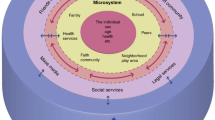
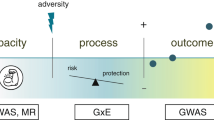
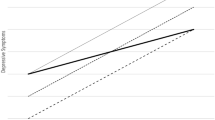
Abstract
Most studies on the relationship between childhood adversity and negative outcomes across the lifespan have focused on individual exposures or outcomes—an approach that has been limited in its ability to elucidate mechanisms or causality. We propose a new framework for examining the relationship between childhood adversity and negative outcomes—the bio-exposome. In this model, we aim to understand the interconnections between every aspect of biology and the exposome, and the way disparate biological and exposome factors shape, and are shaped by, one another. Once we understand when, in which contexts and towards whom stress calibration interventions should be targeted, through examination of the bio-exposome, we could facilitate prevention of some of the major causes of morbidity across the lifespan. To examine the bio-exposome, we offer a new research agenda that embraces complexity science, large datasets and collaboration across a wide range of scientific disciplines.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$59.00 per year
only $4.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Lambert, H. K., Meza, R., Martin, P., Fearey, E. & McLaughlin K. A. in Evidence-Based Treatments for Trauma Related Disorders in Children and Adolescents (eds. Landolt, M. A. et al.) 49–66 (Springer, 2017).
McLaughlin, K. A. & Sheridan, M. A. Beyond cumulative risk: a dimensional approach to childhood adversity. Curr. Dir. Psychol. Sci. 25, 239–245 (2016).
Keenan, K., Hipwell, A. E., Class, Q. A. & Mbayiwa, K. Extending the developmental origins of disease model: impact of preconception stress exposure on offspring neurodevelopment. Dev. Psychobiol. 60, 753–764 (2018).
Combet, E. & Buckton, C. Micronutrient deficiencies, vitamin pills and nutritional supplements. Medicine 43, 66–72 (2015).
Kaniasty, K. Social support, interpersonal, and community dynamics following disasters caused by natural hazards. Curr. Opin. Psychol. 32, 105–109 (2020).
Lacey, R. E., Howe, L. D., Kelly-Irving, M., Bartley, M. & Kelly, Y. The clustering of adverse childhood experiences in the Avon Longitudinal Study of Parents and Children: are gender and poverty important? J. Interpers. Violence 37, 2218–2241 (2020).
Penkler, M., Hanson, M., Biesma, R. & Müller, R. DOHaD in science and society: emergent opportunities and novel responsibilities. J. Dev. Origins Health Dis. 10, 268–273 (2019).
Jazwiec, P. A. & Sloboda, D. M. Nutritional adversity, sex and reproduction: 30 years of DOHaD and what have we learned? J. Endocrinol. 242, T51–T68 (2019).
Merz, M. P. & Turner, J. D. Is early life adversity a trigger towards inflammageing? Exp. Gerontol. 150, 111377 (2021).
McEwen, C. A. Connecting the biology of stress, allostatic load and epigenetics to social structures and processes. Neurobiol. Stress 17, 100426 (2022).
Reid, B. & Danese, A. Challenges in researching the immune pathways between early life adversity and psychopathology. Dev. Psychopathol. 32, 1597–1624 (2020).
McEwen, B. S. Stress, adaptation, and disease. Allostasis and allostatic load. Ann. NY Acad. Sci. 840, 33–44 (1998).
Ellis, B. J. & Del Giudice, M. Developmental adaptation to stress: an evolutionary perspective. Annu. Rev. Psychol. 70, 111–139 (2019).
Shiels, P. G. et al. Manipulating the exposome to enable better ageing. Biochem. J. 478, 2889–2898 (2021).
McEwen, B. S. & Stellar, E. Stress and the individual: mechanisms leading to disease. Arch. Intern. Med. 153, 2093–2101 (1993).
Del Giudice, M., Ellis, B. J. & Shirtcliff, E. A. The adaptive calibration model of stress responsivity. Neurosci. Biobehav. Rev. 35, 1562–1592 (2011).
Scheeringa, M. S. Reexamination of diathesis stress and neurotoxic stress theories: A qualitative review of pre-trauma neurobiology in relation to posttraumatic stress symptoms. Int. J. Methods Psychiatr. Res. 30, e1864 (2021).
McCrory, E. J. & Viding, E. The theory of latent vulnerability: reconceptualizing the link between childhood maltreatment and psychiatric disorder. Dev. Psychopathol. 27, 493–505 (2015).
Lahti-Pulkkinen, M., Räikkönen, K., Bhattacharya, S. & Reynolds, R. M. Maternal body mass index in pregnancy and mental disorders in adult offspring: a record linkage study in Aberdeen, Scotland. Sci. Rep. 11, 15132 (2021).
McKerracher, L., Moffat, T., Barker, M., Williams, D. & Sloboda, D. M. Translating the developmental origins of health and disease concept to improve the nutritional environment for our next generations: a call for a reflexive, positive, multi-level approach. J. Dev. Origins Health Dis. 10, 420–428 (2019).
Lang, J. et al. The hallmarks of childhood abuse and neglect: a systematic review. PLoS ONE 15, e0243639 (2020).
Shiels, P. G., Buchanan, S., Selman, C. & Stenvinkel, P. Allostatic load and ageing: linking the microbiome and nutrition with age-related health. Biochem. Soc. Trans. 47, 1165–1172 (2019).
Geronimus, A. T. et al. Weathering in Detroit: Place, race, ethnicity, and poverty as conceptually fluctuating social constructs shaping variation in allostatic load. Milbank Q. 98, 1171–1218 (2020).
Sandifer, P. A., Juster, R.-P., Seeman, T. E., Lichtveld, M. Y. & Singer, B. H. Allostatic load in the context of disasters. Psychoneuroendocrinology 140, 105725 (2022).
Leimert, K. B. & Olson, D. M. Racial disparities in pregnancy outcomes: genetics, epigenetics, and allostatic load. Curr. Opin. Physiol. 13, 155–165 (2020).
Wild, C. P. Complementing the genome with an ‘exposome’: the outstanding challenge of environmental exposure measurement in molecular epidemiology. Cancer Epidemiol. Biomarkers Prev. 14, 1847–1850 (2005).
Engel, G. L. The need for a new medical model: a challenge for biomedicine. Science 196, 129–136 (1977).
van Wietmarschen, H. A., Wortelboer, H. M. & van der Greef, J. Grip on health: a complex systems approach to transform health care. J. Eval. Clin. Pract. 24, 269–277 (2018).
Norris, S. A. et al. Nutrition in adolescent growth and development. Lancet 399, 172–184 (2022).
Krakau, L. et al. Personality functioning as a mediator of adult mental health following child maltreatment. J. Affect. Disord. 291, 126–134 (2021).
Skinner, E. & Zimmer-Gembeck, M. The Development of Coping (Springer, 2016).
Ioannidis, K., Askelund, A. D., Kievit, R. A. & van Harmelen, A.-L. The complex neurobiology of resilient functioning after childhood maltreatment. BMC Med. 18, 32 (2020).
Cain, D. W. & Cidlowski, J. A. Immune regulation by glucocorticoids. Nat. Rev. Immunol. 17, 233 (2017).
Shiels, P. G., McGuinness, D., Eriksson, M., Kooman, J. P. & Stenvinkel, P. The role of epigenetics in renal ageing. Nat. Rev. Nephrol. 13, 471 (2017).
Craig J. M., Sutton E. K. & Jahanfar S. in Developmental and Fetal Origins of Differences in Monozygotic Twins From Genetics to Environmental Factors (eds. Matias, A. & Blickstein. I) 320–343 (Academic Press, 2020).
Bronfenbrenner, U. in International Encyclopaedia of Education 1643–1647 (Elsevier, 1994).
Bronfenbrenner, U. The Ecology of Human Development: Experiments by Nature and Design (Harvard Univ. Press, 1979).
Teicher M. H. & Samson J. A. Enduring neurobiological effects of childhood abuse and neglect. J. Child Psychol. Psychiatry 57, 241–266 (2016).
Colich, N. L., Rosen, M. L., Williams, E. S. & McLaughlin, K. A. Biological aging in childhood and adolescence following experiences of threat and deprivation: a systematic review and meta-analysis. Psychol. Bull. 146, 721–764 (2020).
Young-Southward, G., Svelnys, C., Gajwani, R., Bosquet Enlow, M. & Minnis, H. Child maltreatment, autonomic nervous system responsivity, and psychopathology: current state of the literature and future directions. Child Maltreat. 25, 3–19 (2020).
Danese, A. & Baldwin, J. R. Hidden wounds? Inflammatory links between childhood trauma and psychopathology. Annu. Rev. Psychol. 68, 517–544 (2017).
Hawton, K., Saunders, K. E. & O’Connor, R. C. Self-harm and suicide in adolescents. Lancet 379, 2373–2382 (2012).
van Harmelen, A.-L. et al. Friendships and family support reduce subsequent depressive symptoms in at-risk adolescents. PLoS ONE 11, e0153715 (2016).
Van Harmelen, A.-L., Blakemore, S.-J., Goodyer, I. & Kievit, R. The interplay between adolescent friendship quality and resilient functioning following childhood and adolescent adversity. Advers. Resil. Sci. 2, 37–50 (2021).
Reijman, S. et al. Salivary α-amylase reactivity to infant crying in maltreating mothers. Child Psychiatry Hum. Dev. 46, 589–599 (2015).
Rizeq, J. et al. Vulnerability pathways to mental health outcomes in children and parents during COVID-19. Curr. Psychol. 42, 17348–17358 (2023).
Nelles-McGee, T., Khoury, J., Kenny, M., Joshi, D. & Gonzalez, A. Biological embedding of child maltreatment: a systematic review of biomarkers and resilience in children and youth. Psychol. Trauma 14, S50–S62 (2022).
Scheuplein, M. & Van Harmelen, A.-L. The importance of friendships in reducing brain responses to stress in adolescents exposed to childhood adversity: a pre-registered systematic review. Curr. Opin. Psychol 45, 101310 (2022).
König, M. et al. The stress-buffering role of friendships in young people with childhood threat experiences: a preliminary report. Eur. J. Psychotraumatol. 14, 2281971 (2023).
Liao, J. et al. Residential exposure to green space and early childhood neurodevelopment. Env. Int. 128, 70–76 (2019).
Molnar, B. E. et al. Neighborhood-level social processes and substantiated cases of child maltreatment. Child Abuse Neglect 51, 41–53 (2016).
Ellaway, A., Dundas, R., Robertson, T. & Shiels, P. G. More miles on the clock: neighbourhood stressors are associated with telomere length in a longitudinal study. PLoS ONE 14, e0214380 (2019).
Betancourt, T. S. et al. Psychosocial adjustment and mental health in former child soldiers–a systematic review of the literature and recommendations for future research. J. Child Psychol. Psychiatry 54, 17–36 (2013).
Adhikari, R. P. et al. Protective and risk factors of psychosocial wellbeing related to the reintegration of former child soldiers in Nepal. Intervention 12, 367–378 (2014).
Vermeulen, R., Schymanski, E. L., Barabási, A.-L. & Miller, G. W. The exposome and health: where chemistry meets biology. Science 367, 392–396 (2020).
Rupia, E. J., Binning, S. A., Roche, D. G. & Lu, W. Fight-flight or freeze-hide? Personality and metabolic phenotype mediate physiological defence responses in flatfish. J. Animal Ecol. 85, 927–937 (2016).
Robles, T. F. Social relationships and the immune system during development. J. Child Psychol. Psychiatry 62, 539–559 (2021).
Finlay, S. et al. Adverse childhood experiences and allostatic load: a systematic review. Neurosci. Biobehav. Rev. 136, 104605 (2022).
Krieger, N. & Davey Smith, G. The tale wagged by the DAG: broadening the scope of causal inference and explanation for epidemiology. Int. J. Epidemiol. 45, 1787–1808 (2016).
Ellis, B. J. et al. Hidden talents in harsh environments. Dev. Psychopathol. 34, 95–113 (2022).
Shakiba, N., Ellis, B. J., Bush, N. R. & Boyce, W. T. Biological sensitivity to context: a test of the hypothesized U-shaped relation between early adversity and stress responsivity. Dev. Psychopathol. 32, 641–660 (2020).
van de Ven, P. The journey of sensemaking and identity construction in the aftermath of trauma: peer support as a vehicle for coconstruction. J. Community Psychol. 48, 1825–1839 (2020).
Dai, L., Schurgers, L., Shiels, P. G. & Stenvinkel, P. A biomimetic natural sciences approach to understanding the mechanisms of ageing in burden of lifestyle diseases. Clin. Sci. 135, 1251–1272 (2021).
Ziker, J. P. & Snopkowski, K. Life-history factors influence teenagers’ suicidal ideation: a model selection analysis of the canadian national longitudinal survey of children and youth. Evol. Psychol. 18, 1474704920939521 (2020).
Rizeq, J. & McCann, D. The cognitive, emotional, and behavioral sequelae of trauma exposure: An integrative approach to examining trauma’s effect. Psychol. Trauma 15, 313–321 (2023).
Turecki, G. & Meaney, M. J. Effects of the social environment and stress on glucocorticoid receptor gene methylation: a systematic review. Biol. Psychiatry 79, 87–96 (2016).
Lang, J. et al. Adverse childhood experiences, epigenetics and telomere length variation in childhood and beyond: a systematic review of the literature. Eur. Child Adolesc. Psychiatry 29, 1329–1338 (2020).
Soares, S., Rocha, V., Kelly-Irving, M., Stringhini, S. & Fraga, S. Adverse childhood events and health biomarkers: a systematic review. Front. Public Health 9, 649825 (2021).
Koenig, A. M. et al. Intergenerational gene × environment interaction of FKBP5 and childhood maltreatment on hair steroids. Psychoneuroendocrinology 92, 103–112 (2018).
Womersley, J. S. et al. Childhood trauma, the stress response and metabolic syndrome: a focus on DNA methylation. Eur. J. Neurosci. 55, 2253–2296 (2022).
Craven, H. et al. Socioeconomic position links circulatory microbiota differences with biological age. Sci. Rep. 11, 12629 (2021).
Jennings, J. R., Pardini, D. A. & Matthews, K. A. Heart rate, health, and hurtful behavior. Psychophysiology 54, 399–408 (2017).
Saha, S. K. et al. Correlation between oxidative stress, nutrition, and cancer initiation. Int. J. Mol. Sci. 18, 1544 (2017).
Shalev, A. et al. Cortisol response to stress as a predictor for suicidal ideation in youth. J. Affect. Disord. 257, 10–16 (2019).
Norman, R. E. et al. The long-term health consequences of child physical abuse, emotional abuse, and neglect: a systematic review and meta-analysis. PLoS Med. 9, e1001349 (2012).
Walker, S. P., Chang, S. M., Vera-Hernández, M. & Grantham-McGregor, S. Early childhood stimulation benefits adult competence and reduces violent behavior. Pediatrics 127, 849–857 (2011).
Gu, J., Strauss, C., Bond, R. & Cavanagh, K. How do mindfulness-based cognitive therapy and mindfulness-based stress reduction improve mental health and wellbeing? A systematic review and meta-analysis of mediation studies. Clin. Psychol. Rev. 37, 1–12 (2015).
Lacey, R. E. & Minnis, H. Twenty years of research with adverse childhood experience scores–Advantages, disadvantages and applications to practice. J. Child Psychol. Psychiatry 61, 116–130 (2020).
Gerin, M. I., Hanson, E., Viding, E. & McCrory, E. J. A review of childhood maltreatment, latent vulnerability and the brain: implications for clinical practice and prevention. Adopt. Foster. 43, 310–328 (2019).
Belsky, J. The differential susceptibility hypothesis: sensitivity to the environment for better and for worse. JAMA Pediatr. 170, 321–322 (2016).
Iimura, S. & Kibe, C. Highly sensitive adolescent benefits in positive school transitions: evidence for vantage sensitivity in Japanese high-schoolers. Dev. Psychol. 56, 1565–1581 (2020).
Neumann, C. S., Johansson, P. T. & Hare, R. D. The Psychopathy Checklist-Revised (PCL-R), low anxiety, and fearlessness: a structural equation modeling analysis. Personal. Disord. 4, 129–137 (2013).
Gajwani, R. & Minnis, H. Double jeopardy: implications of neurodevelopmental conditions and adverse childhood experiences for child health. Eur. Child Adolesc. Psychiatry 32, 1–4 (2023).
Northover, C., Thapar, A., Langley, K., Fairchild, G. & Van Goozen, S. H. Cortisol levels at baseline and under stress in adolescent males with attention-deficit hyperactivity disorder, with or without comorbid conduct disorder. Psychiatry Res. 242, 130–136 (2016).
Rizeq, J., Kennedy, M., Kreppner, J., Maughan, B. & Sonuga-Barke, E. Understanding the prospective associations between neuro-developmental problems, bullying victimization, and mental health: lessons from a longitudinal study of institutional deprivation. Dev. Psychopathol. https://doi.org/10.1017/S095457942200089X (2022).
Slavich, G. M. Social safety theory: a biologically based evolutionary perspective on life stress, health, and behavior. Annu. Rev. Clin. Psychol. 16, 265–295 (2020).
Dinkler, L. et al. Maltreatment-associated neurodevelopmental disorders: a co-twin control analysis. J. Child Psychol. Psychiatry 58, 691–701 (2017).
Gajwani, R. et al. Mania symptoms in a Swedish longitudinal population study: the roles of childhood trauma and neurodevelopmental disorders. J. Affect. Disord. 280, 450–456 (2021).
McCrory, E., Foulkes, L. & Viding, E. Social thinning and stress generation after childhood maltreatment: a neurocognitive social transactional model of psychiatric vulnerability. Lancet Psychiatry 9, 828–837 (2022).
Bradley, R. H. Home environment and self-efficacy beliefs among native american, african american, and latino adolescents. Family Process 58, 418–430 (2019).
Ellaway, A., Dundas, R., Olsen, J. R. & Shiels, P. G. Perceived neighbourhood problems over time and associations with adiposity. Int. J. Environ. Res. Public Health 15, 1854 (2018).
Ge, T. & Wang, L. Multidimensional child poverty, social relationships and academic achievement of children in poor rural areas of China. Child. Youth Serv. Rev. 103, 209–217 (2019).
Berlemann, M. & Steinhardt, M. F. Climate change, natural disasters, and migration—a survey of the empirical evidence. CESifo Econ. Stud. 63, 353–385 (2017).
Lu, S. Family migration and youth psychosocial development: An ecological perspective. Child. Youth Serv. Rev. 113, 104953 (2020).
Walker, S. P. et al. Cognitive, psychosocial, and behaviour gains at age 31 years from the Jamaica early childhood stimulation trial. J. Child Psychol. Psychiatry 63, 626–635 (2022).
Ellis, B. J., Sheridan, M. A., Belsky, J. & McLaughlin, K. A. Why and how does early adversity influence development? Toward an integrated model of dimensions of environmental experience. Dev. Psychopathol. 34, 447–471 (2022).
Reuben, A. et al. Lest we forget: comparing retrospective and prospective assessments of adverse childhood experiences in the prediction of adult health. J. Child Psychol. Psychiatry 57, 1103–1112 (2016).
Richens, J. G., Lee, C. M. & Johri, S. Improving the accuracy of medical diagnosis with causal machine learning. Nat. Commun. 11, 3923 (2020).
Kuranova, A. et al. Measuring resilience prospectively as the speed of affect recovery in daily life: a complex systems perspective on mental health. BMC Med. 18, 36 (2020).
Kievit, R. A., Davis, S. W., Griffiths, J., Correia, M. M. & Henson, R. N. A watershed model of individual differences in fluid intelligence. Neuropsychologia 91, 186–198 (2016).
Author information
Authors and Affiliations
Contributions
All authors have been part of an active and longstanding group discussing and debating the model discussed in the paper, contributed towards the drafting of the paper and agreed the final draft. H.M. wrote the initial draft and P.G.S., as senior author, worked closely with H.M. to build the initial model.
Corresponding author
Ethics declarations
Competing interests
A.L.v.H. acknowledges consulting fees from the Augeo foundation. All other authors declare no competing interests.
Peer review
Peer review information
Nature Mental Health thanks Andre Kerber, Rosemarie de la Rosa and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Minnis, H., van Harmelen, AL., Gajwani, R. et al. The bio-exposome: intracellular processes, stress physiology and the environment. Nat. Mental Health 2, 132–140 (2024). https://doi.org/10.1038/s44220-023-00180-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s44220-023-00180-3