Abstract
Study design:
Retrospective study.
Objectives:
The objective of this study was to find out whether ossification of posterior longitudinal ligament (OPLL) characteristics, including size, shape and subtype, can be used to diagnose myelopathy using somatosensory evoked potential (SEP) in cervical OPLL patients.
Setting:
Yonsei University College of Medicine, Seoul, Korea.
Methods:
We retrospectively reviewed the medical records of 153 cervical OPLL patients who underwent SEP study. OPLL anterior–posterior (AP) diameter, area and involved longitudinal vertebral level were measured. OPLL was classified into subtypes according to longitudinal continuity and shape. Correlation analysis and receiver operating curve were used.
Results:
Tibial SEP latency was significantly correlated with OPLL AP diameter (P=0.001), diameter occupying ratio (P=0.019), area (P=0.007), area occupying ratio (P=0.008), involved longitudinal vertebral level (P=0.028) and space available for the spinal cord (P=0.019). The cutoff values that were diagnostic for SEP prolongation suggesting myelopathy were 4.91 mm for OPLL AP diameter, 6.02 mm for space available for the spinal cord, 44.5% for diameter occupying ratio, 63.4 mm2 for area, 36.1% for area occupying ratio and level 2 for the involved longitudinal vertebral level.
Conclusions:
Our results revealed that tibial SEP latency was significantly correlated with OPLL size and suggested cutoff values of OPLL diameter (4.91 mm, 44.5%) and area (63.4 mm2, 36.1%) for early diagnosis of myelopathy. These results can help to establish treatment plans.
Similar content being viewed by others
Introduction
Ossification of posterior longitudinal ligament (OPLL) is pathological calcification of the posterior longitudinal ligament. This condition can compress the spinal cord and lead to myelopathy. The prevalence of cervical OPLL is known to be higher in Asians than other races, and it has recently shown an increase in prevalence due to greater detection on computed tomography (CT) compared with plain radiographs.1 The pathomechanism of myelopathy of the OPLL remains obscure. Myelopathy can be diagnosed when there are neurological symptoms, abnormal findings on imaging studies such as magnetic resonance imaging (MRI) or electrodiagnostic studies, such as somatosensory evoked potential (SEP). Some trials have investigated the correlation between OPLL characteristics, such as anterior–posterior (AP) diameter and subtype, and intramedullary high signal change on T2-weighted MRI.2, 3 Other studies have focused on the correlation between OPLL characteristics and clinical symptoms, such as weakness, sensory change, pain and postoperative outcomes.4, 5, 6, 7, 8 However, there have been no trials to demonstrate the correlation between preoperative SEP and OPLL characteristics. Almost all studies about the relationship between SEP and OPLL have focused on intraoperative SEP monitoring.
SEP is a noninvasive electrophysiological study used to monitor somatosensory pathways, including spinal cord integrity. It has been used to monitor neurological conditions in spinal cord and brain injuries.9, 10, 11, 12 SEP has also been widely used for vertebral and brain surgery to monitor spinal cord integrity during operative treatment and to predict postoperative outcomes.13, 14 Some previous studies suggested that SEP could predict later compressive myelopathy earlier than other methods, such as neurological symptom or MRI finding.9, 15 Thus to find out the correlation between SEP and OPLL characteristics and to suggest cutoff value of OPLL size would be helpful for early detection of myelopathy in cervical OPLL characteristics.
To our knowledge, no prior studies have investigated the correlation between OPLL characteristics and preoperative SEP. Therefore, the objective of this study was to demonstrate the correlation between size, shape, subtype of OPLL and SEP in cervical OPLL patients and to suggest the cutoff value of OPLL size to predict myelopathy on SEP study.
Materials and methods
Study participants
We retrospectively reviewed the medical records of 153 cervical OPLL patients who underwent SEP study between January 2012 and October 2014 at our University Hospital. The diagnosis of OPLL was confirmed by CT. All patients underwent cervical plain radiography, CT and MRI. Exclusion criteria were as follows: (1) a previous history of cervical operation; (2) a combined diagnosis of ossification of the ligament flavum; (3) combined thoracic OPLL; and (4) insufficient radiological or medical records. Demographic data, including age, sex, neurological symptoms and electrodiagnostic findings, were recorded. This study was approved by the Institutional Review Board for Clinical Studies at our institution.
OPLL size measurement and OPLL type classification
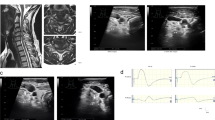
The AP diameter of the spinal canal and OPLL were measured on CT at the maximal compression level (Figure 1a). The OPLL AP diameter occupying ratio was calculated with the following equation: OPLL AP diameter/spinal canal AP diameter × 100. The spinal canal area and OPLL area were measured at the maximal compression level (Figure 1b). The OPLL area occupying ratio was calculated with the following formula: OPLL area/spinal canal area × 100. The OPLL size, including diameter and area, was measured with a Picture archiving and communication system (Centricity PACS Radiology RA1000 Workstation, GE Healthcare, Barrington, IL, USA). An experienced radiologist interpreted the radiological findings and measured the diameter and area of the OPLL three times; the values were then averaged. The extent of the OPLL level was measured at levels 1–7 on sagittal-view CT. The type of OPLL was classified as continuous, segmental, mixed or localized according to longitudinal continuity, and the shape of OPLL was classified as plateau or hill on sagittal-view CT by the criteria of previous studies.3, 16 Axial shape was classified as single-layer sign, double-layer sign or c sign on axial-view CT by the criteria of previous studies.3, 16
Electrophysiological evaluation—SEP
SEP of the median and tibial nerves was performed when patients were diagnosed with cervical OPLL by radiological evaluation. Patients were positioned supine and relaxed on a secured bed in a silent examination room kept at 22–24 °C. Skin temperature was maintained at 32–36 °C. The skin was cleansed and covered with electrolyte gel. SEP was recorded by stimulation of the bilateral median nerves at the wrist and bilateral posterior tibial nerves at the ankle by using bar electrode. Recordings were performed via needle electrodes from the scalp at C3 (right median nerve stimulation), C4 (left median nerve stimulation) and Cz (right and left tibial nerve stimulation) positions and from a reference electrode at Fz according to the 10–20 international EEG system. The stimulation intensity was arranged to produce minimal twitching movement in the thumb for the median nerve and in the first toe for the tibial nerve. The cortical parietal response N20 for the median nerve and the cortical P40 response for the tibial nerve were recorded. The process was repeated at least twice, and the results were averaged for each recording session. The EMG equipment (Medelec Synergy; Oxford Instruments Medical Ltd., Surrey, UK) used for SEP evaluation included a built-in monitor, bipolar stimulator and discoid electrodes. The following settings were used: 0.05-ms stimulation duration, 3 Hz repetition rate, 250 repetitions, 20–2000 Hz band filter width, 2 μV per division sensitivity, and 10 ms per division sweep speed.
To find the cutoff value for deteriorated spinal cord integrity, we calculated abnormal tibial SEP separately for each patient, considering the patient’s height, using our laboratory tibial SEP data on the basis of previous research.17
Statistical analysis
Statistical analysis was performed with SPSS 20.0 for Windows. Simple correlation analysis was used to assess the relationships between OPLL size, including AP diameter, area, involved longitudinal vertebral level and SEP. Student’s t-tests and analysis of variance were used to compare means between different OPLL types. To identify the predictive value of OPLL size for the risk of SEP prolongation, which suggests myelopathy, receiver operating characteristic (ROC) curves were used with analysis of the area under the curve (AUC) and determination of sensitivity and specificity, as well as identification of a cutoff value of the highest sensitivity and specificity. Student’s t-test was used to compare means of OPLL size between symptomatic and asymptomatic patients. A P-value of less than 0.05 was considered statistically significant.
Results
Demographic data
Study patients included 119 men and 34 women with a mean age of 54±10 years. Mean median SEP latency was 20.41±2.41 ms, and tibial SEP latency was 43.97±6.50 ms. Mean OPLL AP diameter was 5.75±1.80 mm, space available for the spinal cord (SAC) was 6.30±2.02 mm and OPLL diameter occupying ratio was 47.88±13.77%. Mean OPLL area was 61.62±30.83 mm2, residual canal area was 143.14±41.26 mm2 and OPLL area occupying ratio was 30.27±13.85%. The involved longitudinal vertebral level was 4.1±1.5 level. The type of OPLL by continuity was classified as follows: segmental, n=46 (30%); continuous, n=41 (27%), mixed, n=58 (40%); and localized, n=8 (5%). The OPLL shape was a plateau in 138 patients (90%) and a hill in 18 patients (10%). Axial shape was distributed as a single-layer sign in 122 patients (80%), double-layer sign in 21 patients (14%) and c sign in 10 patients (7%). The demographic data and types of OPLL of the 153 patients with cervical OPLL are summarized in Table 1.
Analysis of the relationship between OPLL characteristics and SEP
Correlation analysis revealed that tibial SEP latency was positively correlated with both the OPLL AP diameter (r=0.264, P=0.001) and OPLL AP diameter occupying ratio (r=0.251, P=0.019) weekly. The OPLL area (r=0.218, P=0.007) and OPLL area occupying ratio (r=0.215, P=0.008) were also weekly positively correlated with tibial SEP. The involved longitudinal vertebral level of the OPLL was significantly associated with tibial SEP prolongation (r=0.178, P=0.028). In addition, SAC was negatively correlated with SEP prolongation (r=−0.190, P=0.019) (Table 2, Figure 2). However, the correlation between OPLL size and median SEP was not significant.
The SEP latency did not differ significantly between plateau- and hill-shape patients. Analyses of variance revealed no significant differences in SEP latency between patients classified by longitudinal continuity (segmental, continuous, mixed and localized types) or axial shape (single layer, double layer and c sign).
Cutoff value of OPLL size that were diagnostic for SEP prolongation suggesting myelopathy
The ROC curve analysis showed that the cutoff value of OPLL AP diameter to predict deteriorated spinal cord integrity, suggesting myelopathy was 4.91 mm. AUC was established at the level of 0.624. Sensitivity was 78.12%; and specificity, 44.64%. The cutoff value of SAC was 6.02 mm. The AUC was detected at the level of 0.607. Sensitivity was 59.38%; and specificity, 69.64%. The cutoff value of the OPLL AP diameter occupying ratio was 44.5%. The AUC was detected at the level of 0.63. Sensitivity was 65.62%; and specificity, 60.71%. The cutoff value of OPLL area was 63.4 mm2 (AUC, 0.592; sensitivity, 48.96%; specificity, 67.86%). The cutoff value of the OPLL area occupying ratio was 36.1% (AUC, 0.603; sensitivity, 44.79%; specificity, 80.36%). The cutoff value of the involved longitudinal vertebral level was 2 (AUC, 0.622; sensitivity, 90.62%; specificity, 28.57%; Figure 3).
ROC curve for (a) OPLL AP diameter (cutoff value, 4.91 mm; AUC, 0.624; sensitivity, 78.12%; specificity, 44.64%); (b) space available for the spinal cord (cutoff value, 6.02 mm; AUC, 0.607; sensitivity, 59.38%; specificity, 69.64%); (c) AP diameter occupying ratio (cutoff value, 44.5%; AUC, 0.630; sensitivity, 65.62%; specificity, 60.71%); (d) OPLL area occupying ratio (cutoff value, 36.1%; AUC, 0.603; sensitivity, 44.79%; specificity, 80.36%). A full color version of this figure is available at the Spinal Cord journal online.
Comparison of OPLL size between symptomatic and asymptomatic cervical OPLL patients
The OPLL diameter and area were significantly different between symptomatic and asymptomatic patients with cervical OPLL (Table 3). The mean values of OPLL size in symptomatic patient group were higher than the cutoff value of OPLL size for myelopathy diagnosis using SEP.
Discussion
Our results demonstrated that OPLL AP diameter, OPLL AP diameter occupying ratio, OPLL area, OPLL area occupying ratio and involved longitudinal vertebral level were significantly positively correlated with tibial SEP latency. In addition, SAC was significantly negatively correlated with tibial SEP latency. ROC curve analysis showed that the cutoff values that were diagnostic for SEP prolongation were 4.91 mm for OPLL AP diameter, 6.02 mm for SAC, 44.5% for OPLL AP diameter occupying ratio, 63.4 mm2 for OPLL area, 36.1% for OPLL area occupying ratio and level 2 for the involved longitudinal vertebral level. These findings suggest that OPLL size, including diameter, area and length, is important predictive factor to detect myelopathy, and SEP is recommended for evaluation of OPLL-induced myelopathy even without neurological deficit at the time of OPLL diagnosis.
The pathomechanism of OPLL-induced myelopathy remains unclear. Some patients who have a large OPLL do not exhibit myelopathy for long periods; in contrast, some patients with a small OPLL have neurological symptoms. Thus it is very difficult to predict the natural course of OPLL, future neurological deficits and myelopathy. So the decision regarding whether or not to operate and the confirmation of operative timing are also difficult, and the indications and timing of surgical treatment remain controversial.18 There have been some trials to find out the correlations between OPLL characteristics, including size and myelopathy, using the neurological symptoms or MRI finding. Previous research focusing on the real diameter of the OPLL and SAC suggests that less than 6 or 8 mm of SAC is associated with poor outcomes while >14 mm of SAC is correlated with good outcomes.5, 6 A 10-year long-term follow-up study found that an OPLL AP diameter occupying ratio of >60% is predictive of poor outcomes.4 Other authors have suggested that an MRI finding of intramedullary high-signal intensity on T2-weighted images is correlated with the OPLL occupying ratio, and patients with abnormal MRI findings had an OPLL occupying ratio of about 50%.2 Our results demonstrate that the cutoff value of SAC is 6 mm, and the cutoff value of the AP diameter occupying ratio is 44%. These values are smaller than those reported in previous studies, which suggests that SEP can predict myelopathy earlier than neurological symptoms or MRI findings.
On our results, tibial SEP is significantly correlated with OPLL size, but the correlation between OPLL size and median SEP was not significant. SEP study is a noninvasive means of assessing somatosensory system including peripheral nerve, spinal cord and cerebral cortex. At the central nervous system level, SEP signals propagate rostally via the dorsal column pathway, where tibial SEP passes through the gracile fasciculus and median SEP passes through the cuneate fasciculus. On an anatomical basis, the gracilis fasciculus arises from the fibers medial to the cuneate fasciculus. This anatomical difference in pathway could be the reason for different results between tibial and median SEP. Tibial SEP passes through more medial and central dorsal column pathway, and it would be more susceptible to compression by OPLL.
SEP has been used to monitor myelopathy for several cervical spinal cord diseases.9, 15 A study of presymptomatic spondylolytic spinal cord compression patients reported that SEP and motor evoked potential (MEP) monitoring can predict later compressive myelopathy and that electrodiagnostic monitoring is superior to MRI for early detection of myelopathy. These results are similar to our findings. However, this is the first study to report the correlation between OPLL and SEP in OPLL patients. A previous study used evoked spinal cord potentials (ESCPs) to detect spinal cord dysfunction in cervical OPLL patients, and the authors suggested that ESCPs could detect myelopathy that could not be observed with other methods, including roentgenogram, myelopathy and CT.19 Their results are consistent with our findings that electrophysiological methods are appropriate for identifying spinal cord deterioration; however, ESCP is a more invasive monitoring method for spinal cord dysfunction than SEP.
The natural course of OPLL has not been determined yet. Prediction for future progression to myelopathy has been studied, but demographic predictors, such as age, family history, and OPLL subtype, remain controversial.18 However, many previous studies that considered OPLL size in myelopathy prediction reported a meaningful correlation between these two parameters. In our study, we suggest OPLL size to diagnose pmyelopathy earlier than previous studies with SEP, thus it can help to establish treatment plans for cervical OPLL patients with conservative or operational options. We recommend preoperative SEP monitoring to find myelopathy earlier in presymptomatic cervical OPLL patients. If SEP study is not available, the physician can assume that an OPLL AP diameter >5 mm, SAC<6 mm, AP diameter occupying ratio >45% and area occupying ratio >36% are indicators of myelopathy in cervical OPLL patients.
Our study has several limitations. The primary limitation is that we used 1-dimensional (1D) and 2-dimensional (2D) methods to measure OPLL size. Almost all previous studies used 1D or 2D methods to measure OPLL size. However, one recent study used a 3-dimensional (3D) method to measure OPLL, and they found a significant relationship between OPLL 3D volume and Japanese Orthopedic Association Score.20 They suggested that the 3D method is superior to the 1D method. However, that study only compared OPLL size and neurological symptoms, and the number of included patients was relatively small. Further research on the correlation between SEP and OPLL size measurement with a 3D method is needed. The second limitation is that we only used SEP for the electrodiagnostic test. Some previous research has shown that combined SEP and MEP monitoring of cervical spondylotic myelopathy and orthopedic surgery is meaningful and sometimes superior to MEP or SEP monitoring alone. Thus further studies with combined SEP and MEP should be performed. Finally, we did not consider dynamic factors and spine alignment for the development of myelopathy. These two factors have been reported to correlate with myelopathy. However, this is a retrospective study, and there was no sufficient data to consider those factors. Future prospective research about the correlation between SEP and OPLL including dynamic factors and alignment is recommended.
Despite these limitations, this is the first study to investigate the relationship between OPLL characteristics and SEP. Our results demonstrated meaningful early diagnosis of myelopathy after OPLL by using SEP and suggest cutoff values of OPLL diameter and area. These results can help to establish the appropriate treatment plan, including surgical timing. Further research with a larger sample size and a more precise OPLL measurement method are needed.
Data archiving
There were no data to deposit.
References
Fujimori T, Le H, Hu SS, Chin C, Pekmezci M, Schairer W et al. Ossification of the posterior longitudinal ligament of the cervical spine in 3161 patients. Spine 2015; 40: E394–E403.
Choi BW, Hum TW . Significance of intramedullary high signal intensity on magnetic resonance imaging in patients with cervical ossification of the posterior longitudinal ligament. Clin Orthop Surg 2015; 7: 465–469.
Epstein NE . What you need to know about ossification of the posterior longitudinal ligament to optimize cervical spine surgery: a review. Surg Neurol Int 2014; 5: S93–s118.
Matsunaga S, Nakamura K, Seichi A, Yokoyama T, Toh S, Ichimura S et al. Radiographic predictors for the development of myelopathy in patients with ossification of the posterior longitudinal ligament: a multicenter cohort study. Spine (Phila Pa 1976) 2008; 33: 2648–2650.
Matsunaga S, Kukita M, Hayashi K, Shinkura R, Koriyama C, Sakou T et al. Pathogenesis of myelopathy in patients with ossification of the posterior longitudinal ligament. J Neurosurg 2002; 96: 168–172.
Koyanagi I, Imamura H, Fujimoto S, Hida K, Iwasaki Y, Houkin K . Spinal canal size in ossification of the posterior longitudinal ligament of the cervical spine. Surg Neurol 2004; 62: 286–291 discussion 291.
Fujimori T, Iwasaki M, Okuda S, Takenaka S, Kashii M, Kaito T et al. Long-term results of cervical myelopathy due to ossification of the posterior longitudinal ligament with an occupying ratio of 60% or more. Spine (Phila Pa 1976) 2014; 39: 58–67.
Iwasaki M, Okuda S, Miyauchi A, Sakaura H, Mukai Y, Yonenobu K et al. Surgical strategy for cervical myelopathy due to ossification of the posterior longitudinal ligament: Part 1: clinical results and limitations of laminoplasty. Spine (Phila Pa 1976) 2007; 32: 647–653.
Bednarik J, Kadanka Z, Dusek L, Kerkovsky M, Vohanka S, Novotny O et al. Presymptomatic spondylotic cervical myelopathy: an updated predictive model. Eur Spine J 2008; 17: 421–431.
Hwang P, Sohn MK, Kim CS, Jee S . Tibial somatosensory evoked potential can prognosticate for ambulatory function in subacute hemiplegic stroke. J Clin Neurosci 2016; 26: 122–125.
Han EY, Jung HY, Kim MO . Absent median somatosensory evoked potential is a predictor of type I complex regional pain syndrome after stroke. Disabil Rehabil 2014; 36: 1080–1084.
Cidem M, Sahin Z, Aydin T, Aysal F . Somatosensory evoked potential findings in ankylosing spondylitis. Eurasian J Med 2014; 46: 42–46.
Pelosi L, Lamb J, Grevitt M, Mehdian SM, Webb JK, Blumhardt LD . Combined monitoring of motor and somatosensory evoked potentials in orthopaedic spinal surgery. Clin Neurophysiol 2002; 113: 1082–1091.
Garcia RM, Qureshi SA, Cassinelli EH, Biro CL, Furey CG, Bohlman HH . Detection of postoperative neurologic deficits using somatosensory-evoked potentials alone during posterior cervical laminoplasty. Spine J 2010; 10: 890–895.
Bednarik J, Kadanka Z, Vohanka S, Novotny O, Surelova D, Filipovicova D et al. The value of somatosensory and motor evoked potentials in pre-clinical spondylotic cervical cord compression. Eur Spine J 1998; 7: 493–500.
Sartip KA, Dong T, Ndukwe M, East JE, Graves JA, Davis B et al. Ossification of the posterior longitudinal ligament: imaging findings in the era of cross-sectional imaging. J Comput Assist Tomogr 2015; 39: 835–841.
Chiappa KH . Evoked Potentials in Clinical Medicine, 3rd edn. Lippincott Williams & Wilkins, Raven press, NY, USA,. 1997.
Yonenobu K . Is surgery indicated for asymptomatic or mildly myelopathic patients with significant ossification of the posterior longitudinal ligament? Spine (Phila Pa 1976) 2012; 37: E315–E317.
Shinomiya K, Furuya K, Sato R, Okamoto A, Kurosa Y, Fuchioka M . Electrophysiologic diagnosis of cervical OPLL myelopathy using evoked spinal cord potentials. Spine (Phila Pa 1976) 1998; 13: 1225–1233.
Lee N, Ji GY, Shin HC, Ha Y, Jang JW, Shin DA . Usefulness of 3-dimensional measurement of ossification of the posterior longitudinal ligament (OPLL) in patients with OPLL-induced myelopathy. Spine (Phila Pa 1976) 2015; 40: 1479–1486.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Yoon, S., Park, T., Eun, N. et al. The cutoff value of ossification of posterior longitudinal ligament (OPLL) for early diagnosis of myelopathy using somatosensory evoked potential in cervical OPLL patients. Spinal Cord 55, 606–611 (2017). https://doi.org/10.1038/sc.2017.15
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sc.2017.15
This article is cited by
-
Intraoperative evoked potentials in patients with ossification of posterior longitudinal ligament
Journal of Clinical Monitoring and Computing (2022)
-
Correlation between preoperative somatosensory evoked potentials and intraoperative neurophysiological monitoring in spinal cord tumors
Journal of Clinical Monitoring and Computing (2021)