Abstract
Introduction:
A novel pattern of transcranial magnetic stimulation (TMS) abnormalities in cervical spondylotic myelopathy (CSM) comprising abnormal central motor conduction time (CMCT) to the upper limbs and normal CMCT to the lower limbs was observed. CSM was more severe radiologically and tended to be more severe clinically when this pattern was encountered.
Case Presentation:
To further characterize this observation, 414 consecutive TMS evaluations of cervical spondylosis were reviewed. Those cases in which (a) CMCT was abnormal at the upper and (b) normal at the lower limbs and (c) a cervical spine magnetic resonance imaging (MRI) was available (ULabnormal group) were included for further analysis. Cases where CMCT was abnormal at the lower limbs only (LLabnormal) were used for comparison. MRI-measured sagittal and parasagittal diameters of the spinal canal at all intervertebral levels and cervical spinal cord T2 hyperintensities were compared between these groups. Four patients fulfilled all inclusion criteria in each group. In ULabnormal, all patients had T2 hyperintensities, compared to none in LLabnormal (P=0.004). The C6–7 right (6 mm±1.05 vs 8.48 mm±4.01, P=0.05) and left (6.58 mm±1.39 vs 9.17 mm±5.03, P=0.06) parasagittal spinal canal diameters tended to be smaller in ULabnormal. The modified Japanese Orthopaedic Association scale tended to be lower in ULabnormal (11.5±2.65 vs 15.75±0.96, P=0.13).
Discussion:
CMCT abnormalities isolated to the upper limbs constitute a less frequent pattern of involvement, which may correlate with more severe CSM.
Similar content being viewed by others
Introduction
Central motor conduction time (CMCT), measured by transcranial magnetic stimulation (TMS), is an estimate of the conduction time of corticospinal fibers between motor cortex and spinal (or bulbar) motor neurons. It includes the times for excitation of cortical cells, conduction via the corticospinal tract and excitation of the motor neuron sufficient to exceed its firing threshold. The estimate is made by subtracting the spinal motor neuron-to-muscle latency from the cortex-to-muscle latency.1 CMCT is sensitive in detecting cervical spondylotic myelopathy (CSM), which is caused by compression of the cervical spinal cord by spondylotic changes, while it correlates with the severity of cord compression.1–3 CMCT to lower limb muscles is more sensitive and is earlier affected, while upper limb CMCT is affected later in the course of CSM and indicates more severe disease.4,5
It was observed in a minority of patients seen in our laboratory that CMCT to the upper limbs was abnormal, while that to the lower limbs was normal. This pattern of involvement correlated with more severe disease radiologically, with a trend toward more severe clinical symptoms. Here we discuss this unexpected finding.
Materials and methods
All TMS evaluations of cervical spondylosis conducted between October 2008 and July 2016 were retrospectively analysed. Those cases were included for further analysis where (a) CMCT was measured at all four limbs, (b) lower limb CMCT was normal, (c) upper limb CMCT was abnormal and (d) a cervical spinal MRI was available to allow the study of spine anatomy in relation to TMS results. This last inclusion criterion was imposed by the fact that many patients had been referred for TMS evaluation alone; therefore, a spine MRI was not always available.
TMS evaluation comprised determination of CMCT at the upper and lower limbs bilaterally, according to the F-Wave method.6 Motor evoked potentials were recorded from the abductors digiti minimi at the upper limbs and from the extensors digitorum brevis or the tibialis anteriors at the lower limbs, using a circular coil (MC-125, MagVenture, Farum, Denmark) positioned over the vertex. CMCT at the abductors digiti minimis was considered abnormal if it was higher than 9.81 ms or the inter-side difference was higher than 2.78 ms. For the extensors digitorum brevis, an abnormal test was defined as CMCT >17.7 ms or inter-side difference >3.35 ms, and for the tibialis anteriors as CMCT >18.61 ms or inter-side difference >3.49 ms (laboratory-established normal values).
For spinal canal measurements, axial T2 MRI sections were made at the intervertebral levels from C2–3 to C6–7. The slice planes were set parallel to each intervertebral disc space. Spinal canal diameters were measured at all levels with the aid of image-processing software.7 The midline anteroposterior diameter was measured at the midline as the distance between the posterior margin of the intervertebral disc and the anterior margin of the ligamentum flavum (dmri_midline). Lateral anteroposterior diameters were also measured at 50% of the distance between the midline and the left (dmri_left) and right (dmri_right) border of the spinal canal (Figure 1). The lateral borders were set at the interior margins of the pedicles. MRI was further evaluated for spinal cord T2 signal hyperintensities, while radicular lesions were screened by electromyogram.
Patients in whom (a) CMCT was measured at all four limbs, (b) lower limb CMCT was abnormal, (c) upper limb CMCT was normal and (d) a cervical spinal MRI was available were recruited for comparison. The same TMS and MRI parameters were measured in this second group and were compared between the two groups: patients with abnormal CMCT at the upper limbs (ULabnormal) or the lower limbs (LLabnormal). The aim was to identify anatomical characteristics in the cervical spine MRI that could account for the selective involvement of the upper or lower limb CMCT in TMS and to correlate said MRI characteristics with different pathophysiological mechanisms, if possible.
Patients with abnormal CMCT at both the upper and lower limbs were not included in the analysis. This pattern of involvement might have occurred as a result of (a) a long-standing lesion initially affecting the lower limbs, which by time progressed to the upper limbs, or of (b) a lesion that initially affected the upper limbs, as in the ULabnormal group, but progressed to the lower limbs by time, or of (c) a severe lesion, which affected all four limbs right from the beginning. As it would not be possible to differentiate between these cases on the basis of a single TMS and MRI evaluation for each patient, this group could have confounded our results.
Two-tailed Fisher’s exact test was used for between-group comparisons. Rates were compared with the Barnard test.8 All calculations were done in LibreOffice 5.2.
Results
Of the 414 consecutive TMS evaluations performed, isolated upper limb CMCT abnormalities were seen in 15 patients (3.7%), isolated lower limb abnormalities in 62 (15%), combined upper and lower limb abnormalities in 141 (34%), while the remaining 196 evaluations (47.3%) were normal. However, all inclusion criteria were fulfilled by four patients with isolated upper limb abnormalities (ULabnormal group) and four patients with isolated lower limb abnormalities (LLabnormal group). The demographic, clinical (expressed in terms of the modified Japanese Orthopaedic Association assessment scale (modified-JOA)9) and TMS evaluations of patients in the ULabnormal and LLabnormal groups are shown in Table 1. The MRI measurements are shown in Table 2.
Table 3 compares modified-JOA and MRI measurements between the two groups. Spinal canal diameters (dmri_right, dmri_midline and dmri_left) were not significantly different at C2–3, C3–4, C4–5 and C5–6. At C6–7 dmri_right and dmri_left were smaller in ULabnormal with borderline statistical significance (P=0.05 and P=0.06, respectively). dmri_midline was not significantly different. Furthermore, all patients in ULabnormal had T2 hyperintensities, compared to none in LLabnormal (P=0.004).
The level of T2 hyperintensities in the ULabnormal group did not always correspond to the level of maximum stenosis in MRI. In patient #1 (Table 2), maximum stenosis was seen at level C6–7, while T2 hyperintensity was evident as C3–4. In patient #2, hyperintensity was evident at C3–4, while maximum stenosis occurred at C5–6 and C6–7. In patient #3, hyperintensity was seen at C5–6, while significant stenosis occurred at C4–5, C5–6 and C6–7 (maximum at C6–7). In patient #4, hyperintensity was found at C3–4 and stenosis was observed at all C3–4, C4–5, C5–6, C6–7 levels (maximum at C5–6).
There were no findings of radicular lesions in the electromyogram in any of the patients, with the exception of patient #4 in ULabnormal, where EMG yielded signs of chronic denervation and re-innervation at the level of C8, on the right (a discrete maximum voluntary contraction diagram and high-amplitude motor unit were recorded from the abductors digiti minimi).
Modified-JOA was not significantly different between the two groups, although there was a trend toward higher values (better clinical condition) in LLabnormal.
Discussion
It is commonly accepted that CMCT to lower limb muscles is more sensitive and is earlier affected, while upper limb CMCT is affected later. This belief is supported by many published studies.2–5 Under time pressure one might opt to study CMCT to the lower limbs only, assuming that if it is normal then CMCT to the upper limbs should be normal as well. Here initial evidence is provided against this belief. In a small, yet measurable, subset of patients, CSM manifested with isolated upper limb TMS abnormalities, leaving the lower limb CMCT unaffected. In these patients, T2 hyperintensities were significantly more frequent in the cervical spine MRI. The spinal canal was narrower laterally, at the C6–7 level, albeit with borderline statistical significance, while modified-JOA tended to be, but was not significantly, lower in this group.
The number of patients that presented with this pattern of TMS abnormalities was small, which limits the statistical power of these findings. However, the present series of TMS evaluations in suspected CSM (414 patients) is among the biggest found in the literature; therefore, the small number of patients with isolated upper limb abnormalities may reflect the rarity of this condition. Four such patients were included in this study, while in another 11, the results of TMS evaluation were consistent with this pattern, but cervical MRI was not available. Thus, the frequency of isolated upper limb CMCT abnormalities in this series ranges from a validated lower limit of 1% (4/414) to a potential upper limit of 3.7% (15/414).
The number of patients with isolated lower limb CMCT abnormalities, who served as comparators, was also small (four patients); because CMCT to the lower limbs is affected first, as discussed above, more patients would have been expected to have presented with this pattern of TMS findings, compared to those with isolated upper limb abnormalities. Indeed, 62 patients (15%) in total fulfilled the TMS criteria, but MRI was not available in most of them. Thus, only four fulfilled all inclusion criteria for this study. The small number of patients fulfilling all inclusion criteria may have been responsible for the borderline significance found in MRI measurements and in clinical scores. Confirmation of these findings in larger studies is thus warranted.
The pathophysiological underpinnings of the different patterns of involvement of upper and lower limb pyramidal tracts in CSM are not entirely clear. Blood flow supply changes to the cervical cord, leading to ischemia, demyelination and axon loss have all been implicated in the pathogenesis of CSM.4,9,10,11 It has been suggested that when there is a close correlation between MRI and TMS findings, the most important etiological factor may be probably a segmental demyelination of central motor pathways due to a direct mechanical spinal cord compression. When discrepancies between the level of spinal cord compression documented by neuroimaging studies and the level of spinal cord dysfunction revealed by TMS are observed,3,12 more mechanisms come into play. A more caudal functional involvement of the cervical cord revealed by TMS has been attributed to compromised blood flow to the spinal cord. The anterolateral regions of lower cervical segments are blood supplied almost exclusively from the anterior spinal artery, whereas the higher cervical segments that are located between the cervical and intracranial arterial territories have more sources of blood. Therefore, the frequent involvement of lower cervical segments may depend on their higher vulnerability to ischemic damage. When cervical spondylotic compression involves the anterior spinal artery, the major damage is of vascular origin and localized to lower cervical segments, independent from the level of spondylotic degenerative changes.3,13 These observations are mostly based on evidence stemming from small, older anatomical studies; hence, more work is needed to fully elucidate the pathophysiological mechanisms involved in CSM. Nevertheless, histological findings of both spinal cord ischemia and demyelination have been found in autopsy material in said studies,14,15 alluding to the fact that both these mechanisms come into play in CSM.
We hypothesize that ischemic damage is the major pathogenetic factor in patients presenting with isolated upper limb CMCT abnormalities. Older anatomical studies, from the work of Adamkievicz in 188116 up to that of Chakravorty in 1969,17 have suggested that the cervical radicular arteries, that is, the arteries accompanying the nerve roots entering through the intervertebral foramina, also supply the spinal cord. Dye injected into these arteries was capable of filling both anterior and posterior arteries in the cervical cord.17 These radicular arteries may originate from any branch of the subclavian artery in the neck, that is, from the vertebral, costocervical and thyrocervical trunks. In the upper six segments, they can arise from the vertebrals or from the ascending cervical branch of the thyrocervical trunk, and the spinal branches of these two vessels always anastomose.17
In most cases the cervical radicular arteries are two or three; in up to two-third of cases there is only one such artery.17 They accompany more frequently the C4–6 nerve roots and very rarely C3, C7 and C8.17 It has been suggested that if the radicular artery (or arteries) is occluded the risk of spinal cord ischemia increases. The risk is greater if there is only a single major radicular artery, which is occluded and is more likely to occur at the presence of lateral disc protrusion, when the protruding disc is in closer anatomical relation to said artery.17
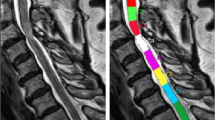
Indeed, in our ULabnormal patients, the lateral diameters of the spinal canal (dmri_right and dmri_left) at C6–7 were smaller compared to LLabnormal, albeit with borderline statistical significance, while there was a trend for lower diameters at C5–6, where most radicular arteries are found. Figure 1 shows an axial T2 MRI section of ULabnormal patient #4 at the C5–6 level. Significant right lateral stenosis is obvious, while CMCT is also prolonged at the right upper limb. The role of ischemia in this pattern of TMS abnormalities is further supported by the fact that in three of the four ULabnormal patients, T2 hyperintensities were seen rostral (C3–4) to the level of stenosis (C4–5, C5–6 and/or C6–7). In the remaining patient, stenosis extended from C3–4 to C6–7. LLabnormal patients, in whom T2 hyperintensities were not seen in the MRI, also had considerable stenosis, but less than that in ULabnormal patients at C5–6 and C6–7. The mean spinal canal diameters at all intervertebral levels for the two groups are shown side-by-side for comparison in Figure 2. At C5–6 and C6–7 mean diameters in ULabnormal are smaller compared to LLabnormal, with borderline statistical significance.
Comparison of mean spinal canal diameters and T2 hyperintensities at all intervertebral levels for the ULabnormal and LLabnormal groups. At C5–6 and C6–7 mean diameters in ULabnormal are smaller, especially ULabnormal dmri_right and dmri_left, for which the differences are borderline significant. T2 hyperintensities are significantly more frequent in ULabnormal.
In conclusion, we have observed that in a small subset of patients with CSM, TMS discloses isolated upper limb CMCT abnormalities. This pattern of involvement correlates with more severe disease, both clinically and radiologically, and should be sought by neurophysiologists, who should not limit their evaluation to the, commonly accepted as more sensitive to CSM, lower limbs. As this was a relatively rare finding in the present series, confirmation by other researchers is warranted, while more detailed studies are needed to elucidate its pathophysiological underpinnings.
References
Chen R, Cros D, Curra A, Di Lazzaro V, Lefaucheur JP, Magistris MR et al. The clinical diagnostic utility of transcranial magnetic stimulation: report of an IFCN committee. Clin Neurophysiol 2008; 119: 504–532.
Deftereos SN, Kechagias E, Ioakeimidou C, Georgonikou D . Transcranial magnetic stimulation but not MRI predicts long-term clinical status in cervical spondylosis: a case series. Spinal Cord 2015; 53 (Suppl 1): S16–S18.
Nardone R, Höller Y, Brigo F, Frey VN, Lochner P, Leis S et al. The contribution of neurophysiology in the diagnosis and management of cervical spondylotic myelopathy: a review. Spinal Cord 2016; 54: 756–766.
Lo YL . The role of electrophysiology in the diagnosis and management of cervical spondylotic myelopathy. Ann Acad Med Singapore 2007; 36: 886–893.
Brunhölzl C, Claus D . Central motor conduction time to upper and lower limbs in cervical cord lesions. Arch Neurol 1994; 51: 245–249.
Hallett M, Chokroverty S . In: Magnetic Stimulation in Clinical Neurophysiology 2nd edn. Butterworth-Heinemann: Philadelphia, 2005, pp 106–120.
Ginkgo CADx v3.7.1. Available at http://ginkgo-cadx.com. Accessed on 31 October 2016.
Mehta CR, Hilton JF . Exact power of conditional and unconditional tests: going beyond the 2×2 contingency table. Am Stat 1993; 47: 91–98.
Benzel EC, Lancon J, Kesterson L, Hadden T . Cervical laminectomy and dentate ligament section for cervical spondylotic myelopathy. J Spinal Disord 1991; 4: 286–295.
Fehlings MG, Skaf G . A review of the pathophysiology of cervical spondylotic myelopathy with insights for potential novel mechanisms drawn from traumatic spinal cord injury. Spine 1998; 23: 2730–2737.
Gooding MR, Wilson CB, Hoff JT . Experimental cervical myelopathy: effects of ischemia and compression in the canine cervical cord. J Neurosurg 1975; 43: 9–17.
Blisard KS, Follis F, Wong R, Miller KB, Wernly JA, Seremin OU . Degeneration of axons in the corticospinal tract secondary to spinal cord ischemia in rats. Paraplegia 1995; 33: 136–140.
Di Lazzaro V, Oliviero A . Evaluation of myelopathy, radiculopathy and thoracic nerve. In: Hallett M, Chokroverty S (eds). Magnetic Stimulation in Clinical Neurophysiology. Butterworth-Heinemann: Philadelphia, PA, USA, 2005, pp 105–127.
Mair WG, Druckman R . The pathology of spinal cord lesions and their relation to the clinical features in protrusion of cervical intervertebral discs; a report of four cases. Brain 1953; 76: 70–91.
Allen KL . Neuropathies caused by bony spurs in the cervical spine with special reference to surgical treatment. J Neurol Neurosurg Psychiatry 1952; 15: 20–36.
Adamkiewicz A . Die Blutgefässe des menschlichen Rückenmarkes. II. Die Gefässe der Rückenmarksoberfläche. Sitzber Akad Wiss Wien Math-Naturwiss 1881; 84: 469–502.
Chakravorty BG . Arterial supply of the cervical spinal cord and its relation to the cervical myelopathy in spondylosis. Ann R Coll Surg Engl 1969; 45: 232–251.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no conflict of interest.
Rights and permissions
About this article
Cite this article
Deftereos, S. Abnormal central motor conduction at the upper but not lower limbs correlates with severe cervical spondylosis: discussion of an unexpected observation. Spinal Cord Ser Cases 3, 17009 (2017). https://doi.org/10.1038/scsandc.2017.9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/scsandc.2017.9