Abstract
Study design:
Case study.
Objectives:
Subacute myelo-optico-neuropathy (SMON) is a severe neuro-degenerative disorder caused by poisoning due to over-dose and prolonged oral administration of clioquinol; this disorder was more frequent during 1957–1970. It is characterized by axonal degeneration and gliosis in the cervical gracile fasciculus. Recently, copper-deficient myelo-neuropathies presenting similar symptoms (that is, painful dysesthesia/paresthesia in the lower limbs, ataxia, spastic paraplegia, autonomic disorders and visual impairment) were reported. Magnetic resonance imaging (MRI) of these patients detected T2-weighted hyperintensities in the cervical spinal cord. An unbalanced zinc–copper metabolism was suggested as one of the candidate pathogenesis of clioquinol toxicity because of its metal-chelating ability. The aim of this study was to present MRI findings of old SMON patients and to compare them with those of current copper-deficient myelo-neuropathies.
Setting:
Japan.
Methods:
We conducted and analyzed cervical and brain MRIs of seven old SMON patients who contracted the disorder during the 1960s. Serum iron, magnesium, copper, zinc and ceruloplasmin levels were also measured.
Results:
Cervical T2-weighted MRIs showed mild volume loss and faint hyperintensities in the dorsal columns, which might reflect residual gliosis. Brain fast fluid-attenuated inversion-recovery images and tractography were normal. Current levels of serum copper and zinc were within almost normal ranges.
Conclusion:
Although fainter, the abnormal T2 MRI signals we observed were similar to and occurred in the same locations as those reported in copper-deficient myelo-neuropathy patients. We suggest that these findings are useful to study the mechanism of clioquinol toxicity before using it to treat neurodegenerative diseases such as Alzheimer's disease.
Similar content being viewed by others
Introduction
Subacute myelo-optico-neuropathy (SMON) is a neuro-degenerative disorder caused by poisoning due to over-dose and prolonged oral administration of clioquinol. This was shown by an epidemiological study in Japan in 1971,1 and confirmed by a series of animal experiments.2 It is characterized by symptoms of severe myelo-neuropathy: painful dysesthesia and paresthesia (such as tingling, stinging, fastening, cold, and sticking sensations) initiating in and moving upwards from the feet, loss of sensations, gait disturbance with ataxic and spastic paraplegia, autonomic disorders, and visual impairment, which almost invariably followed a severe abdominal pain (and sometimes led to loss of consciousness and opisthotonus), constipation, and diarrhea.3, 4, 5 In Japan, there are still more than 2500 SMON patients suffering from severe dysesthesia/paresthesia and ataxic paraplegia.6 Many autopsy case reports confirmed that the characteristic pathological finding of SMON was a ‘dying back neuropathy’ in the upper cervical gracile fasciculus (the axon terminals of dorsal root neurons) and the lateral funiculus (the long peripheral terminals of the pyramidal tract). In SMON patients who died at an early stage of the disease, active gliosis and axonal degeneration were detected in the gracile fasciculus (Goll fasciculus), and both the lateral funiculus and dorsal root ganglion were also severely affected.7 In another autopsy case of SMON, performed 42 years after the onset of the disease, gliosis was observed in an atrophic gracile fasciculus, but not in the lumbar lateral funiculus.8
A recent study of patients with copper deficiency related to hyperzincemia,9, 10 called ‘human swayback’, presenting with symptoms of severe sensory ataxic myelo-optic-neuropathy and urinary incontinence, showed abnormal magnetic resonance imaging (MRI) findings in their cervical spinal cords. Although the mechanism by which clioquinol toxicity developed into severe myelo-optico-neuropathy remains undefined, Kumar11 and Schaumburg12 suggested that it might be similar to that involved in the copper deficiency related to hyperzincemia reported in recent studies,13, 14, 15, 16 as some of the hypocupremia patients presented with severe dysesthesia in their lower extremities.
As the peak period of the SMON epidemic in Japan was about 10 years before development of MRI technology, SMON patients were not examined with MRI at that time. Even now, we are not aware of any literature presenting the MRI findings of SMON patients. To compare SMON with the copper-deficient myelo-neuropathies, we took and analyzed cervical and brain MRIs of some of the surviving SMON patients. We also present their clinical laboratory data, including serum copper, zinc and ceruloplasmin levels.
Methods
Seven SMON patients diagnosed by their neurological manifestations and confirmed histories of intake of high/prolonged doses of oral clioquinol (1.2–2.4 g per day, for 4 weeks to 6 months), who were followed up in our hospital, participated in this study (average age 72.6±9.3 years). Patient characteristics are given in Table 1. All seven patients have survived more than 40 years after the onset of their symptoms, and still have the typical symptoms of SMON: severe dysesthesia, paresthesia, loss of sensation in the lower extremities, sensory ataxia, spastic paralysis and/or visual impairments. All of them have normal scores on the Mini-Mental State Examination (28.8±1.6). At the same time, we also measured serum levels of copper, zinc, iron and ceruloplasmin.
The patients underwent MR imaging for the head and cervical regions on a 3-T MRI scanner (Achieva 3.0T; Philips Medical Systems, Best, The Netherlands) using eight-channel head coils. The imaging sequences for the head region included axial spin-echo T1-weighted (repetition time (TR)/echo time (TE)/number of signal intensity acquisition (NSA) 450 ms/10 ms/1, matrix 320 × 320), turbo spin-echo T2-weighted (TR/TE/NSA 4060 ms/80 ms/2, turbo factor 9, matrix 512 × 512), fast fluid-attenuated inversion-recovery (TR/TE/NSA 9000 ms/120 ms/1, inversion time (TI) 2500 ms, turbo factor 15, matrix 352 × 352) and diffusion-tensor images. For diffusion-tensor imaging we used a single-shot, spin-echo, echo-planar technique; the parameters were TR/TE/NSA 8500 ms/95 ms/2, motion-probing gradient in six directions, b value 1000 s mm−2, matrix 128 × 128, voxel size 1.8 × 1.8 × 2.0 mm3 and no intersection gap. The field of view was 23 cm on all conventional MR images. The imaging sequences for the cervical region included sagittal and axial spin-echo T1-weighted (TR/TE/NSA 450 ms/10 ms/4, slice thickness 3 mm) and turbo spin-echo T2-weighted images (TR/TE/NSA 3000 ms/90 ms/2, turbo factor 17, slice thickness 3 mm). The diffusion-tensor imaging data were transferred to an offline workstation (Precision 530; Dell, Round Rock, TX, USA); Philips Research Imaging Development Environment (PRIDE) software (Philips Medical Systems) was used for image analysis. Fiber tracking was performed with FiberTracking V4.1 (PRIDE) on the same workstation.
Results
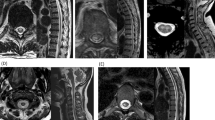
Although we did not have data from their peak disease period, the patients’ current serum copper, zinc, iron and ceruloplasmin levels were all within the normal range (Table 1). We first examined T2-weighted and fast fluid-attenuated inversion-recovery images to see whether there were specific signal changes in the cervical dorsal column, where the severest degeneration was observed in previous autopsies of SMON patients. In three of the seven patients, no abnormal findings on the spinal cord were observed. In the other four patients, we observed medium-sized depressions in the cervical dorsal columns, possibly related to volume loss in the gracile fasciculus. A slight T2-weighted hyperintensity on the cervical spinal cord of a 56-year-old female patient is shown in Figure 1. In the other three patients, such abnormal signals were also observed at the same level of the cervical spinal cord, but they were much weaker than in the first case. Three patients (one with T2-weighted hyperintensity and two without any abnormal findings) exhibited mild compression damages with spondylotic changes or disc herniations in their cervical spinal cords. There were no abnormal findings detected on the brain MRIs, except mild brain atrophies or nonspecific ischemic changes that are probably attributable to aging. In MR tractography, the higher cortico-spinal tracts and visual cortex-related fibers were normally traced.
MRI of a 56-year-old female SMON patient. (a) T2-weighted image of the cervical spinal cord shows a longitudinal, faint hyperintensity in the sagittal image (arrowhead), (b) which was located on the dorsal column of the spinal cord in the axial image (arrowhead). (c) Brain MRI FLAIR image appears normal. (d) MR tractography shows normal tracing of the pyramidal tract above the brain stem.
Discussion
In this MRI study of old, surviving SMON patients, we found slight hyperintense signals in the cervical spinal cord and medium-sized depressions in their cervical dorsal column. As only old SMON survivors were able to participate in this study, these mild findings might be consistent with a decrease in or cessation of the degeneration followed by gliosis after they had stopped taking clioquinol. On the other hand, the relatively low severity of their symptoms possibly enabled these patients to survive longer, and thus to have a chance for these MRI examinations. Alternatively, such low signals might be considered as an artifact of the 3-T MRI or over-estimation.
The first reported cervical MRI of a copper-deficient myelo-neuropathy patient17 showed a T2-weighted hyperintensity in the dorsal column of the cervical spinal cord (C1–C7). In 11 of 25 clinically diagnosed patients, Kumar et al.18 also found T2-weighted hyperintensities at almost the same portions (central dorsal midline cord involving the dorsal columns) of the spinal cord (spread out from cervical to thoracic, mainly C2–C7 level). Some signals were quite strong and some were faint, which might depend on the severity or the duration of neurological symptoms. In addition, Spinazzi et al.9 presented a patient with copper deciency myelopathy induced by parenteral zinc overloading during chronic hemodialysis with a longitudinal midline central and dorsal lesion in the lower cervical spinal cord. The abnormal T2 hyperintensity signals of these patients disappeared18 or declined9, 10 as the symptoms recovered when they were treated with copper supplements. The T2 hyperintensities we observed on the cervical spinal cord images of old SMON survivors (4 out of 7) were all much fainter but in the same positions, cervical mid-dorsal columns, and were accompanied by mild volume loss. We considered that these fainter signals might reflect the duration and severity of the clioquinol-induced neuronal damages; the patho-mechanism by which the ‘iatrogenic’ over-dose and prolonged administration of clioquinol induced SMON may be related to a hyperzincemia-induced copper deficiency, which is described as the most probable theory by Kumar and Knopman11 and Nations et al.13
The previously reported hyperintensities seen in T2-weighted MRIs from patients with copper deficiency related to hyperzincemia might partly represent an active phase of SMON.9 The brain MRIs of all seven SMON patients, as well as the tract-tracing study of their higher cortico-spinal tract and visual cortex-related fibers, were all normal. It was suggested that these portions were quite mildly (or not at all) affected, and had recovered, over a long period, well after they stopped receiving clioquinol. An autopsy record of an SMON patient in our hospital described memory loss, cognitive dysfunction and/or character changes before his death. However, given the lack of brain abnormalities in the current patients, it is difficult to confidently suggest that these psychotic symptoms are a direct result of clioquinol in the central nervous system or other factors related to comorbid metabolic disorders.
Conclusion
Finally, we believe that it is worthwhile to examine MRI evidence in the old SMON patients in order to compare them with those of hyperzincemia-induced copper-deficient myelo-neuropathy patients, which might partly mimic the patho-mechanism of SMON. Even though its toxic mechanism still remains incompletely understood, clioquinol is currently being considered as therapy for other neurodegenerative diseases such as Alzheimer's and Parkinson's diseases in the near future.19, 20 Thus, it is important to further our understanding of the mechanism of clioquinol toxicity before using it therapeutically for neurodegenerative diseases as ‘a new therapy’.
Consent
Written informed consent was obtained from all patients for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
References
Tsubaki T, Honma Y, Hoshi M . Neurological syndrome associated with clioquinol. Lancet 1971; 1: 696–697.
Tateishi J, Kuroda S, Ikeda H, Otsuki S . Neurotoxity of iodoxyquinoline: a further study on beagle dogs. Jpn J Med Sci Biol 1975; 28 (Suppl): 187–195.
Sobue I . Clinical aspects of subacute myelo-optico-neuropathy (SMON). In: Vinken PJ BG, Cohen MM et al (eds). Handbook of clinical neurology, vol. 37 Elsevier: Amsterdam, 1979. pp 115–139.
Shiraki H In: Vinken PJ, Bruyn GW, Cohen MM, et al. (eds). Handbook of Clinical Neurology. Elsevier: Amsterdam, 1979. pp 141–198.
Nakae K, Yamamoto S, Igata A . Subacute myelo-optico-neuropathy (S.M.O.N.) in Japan. A community survey. Lancet 1971; 2: 510–512.
Konagaya M, Matsumoto A, Takase S, Mizutani T, Sobue G, Konishi T et al. Clinical analysis of longstanding subacute myelo-optico-neuropathy: sequelae of clioquinol at 32 years after its ban. J Neurol Sci 2004; 218: 85–90.
Shiraki H . Neuropathology of subacute myelo-optico-neuropathy, ‘SMON’. Jpn J Med Sci Biol 1971; 24: 217–243.
Konno H, Takase S . Neuropathology of longstanding subacute myelo-optico-neuropathy (SMON)]. Shinkei Naika 2005; 63: 162–169.
Spinazzi M, De Lazzari F, Tavolato B, Angelini C, Manara R, Armani M . Myelo-optico-neuropathy in copper deficiency occurring after partial gastrectomy. Do small bowel bacterial overgrowth syndrome and occult zinc ingestion tip the balance? J Neurol 2007; 254: 1012–1017.
Spain RI, Leist TP, De Sousa EA . When metals compete: a case of copper-deficiency myeloneuropathy and anemia. Nat Clin Pract Neurol 2009; 5: 106–111.
Kumar N, Knopman DS . SMON, clioquinol, and copper. Postgrad Med J 2005; 81: 227.
Schaumburg H, Herskovitz S . Copper deficiency myeloneuropathy: a clue to clioquinol-induced subacute myelo-optic neuropathy? Neurology 2008; 71: 622–623.
Nations SP, Boyer PJ, Love LA, Burritt MF, Butz JA, Wolfe GI et al. Denture cream: an unusual source of excess zinc, leading to hypocupremia and neurologic disease. Neurology 2008; 71: 639–643.
Kumar N, Gross Jr JB, Ahlskog JE . Myelopathy due to copper deficiency. Neurology 2003; 61: 273–274.
Hedera P, Fink JK, Bockenstedt PL, Brewer GJ . Myelopolyneuropathy and pancytopenia due to copper deficiency and high zinc levels of unknown origin: further support for existence of a new zinc overload syndrome. Arch Neurol 2003; 60: 1303–1306.
Hedera P, Peltier A, Fink JK, Wilcock S, London Z, Brewer GJ . Myelopolyneuropathy and pancytopenia due to copper deficiency and high zinc levels of unknown origin. II. The denture cream is a primary source of excessive zinc. Neurotoxicology 2009; 30: 996–999.
Schleper B, Stuerenburg HJ . Copper deficiency-associated myelopathy in a 46-year-old woman. J Neurol 2001; 248: 705–706.
Kumar N, Ahlskog JE, Klein CJ, Port JD . Imaging features of copper deficiency myelopathy: a study of 25 cases. Neuroradiology 2006; 48: 78–83.
Gouras GK, Beal MF . Metal chelator decreases Alzheimer beta-amyloid plaques. Neuron 2001; 30: 641–642.
Kaur D, Yantiri F, Rajagopalan S, Kumar J, Mo JQ, Boonplueang R et al. Genetic or pharmacological iron chelation prevents MPTP-induced neurotoxicity in vivo: a novel therapy for Parkinson's disease. Neuron 2003; 37: 899–909.
Acknowledgements
We thank the patients for their contribution to this study, Jason Cole for medical English editing, and Tomoko Nakayama and Akiko Hamada for their helpful secretarial assistance. This study was financially supported by the Ministry of Health, Labor, and Welfare of Japan.
Authors' contributions: EK was the primary neurologist, conceived the original study, organized and analyzed the data, prepared the draft of the paper, and supervised the entire study. TH contributed to the original idea and helped write and edit the final draft of the paper. SY, YU and YM were consulting neurologists, evaluated MRI data, and assisted with paper editing. TH was the chief consultant regarding neuro-radiological evaluations. MU analyzed the data and helped write and edit the paper.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Kimura, E., Hirano, T., Yamashita, S. et al. Cervical MRI of subacute myelo-optico-neuropathy. Spinal Cord 49, 182–185 (2011). https://doi.org/10.1038/sc.2010.68
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sc.2010.68
Keywords
This article is cited by
-
Is the clinical course of non-arteritic ischemic optic neuropathy associated with oxidative damage and the dynamics of the antioxidant response?
International Ophthalmology (2023)
-
Clioquinol inhibits dopamine-β-hydroxylase secretion and noradrenaline synthesis by affecting the redox status of ATOX1 and copper transport in human neuroblastoma SH-SY5Y cells
Archives of Toxicology (2021)