; 107: 1665.
Hoefert S, Eufinger H . Sunitinib may raise the risk of bisphosphonate-related osteonecrosis of the jaw: Presentation of three cases. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2010; 110: 463.
Brunello A, Saia G, Bedogni A et al. Worsening of osteonecrosis of the jaw during treatment with sunitinib in a patient with met-astatic renal cell carcinoma. Bone 2009; 44: 173.
Ayllon J, Launay-Vacher V. Osteonecrosis of the jaw under bisphosphonate and anti-angiogenic therapies: Cumulative toxic-ity profile? Ann Oncol 2009; 20: 600.
Bozas G, Roy A, Ramasamy V et al. Osteonecrosis of the jaw after a single bisphosphonate infusion in a patient with metastatic renal cancer treated with sunitinib. Onkologie 2010; 33: 321.
Agrillo A, Nastro-Siniscalchi E, Facchini A et al. Osteonecrosis of the jaws in patients assuming bisphosphonates and sunitinib: Two case reports. Eur Rev Med Pharmacol Sci 2012; 16: 952.
Lescaille G, Coudert A E, Baaroun V et al. Clinical study evaluating the effect of bevacizumab on the severity of zoledronic acid-related osteonecrosis of the jaw in cancer patients. Bone 2014; 58: 103–107.
https://www.hpra.ie/docs/default-source/Safety-Notices/avastin_dhcp-nov-2010-pdf_081210.pdf?sfvrsn=0.
MHRA. Drug safety update: Bevacizumab and sunitinib: risk of osteonecrosis of the jaw. 2011. Available online at http://www.mhra.gov.uk/Safetyinformation/DrugSafetyUpdate/CON105745 (accessed August 2015).
British National Formulary 67. March-September 2014. pp 575–576.
Guarneri V, Miles D, Robert N et al. Bevacizumab and osteonecrosis of the jaw: incidence and association with bisphosphonate therapy in three large prospective trials in advanced breast cancer. Breast Cancer Res Treat 2010; 122: 181–188.
McArthur H L, Estilo C, Huryn J et al. Osteonecrosis of the jaw (ONJ) among intravenous (IV) bisphosphonate and/or bevacizumab-treated patients at Memorial Sloan-Kettering Cancer Centre (MSKCC). J Clin Oncol 2008; 26: 9588.
Aragon-Ching J B, Ning Y M, Chen C C et al. Higher incidence of osteonecrosis of the jaw (ONJ) in patients with metastatic castration resistant prostate cancer treated with anti-angiogenic agents. Cancer Invest 2009; 27: 221–226.
Kollmannsberger C, Soulieres D, Wong R, Scalera A, Gaspo R, Bjarnason G . Sunitinib therapy for metastatic renal cell carci-noma: recommendations for management of side effects. Can Urol Assoc J 2007; 1: 41–54.
Mendel D B, Laird A D, Xin X et al. In vivo antitumour activity of SU11248, a novel tyrosine kinase inhibitor targeting vascular endothelial growth factor and platelet-derived growth factor receptors: determination of a pharmacokinetic/pharmaco-dynamic relationship. Clin Cancer Res 2003; 9: 327–337.
Pawson T . Regulation and targets of receptor tyrosine kinases. Eur J Cancer 2002; 38 (Suppl 5): S3–S10.
Nicolatou-Galitis O, Migkou M, Psyrri A et al. Gingival bleeding and jaw bone necrosis in patients with metastatic renal cell carcinoma receiving sunitinib: report of 2 cases with clinical implications. Oral Surg Oral Med Oral Pathol Oral Radiol 2012; 113: 234–238.
Bozas G, Allgar V, Greenwood G, Maraveyas A . Osteonecrosis of the jaw in patients treated with sunitinib and zoledronic acid. J Clin Oncol 2011; 29 (Suppl): abstract e15116.
Hart C D, De Boer R H . Profile of cabozantinib and its potential in the treatment of advanced medullary thyroid cancer. Onco Targets Ther 2013; 6: 1–7.
Elisei R, Schlumberger M J, Muller S P et al. Cabozantinib in progressive medullary thyroid cancer. J Clin Oncol 2013; 10.1200/JCO.2012.48.4659. [Epub ahead of print].
Marino R, Orlandi F, Arecco F et al. Osteonecrosis Of The Jaw In A Patient Receiving Cabozantinib. Aust Dent J 2014; 10.1111/adj.12254. [Epub ahead of print].
Bruland Ø S, Nilsson S, Fisher D R, Larsen R H . High-linear energy transfer irradiation targeted to skeletal metastases by the alpha-emitter 223Ra: adjuvant or alternative to conventional modalities? Clin Cancer Res 2006; 12: 6250s–6257s.
Lewington V J . Bone-seeking radionuclides for therapy. J Nucl Med 2005; 46 (Suppl 1): 38S–47S.
Henriksen G, Breistøl K, Bruland Ø S, Fodstad Ø, Larsen R H. Significant antitumour effect from bone-seeking, alpha-particle-emitting (223)Ra demonstrated in an experimental skeletal metastases model. Cancer Res 2002; 62: 3120–3125.
Liepe K . Alpharadin, a 223Ra-based alpha-particle-emitting pharmaceutical for the treatment of bone metastases in patients with cancer. Curr Opin Investig Drugs 2009; 10: 1346–1358.
McDevitt M R, Sgouros G, Finn R D et al. Radioimmunotherapy with alpha-emitting nuclides. Eur J Nucl Med 1998; 25: 1341–1351.
Parker et al. Alpha Emitter Radium-223 and Survival in Metastatic Prostate Cancer. N Engl J Med 2013; 369: 213–223.
University of Birmingham. TRAPEZE Trial. Available online at http://www.birmingham.ac.uk/research/activity/mds/trials/crctu/trials/trapeze/index.aspx (accessed August 2015).
Reiser J, Adair B, Reinheckel T . Specialized roles for cysteine cathepsins in health and disease. J Clin Invest 2010; 120: 3421–3431.
Leung P, Pickarski M, Zhuo Y, Masarachia P J, Duong L T . The effects of the cathepsin K inhibitor odanacatib on osteoclastic bone resorption and vesicular trafficking. Bone 2011; 49: 623–635.
The Health and Social Care Information Centre. Adult Dental Health Survey 2009 - Summary report and thematic series [NS]. Available online at http://www.hscic.gov.uk/pubs/dentalsurveyfullreport09 (accessed August 2015).
Nabil S, Samman N . Risk factors for osteoradionecrosis after head and neck radiation: a systematic review. Oral Surg Oral Med Oral Pathol Oral Radiol 2012; 113: 54–69.
Eisbruch A, Harris J, Garden A S et al. Multi-institutional trial of accelerated hypofraction-ated intensity-modulated radiation therapy for early-stage oro-pharyngeal cancer (RTOG 00–22). Int J Radiat Oncol Biol Phys 2010; 76: 1333–1338.
Nabil S, Samman N . Incidence and prevention of osteoradionecrosis after dental extraction in irradiated patients: a systematic review. Int J Oral Maxillofac Surg 2011; 40: 229–243.
The Health and Social Care Information Centre. Complexity and maintenance - a report from teh Adult Dental Health Survey 2009. 2011. Available online at http://www.hscic.gov.uk/catalogue/PUB01086/adul-dent-heal-surv-summ-them-the4-2009-rep6.pdf (accessed August 2015).
Author information
Authors and Affiliations
Corresponding author
Additional information
Refereed Paper
Rights and permissions
About this article
Cite this article
Patel, V., Kelleher, M., Sproat, C. et al. New cancer therapies and jaw necrosis. Br Dent J 219, 203–207 (2015). https://doi.org/10.1038/sj.bdj.2015.680
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bdj.2015.680
This article is cited by
-
The presenting dental status of solid tumours with bone metastases requiring bone-targeting agents - part 3: prostate cancer
British Dental Journal (2022)
-
Deciphering novel chemotherapy and its impact on dentistry
British Dental Journal (2020)
-
MRONJ risk of adjuvant bisphosphonates in early stage breast cancer
British Dental Journal (2018)
-
Otorhinolaryngological Toxicities of New Drugs in Oncology
Advances in Therapy (2017)