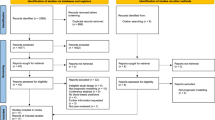
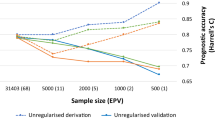
Abstract
Around a quarter of people who experience a first episode of psychosis (FEP) will develop treatment-resistant schizophrenia, but there are currently no established clinically useful methods to predict this from baseline. We aimed to explore the predictive potential for clozapine use as a proxy for treatment-resistant schizophrenia of routinely collected, objective biomedical predictors at FEP onset, and to validate the model externally in a separate clinical sample of people with FEP. We developed and externally validated a forced-entry logistic regression risk prediction model for clozapine treatment, or MOZART, to predict up to 8-year risk of clozapine use from FEP using routinely recorded information including age, sex, ethnicity, triglycerides, alkaline phosphatase levels and lymphocyte counts. We also produced a least-absolute shrinkage and selection operator (LASSO) based model, additionally including neutrophil count, smoking status, body mass index and random glucose levels. The models were developed using data from two United Kingdom (UK) psychosis early intervention services and externally validated in another UK early intervention service. Model performance was assessed by discrimination and calibration. We developed the models in 785 patients and validated them externally in 1,110 patients. Both models predicted clozapine use well during internal validation (MOZART: C statistic, 0.70 (95% confidence interval, 0.63–0.76); LASSO: 0.69 (0.63–0.77)). At external validation, discrimination performance reduced (MOZART: 0.63 (0.58–0.69); LASSO: 0.64 (0.58–0.69)) but recovered after re-estimation of the lymphocyte predictor (0.67 (0.62–0.73)). Calibration plots showed good agreement between observed and predicted risk in the forced-entry model. We also present a decision-curve analysis and an online data visualization tool. The use of routinely collected clinical information including blood-based biomarkers taken at FEP onset can help to predict the individual risk of clozapine use, and should be considered equally alongside other potentially useful information such as symptom scores in large-scale efforts to predict psychiatric outcomes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$59.00 per year
only $4.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The source data for this work is anonymized patient records from three UK NHS Trusts: the CPFT Research Database, SLaM CRIS and the Birmingham EIP. Data from these Trusts are only available to clinicians and clinical researchers with clinical contracts with the Trusts. The data are securely held on clinical systems and available following ethical approval to preserve patient confidentiality. Therefore, the raw data cannot be shared. However, we developed an online data visualization tool (https://eosimo.shinyapps.io/trs_app/) for both the original and recalibrated MOZART models, which allows interactive exploration of the effect of each predictor and their combinations on the risk of clozapine use based on the predictors included in this study.
Code availability
R code for data extraction and analysis is available upon request to the corresponding author.
References
Menezes, N., Arenovich, T. & Zipursky, R. A systematic review of longitudinal outcome studies of first-episode psychosis. Psychol. Med. 36, 1349–1362 (2006).
Osimo, E. F. et al. Inflammatory and cardiometabolic markers at presentation with first episode psychosis and long-term clinical outcomes: A longitudinal study using electronic health records. Brain Behav. Immun. 91, 117–127 (2021).
Siskind, D. et al. Rates of treatment-resistant schizophrenia from first-episode cohorts: systematic review and meta-analysis. Br. J. Psychiatry 220, 115–120 (2022).
Howes, O. D., Thase, M. E., & Pillinger, T. Treatment resistance in psychiatry: state of the art and new directions. Mol. Psychiatry 27, 58–72 (2021).
Kennedy, J. L., Altar, C. A., Taylor, D. L., Degtiar, I. & Hornberger, J. C. The social and economic burden of treatment-resistant schizophrenia: a systematic literature review. Int. Clin. Psychopharmacol. 29, 63–76 (2014).
Mizuno, Y., McCutcheon, R. A., Brugger, S. P. & Howes, O. D. Heterogeneity and efficacy of antipsychotic treatment for schizophrenia with or without treatment resistance: a meta-analysis. Neuropsychopharmacology 45, 622–631 (2020).
Howes, O. D. et al. Adherence to treatment guidelines in clinical practice: study of antipsychotic treatment prior to clozapine initiation. Br. J. Psychiatry 201, 481–485 (2012).
Barnes, T. R. et al. Evidence-based guidelines for the pharmacological treatment of schizophrenia: updated recommendations from the British Association for Psychopharmacology. J. Psychopharmacol. 34, 3–78 (2020).
McGuire, P. & Dazzan, P. Does neuroimaging have a role in predicting outcomes in psychosis? World Psychiatry 16, 209–210 (2017).
Wimberley, T. et al. Predictors of treatment resistance in patients with schizophrenia: a population-based cohort study. Lancet Psychiatry 3, 358–366 (2016).
Demjaha, A. et al. Antipsychotic treatment resistance in first-episode psychosis: prevalence, subtypes and predictors. Psychol. Med. 47, 1981–1989 (2017).
Chan, S. et al. Predictors of treatment resistant schizophrenia-spectrum disorder: 10-year retrospective study of first-episode psychosis (A56). Early Interv. Psychiatry 8, 78 (2014).
Bozzatello, P., Bellino, S. & Rocca, P. Predictive factors of treatment resistance in first episode of psychosis: a systematic review. Front. Psychiatry 10, 67 (2019).
Lally, J. et al. Two distinct patterns of treatment resistance: clinical predictors of treatment resistance in first-episode schizophrenia spectrum psychoses. Psychol. Med. 46, 3231–3240 (2016).
Üçok, A. et al. Correlates of clozapine use after a first episode of schizophrenia: results from a long-term prospective study. CNS Drugs 30, 997–1006 (2016).
Smart, S., Kępińska, A., Murray, R. & MacCabe, J. Predictors of treatment resistant schizophrenia: a systematic review of prospective observational studies. Psychol. Med. 51, 44–53 (2021).
Dwyer, D. B., Falkai, P. & Koutsouleris, N. Machine learning approaches for clinical psychology and psychiatry. Annu Rev. Clin. Psychol. 14, 91–118 (2018).
Hippisley-Cox, J., Coupland, C. & Brindle, P. Development and validation of QRISK3 risk prediction algorithms to estimate future risk of cardiovascular disease: prospective cohort study. BMJ 357, j2099 (2017).
Perry, B. I. et al. Development and external validation of the Psychosis Metabolic Risk Calculator (PsyMetRiC): a cardiometabolic risk prediction algorithm for young people with psychosis. Lancet Psychiatry 8, 589–598 (2021).
Perry, B. I., McIntosh, G., Weich, S., Singh, S. & Rees, K. The association between first-episode psychosis and abnormal glycaemic control: systematic review and meta-analysis. Lancet Psychiatry 3, 1049–1058 (2016).
Pillinger, T., Beck, K., Stubbs, B. & Howes, O. D. Cholesterol and triglyceride levels in first-episode psychosis: systematic review and meta-analysis. Br. J. Psychiatry 211, 339–349 (2017).
Pillinger, T. et al. A meta-analysis of immune parameters, variability, and assessment of modal distribution in psychosis and test of the immune subgroup hypothesis. Schizophr. Bull. 45, 1120–1133 (2019).
Nettis, M. A. et al. Metabolic-inflammatory status as predictor of clinical outcome at 1-year follow-up in patients with first episode psychosis. Psychoneuroendocrinology 99, 145–153 (2019).
Legge, S. et al. Clinical indicators of treatment-resistant psychosis. Br. J. Psychiatry 216, 259–266 (2020).
Wimberley, T. et al. Polygenic risk score for schizophrenia and treatment-resistant schizophrenia. Schizophr. Bull. 43, 1064–1069 (2017).
Trubetskoy, V. et al. Mapping genomic loci implicates genes and synaptic biology in schizophrenia. Nature 604, 502–508 (2022).
Pardiñas, A. F. et al. Interaction testing and polygenic risk scoring to estimate the association of common genetic variants with treatment resistance in schizophrenia. JAMA Psychiatry 79, 260–269 (2022).
Steyerberg, E. W. et al. Prognosis Research Strategy (PROGRESS) 3: prognostic model research. PLoS Med. 10, e1001381 (2013).
Wolff, R. F. et al. PROBAST: a tool to assess the risk of bias and applicability of prediction model studies. Ann. Intern. Med. 170, 51–58 (2019).
Morlán-Coarasa, M. J. et al. Incidence of non-alcoholic fatty liver disease and metabolic dysfunction in first episode schizophrenia and related psychotic disorders: a 3-year prospective randomized interventional study. Psychopharmacology 233, 3947–3952 (2016).
Perry, B. I. et al. Dysglycaemia, inflammation and psychosis: findings from the UK ALSPAC birth cohort. Schizophr. Bull. 45, 330–338 (2019).
Pillinger, T. et al. Impaired glucose homeostasis in first-episode schizophrenia: a systematic review and meta-analysis. JAMA Psychiatry 74, 261–269 (2017).
Machado, M. V. & Diehl, A. M. Pathogenesis of nonalcoholic steatohepatitis. Gastroenterology 150, 1769–1777 (2016).
Dix, H. M., Robinson, E. M. & Dillon, J. F. in Textbook of Addiction Treatment (eds. el-Guebaly, N., et al.) 1099–1111 (Springer, 2021).
Van de Mortel, T. F. Faking it: social desirability response bias in self-report research. Aust. J. Adv. Nurs. 25, 40–48 (2008).
Moody, G. & Miller, B. J. Total and differential white blood cell counts and hemodynamic parameters in first-episode psychosis. Psychiatry Res. 260, 307–312 (2018).
Garcia‐Rizo, C. et al. Blood cell count in antipsychotic‐naive patients with non‐affective psychosis. Early Interv. Psychiatry 13, 95–100 (2019).
Perry, B. I. et al. Associations of immunological proteins/traits with schizophrenia, major depression and bipolar disorder: a bi-directional two-sample Mendelian randomization study. Brain Behav. Immun. 97, 176–185 (2021).
Bunders, M., Cortina-Borja, M. & Newell, M.-L. Age-related standards for total lymphocyte, CD4+ and CD8+ T cell counts in children born in Europe. Pediatr. Infect. Dis. J. 24, 595–600 (2005).
Lang, X. et al. Differences in patterns of metabolic abnormality and metabolic syndrome between early-onset and adult-onset first-episode drug-naive schizophrenia patients. Psychoneuroendocrinology 132, 105344 (2021).
Psychosis and Schizophrenia in Adults: Prevention and Management CG178 (National Institute for Health and Care Excellence, 2014). https://www.nice.org.uk/guidance/cg178
National Clinical Audit of Psychosis – National Report for the Early Intervention in Psychosis Audit 2019/2020. London (Royal College of Psychiatrists, 2020). www.rcpsych.ac.uk/NCAP
Psychosis and Schizophrenia in Adults QS80 (National Institute for Health and Care Excellence, 2015). https://www.nice.org.uk/guidance/qs80
Haw, C. & Stubbs, J. Off-label use of antipsychotics: are we mad? Expert Opin. Drug Saf. 6, 533–545 (2007).
Hodgson, R. & Belgamwar, R. Off-label prescribing by psychiatrists. Psychiatric Bull. 30, 55–57 (2006).
Pardiñas, A. F. et al. Common schizophrenia alleles are enriched in mutation-intolerant genes and in regions under strong background selection. Nat. Genet. 50, 381–389 (2018).
Chan, S. K. W. et al. Predictors of treatment-resistant and clozapine-resistant schizophrenia: a 12-year follow-up study of first-episode schizophrenia-spectrum disorders. Schizophrenia Bull. 47, 485–494 (2021).
Riley, R. D. et al. Minimum sample size for developing a multivariable prediction model: PART II—binary and time‐to‐event outcomes. Stat. Med. 38, 1276–1296 (2019).
Cardinal, R. N. Clinical records anonymisation and text extraction (CRATE): an open-source software system. BMC Med. Inf. Decis. Making 17, 50 (2017).
McGorry, P. D. Early intervention in psychosis: obvious, effective, overdue. J. Nerv. Ment. Dis. 203, 310–318 (2015).
Meltzer, H. Y. Treatment-resistant schizophrenia-the role of clozapine. Curr. Med. Res. Opin. 14, 1–20 (1997).
R Core Team R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2022).
Ensor, J., Martin, E. C. & Riley, R. D. pmsampsize (2021). https://cran.r-project.org/web/packages/pmsampsize/index.html
Van Buuren, S. & Groothuis-Oudshoorn, K. mice: multivariate imputation by chained equations in R. J. Stat. Softw. 45, 1–67. (2011).
Robin, X. et al. pROC: an open-source package for R and S+ to analyze and compare ROC curves. BMC Bioinf. 12, 77 (2011).
Van Calster, B. et al. A calibration hierarchy for risk models was defined: from utopia to empirical data. J. Clin. Epidemiol. 74, 167–176 (2016).
Schomaker, M. & Heumann, C. Model selection and model averaging after multiple imputation. Comput. Stat. Data Anal. 71, 758–770 (2014).
Eekhout, I., Van De Wiel, M. A. & Heymans, M. W. Methods for significance testing of categorical covariates in logistic regression models after multiple imputation: power and applicability analysis. BMC Med. Res. Method. 17, 129 (2017).
Radchenko, P. & James, G. M. Variable inclusion and shrinkage algorithms. J. Am. Stat. Assoc. 103, 1304–1315 (2008).
Friedman, J., Hastie, T. & Tibshirani, R. Regularization paths for generalized linear models via coordinate descent. J. Stat. Softw. 33, 1–22 (2010).
Steyerberg, E. W. Clinical Prediction Models (Springer, 2019).
Steyerberg, E. W., Borsboom, G. J., van Houwelingen, H. C., Eijkemans, M. J. & Habbema, J. D. F. Validation and updating of predictive logistic regression models: a study on sample size and shrinkage. Stat. Med. 23, 2567–2586 (2004).
Vickers, A. J. & Elkin, E. B. Decision curve analysis: a novel method for evaluating prediction models. Med. Decis. Making 26, 565–574 (2006).
Vickers, A. J., van Calster, B. & Steyerberg, E. W. A simple, step-by-step guide to interpreting decision curve analysis. Diagn. Progn. Res. 3, 18 (2019).
Chang, W. et al. shiny: Web Application Framework for R v.1.7.2 (2022). https://cran.r-project.org/web/packages/shiny/index.html
Acknowledgements
This work was funded by a Clinical PhD Fellowship to E.F.O. jointly funded by the NIHR Imperial BRC and the UK Research and Innovation Medical Research Council London Institute of Medical Sciences. B.I.P. acknowledges funding support from the NIHR (doctoral research fellowship, DRF-2018-11-ST2-018). R.U. received funding support from the NIHR (HTA grant 127700) and Medical Research Council (Therapeutic Target Validation in Mental Health grant MR/S037675/1). G.M.K. received funding support from the Wellcome Trust (grant 201486/Z/16/Z), the MQ: Transforming Mental Health (grant MQDS17/40), the Medical Research Council UK (grants MC_PC_17213; MR/S037675/1; and MR/W014416/1), and the British Medical Association Foundation (J Moulton grant 2019). R.N.C. acknowledges support from the Medical Research Council (grants MC_PC_17213, MR/W014386/1). This research was supported in part by the NIHR Imperial BRC and NIHR Cambridge BRC (BRC-1215-20014); J.P. and P.B.J. acknowledge funding from the NIHR ARC EoE; the views expressed are those of the authors and not necessarily those of the NHS, the NIHR or the Department of Health and Social Care. The funding bodies had no role in design or conduct of the study; collection, management, analysis or interpretation of the data; preparation, review or approval of the manuscript; or the decision to submit the manuscript for publication.
Author information
Authors and Affiliations
Contributions
E.F.O. and B.I.P. designed the study and selected the predictors and outcome variables under the supervision of R.U. and G.M.K., and with input from P.M., G.K.M., J.P., P.B.J., R.N.C. and O.D.H.; E.F.O. had access to all datasets, collected the data and performed the statistical analyses, in close discussion with B.I.P. and the wider supervisory team. M.P. and O.D.H. supported E.F.O. in data collection and analysis for the SLaM cohort. J.L. and R.N.C. supported E.F.O. in data collection and analysis for the Cambridge cohort. A.K. and R.U. supported E.F.O. in data collection and analysis for the Birmingham cohort. E.F.O. wrote the first draft of the manuscript, with constant support from B.I.P. All other authors contributed to the drafting, re-drafting and perfecting of the manuscript, including responses to reviewers’ comments.
Corresponding author
Ethics declarations
Competing interests
O.D.H. is a part-time employee of H. Lundbeck A/S. He has received investigator-initiated research funding from and/or participated in advisory/speaker meetings organized by Angelini, Autifony, Biogen, Boehringer Ingelheim, Eli Lilly, Heptares, Global Medical Education, Invicro, Janssen, H. Lundbeck A/S, Neurocrine, Otsuka, Sunovion, Recordati, Roche and Viatris/Mylan. O.D.H. has a patent for the use of dopaminergic imaging. R.N.C. consults for Campden Instruments and receives royalties from Cambridge Enterprise, Routledge and Cambridge University Press. The other authors declare no competing interests.
Peer review
Peer review information
Nature Mental Health thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 PubMed results for ‘risk AND prediction AND psychosis’ by year.
Generated with PubMed by Year. Available from http://esperr.github.io/pubmed-by-year/.
Extended Data Fig. 2 Internal validation: distribution of predicted probabilities for MOZART.
X axis: predicted probability.
Extended Data Fig. 3 Internal validation: distribution of predicted probabilities for the LASSO model.
X axis: predicted probability.
Extended Data Fig. 4 Internal validation: calibration plot for MOZART.
Model calibration is the extent to which outcomes predicted by the model are similar to those observed in the validation dataset. Calibration plots illustrate agreement between observed proportion (y axis) and predicted risk (x axis). Perfect agreement would trace the red line. Model calibration is shown by the continuous black line. Triangles denote grouped observations for participants at deciles of predicted risk, with 95% CIs indicated by the vertical black lines. Axes range between 0 and 0.3 since very few individuals received predicted probabilities greater than 0.3. N=785 participants in pooled development sample.
Extended Data Fig. 5 Internal validation: calibration plot for the LASSO model.
Model calibration is the extent to which outcomes predicted by the model are similar to those observed in the validation dataset. Calibration plots illustrate agreement between observed proportion (y axis) and predicted risk (x axis). Perfect agreement would trace the red line. Model calibration is shown by the continuous black line. Triangles denote grouped observations for participants at deciles of predicted risk, with 95% CIs indicated by the vertical black lines. Axes range between 0 and 0.3 since very few individuals received predicted probabilities greater than 0.3. N=785 participants in pooled development sample.
Extended Data Fig. 6 Internal validation: distribution of predicted probabilities for M3.
X axis: predicted probability.
Extended Data Fig. 7 Internal validation: calibration plot for M3.
Calibration plots illustrate agreement between observed proportion (y axis) and predicted risk (x axis). Perfect agreement would trace the red line. Model calibration is shown by the continuous black line. Triangles denote grouped observations for participants at deciles of predicted risk, with 95% CIs indicated by the vertical black lines. Axes range between 0 and 0.3 since very few individuals received predicted probabilities greater than 0.3. N=785 participants in pooled development sample.
Extended Data Fig. 8 Internal validation: distribution of predicted probabilities for M4.
X axis: predicted probability.
Extended Data Fig. 9 Internal validation: calibration plot for M4.
Calibration plots illustrate agreement between observed proportion (y axis) and predicted risk (x axis). Perfect agreement would trace the red line. Model calibration is shown by the continuous black line. Triangles denote grouped observations for participants at deciles of predicted risk, with 95% CIs indicated by the vertical black lines. Axes range between 0 and 0.3 since very few individuals received predicted probabilities greater than 0.3. N=785 participants in pooled development sample.
Supplementary information
Supplementary Information
Supplementary notes and Tables 1–5.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Osimo, E.F., Perry, B.I., Mallikarjun, P. et al. Predicting treatment resistance from first-episode psychosis using routinely collected clinical information. Nat. Mental Health 1, 25–35 (2023). https://doi.org/10.1038/s44220-022-00001-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s44220-022-00001-z
This article is cited by
-
Management von Therapieresistenzen – therapieresistente Schizophrenie
Der Nervenarzt (2024)
-
Development of an individualized risk calculator of treatment resistance in patients with first-episode psychosis (TRipCal) using automated machine learning: a 12-year follow-up study with clozapine prescription as a proxy indicator
Translational Psychiatry (2024)