Abstract
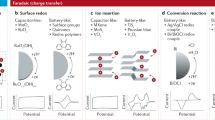
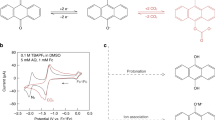
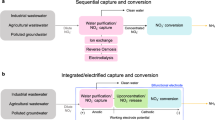
Electrochemical separations are powerful platforms for the sustainable recovery of critical elements, environmental remediation and downstream processing. However, the recent development of electroseparations has primarily focused on heterogeneous adsorbents, which face the challenge of intermittent electroswing operation. Here we present a redox-mediated electrochemical liquid–liquid extraction separation platform that translates selective single-site binding to a fully continuous separation scheme. A redox-active extractant is molecularly designed with controllable hydrophobicity to maximize organic phase retention. The redox flow design enables fully electrified continuous operation with no external chemical input, achieving the selective recovery of precious metals from multicomponent streams. We demonstrate an atomic efficiency of over 90% and over 100:1 selectivity for practical critical metal leach streams, and 16-fold up-concentration for gold and platinum group metals from varied feedstocks including electronic waste, catalytic converter waste and mining streams. Our work is envisioned as a pathway towards a broader class of industrially applicable liquid–liquid extraction-based electrochemical separations.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data are available within the paper and the Supplementary Information. Source data are provided with this paper.
References
Srimuk, P., Su, X., Yoon, J., Aurbach, D. & Presser, V. Charge-transfer materials for electrochemical water desalination, ion separation and the recovery of elements. Nat. Rev. Mater. 5, 517–538 (2020).
Alkhadra, M. A. et al. Electrochemical methods for water purification, ion separations and energy conversion. Chem. Rev. 122, 13547–13635 (2022).
Su, X. et al. Asymmetric Faradaic systems for selective electrochemical separations. Energy Environ. Sci. 10, 1272–1283 (2017).
Kim, K., Raymond, D., Candeago, R. & Su, X. Selective cobalt and nickel electrodeposition for lithium-ion battery recycling through integrated electrolyte and interface control. Nat. Commun. 12, 6554 (2021).
High, M. et al. Precursor engineering of hydrotalcite-derived redox sorbents for reversible and stable thermochemical oxygen storage. Nat. Commun. 13, 5109 (2022).
Li, X., Zhao, X. H., Liu, Y. Y., Hatton, T. A. & Liu, Y. Y. Redox-tunable Lewis bases for electrochemical carbon dioxide capture. Nat. Energy 7, 1065–1075 (2022).
Liu, Y. Y., Ye, H. Z., Diederichsen, K. M., Van Voorhis, T. & Hatton, T. A. Electrochemically mediated carbon dioxide separation with quinone chemistry in salt-concentrated aqueous media. Nat. Commun. 11, 2278 (2020).
Cotty, S. et al. Electrochemical recycling of homogeneous catalysts. Sci. Adv. 8, eade3094 (2022).
Massen-Hane, M., Diederichsen, K. M. & Hatton, T. A. Engineering redox-active electrochemically mediated carbon dioxide capture systems. Nat. Chem. Eng. 1, 35–44 (2024).
Wilcox, J. An electro-swing approach. Nat. Energy 5, 121–122 (2020).
Kim, N., Elbert, J., Kim, C. & Su, X. Redox-copolymers for nanofiltration-enabled electrodialysis. ACS Energy Lett. 8, 2097–2105 (2023).
Kim, N., Lee, J. & Su, X. Precision tuning of highly selective polyelectrolyte membranes for redox-mediated electrochemical separation of organic acids. Adv. Funct. Mater. 33, 2211645 (2023).
Su, X., Chen, Z., St-Pierre, J. & Vasiljevic, N. Electrochemistry for recycling. Electrochem. Soc. Interface 30, 41–43 (2021).
Chen, R., Sheehan, T., Ng, J. L., Brucks, M. & Su, X. Capacitive deionization and electrosorption for heavy metal removal. Environ. Sci. Water Res. Technol. 6, 258–282 (2020).
Su, X. & Hatton, T. A. Redox-electrodes for selective electrochemical separations. Adv. Colloid Interface Sci. 244, 6–20 (2017).
Chen, R. L. et al. Structure and potential-dependent selectivity in redox-metallopolymers: electrochemically mediated multicomponent metal separations. Adv. Funct. Mater. 31, 2009307 (2021).
Kim, K. et al. Electrochemical approaches for selective recovery of critical elements in hydrometallurgical processes of complex feedstocks. iScience 24, 102374 (2021).
Candeago, R. et al. Semiconducting polymer interfaces for electrochemically assisted mercury remediation. ACS Appl. Mater. Interfaces 12, 49713–49722 (2020).
Cotty, S. R., Kim, N. & Su, X. Electrochemically mediated recovery and purification of gold for sustainable mining and electronic waste recycling. ACS Sustain. Chem. Eng. 11, 3975–3986 (2023).
Guo, Z.-Y. et al. Development of electrochemical lithium extraction based on a rocking chair system of LiMn2O4/Li1–xMn2O4: self-driven plus external voltage driven. Sep. Purif. Technol. 259, 118154 (2021).
Xu, T. & Huang, C. Electrodialysis-based separation technologies: a critical review. AlChE J. 54, 3147–3159 (2008).
Brown, C. G. & Sherrington, L. G. Solvent extraction used in industrial separation of rare earths. J. Chem. Technol. Biotechnol. 29, 193–209 (1979).
El-Nadi, Y. A. Solvent extraction and its applications on ore processing and recovery of metals: classical approach. Sep. Purif. Rev. 46, 195–215 (2017).
Mooiman, M. B. The solvent extraction of precious metals: a review. In Proc. 17th International Precious Metals Conference 411–434 (International Precious Metals Institute, 1993).
Yordanov, A. T. & Roundhill, D. M. Solution extraction of transition and post-transition heavy and precious metals by chelate and macrocyclic ligands. Coord. Chem. Rev. 170, 93–124 (1998).
Mahandra, H., Faraji, F. & Ghahreman, A. Novel extraction process for gold recovery from thiosulfate solution using phosphonium ionic liquids. ACS Sustain. Chem. Eng. 9, 8179–8185 (2021).
Towler, G. & Sinnott, R. in Chemical Engineering Design 3rd edn (eds Towler, G. & Sinnott, R.) 631–733 (Butterworth-Heinemann, 2022).
Kislik, V. S. in Solvent Extraction (ed. Kislik, V. S.) 113–156 (Elsevier, 2012).
Vidal, O. in Mineral Resources and Energy (ed. Vidal, O.) 27–52 (Elsevier, 2018).
Sovacool, B. K. et al. Sustainable minerals and metals for a low-carbon future. Science 367, 30–33 (2020).
Butt, C. R. M. & Hough, R. M. Why gold is valuable. Elements 5, 277–280 (2009).
Murphy, K. Gold RRS 2022—Surge in recent discoveries. S&P Global Market Intelligence https://www.spglobal.com/marketintelligence/en/news-insights/research/gold-rrs-2022-surge-in-recent-discoveries (2022).
Calvo, G., Mudd, G., Valero, A. & Valero, A. Decreasing ore grades in global metallic mining: a theoretical issue or a global reality? Resources 5, 36 (2016).
Moreau, V., Dos Reis, P. C. & Vuille, F. Enough metals? Resource constraints to supply a fully renewable energy system. Resources 8, 29 (2019).
Cui, J. R. & Zhang, L. F. Metallurgical recovery of metals from electronic waste: a review. J. Hazard. Mater. 158, 228–256 (2008).
Zadra, J. B., Engel, A. L. & Heinen, H. J. Process for Recovering Gold and Silver from Activated Carbon by Leaching and Electrolysis (US Department of the Interior, Bureau of Mines, 1952).
Ford, P. et al. Economics of end-of-life materials recovery: a study of small appliances and computer devices in Portugal. Environ. Sci. Technol. 50, 4854–4862 (2016).
Beyuo, M. & Abaka-Wood, G. ZADRA elution circuit optimisation and operational experience at the CIL Plant of Gold Fields Ghana Limited. In Proc. 4th UMaT Biennial International Mining and Mineral Conference 161–167 (UMaTBIC, 2016).
Cutting cobalt. Nat. Energy 5, 825 (2020).
Babu, B. R., Parande, A. K. & Basha, C. A. Electrical and electronic waste: a global environmental problem. Waste Manag. Res. 25, 307–318 (2007).
Ding, Y. J. et al. Recovery of precious metals from electronic waste and spent catalysts: a review. Resour. Conserv. Recycl. 141, 284–298 (2019).
Yu, M. L., Wang, K. & Vredenburg, H. Insights into low-carbon hydrogen production methods: green, blue and aqua hydrogen. Int. J. Hydrog. Energy 46, 21261–21273 (2021).
Shestakova, M. & Sillanpää, M. Removal of dichloromethane from ground and wastewater: a review. Chemosphere 93, 1258–1267 (2013).
Priya, V. S. & Philip, L. Treatment of volatile organic compounds in pharmaceutical wastewater using submerged aerated biological filter. Chem. Eng. J. 266, 309–319 (2015).
Toth, A. J. et al. Novel method for the removal of organic halogens from process wastewaters enabling water reuse. Desalin. Water Treat. 130, 54–62 (2018).
Alder, C. M. et al. Updating and further expanding GSK’s solvent sustainability guide. Green Chem. 18, 3879–3890 (2016).
Larsen, C. et al. A tool for identifying green solvents for printed electronics. Nat. Commun. 12, 4510 (2021).
Lu, P. Y. & Alkire, R. C. Mass transfer in parallel plate electrolyzers with two‐phase liquid‐liquid flow. J. Electrochem. Soc. 131, 1059 (1984).
Alkire, R. & Köhler, J. Indirect electrochemical epoxidation of hexene in a liquid-liquid electrolyte. J. Appl. Electrochem. 18, 405–409 (1988).
Papageorgiou, N., Maier, W. F. & Grätzel, M. An iodine/triiodide reduction electrocatalyst for aqueous and organic media. J. Electrochem. Soc. 144, 876 (1997).
Masahiro, T., Akitsugu, O. & Taijiro, O. The chemical behavior of low valence sulfur compounds. VIII. The oxidation of sodium thiosulfate with ozone. Bull. Chem. Soc. Jpn 46, 3785–3789 (1973).
Rumble, J. R. CRC Handbook of Chemistry and Physics (CRC Press, 2017).
Sarla, M., Pandit, M., Tyagi, D. K. & Kapoor, J. C. Oxidation of cyanide in aqueous solution by chemical and photochemical process. J. Hazard. Mater. 116, 49–56 (2004).
Pei, Z. B. et al. Review of the I−/I3− redox chemistry in Zn-iodine redox flow batteries. Mater. Res. Bull. 141, 111347 (2021).
Li, X. et al. Symmetry-breaking design of an organic iron complex catholyte for a long cyclability aqueous organic redox flow battery. Nat. Energy 6, 873–881 (2021).
Orita, A., Verde, M. G., Sakai, M. & Meng, Y. S. A biomimetic redox flow battery based on flavin mononucleotide. Nat. Commun. 7, 13230 (2016).
Geankoplis, C. J., Hersel, A. A. & Lepek, D. H. Prentice Hall International Series in the Physical and Chemical Engineering Sciences (Prentice Hall, 2018).
Baker, D. A., East, G. C. & Mukhopadhyay, S. K. Synthesis and characterization of some disulfonyl azides as potential crosslinking agents for textile fibers. J. Appl. Polym. Sci. 79, 1092–1100 (2001).
Acknowledgements
This paper is based on work supported by the US Department of Energy, Office of Basic Energy Sciences under award no. DOE DE-SC0021409. This work is also partially supported by the NSF DMREF grant 2323988.
Author information
Authors and Affiliations
Contributions
S.R.C., A.F., J.E. and X.S. conceptualized and designed this work. S.R.C. and A.F. performed the experiments. S.R.C. and X.S. wrote the original paper. X.S. supervised the work and secured the funding. All authors analyzed the data and edited the paper.
Corresponding author
Ethics declarations
Competing interests
The authors have applied for a provisional patent based on the technology proposed in the paper.
Peer review
Peer review information
Nature Chemical Engineering thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–38 and Discussion.
Source data
Source Data Fig. 2
Statistical source data.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 4
Statistical source data.
Source Data Fig. 5
Statistical source data.
Source Data Fig. 6
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cotty, S.R., Faniyan, A., Elbert, J. et al. Redox-mediated electrochemical liquid–liquid extraction for selective metal recovery. Nat Chem Eng 1, 281–292 (2024). https://doi.org/10.1038/s44286-024-00049-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s44286-024-00049-x
This article is cited by
-
Electrochemical concentration pumping in liquid–liquid extractions
Nature Chemical Engineering (2024)