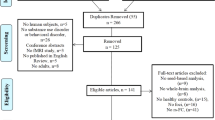
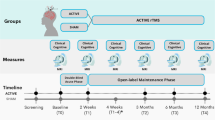
Abstract
Cocaine use disorder (CUD) is prevalent, and repetitive transcranial magnetic stimulation (rTMS) shows promise in reducing cravings. However, the association between a consistent CUD-specific functional connectivity signature and treatment response remains unclear. Here we identify a validated functional connectivity signature from functional magnetic resonance imaging to discriminate CUD, with successful independent replication. We found increased connectivity within the visual and dorsal attention networks and between the frontoparietal control and ventral attention networks, alongside reduced connectivity between the default mode and limbic networks in patients with CUD. These connections were associated with drug use history and cognitive impairments. Using data from a randomized clinical trial, we also established the prognostic value of these functional connectivities for rTMS treatment outcomes in CUD, especially involving the frontoparietal control and default mode networks. Our findings reveal insights into the neurobiological mechanisms of CUD and link functional connectivity biomarkers with rTMS treatment response, offering potential targets for future therapeutic development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$59.00 per year
only $4.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data supporting the results in this study are available within the paper and its Supplementary Information. Part of this study analyzed data from a clinical trial and did not generate new clinical trial data. The datasets used in this study are publicly available. The SUDMEX-CONN dataset is available from Zenodo (https://zenodo.org/record/5123331). The SUDMEX-TMS dataset is also available from Zenodo (https://zenodo.org/record/7126853). The UCLA-CNP dataset is available from OpenNeuro (https://openneuro.org/datasets/ds000030/versions/1.0.0). The NYU dataset is available from the International Neuroimaging Data-sharing Initiative (https://fcon_1000.projects.nitrc.org/indi/retro/nyuCocaine.html).
Code availability
All statistical analyses and machine learning models were implemented in Python 3.7. The packages used in this study are publicly available. The XGBoost classification analysis was implemented using the xgboost 1.6.2 package (https://xgboost.readthedocs.io/en/stable/python/python_intro.html). The RVM regression was done using the sklearn 0.24.2 package (https://scikit-learn.org/stable/index.html). All other statistical analysis methods were implemented using the scipy 1.10.1 package (https://docs.scipy.org/doc/scipy/reference/index.html). Main code for analysis is provided at https://github.com/zhangyubrain/FCN-CUD-Phenotyping-rTMS. Resting-state functional MRI data were processed with fMRIPrep 20.2.3 (https://hub.docker.com/r/nipreps/fmriprep/tags). Internal operations of fMRIPrep 20.2.3 use the following software: Advanced Normalization Tools 2.3.3, Nipype 1.6.1, FSL 5.0.9, FreeSurfer 6.0.1, AFNI 20160207.
References
Huang, C. C., Chen, C. C. & Hsu, K. S. In The Neuroscience of Cocaine (ed. Preedy, V. R.) Ch. 44, 439–447 (2017).
2022 National Survey of Drug Use and Health (NSDUH) releases. Substance Abuse and Mental Health Services Administration https://www.samhsa.gov/data/release/2022-national-survey-drug-use-and-health-nsduh-releases (accessed on 5 February 2024).
Poireau, M. et al. Biomarkers of relapse in cocaine use disorder: a narrative review. Brain Sci. 12, 1013 (2022).
Volkow, N. D., Koob, G. & Baler, R. Biomarkers in substance use disorders. ACS Chem. Neurosci. 6, 522–525 (2015).
Kwako, L. E., Bickel, W. K. & Goldman, D. Addiction biomarkers: dimensional approaches to understanding addiction. Trends Mol. Med. 24, 121–128 (2018).
Orru, G., Pettersson-Yeo, W., Marquand, A. F., Sartori, G. & Mechelli, A. Using support vector machine to identify imaging biomarkers of neurological and psychiatric disease: a critical review. Neurosci. Biobehav. Rev. 36, 1140–1152 (2012).
Pando-Naude, V. et al. Gray and white matter morphology in substance use disorders: a neuroimaging systematic review and meta-analysis. Transl. Psychiatry 11, 29 (2021).
Wilcox, C. E., Teshiba, T. M., Merideth, F., Ling, J. & Mayer, A. R. Enhanced cue reactivity and fronto-striatal functional connectivity in cocaine use disorders. Drug Alcohol Depend. 115, 137–144 (2011).
Seminowicz, D. A. et al. Effective treatment of chronic low back pain in humans reverses abnormal brain anatomy and function. J. Neurosci. 31, 7540–7550 (2011).
Rowe, J. B. Connectivity analysis is essential to understand neurological disorders. Front. Syst. Neurosci. 4, 144 (2010).
Yang, Y. et al. Functional connectivity and structural changes of thalamic subregions in episodic migraine. J. Headache Pain 23, 119 (2022).
Rowe, J. B. et al. Is the prefrontal cortex necessary for establishing cognitive sets? J. Neurosci. 27, 13303–13310 (2007).
Kelly, C. et al. Reduced interhemispheric resting state functional connectivity in cocaine addiction. Biol. Psychiatry 69, 684–692 (2011).
Sakoglu, U. et al. Classification of cocaine-dependent participants with dynamic functional connectivity from functional magnetic resonance imaging data. J. Neurosci. Res. 97, 790–803 (2019).
McHugh, M. J., Gu, H., Yang, Y., Adinoff, B. & Stein, E. A. Executive control network connectivity strength protects against relapse to cocaine use. Addict. Biol. 22, 1790–1801 (2017).
Worhunsky, P. D. et al. Functional brain networks associated with cognitive control, cocaine dependence, and treatment outcome. Psychol. Addict. Behav. 27, 477–488 (2013).
Hobkirk, A. L., Bell, R. P., Utevsky, A. V., Huettel, S. & Meade, C. S. Reward and executive control network resting-state functional connectivity is associated with impulsivity during reward-based decision making for cocaine users. Drug Alcohol Depend. 194, 32–39 (2019).
Rashid, B. & Calhoun, V. Towards a brain-based predictome of mental illness. Hum. Brain Mapp. 41, 3468–3535 (2020).
Wang, J. M. et al. In cocaine dependence, neural prediction errors during loss avoidance are increased with cocaine deprivation and predict drug use. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 4, 291–299 (2019).
Collins, G. S., Ogundimu, E. O. & Altman, D. G. Sample size considerations for the external validation of a multivariable prognostic model: a resampling study. Stat. Med. 35, 214–226 (2016).
Abi-Dargham, A. & Horga, G. The search for imaging biomarkers in psychiatric disorders. Nat. Med. 22, 1248–1255 (2016).
Demjaha, A., Murray, R. M., McGuire, P. K., Kapur, S. & Howes, O. D. Dopamine synthesis capacity in patients with treatment-resistant schizophrenia. Am. J. Psychiatry 169, 1203–1210 (2012).
Liston, C. et al. Default mode network mechanisms of transcranial magnetic stimulation in depression. Biol. Psychiatry 76, 517–526 (2014).
Posner, J. et al. Increased default mode network connectivity in individuals at high familial risk for depression. Neuropsychopharmacology 41, 1759–1767 (2016).
Coles, A. S., Kozak, K. & George, T. P. A review of brain stimulation methods to treat substance use disorders. Am. J. Addict. 27, 71–91 (2018).
Torres-Castano, A. et al. Transcranial magnetic stimulation for the treatment of cocaine addiction: a systematic review. J. Clin. Med. 10, 5595 (2021).
Garza-Villarreal, E. A. et al. Clinical and functional connectivity outcomes of 5-Hz repetitive transcranial magnetic stimulation as an add-on treatment in cocaine use disorder: a double-blind randomized controlled trial. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 6, 745–757 (2021).
Martinotti, G. et al. Repetitive transcranial magnetic stimulation in treatment-seeking subjects with cocaine use disorder: a randomized, double-blind, sham-controlled trial. Prog. Neuropsychopharmacol. Biol. Psychiatry 116, 110513 (2022).
Sagi, O. & Rokach, L. Ensemble learning: a survey. Wiley Interdiscip. Rev. Data Min. Knowl. Discov. 8, e1249 (2018).
Stanford, M. S. et al. Fifty years of the Barratt Impulsiveness Scale: an update and review. Pers. Individ. Dif. 47, 385–395 (2009).
Parker, L. A. et al. Diagnostic biomarkers: are we moving from discovery to clinical application? Clin. Chem. 64, 1657–1667 (2018).
Yang, L., Du, Y., Yang, W. & Liu, J. Machine learning with neuroimaging biomarkers: application in the diagnosis and prediction of drug addiction. Addict. Biol. 28, e13267 (2023).
Schaefer, A. et al. Local-global parcellation of the human cerebral cortex from intrinsic functional connectivity MRI. Cereb. Cortex 28, 3095–3114 (2018).
Yeo, B. T. et al. The organization of the human cerebral cortex estimated by intrinsic functional connectivity. J. Neurophysiol. 106, 1125–1165 (2011).
Üstün, T. B., Kostanjsek, N., Chatterji, S. & Rehm, J. Measuring Health and Disability: Manual for WHO Disability Assessment Schedule WHODAS 2.0 (World Health Organization, 2010).
Derogatis, L. R. & Unger, R. in The Corsini Encyclopedia of Psychology (John Wiley and Sons, 2010) https://doi.org/10.1002/9780470479216.corpsy0970
Cowley, D. S. Alcohol abuse, substance abuse, and panic disorder. Am. J. Med. 92, S41–S48 (1992).
Castillo-Carniglia, A., Keyes, K. M., Hasin, D. S. & Cerda, M. Psychiatric comorbidities in alcohol use disorder. Lancet Psychiatry 6, 1068–1080 (2019).
Evans-Polce, R. J., Kcomt, L., Veliz, P. T., Boyd, C. J. & McCabe, S. E. Alcohol, tobacco, and comorbid psychiatric disorders and associations with sexual identity and stress-related correlates. Am. J. Psychiatry 177, 1073–1081 (2020).
Tipping, M. E. Sparse Bayesian learning and the relevance vector machine. J. Mach. Learn. Res. 1, 211–244 (2001).
Rasgado-Toledo, J., Issa-Garcia, V., Alcala-Lozano, R., Garza-Villarreal, E. A. & Gonzalez-Escamilla, G. Cortical and subcortical connections change after repetitive transcranial magnetic stimulation therapy in cocaine use disorder and predict clinical outcome. Preprint at medRxiv https://doi.org/10.1101/2022.09.29.22280253 (2022).
Tiffany, S. T., Singleton, E., Haertzen, C. A. & Henningfield, J. E. The development of a cocaine craving questionnaire. Drug Alcohol Depend. 34, 19–28 (1993).
Wang, W., Zhornitsky, S., Zhang, S. & Li, C. R. Noradrenergic correlates of chronic cocaine craving: neuromelanin and functional brain imaging. Neuropsychopharmacology 46, 851–859 (2021).
Zhornitsky, S. et al. Reward-related responses and ronic craving in cocaine addiction: an imaging study of the monetary incentive delay task. Int. J. Neuropsychopharmacol. 24, 634–644 (2021).
Camchong, J. et al. Frontal hyperconnectivity related to discounting and reversal learning in cocaine subjects. Biol. Psychiatry 69, 1117–1123 (2011).
Cisler, J. M. et al. Altered functional connectivity of the insular cortex across prefrontal networks in cocaine addiction. Psychiatry Res. 213, 39–46 (2013).
Contreras-Rodriguez, O. et al. Increased corticolimbic connectivity in cocaine dependence versus pathological gambling is associated with drug severity and emotion-related impulsivity. Addict. Biol. 21, 709–718 (2016).
Bell, R. P., Foxe, J. J., Ross, L. A. & Garavan, H. Intact inhibitory control processes in abstinent drug abusers (I): a functional neuroimaging study in former cocaine addicts. Neuropharmacology 82, 143–150 (2014).
Dalley, J. W. et al. Nucleus accumbens D2/3 receptors predict trait impulsivity and cocaine reinforcement. Science 315, 1267–1270 (2007).
Aron, A. R., Robbins, T. W. & Poldrack, R. A. Inhibition and the right inferior frontal cortex. Trends Cogn. Sci. 8, 170–177 (2004).
Lim, K. O., Choi, S. J., Pomara, N., Wolkin, A. & Rotrosen, J. P. Reduced frontal white matter integrity in cocaine dependence: a controlled diffusion tensor imaging study. Biol. Psychiatry 51, 890–895 (2002).
Prisciandaro, J. J. et al. The relationship between years of cocaine use and brain activation to cocaine and response inhibition cues. Addiction 109, 2062–2070 (2014).
Menon, V. Large-scale brain networks and psychopathology: a unifying triple network model. Trends Cogn. Sci. 15, 483–506 (2011).
Geng, X. et al. Salience and default mode network dysregulation in chronic cocaine users predict treatment outcome. Brain 140, 1513–1524 (2017).
Kosten, T. R. et al. Cue-induced brain activity changes and relapse in cocaine-dependent patients. Neuropsychopharmacology 31, 644–650 (2006).
Sevinc, G. & Spreng, R. N. Contextual and perceptual brain processes underlying moral cognition: a quantitative meta-analysis of moral reasoning and moral emotions. PLoS One 9, e87427 (2014).
Gong, J. et al. Disrupted functional connectivity within the default mode network and salience network in unmedicated bipolar II disorder. Prog. Neuropsychopharmacol. Biol. Psychiatry 88, 11–18 (2019).
Herlin, B., Navarro, V. & Dupont, S. The temporal pole: from anatomy to function-a literature appraisal. J. Chem. Neuroanat. 113, 101925 (2021).
Castaneda, A. E., Tuulio-Henriksson, A., Marttunen, M., Suvisaari, J. & Lonnqvist, J. A review on cognitive impairments in depressive and anxiety disorders with a focus on young adults. J. Affect. Disord. 106, 1–27 (2008).
Zhou, Y. et al. Default-mode network disruption in mild traumatic brain injury. Radiology 265, 882 (2012).
Vonmoos, M. et al. Cognitive dysfunctions in recreational and dependent cocaine users: role of attention-deficit hyperactivity disorder, craving and early age at onset. Br. J. Psychiatry 203, 35–43 (2013).
Luthi, A. & Luscher, C. Pathological circuit function underlying addiction and anxiety disorders. Nat. Neurosci. 17, 1635–1643 (2014).
Mackey, S. et al. Mega-analysis of gray matter volume in substance dependence: general and substance-specific regional effects. Am. J. Psychiatry 176, 119–128 (2019).
Kaag, A. M. et al. The relation between gray matter volume and the use of alcohol, tobacco, cocaine and cannabis in male polysubstance users. Drug Alcohol Depend. 187, 186–194 (2018).
Meyerhoff, D. J. Structural neuroimaging in polysubstance users. Curr. Opin. Behav. Sci. 13, 13–18 (2017).
Minnes, S., Lang, A. & Singer, L. Prenatal tobacco, marijuana, stimulant, and opiate exposure: outcomes and practice implications. Addict. Sci. Clin. Pract. 6, 57–70 (2011).
Li, J., Chen, J., Kong, W., Li, X. & Hu, B. Abnormal core functional connectivity on the pathology of MDD and antidepressant treatment: a systematic review. J. Affect. Disord. 296, 622–634 (2022).
Volkow, N. D., Michaelides, M. & Baler, R. The neuroscience of drug reward and addiction. Physiol. Rev. 99, 2115–2140 (2019).
Delli Pizzi, S. et al. GABA content within the ventromedial prefrontal cortex is related to trait anxiety. Soc. Cogn. Affect. Neurosci. 11, 758–766 (2016).
Hanlon, C. A. et al. Developing repetitive transcranial magnetic stimulation (rTMS) as a treatment tool for cocaine use disorder: a series of six translational studies. Curr. Behav. Neurosci. Rep. 4, 341–352 (2017).
Gorelick, D. A., Zangen, A. & George, M. S. Transcranial magnetic stimulation in the treatment of substance addiction. Ann. N. Y. Acad. Sci. 1327, 79–93 (2014).
Mehta, D. D. et al. A systematic review and meta-analysis of neuromodulation therapies for substance use disorders. Neuropsychopharmacology https://doi.org/10.1038/s41386-023-01776-0 (2023).
Zhao, D. et al. Deep magnetic stimulation targeting the medial prefrontal and anterior cingulate cortices for methamphetamine use disorder: a randomised, double-blind, sham-controlled study. Gen. Psychiatr. 36, e101149 (2023).
Martinez, D. et al. Transcranial magnetic stimulation of medial prefrontal and cingulate cortices reduces cocaine self-administration: a pilot study. Front. Psychiatry 9, 80 (2018).
Harel, M. et al. Repetitive transcranial magnetic stimulation in alcohol dependence: a randomized, double-blind, sham-controlled proof-of-concept trial targeting the medial prefrontal and anterior cingulate cortices. Biol. Psychiatry 91, 1061–1069 (2022).
Cho, S. S. & Strafella, A. P. rTMS of the left dorsolateral prefrontal cortex modulates dopamine release in the ipsilateral anterior cingulate cortex and orbitofrontal cortex. PLoS One 4, e6725 (2009).
Hanlon, C. A., Dowdle, L. T. & Henderson, J. S. Modulating neural circuits with transcranial magnetic stimulation: implications for addiction treatment development. Pharmacol. Rev. 70, 661–683 (2018).
Bickel, W. K., Snider, S. E., Quisenberry, A. J., Stein, J. S. & Hanlon, C. A. Competing neurobehavioral decision systems theory of cocaine addiction: from mechanisms to therapeutic opportunities. Prog. Brain Res. 223, 269–293 (2016).
McHugh, R. K., Votaw, V. R., Sugarman, D. E. & Greenfield, S. F. Sex and gender differences in substance use disorders. Clin. Psychol. Rev. 66, 12–23 (2018).
Siddiqi, S. H., Kording, K. P., Parvizi, J. & Fox, M. D. Causal mapping of human brain function. Nat. Rev. Neurosci. 23, 361–375 (2022).
Joutsa, J. et al. Brain lesions disrupting addiction map to a common human brain circuit. Nat. Med. 28, 1249–1255 (2022).
Goldstein, R. Z. et al. Oral methylphenidate normalizes cingulate activity in cocaine addiction during a salient cognitive task. Proc. Natl Acad. Sci. USA 107, 16667–16672 (2010).
Goldstein, R. Z. et al. Anterior cingulate cortex hypoactivations to an emotionally salient task in cocaine addiction. Proc. Natl Acad. Sci. USA 106, 9453–9458 (2009).
Angeles-Valdez, D. et al. The Mexican magnetic resonance imaging dataset of patients with cocaine use disorder: SUDMEX CONN. Sci Data 9, 133 (2022).
Sheehan, D. V. et al. The Mini-International Neuropsychiatric Interview (MINI): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J. Clin. Psychiatry 59, 22–33 (1998).
Black, D. W. & Grant, J. E. DSM-5 Guidebook: The Essential Companion to the Diagnostic and Statistical Manual of Mental Disorders (American Psychiatric Association, 2014).
Medición de la salud y la discapacidad: manual para el cuestionario de evaluación de la discapacidad de la OMS: WHODAS 2.0 (World Health Organization, Servicio Nacional de Rehabilitación, 2015).
Oquendo, M. A. et al. Spanish adaptation of the Barratt impulsiveness scale (BIS-11). Eur. J. Psychiatry 15, 147–155 (2001).
Esteban, O. et al. fMRIPrep: a robust preprocessing pipeline for functional MRI. Nat. Methods 16, 111–116 (2019).
Avants, B. B., Epstein, C. L., Grossman, M. & Gee, J. C. Symmetric diffeomorphic image registration with cross-correlation: evaluating automated labeling of elderly and neurodegenerative brain. Med. Image Anal. 12, 26–41 (2008).
Zhang, Y., Brady, J. M. & Smith, S. Hidden Markov random field model for segmentation of brain MR image. In Medical Imaging 2000: Image Processing Vol. 3979, 1126–1137 (Society of Photo-Optical Instrumentation Engineers, 2000).
Greve, D. N. & Fischl, B. Accurate and robust brain image alignment using boundary-based registration. Neuroimage 48, 63–72 (2009).
Jenkinson, M., Bannister, P., Brady, M. & Smith, S. Improved optimization for the robust and accurate linear registration and motion correction of brain images. Neuroimage 17, 825–841 (2002).
Pruim, R. H. R. et al. ICA-AROMA: a robust ICA-based strategy for removing motion artifacts from fMRI data. Neuroimage 112, 267–277 (2015).
Power, J. D., Barnes, K. A., Snyder, A. Z., Schlaggar, B. L. & Petersen, S. E. Steps toward optimizing motion artifact removal in functional connectivity MRI; a reply to Carp. Neuroimage 76, 439–441 (2013).
Zhao, K. et al. Individualized fMRI connectivity defines signatures of antidepressant and placebo responses in major depression. Mol. Psychiatry 28, 2490–2499 (2023).
Yu, M. et al. Statistical harmonization corrects site effects in functional connectivity measurements from multi-site fMRI data. Hum. Brain Mapp. 39, 4213–4227 (2018).
Poldrack, R. A. et al. A phenome-wide examination of neural and cognitive function. Sci. Data 3, 160110 (2016).
Sharma, U. C. et al. Modified GAN augmentation algorithms for the MRI-classification of myocardial scar tissue in ischemic cardiomyopathy. Front. Cardiovasc. Med. 8, 726943 (2021).
Tang, T., Jiao, D., Chen, T. & Gui, G. Medium- and long-term precipitation forecasting method based on data augmentation and machine learning algorithms. IEEE J. Sel. Top. Appl. Earth Obs. Remote Sens. 15, 1000–1011 (2022).
Iwana, B. K. & Uchida, S. An empirical survey of data augmentation for time series classification with neural networks. PLoS One 16, e0254841 (2021).
Van Dijk, K. R. et al. Intrinsic functional connectivity as a tool for human connectomics: theory, properties, and optimization. J. Neurophysiol. 103, 297–321 (2010).
Acknowledgements
This work was supported in part by National Institutes of Health grants R01MH129694, R21MH130956 and R21AG080425 to Y.Z.; R01MH132784 and K23MH114023 to G.A.F.; DP1MH116506 and R44MH123373 to A.E.; it was also supported by an Alzheimer’s Association grant (AARG-22-972541) as well as Lehigh University Faculty Innovation Grant (FIGAWD35), CORE and Accelerator grants to Y.Z. Portions of this research were conducted on Lehigh University’s Research Computing infrastructure partially supported by National Science Foundation Award 2019035. This work was also supported in part by philanthropic funding and grants from the One Mind Baszucki Brain Research Fund, the SEAL Future Foundation and the Brain and Behavior Research Foundation to G.A.F.
Author information
Authors and Affiliations
Contributions
K.Z. conceptualized and designed the work, performed data analysis and result interpretation and drafted and revised the manuscript. H.X., G.A.F., N.B.C. and A.E. interpreted the data, refined the design of the work and revised the manuscript. D.J.O. and C.J.K. interpreted the data and revised the manuscript. E.A.G.-V. collected the clinical trial data, interpreted the data, refined the design of the work and revised the manuscript. Y.Z. conceptualized and designed the work, oversaw the analysis and interpretation of data and revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
G.A.F. received monetary compensation for consulting work for SynapseBio AI and owns equity in Alto Neuroscience. A.E. reports salary and equity from Alto Neuroscience and holds equity in Akili Interactive and Mindstrong Health. The other authors declare no competing interests.
Peer review
Peer review information
Nature Mental Health thanks James Mahoney, Abraham Zangen and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Statistical difference in the classifier-identified discriminative FCs between CUD patients and healthy controls, examined by two-sample t-tests.
a, All significant hyperconnections (CUD > healthy controls). b, All significant hypoconnections (CUD < healthy controls). Histogram indicated the node strength calculated from the sum of the linked FC importance. VIS, visual network; SMN, somatomotor network; DAN, dorsal attention network; VAN, ventral attention network; LIM, limbic network; FPC, frontoparietal control network; DMN, default mode network. Only the significant t values that survived FDR were shown.
Extended Data Fig. 2 Visualization of the CUD-discriminative FCs involved in active repetitive transcranial magnetic stimulation treatment response specific prediction.
a, The rTMS predictive FC signature. b, We grouped the importance of predictive FCs into the seven typical networks including visual network (VIS), somatomotor network (SMN), dorsal attention network (DAN), ventral attention network (VAN), limbic network (LIM), frontoparietal control network (FPC), and default mode network (DMN).
Extended Data Fig. 3 Association between the discriminative FCs and treatment-outcome-predictive FCs.
a,b, Correlation between the top 2 active rTMS treatment response predictive FCs and active rTMS VAS score change. These two FCs were between the orbitofrontalcortex and anterior cingulate cortex, and between the middle temporal cortex and superior orbitofrontalcortex. Error bars = s.e.m. represent the Pearson’s correlation between FCs and VAS score changes based on the two-sided test against the alternative hypothesis that r ≠ 0. c,d, Correlation between these two FCs and sham rTMS VAS score change. e,f, These two FCs distribution between CUD and HC in the discovery and independent cohorts. The difference of FCs were comfirmed by independent samples t-test (two-sided test against the alternative hypothesis that t ≠ 0). The data in discovery cohort was augmented twice. These two FCs were significantly and specifically correlated to the VAS score change and significantly different between CUD and HC. The boxplots show the interquartile range (IQR; first quartile, 25th percentile; third quartile, 75th percentile), and the whiskers indicate Q1 − (1.5 × IQR) or Q3 + (1.5 × IQR). The line within the boxplot represents the median. The sample sizes for all panels were n = 213 (augmented FC from 71 subjects) for the CUD in discovery cohort, n = 174 (augmented FC from 58 subjects) for the HC in discovery cohort, n = 82 for the CUD in independent cohort, n = 81 for the HC in independent cohort. g, Venn diagram indicating the association between discriminative and abnormal FCs (551) with active rTMS treatment outcome. Discriminative atypical FCs were defined as the discriminative FCs identified by our classification models and the significantly atypical FCs detected by two-sample t-tests (two-sided test against the alternative hypothesis that t ≠ 0) comparing CUD and HC subjects, with those surviving FDR correction (pfdr < 0.05). The number of discriminative atypical FCs was equal to the sum of hyperconnections and hypoconnections. Deeper bluer shading indicates larger treatment predictive weights. The red numbers in the red rectangle represent the overlapping numbers between the top 100 treatment predictive FCs and all discriminative atypical FCs in descending order.
Extended Data Fig. 4 Illustration of our proposed analytical framework.
a, Region of interests (ROIs) level time series were extracted from fMRI BOLD signals based on the Schaefer atlas. Functional connectivity was calculated by Pearson’s correlation in time series between any pair of ROIs. b, The functional connectivity features were used to train the XGBoost model to classify the subjects into CUD patients or healthy controls on discovery cohort. The performance was cross-validated. Obtained diagnostic (discriminative) pattern was applied directly on the independent cohort to demonstrate the generalizability of its diagnostic power. c, Utilizing discriminative pattern as a mask to select the discriminative functional connectivity (FC) features from rTMS dataset, a relevance vector machine (RVM) model was employed to predict changes in visual analog scale (VAS) scores for patients undergoing repetitive transcranial magnetic stimulation (rTMS) treatment.
Supplementary information
Supplementary Information
Supplementary Figs. 1–12 and Tables 1–8.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhao, K., Fonzo, G.A., Xie, H. et al. Discriminative functional connectivity signature of cocaine use disorder links to rTMS treatment response. Nat. Mental Health 2, 388–400 (2024). https://doi.org/10.1038/s44220-024-00209-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s44220-024-00209-1