Abstract
Introduction:
Spinal epidural abscess (SEA) is a rare but serious cause of back pain in the critical care setting. It occurs most commonly in adults in their fifth and sixth decades of life. Risk factors include diabetes mellitus, alcoholism, AIDS or other immunocompromised states, cancer, intravenous drug use, trauma and spinal surgery. The clinical presentation can be non-specific but the classical triad includes back pain, fever and neurological deficits. Magnetic resonance imaging (MRI) with gadolinium is the diagnostic imaging modality of choice.
Case presentation:
Here we report a case of SEA in a 63-year-old man with type II diabetes who presented with severe low back pain. He was found to have SEA likely secondary to a hip joint injection. The diagnosis was delayed due an earlier non-gadolinium-enhanced MRI of the spine showing no epidural abscess.
Discussion:
This case stresses the need for the definitive diagnostic study, MRI with gadolinium, in patients whose SEA is high on the list of differential diagnoses.
Similar content being viewed by others
Introduction
Spinal epidural abscess (SEA) is a rare but serious cause of back pain in the critical care setting. The first case of SEA was described in the medical literature in 1761. At the beginning of the twentieth century, mortality for SEA approached 100%, whereas today mortality is 15–20%.1 SEA represents an often pyogenic infection of the epidural space requiring prompt identification and treatment to prevent serious neurologic sequelae or death. Other back pain emergencies include cauda equine syndrome, abdominal aortic dissection and spinal hematoma.
Epidural abscesses most commonly occur during the fifth and sixth decades of life. Risk factors include diabetes, alcoholism, immune-compromised states, intravenous drug use, trauma and surgery.
The clinical presentation can be non-specific but the classical triad includes back pain, fever and neurological deficits. Magnetic resonance imaging (MRI) with gadolinium is the diagnostic imaging modality of choice. Here we report a case of SEA likely secondary to a hip joint injection that was not diagnosed with MRI alone.
Case presentation
A 63-year-old man with a medical history of degenerative joint disease, psoriatic arthritis and diabetes mellitus presented to the emergency department with his chronic lower back pain. An X-ray was performed demonstrating degenerative changes with disk space narrowing but no acute fracture (Figure 1). He was given intravenous dexamethasone, ketorolac and hydromorphone with some improvement of symptoms, and he was discharged home with meloxicam, oral steroids and oxycodone/acetaminophen. Later that same day, he went to an outpatient clinic where he received a steroid injection in his left hip.
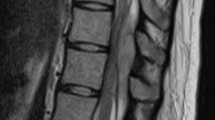
He returned to the emergency department a week later with worsening back pain and inability to ambulate. The pain was sharp, located in the left lower back and flank, and radiated to his left groin. An MRI without contrast of the thoracic and lumbar spine was performed (Figure 2) demonstrating severe degenerative disk disease with collapse of the disk spaces at L3-4, L4-5, L5-S1. In addition, there was a non-specific abnormal signal in those disks.
Non-contrast MRI of the spine. An MRI without contrast was performed demonstrating severe degenerative disk disease with collapse of the disk space at L3-4, L4-5 and L5-S1. In addition, there was an abnormal signal in all of those disks with increased signal on T2-weighted imaging but no epidural abscess.
On admission, he also admitted to fever, diarrhea, weight loss, nausea and anorexia.
He was admitted to the medical floors and started on pain medications and muscle relaxants. On day 2 of his hospital stay he became hypotensive, febrile, with abdominal distention. He also developed bloody diarrhea, nausea, vomiting and epigastric pain. The hypotension persisted despite 7 l of normal saline and his lactate was elevated (3.7 mmol l−1). He was transferred to the intensive care unit shortly thereafter for continuing care.
An abdominal X-ray was performed that morning that revealed dilated loops of bowel concerning for a small bowel obstruction. Blood cultures demonstrated 2/2 bottles positive for methicillin-sensitive Staphylococcus aureus (MSSA) and a chest X-ray demonstrated multiple nodular opacities concerning for septic emboli (Figures 3 and 4).
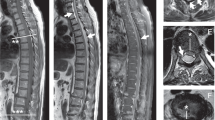
A central line was placed and vasopressor therapy was started. A nasogastric tube was inserted to decompress the stomach, and broad-spectrum antibiotics were initiated. A transesophageal echocardiogram was negative for valvular vegetations. His low back pain was persistent; so on day 4 in the hospital, a repeat MRI was obtained but with the addition of gadolinium (Figures 5 and 6).
MRI spine with gadolinium. MRI with gadolinium demonstrating an epidural contrast-enhancing mass extending from mid L1 to upper L5 and is most consistent with epidural pannus and abscess. It is resulting in mild-to-moderate central canal stenosis, particularly at L3 and L4 and hyperintensity of the disc space at L3-4 suggestive of a disc space infection at this level. The vertebral body of inferior L3 is irregular in signal, consistent with osteomyelitis.
Case continued
Neurosurgery was consulted and percutaneous drainage of the epidural abscess was performed. Definitive surgery intervention was recommended only if he developed any signs of worsening neurological deficits. His back pain improved after several days of antibiotics. Interventional radiology percutaneously drained both psoas abscesses. A left hip arthrocentesis was performed and the joint fluid also grew MSSA. His blood cultures were eventually sterilized after 1 week of antibiotics. An Esophagogastroduodenoscopy demonstrated multiple duodenal and gastric ulcers. The small bowel obstruction improved and his diet was advanced. Vasopressors were discontinued and he was able to leave the intensive care unit.
Diagnosis
-
T4-T5 osteomyelitis, L3-L4 diskitis and multi-level lumbar epidural abscess with mild cord compression
-
MSSA bacteremia with urinary tract infection and pulmonary septic emboli
-
Left hip MSSA septic arthritis
-
Bilateral MSSA psoas abscesses
The patient’s outpatient hip injection was thought to be the causative factor.
Discussion
SEA is a rare condition (0.2–2.8 cases per 10 000 per year) and is an emergent cause of low back pain in the acute care setting.2 It occurs most commonly in adults in their fifth and sixth decades of life. Risk factors include diabetes mellitus, alcoholism, AIDS or other immunocompromised states, cancer, intravenous drug use, trauma and spinal surgery.3 However, up to 20% of patients will have no predisposing factors.4 The patient presented had the predisposing factor of older age and risk factors of diabetes, and a traumatic procedure.
The majority of SEA occur posteriorly (80%), and are usually related to a distant infectious focus such as dental abscesses, pharyngitis or skin and soft tissue infections.5 Anterior infections (20% of cases) are generally related to osteomyelitis or diskitis. The thoracic and lumbar spine account for most infections with only 20% of epidural abscesses occurring in the cervical spine.6
The classic triad for the presentation of SEA is fever, back pain and neurological deficits although commonly the presentation is non-specific. Back pain is the most common presenting symptom followed by motor weakness, radicular pain and fever.7–9
A chart review of 46 patients with SEA over a 10-year period demonstrated the following risk factors: diabetes (46%), frequent venous puncture (35%), spinal trauma (24%) and history of spinal surgery (22%).8
Erythrocyte sedimentation rate was also elevated uniformly (a mean of 86.6 mm h−1) and as in this case, Staphylococcus aureus was the most common causative organism (39%). The presence of thrombocytopenia, extremely high erythrocyte sedimentation rate (>110 mm h−1) and cervical spine epidural abscesses portended a poorer outcome.
Once there is clinical suspicion for SEA, it is crucial to promptly diagnose and initiate treatment. In one study, out of 10 patients where the diagnosis was not made on hospital admission, 4 became paraplegic and 3 died. The authors found that gadolinium-enhanced MRI was the most useful diagnostic test, and recommend that contrast-enhanced MRI (CEMRI) be performed promptly in any patient with clinical features suggesting SEA since early surgical drainage and appropriate antibiotics contribute to a favorable outcome.10
The challenge in this case was that the definitive study—MRI with gadolinium was delayed due an earlier non-CEMRI showing no epidural abscess. This delay in diagnosis led to delay in treatment. In a small study comparing CEMRI to non-contrast MRI, contrast enhancement was thought to be more sensitive for the diagnosis of SEA especially if it involved multiple disk spaces as in this case.11,12
In one British case series, the diagnosis of spinal epidural empyema was made with gadolinium-enhanced MRI in eight of nine cases and S. aureus was the causative organism in all cases.13 Lumbar puncture to determine cerebrospinal fluid protein concentrations is not needed for diagnosis and carries the risk of spreading bacteria into the subarachnoid space with consequent meningitis; therefore, it should not be performed. CEMRI has also replaced computed tomography myelography as the diagnostic procedure of choice.1
As any delay in treatment may contribute to irreversible neurological damage or death, once the diagnosis is suspected, neurosurgery consultation is recommended. A blood culture should be obtained (60% of patients have bacteremia) to confirm the diagnosis.9
There are two patterns that are commonly seen on gadolinium-enhanced MRI. The first is a homogenous enhancement of the abnormal area seen in the phlegmonous stage, which correlates with granulomatous-thickened tissue with embedded microabscesses without a significant pus collection. The second pattern is a fluid abscess surrounded by peripheral inflammation that enhances in CEMRI.14 CEMRI is also useful in distinguishing epidural abscess from the adjacent compressed thecal sac and other potential compressive lesions such as a herniated disc.
In T1-weighted MRI images, SEA appears as decreased signal in the vertebral body with loss of cortical margins and continuity.15 In T2-weighted images, the abscesses lead to increased signal in the affected intervertebral disc and vertebral body.16
This case underlies the importance of obtaining the definitive study (MRI with gadolinium) in patients with risk factors and clinical presentation concerning for SEA if they are able to tolerate contrast. Any delay in establishing the diagnosis can contribute to significant morbidity and mortality in these patients.
Treatment for SEA usually involves 4–6 weeks of antibiotics and surgical decompression or percutaneous drainage. In certain patients, conservative management is warranted.
References
Reihsaus E, Waldbaur H, Seeling W . Spinal epidural abscess: a meta-analysis of 915 patients. Neurosurg Rev 2000; 23: 175–204, discussion 205.
Martin RJ, Yuan HA . Neurosurgical care of spinal epidural, subdural, and intramedullary abscesses and arachnoiditis. Orthop Clin North Am 1996; 27: p 125–136.
Chao D, Nanda A . Spinal epidural abscess: a diagnostic challenge. Am Fam Physician 2002; 65: 1341–1346.
Vilke GM, Honingford EA . Cervical spine epidural abscess in a patient with no predisposing risk factors. Ann Emerg Med 1996; 27: 777–780.
Baker AS, Ojemann RG, Swartz MN, Richardson EP . Spinal epidural abscess. N Engl J Med 1975; 293: 463–468.
Kabbara A, Rosenberg SK, Untal C . Methicillin-resistant Staphylococcus aureus epidural abscess after transforaminal epidural steroid injection. Pain Physician 2004; 7: 269–272.
Sendi P, Bregenzer T, Zimmerli W . Spinal epidural abscess in clinical practice. QJM 2008; 101: 1–12.
Tang HJ, Lin HJ, Liu YC, Li CM . Spinal epidural abscess--experience with 46 patients and evaluation of prognostic factors. J Infect 2002; 45: 76–81.
Darouiche RO, Hamill RJ, Greenberg SB, Weathers SW, Musher DM . Bacterial spinal epidural abscess. Review of 43 cases and literature survey. Medicine 1992; 71: 369–385.
Teman AJ . Spinal epidural abscess. Early detection with gadolinium magnetic resonance imaging. Arch Neurol 1992; 49: 743–746.
Sadato N, Numaguchi Y, Rigamonti D, Kodama T, Nussbaum E, Sato S et al. Spinal epidural abscess with gadolinium-enhanced MRI: serial follow-up studies and clinical correlations. Neuroradiology 1994; 36: 44–48.
Rigamonti D, Liem L, Sampath P, Knoller N, Namaguchi Y, Schreibman DL et al. Spinal epidural abscess: contemporary trends in etiology, evaluation, and management. Surg Neurol 1999; 52: 189–196, discussion 197.
Pilkington SA, Jackson SA, Gillett GR . Spinal epidural empyema. Br J Neurosurg 2003; 17: 196–200.
Numaguchi Y, Rigamonti D, Rothman MI, Sato S, Mihara F, Sadato N . Spinal epidural abscess: evaluation with gadolinium-enhanced MR imaging. Radiographics 1993; 13: 545–559, discussion 559–60.
Sandhu FS, Dillon WP . Spinal epidural abscess: evaluation with contrast-enhanced MR imaging. AJNR Am J Neuroradiol 1991; 12: 1087–1093.
Pradilla G, Ardila GP, Hsu W, Rigamonti D . Epidural abscesses of the CNS. Lancet Neurol 2009; 8: 292–300.
Acknowledgements
CC-M takes responsibility for the content of the manuscript, including the case compilation, description and literature review. MP and MS provided mentorship and constructive review during the writing of this case.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Chima-Melton, C., Pearl, M. & Scheiner, M. Diagnosis of spinal epidural abscess: a case report and literature review. Spinal Cord Ser Cases 3, 17013 (2017). https://doi.org/10.1038/scsandc.2017.13
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/scsandc.2017.13