Abstract
Study design:
Retrospective cohort study.
Objectives:
Anejaculation is commonly found in spinal cord injured (SCI) men. Clinical treatments and assisted reproductive techniques allow SCI men to father children but few home pregnancies have been reported. The objective of this paper is to evaluate the results from the last 20 years’ of treatment with penile vibratory stimulation (PVS) and vaginal self-insemination at home in SCI men and their partners.
Setting:
The data originate from two European centers and one American center.
Methods:
A total of 140 SCI men with anejaculation and their healthy partners were available for this analysis. Men who obtained antegrade ejaculation by PVS and had motile sperm in the ejaculate were offered the possibility of PVS combined with vaginal self-insemination at home. Couples were instructed to perform PVS and to instill the ejaculate intravaginally. Outcome measures were pregnancy rate per couple, number of live births, total motile sperm count and time to pregnancies.
Results:
Median total motile sperm count was 29 million (range, 1–92 million). In all, 60 of the 140 couples (43% pregnancy rate) achieved 82 pregnancies. Seventy-two of the pregnancies resulted in live births with the delivery of 73 healthy babies. Median time to first pregnancy was 22.8 months (6.0–98.4). No complications were reported.
Conclusion:
PVS combined with vaginal self-insemination may be performed as a viable, inexpensive option for assisted conception in couples in whom the SCI male partner has an adequate total motile sperm count and the female partner is healthy.
Similar content being viewed by others
Introduction
Contrary to women with spinal cord injury (SCI), the ability to procreate naturally is lost in the majority of SCI males due to anejaculation and abnormal semen characteristics.1 However, successful pregnancies in partners of SCI men have been reported after ejaculation induced by penile vibratory stimulation (PVS), electroejaculation (EEJ) or surgical sperm retrieval followed by the use of assisted reproductive techniques (ART).1 ART includes intrauterine insemination (IUI), and in vitro fertilization (IVF) with or without intracytoplasmic sperm injection (ICSI).
PVS can also be combined with vaginal self-insemination in selected patients. This combination is inexpensive and allows the couple to attempt pregnancy by themselves in their own home. If reasonable rates of pregnancy can be seen with this combination, the cost saving and retention of the privacy of procreation would seem to be optimal.
In 1984, Brindley2 reported the first seven home pregnancies following PVS and vaginal self-insemination with delivery of five healthy babies (one ongoing/one spontaneous abortion). Since that initial report, only a limited number of pregnancies have been reported from PVS procedures combined with self-insemination at home.3, 4, 5, 6, 7 Most studies reported that multiple ovulation cycles were used to achieve the home pregnancies and the overall pregnancy rate per couple is ranging from 25 to 61%.3, 4, 5, 6, 7
The purpose of this study is to present the last 20 years’ experience from penile PVS and vaginal self-insemination at home in SCI men and their partners. The data originate from two European centers and one American center.
Materials and methods
Couples with an SCI male partner presenting for infertility treatment between 1988 and 2008 were evaluated by a retrospective chart review. Only men who obtained antegrade ejaculation by PVS and had motile sperm in the ejaculate were offered together with their partners the possibility of PVS combined with vaginal self-insemination at home. The criteria for the female partner were a normal gynecological history and regular menstrual cycles. Pregnancy outcome data of 140 SCI men and their partners were available for this analysis.
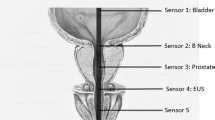
PVS was performed by placing a vibrator applicator on either the dorsum or frenulum of the glans penis, thus activating the ejaculatory reflex mechanically through the dorsal nerve of the penis (Figure 1). PVS was performed for 2–3 min or until ejaculation occurred. If the patient did not ejaculate, a new attempt was made after a short break. Patients were considered non-responders after six failed PVS attempts. The ejaculate was collected in a cup and the urethra milked manually to ensure that as much semen as possible was collected. Baseline raw semen analysis was performed in all men, and the total sperm motility count per ejaculate was calculated.
For penile vibratory stimulation the device is placed on the glans penis. The FERTI CARE vibrator (Multicept A/S, Frederiksberg, Denmark) depicted here was developed for SCI men as the amplitude and frequency can be set at exactly 2.5 mm and 100 Hz that is optimal for inducing ejaculation.8
After the initial evaluation the couples were instructed on how to perform PVS at home by their treating physician at one of the three participating centers. The timing of ovulation was identified by monitoring the basal body temperature. At the time of ovulation PVS was performed and the ejaculate was collected in a non-spermicidal container and instilled intravaginally in the female partner with a 10 ml syringe. The outcome measures were live birth rate, pregnancy rate per couple, total motile sperm count and time to pregnancies.
The Mann–Whitney rank test for non-paired data was used to asses differences between the pregnancy group and non-pregnancy group related to duration of spinal cord lesion, male age, female age or total sperm motility count. A P-value <0.05 was considered as level of statistical significance.
All applicable institutional and governmental regulations concerning the ethical use of human volunteers were followed during the course of research in all the three centers involved.
Results
The median age of the SCI men was 30 years (range, 22–44), and the median duration of their spinal cord lesion was 7 years (range, 1–22), with level of lesion ranging from C2 to T9. The median age of women was 28 years (range, 19–39).
The median total motile sperm count was 29 million (range, 1–92 million). Overall, 60 of the 140 couples (43% pregnancy rate) achieved 82 pregnancies. Seventy-two of the pregnancies resulted in live births with the delivery of 73 healthy babies. Ten miscarriages were noted. A total of 57 couples (41% live birth rate) had children with 41 couples having one child each, 15 couples having two children and one couple having one pair of twins. The median time to first pregnancy was 22.8 months (range, 6.0–98.4 months). No statistically significantly differences were seen between the pregnancy group compared with the non-pregnancy group in relation to duration of spinal cord lesion (P=0.77), male age (P=0.68), female age (P=0.45), or total sperm motility count (P=0.87). No complications from PVS and home insemination procedures were reported.
Discussion
This is the largest study reporting pregnancy rates and live births in couples with an SCI male partner using the non-invasive and inexpensive combination of PVS and vaginal self-insemination performed by the patients in their own home. In selected couples, we have shown a pregnancy rate per couple of 43%. More importantly, 57 out of 140 couples achieved at least one live birth (41% live birth rate). This is comparable to previous results of IUI in SCI men4, 6, 7, 9 and in the low end of results reported from IVF/ICSI that have shown pregnancy rates per couple between 38 and 100%.3, 4, 5, 6, 7, 10, 11, 12 Also, the results are similar to the findings of a smaller study recently published by Kathiresan et al.,13 where PVS followed by intravaginal insemination was undertaken in 45 selected couples. Seventeen of these couples (38% pregnancy rate) achieved 20 pregnancies resulting in 18 live births.
The method of self-insemination at home cannot be offered to all couples with an SCI male partner as the prerequisites are both an ability to ejaculate in response to PVS treatment and motile sperm in the ejaculate in the male as well as the healthy female partner.
Semen parameters are reduced in many SCI men1 and the fertility potential of an SCI man must be verified before vaginal self-insemination is attempted. There are no definitive guidelines on the subject, but self-insemination and/or IUI is usually recommended when at least four million motile sperm are present in the ejaculate, whereas numbers lower than four million call for either IVF either with or without ICSI.9 In this study, we have included men with total motile sperm counts as low as one million.
For PVS to be effective, the dorsal penile nerves must be intact as these comprise the afferent part of the ejaculatory reflex. Also, PVS generally works best in men with an intact reflex arc at the spinal cord level of S2–S4 and T11–L2. The latter is evident in the finding that men with injuries of T10 or above have 75% success rate with PVS, whereas men with lower injuries have only a 15% success rate.14 In the present study, all patients had a level of injury above T10.
Aside from PVS, methods of inducing ejaculation in SCI men include EEJ and surgical sperm retrieval. Both of these techniques are far more invasive than PVS and generally result in fewer motile sperm.15
In EEJ, electricity is delivered through a rectal probe to induce ejaculation, which always requires administration by a doctor. Furthermore, SCI men with incomplete lesions and/or preserved pelvic sensation may require general anesthesia. In contrast to PVS, in which the vast majority of patients have mainly antegrade ejaculation, EEJ often results in some degree of retrograde ejaculation, which must then be collected from the bladder by cathetrization after EEJ.15
Surgical sperm retrieval, which may involve testicular biopsies and/or needle aspiration of sperm from the testis or epididymis16 are invasive procedures that may result in a variety of adverse events including bleeding, infection and prolonged testicular pain. Furthermore, the numbers of sperm obtained from surgical sperm retrieval procedures are usually so low that it commits the couple to higher levels of ART such as IVF/ICSI. Couples with an SCI male partner in whom the semen parameters are poor can certainly benefit from ART, as pregnancy and delivery rates per couple appear to be similar to other infertility couples without an SCI situation.17
In IUI, semen is collected at the clinic and injected into the uterine cavity. As IUI is performed in the clinic, it is less convenient and more expensive than home vaginal self-insemination. Additionally, the technique is sometimes combined with ovarian stimulation. Ovarian stimulation carries a risk of ovarian hyperstimulation syndrome and has an increased rate of multiple pregnancies.18 Both IVF with and without ICSI are more advanced techniques and involve ovarian stimulation followed by ultrasound-guided transvaginal oocyte retrieval. These techniques are feasible with very few motile sperm,9, 19 but also carries with them increased risk for the female partner including risks of developing ovarian hyperstimulation syndrome, as well as potential complications of oocyte retrieval, multiple gestation pregnancy and increased pregnancy losses.20
Aside from invasiveness and convenience, total cost of treatment must also be considered. Both EEJ and surgical sperm retrieval are more expensive than PVS; and although IVF/ICSI are good options when the less invasive options have failed, the price of these procedures is also high (US $8000−US $12 000).
Many health practitioners do not offer PVS and self-insemination to their SCI patients. A number of centers are even using surgical sperm retrieval followed by IVF/ICSI as the first line of treatment.17 Reasons for not offering the less invasive techniques include a lack of equipment and/or lack of knowledge and training, as well as a lack of trust in the effectiveness of these techniques.
This study shows that good results can in fact be obtained with the combination of PVS and self-insemination sparing selected SCI men and their partners both monetary cost and risk of adverse effects from more invasive treatments. Patients suitable for the treatment can be selected by PVS attempts in the clinic followed by a semen analysis. With this simple selection we have shown a live birth rate of 41%. Given its low cost and the non-invasive nature, it is the authors' opinion that it is reasonable to attempt PVS in SCI patients before continuing to more invasive treatments. Even though not all patients can be offered the treatment, all patients should undergo this basic evaluation. In centers that are not able to offer PVS, patients should be referred to centers that can offer this option.
One drawback of the PVS/self-insemination technique is the fairly long median time to pregnancy. In this study, time to pregnancy was 22.8 months. Owing to a lack of data it is unclear what the cause of the long median time to pregnancy is, and this is a weakness of the study. However, the wide range of time to pregnancy of 6.0–98.4 months points to few and/or sporadic conception attempts by some couples as a possible explanation. In any case patients should be informed of the potential for an extended period of attempts to insure the required patience in the treatment. Furthermore, the method may not be ideal with advanced age of the female partner. When successful, however, PVS/self-insemination provides patients with a more natural and intimate method of conception at a much lower cost than other methods of ART. It also offers the opportunity of having more than one child without further medical assistance, as seen in this study with 15 couples having more than one pregnancy resulting in a live childbirth. One weakness of this study is that it is simply a case series and not a randomized trial. Also, the minimal medical assistance associated with PVS/self-insemination as well as the retrospective, long-term nature of the study did not allow us to collect consistent data on patient characteristics such as number of attempted conceptions. In addition, only one basic semen analysis was performed per patient. This means we are unable to identify and compare variables associated with treatment success and time to pregnancy in a reliable way.
Considering our highly selected patient group, it is possible that a higher pregnancy rate could have been seen with one of the more traditional treatment modalities. On the other hand, SCI men with motile sperm counts down to one million were included and the evaluations were based on only one semen sample. This suggests that higher pregnancy rates might be obtained if a higher standard for sperm evaluation was employed, including more than one semen analysis and use of sperm morphology and viability. Also, higher pregnancy rates might be seen if the female partners were evaluated for the absence of any tubal or uterine pathology, especially because there is no available data regarding subsequent diagnosis of female factors in the non-pregnancy group. As described, this study did not find any significant differences between the pregnancy group and the non-pregnancy group regarding spinal cord lesion, male age, female age, or total sperm motility count, and further studies are needed to access the optimal parameters when offering PVS/self-insemination. The study lacks an assessment of how many home attempts each SCI couple made at pregnancy before they succeeded or terminated attempts, and we lack exact knowledge regarding subsequent fertility treatments from all the centers involved in this study. However, based on the data from one of the centers in this study (Murnau), it was determined that the couples who failed PVS/self-insemination proceeded to have a pregnancy rate per couple of 50% following ART.
Based on the largest series reported to date, it is concluded that PVS combined with vaginal self-insemination may be performed as a viable, inexpensive option for assisted conception in couples in whom the SCI male partner has an adequate total motile sperm count and the female partner is healthy. If self-insemination fails, it is possible to proceed to other reproductive treatments. Future research is needed to further assess how different patient and semen variables relate to treatment success and time to pregnancy.
References
Biering-Sørensen F, Sønksen J . Sexual function in spinal cord lesioned men. Spinal Cord 2001; 39: 455–470.
Brindley GS . The fertility of men with spinal injuries. Paraplegia 1984; 22: 337–348.
Dahlberg A, Ruutu M, Hovatta O . Pregnancy results from a vibrator application, electroejaculation, and a vas aspiration programme in spinal-cord injured men. Hum Reprod 1995; 10: 2305–2307.
Nehra A, Werner MA, Bastuba M, Title C, Oates RD . Vibratory stimulation and rectal probe electroejaculation as therapy for patients with spinal cord injury: semen parameters and pregnancy rates. J Urol 1996; 155: 554–559.
Löchner-Ernst D, Mandalka B, Kramer G, Stöhrer M . Conservative and surgical semen retrieval in patients with spinal cord injury. Spinal Cord 1997; 35 (7): 463–468.
Sønksen J, Sommer P, Biering-Sørensen F, Ziebe S, Lindhard A, Loft A et al. Pregnancy after assisted ejaculation procedures in men with spinal cord injury. Arch Phys Med Rehabil 1997; 78 (10): 1059–1061.
Rutkowski SB, Geraghty TJ, Hagen DL, Bowers DM, Craven M, Middleton JW . A comprehensive approach to the management of male infertility following spinal cord injury. Spinal Cord 1999; 37 (7): 508–514.
Sønksen J, Biering-Sørensen F, Kvist Kristensen J . Ejaculation induced by penile vibratory stimulation in men with spinal cord injuries. The importance of the vibratory amplitude. Paraplegia 1994; 32: 651–660.
Ohl DA, Wolf LJ, Menge AC, Christman GM, Hurd WW, Ansbacher R et al. Electroejaculation and assisted reproductive technologies in the treatment of anejaculatory infertility. Fertil Steril 2001; 76: 1249–1255.
Hultling C, Rosenlund B, Levi R, Fridström M, Sjöblom P, Hillensjö T . Assisted ejaculation and in-vitro fertilization in the treatment of infertile spinal cord-injured men: the role of intracytoplasmic sperm injection. Hum Reprod 1997; 12 (3): 499–502.
Heruti RJ, Katz H, Menashe Y, Weissenberg R, Raviv G, Madjar I et al. Treatment of male infertility due to spinal cord injury using rectal probe electroejaculation: the Israeli experience. Spinal Cord 2001; 39 (3): 168–175.
Shieh JY, Chen SU, Wang YH, Chang HC, Ho HN, Yang YS . A protocol of electroejaculation and systematic assisted reproductive technology achieved high efficiency and efficacy for pregnancy for anejaculatory men with spinal cord injury. Arch Phys Med Rehabil 2003; 84 (4): 535–540.
Kathiresan AS, Ibrahim E, Aballa TC, Attia GR, Lynne CM, Brackett NL . Pregnancy outcomes by intravaginal and intrauterine insemination in 82 couples with male factor infertility due to spinal cord injuries. Fertil Steril 2011; 96 (2): 328–331.
Sonksen J . Assisted ejaculation and semen characteristics in spinal cord injured males. Scand J Urol Nephrol Suppl 2003; 213: 1–31.
Ohl DA, Sonksen J, Menge AC, McCabe M, Keller LM . Electroejaculation versus vibratory stimulation in spinal cord injured men: sperm quality and patient preference. J Urol 1997; 157: 2147–2149.
Practice Committee of the American Society for Reproductive Medicine. Sperm retrieval for obstructive azoospermia. Fertil Steril 2006; 86 (Suppl 1): 115–120.
Kafetsoulis A, Brackett NL, Ibrahim E, Attia GR, Lynne CM . Current trends in the treatment of infertility in men with spinal cord injury. Fertil Steril 2006; 86: 781–789.
Cantineau AEP, Cohlen BJ, Heineman MJ . Ovarian stimulation protocols (anti-oestrogens, gonadotrophins with and without GnRH agonists/antagonists) for intrauterine insemination (IUI) in women with subfertility. Cochrane Database Syst Rev 2007; 18: CD005356.
Palermo G, Joris H, Devroey P, Van Steirteghem AC . Pregnancies after intracytoplasmic injection of single spermatozoon into an oocyte. Lancet 1992; 340: 17–18.
Schenker JG, Ezra Y . Complications of assisted reproduction techniques. Fertil Steril 1994; 61: 411–422.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest. Jens Sønksen is shareholder in Multicept A/S, Frederiksberg, Denmark.
Rights and permissions
About this article
Cite this article
Sønksen, J., Fode, M., Löchner-Ernst, D. et al. Vibratory ejaculation in 140 spinal cord injured men and home insemination of their partners. Spinal Cord 50, 63–66 (2012). https://doi.org/10.1038/sc.2011.101
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sc.2011.101
Keywords
This article is cited by
-
Evaluation of a re-engineered device for penile vibratory stimulation in men with spinal cord injury
Spinal Cord (2021)
-
Fertility and sexuality in the spinal cord injury patient
World Journal of Urology (2018)
-
Ejaculation and sperm characteristics in men with cauda equina and conus medullaris syndromes
Spinal Cord (2017)
-
Comparison of three methods of penile vibratory stimulation for semen retrieval in men with spinal cord injury
Spinal Cord (2017)
-
A step-wise approach to sperm retrieval in men with neurogenic anejaculation
Nature Reviews Urology (2015)