Abstract
Study design:
Single group, pretest–post-test study.
Objectives:
To determine the effects of a non-task-specific, voluntary, progressive aerobic exercise training (AET) intervention on fitness and walking-related outcomes in ambulatory adults with chronic motor-incomplete SCI.
Setting:
Rehabilitation research center.
Methods:
Ten ambulatory individuals (50% female; 57.94±9.33 years old; 11.11±9.66 years postinjury) completed voluntary, progressive moderate-to-vigorous intensity AET on a recumbent stepper 3 days per week for 6 weeks. The primary outcome measures were aerobic capacity (VO2peak (volume of oxygen that the body can use during physical exertion)) and self-selected overground walking speed (OGWS). Secondary outcome measures included walking economy, 6-minute walk test (6MWT), daily step counts, Walking Index for Spinal Cord Injury (WISCI-II), Dynamic Gait Index (DGI) and Berg Balance Scale (BBS).
Results:
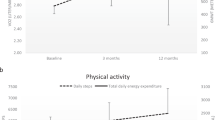
Nine participants completed all testing and training. Significant improvements in aerobic capacity (P=0.011), OGWS (P=0.023), the percentage of VO2peak used while walking at self-selected speed (P=0.03) and daily step counts (P=0.025) resulted following training.
Conclusions:
The results indicate that total-body, voluntary, progressive AET is safe, feasible, and effective for improving aerobic capacity, walking speed, and select walking-related outcomes in an exclusively ambulatory SCI sample. This study suggests the potential for non-task-specific aerobic exercise to improve walking following incomplete SCI and builds a foundation for further investigation aimed at the development of exercise based rehabilitation strategies to target functionally limiting impairments in ambulatory individuals with chronic SCI.
Similar content being viewed by others
Introduction
Aerobic capacity is a health-related component of physical fitness and an indicator of health, morbidity and mortality.1 Following spinal cord injury (SCI), aerobic capacity is significantly diminished, so much so that individuals with SCI are believed to live at the lowest end of the human fitness spectrum.2 The decline in aerobic capacity after SCI is related to a number of factors, including motor level of injury, functional classification, body mass index, age and activity level.3, 4 There is sufficient evidence to show that aerobic exercise training (AET) improves aerobic capacity and mitigates the adverse effects of low fitness after SCI.5, 6, 7, 8 However, aerobic capacity and the effects of AET have not been thoroughly studied in ambulatory adults with incomplete SCI, a large and a growing segment of the SCI population.
Aerobic capacity is a known determinant of normal walking9 and has been suggested as a factor that may influence walking ability in individuals with neurological diseases or disorders (e.g. stroke, multiple sclerosis).10, 11, 12, 13 There is ample evidence supporting the beneficial effects of AET on both aerobic capacity and walking outcomes after stroke14, 15, 16 and multiple sclerosis.17, 18, 19 To date, however, the relationship between aerobic capacity and walking after SCI is not fully understood, and data regarding the effects of AET on these outcomes in ambulatory adults with SCI are lacking. It is reasonable to believe that diminished aerobic capacity may be a factor limiting walking ability after SCI, and, similar to observations from other populations, voluntary (i.e. manual, not assisted or electrically stimulated) AET may result in beneficial effects on fitness and walking outcomes.
The majority of studies aimed at improving walking ability in ambulatory adults with SCI have focused on task-specific (i.e. walking based) strategies, and only a few have assessed changes in fitness following training.20, 21, 22 There is little evidence available regarding the effects of non-task-specific exercise on walking outcomes and fitness in ambulatory adults with SCI. Functional electrical stimulation (FES) cycling is one non-task-specific AET strategy that has been shown to improve fitness after SCI,23, 24 as well as walking outcomes in ambulatory adults with SCI.25, 26 To date, the impact of voluntary, non-task-specific AET has yet to be investigated in this population.
Recumbent stepping is a non-task-specific total-body aerobic exercise that produces reciprocal movements similar to but simpler than walking and relies on similar neural control.27 This exercise modality may target ambulatory deficits via multiple mechanisms, including increased aerobic capacity and muscle strength.10, 28 Recumbent stepping has been shown to increase aerobic capacity and improve walking outcomes in adults with neurologic diseases and is a recommended exercise strategy for rehabilitation of ambulatory impairments, as it is widely accessible, easy to perform and cost-effective.10, 29
Purpose
The purpose of this study was to determine the effects of a 6-week non-task-specific, progressive AET intervention on fitness and walking-related outcomes in ambulatory adults with chronic motor-incomplete SCI. We hypothesized that a voluntary recumbent stepping exercise program, with sufficient focus on aerobic capacity, would result in significant improvements in the primary outcome measures, aerobic capacity (VO2peak (volume of oxygen that the body can use during physical exertion)) and self-selected overground walking speed (OGWS). We further hypothesized that there would be improvements in secondary walking-related outcome measures including walking economy, 6-minute walk test (6MWT), daily step counts, Walking Index for Spinal Cord Injury (WISCI-II), Dynamic Gait Index (DGI) and Berg Balance Scale (BBS).
Materials and methods
Participants
Participants were recruited from a pool of individuals who had previously completed studies at the rehabilitation research center and also from area clinics, support groups and by word of mouth. Twenty-five individuals were screened for eligibility. Ten participants with chronic (>6 months postinjury) motor-incomplete SCI (AIS C, D; C4-T12) volunteered to participate in this single group, pretest–post-test study. Participants received remuneration for their time. Inclusion criteria were as follows: (1) age 18–75 years; (2) a diagnosis of first time SCI including etiology from traumatic or vascular origin; (3) the ability to walk independently for a minimum of 10 m with or without an assistive device; (4) Self-selected walking speed >0.1 and <1.15 m s−1; (5) a medically stable condition allowing for testing of walking function and AET. Before participation, all participants provided a signed informed consent. We certify that all applicable institutional and governmental regulations concerning the ethical use of human volunteers were followed during the course of this research.
AET intervention
Participants completed a 6-week (3 sessions per week; 20 min per session) non-task-specific, progressive AET program. All training was performed on a NuStep T5xr recumbent cross-trainer (NuStep Inc., Ann Arbor, MI, USA). The selected exercise modality required bilateral reciprocal stepping against resistive forces and synchronized upper extremity movement; thus, a total-body workout was achieved. No assistance in performing the exercise was provided by either personnel or electrical stimulation; thus, all AET was voluntary.
The AET intervention was developed to meet the SCI guidelines for aerobic activity30 (20 min, 2 days per week of moderate-to-vigorous exercise) and prepare participants to reach the levels of aerobic exercise recommended by the ACSM and 2008 Physical Activity Guidelines for Americans.1, 31 The primary parameter for progression during the AET was intensity of exercise, expressed as a percentage of VO2 reserve (VO2R), where target VO2R=% intensity × (VO2peak−VO2rest)+VO2rest. Over the course of the training period, the exercise dose was progressed from a starting intensity of 40% VO2R, and increased by at least 5% per week, to a final target exercise intensity of 60–70% VO2R in the final week. Each participant followed an individualized training program, generated based on his or her baseline VO2 values, the identified target intensity and the corresponding metabolic equivalent (MET) levels (1 MET=3.5 ml O2 kg−1 per min (milliliters of oxygen per kilogram body mass per minute)). MET levels were displayed on the NuStep and were used to track intensity and progression of the exercise sessions.
The weekly sessions included two steady-state exercise sessions at the target intensity and one high-intensity interval training session. Two built-in programs were selected for the exercise sessions. The constant power program was used for the steady-state sessions. In this program, the participants exercised at a constant effort; the program adjusted the workload based on the stepping speed and the step length to maintain a constant target MET level. The NuStep profiles program provides five options for different levels of interval training. The built-in interval training program, profile 5, was chosen to allow for 1:1 interval training (1 min of high-intensity exercise and 1 min of active recovery). The interval training intensities were set based on the net target workload for the week (i.e. 40–65% VO2R). During the interval training sessions, the active recovery remained at 20% VO2R; thus, the high-intensity intervals increased from 60 to 110% of pretraining VO2R over the course of the 6 weeks. During all exercise sessions, MET level, heart rate, blood pressure and ratings of perceived exertion (RPE) levels were monitored for safety and to ensure that the participants were exercising at the appropriate intensity.
Outcome measures
The International Classification of Functioning, Disability and Health (ICF) model guided the selection of outcome measures.32 Outcomes related to the components of body functions, body structures, activities and participation were assessed. All outcome measures were collected before and following the AET intervention. OGWS was assessed weekly. To prevent fatigue, the pre- and post-test assessments were spread out over two visits, and participants were given rest periods of 5–10 min between testing at each visit.
Aerobic capacity
Graded exercise tests were completed on the NuStep to assess changes in VO2peak presented as ml O2 kg−1 per min. Following the protocols previously described by Billinger et al.,33 participants completed the modified total-body recumbent stepper exercise test. This device and protocol have been validated for use in healthy, sedentary and poststroke individuals,33, 34 but have yet to be evaluated in individuals with incomplete SCI. Despite this limitation, the decision was made to use the NuStep to maintain consistency between the testing and training of VO2peak. Our decision was further influenced by safety and feasibility concerns to using other common modalities (e.g. treadmill, arm-crank or cycle ergometer). For example, walking impairments may affect the amount of time a participant could spend on a treadmill, thus limiting attainment of VO2peak. In addition, aerobic capacity is often underestimated on arm-crank and cycle ergometer exercise tests, due to the limited muscle mass activated. We believed the participants would more fully tax their cardiorespiratory system by completing a total-body graded exercise test on the NuStep, which requires active recruitment of a larger muscle mass. In addition, utilization of both upper and lower extremities was expected to reduce the risk of local fatigue commonly observed in cycle ergometry exercise tests. We acknowledge that further research is necessary to validate the use of recumbent stepping protocols for assessment of aerobic capacity in individuals with SCI.
Before exercise testing, instructions were given to the participants to refrain from consuming caffeine, food or drink (water was permitted) for at least 3 h and to avoid significant exertion or exercise on the day of the assessment. The protocol and assessments required during the test were described, and participants were given a few minutes to acclimate to the required step rate (80 steps per min) before starting. Once seated, adjustments were made to the seat and arm positions; if needed, hand and leg stabilizers were used. Participants were then fitted with a facemask that allowed for the collection of respiratory gasses. Oxygen uptake was measured using a Quark CPET metabolic cart (COSMED, Rome, Italy). Breath-by-breath cardiorespiratory data were collected and then averaged every 15 s to determine the highest VO2 (VO2peak) achieved during the test.
The modified total-body recumbent stepper exercise test consisted of eight consecutive 2-min stages, where resistance increased at each stage. Each test was supervised by an exercise physiologist, cardiologist and trained personnel to ensure participant safety. A 12-lead electrocardiogram was monitored throughout the test. Vitals (heart rate and blood pressure) and ratings of perceived exertion (Borg RPE scale 6–20) were recorded at the end of each 2-min stage. The participants were instructed to discontinue the test if at any point they felt the need to stop. Otherwise, the exercise test was terminated by the cardiologist or exercise physiologist based on the ACSM’s general indications1 and/or the inability of the participant to maintain stepping cadence of 80 steps per min with verbal cues and encouragement. Peak oxygen consumption was assessed by the attainment of (1) a plateau in VO2, despite an increase in exercise load, (2) RPE >17 and/or (3) respiratory exchange ratio ⩾1.15.
Walking speed
Self-selected OGWS was assessed using a GAITRite portable gait analysis system (CIR Systems Inc./GAITRite, Sparta, NJ, USA). Participants were fitted with a support vest connected to a passive overhead harness as a safety precaution and were instructed to walk at their comfortable walking speed across the walkway. The use of assistive devices and bracing was only permitted as absolutely necessary (i.e. participants were unable to safely walk without them) and maintained for each test for the duration of the study. Trained personnel walked beside the participants if requested or needed to maintain safety. Four trials were completed at each testing point and the average OGWS recorded.
Walking-related outcomes
Guided by published recommendations,35 a number of outcomes were selected to complete a comprehensive assessment of ambulation. The selected outcomes included walking economy, 6-minute walk test (6MWT), daily step counts, WISCI-II, DGI and BBS.
Participants completed a 5-min walking assessment at self-selected speed on a treadmill to assess walking economy. To determine self-selected speed, the treadmill speed was incrementally increased until the participant indicated their comfortable, usual walking speed. Participants completed the post-testing assessment at matched prespeed. While walking on the treadmill, participants were connected to a passive overhead harness, in place as a safety precaution only; no body weight support was provided. Oxygen uptake was measured using a metabolic cart. The VO2 (ml O2 kg−1 per min) data collected during the last minute of the walking test were averaged and normalized by self-selected walking speed to determine walking economy (ml O2 kg−1 per min). The percentage of VO2peak used while walking at self-selected speed (i.e. the relative cost of walking) was also calculated from the VO2 values obtained.
All participants completed a self-paced 6MWT in a hallway to assess submaximal levels of functional capacity (i.e. walking endurance). Total distance walked (meters, m) was recorded. The 6MWT is a valid and a reliable test commonly used to test walking ability in the incomplete SCI population.36, 37
As a participation-level measure, individuals who reported walking as their primary mode of locomotion wore a StepWatch 3.0 Step Activity Monitor (SAM) (Orthocare Innovations, Mountlake Terrace, WA, USA) on their dominant, or less affected, ankle to record daily step counts outside of the intervention. Previous studies have shown that the SAM accurately and reliably captures walking activity in individuals with incomplete SCI.38, 39 Following procedures described by Bowden et al.,39 the settings of each device were manually specified to customize the SAM for each participant at each time point. Participants wore the SAM for 4 days, and step counts were averaged and presented as steps per day.
Finally, three standardized clinical rehabilitation assessments were completed by a licensed physical therapist. The WISCI-II, a 21-level ordinal scale that assesses degrees of assistance used to walk, is a valid and a reliable measure of walking function in individuals with incomplete SCI.40 The DGI is an 8-item measure that evaluates an individual’s capacity to modify walking under different situations and dual tasks that are commonly encountered in daily life. Although commonly used in clinical rehabilitation and research settings to examine gait and balance, the psychometric properties of this measure have not been thoroughly examined after SCI. The BBS is a 14-item instrument that assesses static and dynamic balance abilities, as well as fall risk; it is a valid and a reliable measure for assessing balance after SCI.41, 42
Statistical analysis
Tests for normality (Shapiro–Wilk test) were completed before performing the analyses. Paired-sample t-tests or the nonparametric equivalent, Wilcoxon's matched-pairs signed-ranks test was used to assess pre- to post-test changes in the outcomes. A significance level of α=0.05 was used for all analyses. Effect sizes from the paired-samples t-tests are reported as Cohen’s d, d=M1−M2/SDpooled. Effect sizes for the outcomes requiring nonparametric statistics are reported as r, r=z/√N. An intention-to-treat analysis was performed to include all data from the participants who began the AET. Data were analyzed using IBM SPSS Statistics Version 22 (IBM Corporation, IBM Corp., Armonk, NY, USA).
Results
Ten adults aged 42–72 years of age with chronic (10 months to 33 years postinjury) motor-incomplete SCI (AIS C, D) participated in the AET intervention. All of the individuals were ambulatory; however, two participants were not primary ambulators and predominantly used a manual wheelchair for daily mobility. Demographic characteristics are presented in Table 1. Baseline participant characteristics and changes in the primary outcomes are presented in Table 2. Of the 10 participants who enrolled, nine completed all training visits. One participant withdrew from the study after 3 weeks of training because of issues related to transportation and chronic pain; the last observation was carried forward. All testing and exercise training were safely completed, and the AET intervention (intensity, progression, duration) was feasible.
Primary and secondary outcomes are described below, with summary results presented in Table 3.
Primary outcome measures
Aerobic capacity
Baseline VO2peak values were 20.90±5.54 ml O2 kg−1 per min. As hypothesized, following the AET VO2peak significantly increased to 23.86±4.49 ml O2 kg−1 per min (P=0.01), indicating a large effect (d=1.01). The individual changes in VO2peak ranged from −6.2% to 54.6%, with an average of 17% (Table 2). One participant requested early termination of the post-testing VO2peak assessment due to a flare-up in chronic shoulder pain; thus, a negative change was recorded. The mean change in aerobic capacity following AET represents a 14% increase (Table 3).
OGWS
Statistically and clinically significant improvements were found in self-selected OGWS. After AET, participants walked an average 0.06±0.07 m s−1 faster than at baseline (P=0.02; d=0.86). The individual improvements in self-selected OGWS ranged from −5.5 to 33.3%, with an average of 13% (Table 2). The change in mean OGWS represents a 9% increase in self-selected walking speed (Table 3).
Secondary outcome measures
Walking-related outcomes
Changes in two of the secondary outcomes, percentage of VO2peak at which participants were walking and daily step counts, were statistically significant (Table 3).
Nine participants completed the assessments to determine oxygen consumption at self-selected walking speeds; one participant was unable to walk comfortably on the treadmill and therefore did not complete the test. At pretesting, average walking economy was 0.75±0.31 ml O2 kg−1 per min (range: 0.47–1.45 ml O2 kg−1 per min). There was no significant change following training (post=0.73±0.34 ml O2 kg−1 per min; range: 0.32–1.44 ml O2 kg−1 per min). However, there was a significant change in the relative cost of walking. At pretesting, participants walked at an average 62% of VO2peak (range: 51–87%). Following training, participants walked at 54% of VO2peak (range: 38–65%), which represents a significant reduction (P=0.03, d=0.87) in the percentage of aerobic capacity used while walking at self-selected walking speeds.
There was no significant change in the distance walked during the 6MWT nor did the change meet the previously reported minimal detectable change (45.8 m or a 22% change).43 Only one participant was able to exceed the MDC for the 6MWT. In addition, although trends toward improvement were seen in the BBS and DGI, there were no statistically significant changes in the walking-related clinical rehabilitation tests. WISCI-II scores did not change.
Following training, there was a significant increase in daily step counts for the eight primary ambulators (916±1410 steps per day; P=0.025, r=0.56). Data from the two participants who were primary wheelchair users are not available. One participant did not adhere to wearing the SAM as instructed at baseline and later withdrew from the study. The other participant only walked under supervision with an assistive device in our lab; they elected not to wear the SAM. At baseline, participants walked 5050±1247 steps per day outside the lab (range: 2612–6573 steps per day). After the intervention, the total number of steps taken increased (5967±2174 steps per day; range: 2812–10281 steps per day), indicating an average 23% increase in walking activity in the free-living environment.
Discussion
As hypothesized, the results of this study indicate that AET was effective for improving both aerobic capacity and walking outcomes in a sample of ambulatory adults with incomplete SCI. The findings add to the limited body of evidence supporting exercise training in this sub-population of SCI.
The present findings indicate that aerobic capacity is reduced in these individuals, although not as markedly as previously reported in other SCI sub-populations. Examining aerobic capacity according to recently published reference values for untrained individuals with SCI,4 on average participants fell within the ‘excellent’ fitness category (>15.2 ml O2 kg−1 per min). It is important to point out that the SCI reference values were developed based on data obtained from a sample of primary wheelchair users with primarily complete injuries while performing upper extremity exercise. Thus, it is likely that these reference values are not appropriate for classifying fitness levels in the current sample. Conversely, compared with gender- and age-specific normative percentile rankings, our participants fell in the ‘well below average’ (10th percentile) fitness category, except for one participant who was ‘below average’ (20th percentile).1 This finding calls into question the applicability of either of these reference values for interpreting fitness in the ambulatory SCI population and highlights a need to extend previous research and develop stratified reference values of fitness for men and women with incomplete injuries who are ambulatory.
Exercise training has long been recommended to improve aerobic capacity in individuals with SCI, but this was the first investigation to demonstrate improvements in fitness levels in ambulatory adults with incomplete SCI following a voluntary, total-body recumbent stepping AET program. Despite the relatively short (6-week) duration, VO2peak significantly increased 14%, which is within the generally expected range of training-induced increase (5–30%)1 and slightly lower compared with that recently reported following a 16-week guideline-driven exercise intervention in the SCI population (17% increase).44 It is reasonable to believe that extending the duration of AET may result in greater improvements in aerobic capacity. In addition, increasing the training stimulus either by total minutes per day or days per week would likely result in an increased training effect. Future research should explore exercise dosage and further evaluate the effectiveness of voluntary, guideline-driven AET in the ambulatory SCI population.
Following the AET, both statistically and clinically significant improvements were observed in OGWS. Self-selected speeds increased by an average of 0.06 m s−1 (9%) after the 6-week intervention, meeting the estimated minimally clinically important difference for walking speed after incomplete SCI (0.05 m s−1).45 Five of the 10 participants met or exceeded the minimally clinically important difference for OGWS. Further investigation is necessary to identify participant characteristics (eg, initial walking function, age, fitness level) that may influence changes in walking outcomes following AET. Although larger changes in walking speed have been reported following non-task-specific exercise interventions of longer duration, including FES leg cycling25, 26 and lower extremity resistance training,46, 47 statistically significant findings are limited. Following a 10-week progressive FES cycling case study, an improvement of 0.33 m s−1 was reported.25 In a more recent study, Yasar et al.26 documented a 0.13 m s−1 increase in walking speed following a 16-week FES cycling intervention, but the results were not significant. Although there are a number of benefits associated with FES exercise, we believe voluntary exercise may hold more promise for individuals with SCI who are able to walk. Voluntary, non-task-specific exercise in the form of lower extremity resistance training has been shown to improve walking outcomes in ambulatory adults with SCI. Gregory et al.46 reported marked improvements in self-selected (0.26 m s−1) and fastest-comfortable (0.39 m s−1) walking speed following 12 weeks of lower extremity resistance training. Recently, more modest changes in preferred and maximal walking speed were reported following 4 weeks of resistance training (0.06 and 0.14 m s−1, respectively).47 Importantly, the benefits of non-task-specific training were supported, as the study found that maximal walking speed improved significantly more following non-task-specific lower extremity resistance training compared with task-specific robot-assisted gait training.47 The findings from these studies, in conjunction with the significant findings from our study, support the benefits of voluntary, non-task-specific exercise training for rehabilitation of walking outcomes in ambulatory adults with SCI.
Early studies of the energy cost of locomotion indicate that walking after SCI is a physiologically demanding task, which is supported in our sample. The average walking economy of the participants was within the range previously reported after SCI (0.26–1.02 ml O2 kg−1 per min) and was roughly four to five times greater than that reported in the general population (0.15–0.17 ml O2 kg−1 per min).48, 49, 50 Elevated walking economy may be due to increased oxygen consumption or slow walking speed.50 In the case of our participants, the poor walking economy is likely attributed primarily to slow self-selected walking speeds (0.60 m s−1) compared with normal (1.0–1.67 m s−1), as oxygen consumption was only slightly elevated (13.1 ml O2 kg−1 per min) compared with that reported in the general population (12.0 ml O2 kg−1 per min).50 Further evaluation of oxygen consumption during the walking trial indicated that walking at self-selected speed requires a substantial percentage of peak aerobic capacity and is more physically demanding than in the able-bodied population. In the neurologically healthy population, walking is not a significantly taxing activity; reports suggest that, while walking at a usual pace, oxygen consumption is generally around 30–40% of maximal VO2 values.51, 52, 53 At baseline, the participants in our study walked at 62% (range: 50–87%) of their VO2peak. The high relative cost results from a combination of low VO2peak and elevated oxygen consumption during walking. With training, the average percentage of VO2peak at which the participants were walking was significantly reduced to 54%. It is reasonable to believe that the high relative cost of walking may have a role in limiting walking outcomes after SCI. Therefore, increasing aerobic capacity with AET may compensate for the increased cost of walking and improve functional capacity in ambulatory individuals with SCI.
The AET intervention also resulted in a significant improvement in daily step counts, indicative of increased daily activity and participation in the free-living environment. Despite the high relative cost of walking and slow walking speeds, the participants maintained a higher than average activity level. On average, the participants who were primary ambulators far exceeded the step counts previously reported in the incomplete SCI population.38 Averaging 5050 steps per day at baseline and 5967 steps per day at post-testing, the primary ambulators in this sample fell within the normative step ranges for special populations (1200–8800 steps per day)54 and were above the generally accepted threshold for sedentary lifestyles (<5 000 steps).55 It is important to note that 40% of the participants did not use any assistive devices to walk, and only 20% used a bilateral assistive device; thus, the step counts of our sample may not be representative of the population of ambulatory individuals with incomplete SCI. We acknowledge that the use of assistive devices may have influenced step counts and that the lack of additional data surrounding the daily step activity (i.e. reports on special events, pain, fatigue or sickness that may have influenced step counts) limits our understanding of this outcome.
At baseline, the majority of participants did not engage in any exercise, and most reported a sedentary lifestyle despite the relatively high number of steps taken. After the intervention, in addition to meeting the aerobic exercise guidelines, participants also increased their daily step counts. It is reasonable to hypothesize that the increase in physical activity in addition to increased walking activity in the free-living environment will result in a compounding of health benefits extending beyond the outcomes measured. In the least, changes in free-living physical activity may initiate a perpetual positive cycle whereby increasing fitness decreases the relative cost of walking, resulting in increased activity, thus leading to improved walking function, and may result in greater participation in the community.
Taken together, these results suggest the potential for an non-task-specific, voluntary, total-body AET approach to improve fitness and walking-related outcomes in ambulatory individuals with chronic incomplete SCI. On the basis of the findings, we recommend AET on a recumbent stepper as a feasible alternative or adjuvant to current interventions aimed at improving walking outcomes in ambulatory individuals with incomplete SCI. Compared with other modalities such as locomotor training and robotic-assisted gait training, AET on a recumbent stepper requires fewer personnel, uses less expensive equipment and may be more time efficient, thus increasing the benefits of this option. Further, aerobic exercise is widely accessible and may be more easily translated from the lab to community recreation centers, fitness facilities or home gyms. Most importantly, aerobic exercise can be successfully completed through all stages of one’s life and is known to elicit more than just beneficial changes in fitness and walking outcomes, making AET an attractive option to recommend following SCI.
Limitations
The results of the present study are promising, but several limitations must be addressed when interpreting the findings from this small sample size, non-randomized study. Importantly, the participants were self-selected volunteers, who may be more functional and more motivated to exercise than the general incomplete SCI population. The inclusion criteria for our study limit the interpretation of findings to individuals with chronic incomplete SCI who are able to walk. The lack of an adequate control group is also a limitation. The primary outcome, change in aerobic capacity, was assessed on the NuStep using a protocol that has not yet been validated for use in SCI. Although this protocol appears to be feasible and well tolerated by the participants, the nature of the total-body graded exercise test makes it difficult to compare the aerobic capacity results to previous results. Future research is necessary to validate the use of this exercise protocol for use in individuals with SCI. We do not have adequate follow-up data to determine whether an exercise regimen was maintained following the AET, nor can we determine whether the gains observed following AET were maintained.
Conclusions
The AET intervention was safe, feasible and effective. Following 6 weeks of voluntary, total-body aerobic exercise performed at a moderate–vigorous intensity, meaningful functional improvements in fitness and walking-related outcomes were observed. The major finding of the study is that both aerobic capacity and OGWS significantly increased. Further study with a larger sample is necessary to examine correlations between fitness and walking speed and determine whether aerobic capacity is a determinant of walking after SCI. Statistical improvements were also observed in outcomes relating to walking function, including the percentage of VO2peak at which participants walk and daily step counts. Although no significant improvements were observed in the remaining walking-related outcomes, the results indicate moderate to large effects for these outcomes. Overall, the findings warrant future investigation in a larger cohort, with an adequate control group, as well as comparisons with other established exercise interventions (eg, resistance training, FES cycling). There is a need for further research into the role of non-task-specific AET for improving fitness and walking outcomes in individuals with varying degrees of ambulatory impairment. In addition, there is a need to study the effects of implementing voluntary AET earlier after injury, in attempts to attenuate the effects of physical inactivity on deconditioning and functional limitations. Such investigations could shed light on individual characteristics that influence changes in fitness and walking outcomes to better inform rehabilitation researchers and practitioners.
Data archiving
There were no data to deposit.
References
ACSM. ACSM's Guidelines for Exercise Testing and Prescription 9th edn. Wolters Kluwer/Lippincott Williams & Wilkins, Philadelphia, PA, USA, 2013, 480pp.
Dearwater SR, LaPorte RE, Robertson RJ, Brenes G, Adams LL, Becker D . Activity in the spinal cord-injured patient: an epidemiologic analysis of metabolic parameters. Med Sci Sports Exerc 1986; 18: 541–544.
Janssen TW, Dallmeijer AJ, Veeger DJ, van der Woude LH . Normative values and determinants of physical capacity in individuals with spinal cord injury. J Rehabil Res Dev 2002; 39: 29–39.
Simmons OL, Kressler J, Nash MS . Reference fitness values in the untrained spinal cord injury population. Arch Phys Med Rehabil 2014; 95: 2272–2278.
Ginis KAM, Jorgensen S, Stapleton J . Exercise and sport for persons with spinal cord injury. PM&R 2012; 4: 894–900.
Hicks AL, Martin Ginis KA, Pelletier CA, Ditor DS, Foulon B, Wolfe DL . The effects of exercise training on physical capacity, strength, body composition and functional performance among adults with spinal cord injury: a systematic review. Spinal Cord 2011; 49: 1103–1127.
Jacobs PL, Nash MS . Exercise recommendations for individuals with spinal cord injury. Sports Med 2004; 34: 727–751.
Warburton DE, Eng JJ, Krassioukov A, Sproule S,, the SRT. Cardiovascular health and exercise rehabilitation in spinal cord injury. Top Spinal Cord Inj Rehabil 2007; 13: 98–122.
Barbeau H, Nadeau S, Garneau C . Physical determinants, emerging concepts, and training approaches in gait of individuals with spinal cord injury. J Neurotrauma 2006; 23: 571–585.
Pilutti LA, Hicks AL . Rehabilitation of ambulatory limitations. Phys Med Rehabil Clin N Am 2013; 24: 277–290.
Michael KM, Allen JK, Macko RF . Reduced ambulatory activity after stroke: the role of balance, gait, and cardiovascular fitness. Arch Phys Med Rehabil 2005; 86: 1552–1556.
Patterson SL, Forrester LW, Rodgers MM, Ryan AS, Ivey FM, Sorkin JD et al. Determinants of walking function after stroke: differences by deficit severity. Arch Phys Med Rehabil 2007; 88: 115–119.
Courbon A, Calmels P, Roche F, Ramas J, Rimaud D, Fayolle-Minon I . Relationship between maximal exercise capacity and walking capacity in adult hemiplegic stroke patients. Am J Phys Med Rehabil 2006; 85: 436–442.
Severinsen K, Jakobsen JK, Overgaard K, Andersen H . Normalized muscle strength, aerobic capacity, and walking performance in chronic stroke: a population-based study on the potential for endurance and resistance training. Arch Phys Med Rehabil 2011; 92: 1663–1668.
Stoller O, de Bruin ED, Knols RH, Hunt KJ . Effects of cardiovascular exercise early after stroke: systematic review and meta-analysis. BMC Neurol 2012; 12: 45.
Saunders DH, Sanderson M, Brazzelli M, Greig CA, Mead GE . Physical fitness training for stroke patients. Cochrane Database Syst Rev 2013; 10: CD003316.
Snook EM, Motl RW . Effect of exercise training on walking mobility in multiple sclerosis: a meta-analysis. Neurorehabil Neural Rep 2009; 23: 108–116.
Pearson M, Dieberg G, Smart N . Exercise as a therapy for improvement of walking ability in adults with multiple sclerosis: a meta-analysis. Arch Phys Med Rehabil 2015; 96: 1339–1348.e7.
Latimer-Cheung AE, Pilutti LA, Hicks AL, Martin Ginis KA, Fenuta AM, MacKibbon KA et al. Effects of exercise training on fitness, mobility, fatigue, and health-related quality of life among adults with multiple sclerosis: a systematic review to inform guideline development. Arch Phys Med Rehabil 2013; 94: 1800–28 e3.
Kressler J, Nash MS, Burns PA, Field-Fote EC . Metabolic responses to 4 different body weight-supported locomotor training approaches in persons with incomplete spinal cord injury. Arch Phys Med Rehabil 2013; 94: 1436–1442.
Alexeeva N, Sames C, Jacobs PL, Hobday L, Distasio MM, Mitchell SA et al. Comparison of training methods to improve walking in persons with chronic spinal cord injury: a randomized clinical trial. J Spinal Cord Med 2011; 34: 362–379.
Hoekstra F, van Nunen MP, Gerrits KH, Stolwijk-Swuste JM, Crins MH, Janssen TW . Effect of robotic gait training on cardiorespiratory system in incomplete spinal cord injury. J Rehabil Res Dev 2013; 50: 1411–1422.
Hettinga DM, Andrews BJ . Oxygen consumption during functional electrical stimulation-assisted exercise in persons with spinal cord injury: implications for fitness and health. Sports Med 2008; 38: 825–838.
Davis GM, Hamzaid NA, Fornusek C . Cardiorespiratory, metabolic, and biomechanical responses during functional electrical stimulation leg exercise: health and fitness benefits. Artif Organs 2008; 32: 625–629.
Page SJ, Levine P, Strayer J . An electric stimulation cycling protocol for gait in incomplete spinal cord injury. Arch Phys Med Rehabil 2007; 88: 798–800.
Yasar E, Yilmaz B, Goktepe S, Kesikburun S . The effect of functional electrical stimulation cycling on late functional improvement in patients with chronic incomplete spinal cord injury. Spinal Cord (e-pub ahead of print 17 February 2015; doi:10.1038/sc.2015.19).
Stoloff RH, Zehr EP, Ferris DP . Recumbent stepping has similar but simpler neural control compared to walking. Exp Brain Res 2007; 178: 427–438.
Hass CJ, Garzarella L, de Hoyos DV, Connaughton DP, Pollock ML . Concurrent improvements in cardiorespiratory and muscle fitness in response to total body recumbent stepping in humans. Eur J Appl Physiol 2001; 85: 157–163.
Billinger SA, Mattlage AE, Ashenden AL, Lentz AA, Harter G, Rippee MA . Aerobic exercise in subacute stroke improves cardiovascular health and physical performance. J Neurol Phys Ther 2012; 36: 159–165.
Ginis KA, Hicks AL, Latimer AE, Warburton DE, Bourne C, Ditor DS et al. The development of evidence-informed physical activity guidelines for adults with spinal cord injury. Spinal Cord 2011; 49: 1088–1096.
US Department of Health and Human Services. 2008 Physical Activity Guidelines for Americans. Available at: http://www.health.gov/PAGuidelines. Accessed on 20 January 2015.
WHO Towards a Common Language for Functioning, Disability, and Health: ICF. World Health Organization: Geneva, Switzerland. 2002.
Billinger SA, Tseng BY, Kluding PM . Modified total-body recumbent stepper exercise test for assessing peak oxygen consumption in people with chronic stroke. Phys Ther 2008; 88: 1188–1195.
Billinger SA, Loudon JK, Gajewski BJ . Validity of a total body recumbent stepper exercise test to assess cardiorespiratory fitness. J Strength Cond Res 2008; 22: 1556–1562.
Alexander MS, Anderson KD, Biering-Sorensen F, Blight AR, Brannon R, Bryce TN et al. Outcome measures in spinal cord injury: recent assessments and recommendations for future directions. Spinal Cord 2009; 47: 582–591.
Scivoletto G, Tamburella F, Laurenza L, Foti C, Ditunno JF, Molinari M . Validity and reliability of the 10-m walk test and the 6-min walk test in spinal cord injury patients. Spinal Cord 2011; 49: 736–740.
van Hedel HJ, Wirz M, Dietz V . Assessing walking ability in subjects with spinal cord injury: validity and reliability of 3 walking tests. Arch Phys Med Rehabil 2005; 86: 190–196.
Ishikawa S, Stevens SL, Kang M, Morgan DW . Reliability of daily step activity monitoring in adults with incomplete spinal cord injury. J Rehabil Res Dev 2011; 48: 1187–1194.
Bowden MG, Behrman AL . Step Activity Monitor: accuracy and test-retest reliability in persons with incomplete spinal cord injury. J Rehabil Res Dev 2007; 44: 355–362.
Ditunno JF Jr, Ditunno PL, Scivoletto G, Patrick M, Dijkers M, Barbeau H et al. The Walking Index for Spinal Cord Injury (WISCI/WISCI II): nature, metric properties, use and misuse. Spinal Cord 2013; 51: 346–355.
Wirz M, Muller R, Bastiaenen C . Falls in persons with spinal cord injury: validity and reliability of the Berg Balance Scale. Neurorehabil Neural Rep 2010; 24: 70–77.
Lemay JF, Nadeau S . Standing balance assessment in ASIA D paraplegic and tetraplegic participants: concurrent validity of the Berg Balance Scale. Spinal Cord 2010; 48: 245–250.
Lam T, Noonan VK, Eng JJ, Team SR . A systematic review of functional ambulation outcome measures in spinal cord injury. Spinal Cord 2008; 46: 246–254.
Pelletier CA, Totosy de Zepetnek JO, MacDonald MJ, Hicks AL . A 16-week randomized controlled trial evaluating the physical activity guidelines for adults with spinal cord injury. Spinal Cord 2014; 53: 363–367.
Musselman K . Clinical significance testing in rehabilitation research: what, why, and how? Phys Ther Rev 2007; 12: 287–296.
Gregory CM, Bowden MG, Jayaraman A, Shah P, Behrman A, Kautz SA et al. Resistance training and locomotor recovery after incomplete spinal cord injury: a case series. Spinal Cord 2007; 45: 522–530.
Labruyere R, van Hedel HJ . Strength training versus robot-assisted gait training after incomplete spinal cord injury: a randomized pilot study in patients depending on walking assistance. J Neuroeng Rehabil 2014; 11: 4.
Waters RL, Lunsford BR . Energy cost of paraplegic locomotion. J Bone Joint Surg Am 1985; 67: 1245–1250.
Waters RL, Yakura JS, Adkins RH . Gait performance after spinal cord injury. Clin Orthop Relat Res 1993; 288: 87–96.
Waters RL, Mulroy S . The energy expenditure of normal and pathologic gait. Gait Posture 1999; 9: 207–231.
Blessey RL, Hislop HJ, Waters RL, Antonelli D . Metabolic energy cost of unrestrained walking. Phys Ther 1976; 56: 1019–1024.
Waters RL, Hislop HJ, Perry J, Thomas L, Campbell J . Comparative cost of walking in young and old adults. J Orthop Res 1983; 1: 73–76.
Waters RL, Lunsford BR, Perry J, Byrd R . Energy-speed relationship of walking: standard tables. J Orthop Res 1988; 6: 215–222.
Tudor-Locke C, Craig CL, Aoyagi Y, Bell RC, Croteau KA, De Bourdeaudhuij I et al. How many steps/day are enough? For older adults and special populations. Int J Behav Nutr Phys Act 2011; 8: 80.
Tudor-Locke C, Craig CL, Brown WJ, Clemes SA, De Cocker K, Giles-Corti B et al. How many steps/day are enough? For adults. Int J Behav Nutr Phys Act 2011; 8: 79.
Acknowledgements
This work was supported by the South Carolina Clinical and Translational Research (SCTR) Institute, with an academic home at the Medical University of South Carolina, through NIH Grant Number UL1 TR000062 and TL1 TR000061. Additional support was provided by the South Carolina Spinal Cord Injury Research Fund (Grant No. 13-003) and the South Carolina Spinal Cord Injury Association. We thank the staff Physical Therapist, Dr Lindsay Perry, and the project coordinators, Patrick Morgan and Katy Holthaus, for their assistance with study management and data collection.
Ethical Statement
We certify that all applicable institutional and governmental regulations concerning the ethical use of human volunteers were followed during the course of this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
DiPiro, N., Embry, A., Fritz, S. et al. Effects of aerobic exercise training on fitness and walking-related outcomes in ambulatory individuals with chronic incomplete spinal cord injury. Spinal Cord 54, 675–681 (2016). https://doi.org/10.1038/sc.2015.212
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sc.2015.212
This article is cited by
-
An analysis of the effectiveness of rehabilitation protocols for patients with spinal cord injury: A systematic review
Journal of Public Health (2023)
-
The test-retest reliability of individualized VO2peak test modalities in people with spinal cord injury undergoing rehabilitation
Spinal Cord (2021)
-
Physical activity among individuals with spinal cord injury who ambulate: a systematic scoping review
Spinal Cord (2020)
-
Effects of moderate- and high-intensity aerobic training program in ambulatory subjects with incomplete spinal cord injury–a randomized controlled trial
Spinal Cord (2018)