Abstract
Studies have linked higher digital screen use with poorer mental health. However, there is limited experimental evidence to suggest a causal relationship. In this trial, we aimed to investigate the effects of limiting recreational digital screen use on mental well-being, mood, and biomarkers of stress in healthy young and middle-aged adults. We randomly allocated 89 families (including 164 adults) to participate in an extensive screen media reduction intervention or control. Participants in the intervention group were instructed to decrease their recreational screen use to less than 3 hours/week/person. Intervention compliance was assessed using applications and tv-monitors. Overall subjective mental well-being and mood, and collected daily biomarkers of stress (salivary cortisol and cortisone) was assessed at baseline and 2-week follow-up. Reducing recreational digital screen use resulted in significantly improved self-reported well-being and mood in adults allocated to the intervention compared to control. We observed no intervention effects for biomarkers of stress. (ClinicalTrials.gov: NCT04098913, 23/09/2019).
Similar content being viewed by others
Introduction
The proportion of adults with indicators of poor mental health (i.e., anxiety disorders, depression, and general mental well-being) has increased during the last decade in many countries1,2,3,4,5,6. During this same period, notable changes have occurred in digital technology and how to screen devices are used. Digital screen use has become a major part of people’s lives. Screen media devices provide endless opportunities such as checking the latest news, e-mails, scrolling through social media sites, streaming movies and series, or video calling friends, and much more. Although, each of these activities are not necessarily beneficial or harmful, the ubiquitous availability of digital devices and the high levels of engagement and social expectation to always be available may impact both physical and mental health. The worries about the potential harm continue to be debated heavily among health professionals, educators, and researchers7,8,9.
Digital screen use has been linked to lower self-reported mental health (e.g., increased levels of depression, perceived stress, and negative mood) in adults. A systematic review and meta-analysis found that high screen media use was associated with a 28% increase in the odds of depression based on data from seven longitudinal and 12 cross-sectional studies10. Also, a systematic review and meta-analysis of 37 cross-sectional studies reported a significant positive association between smartphone use and stress and anxiety11. While the findings based on observational studies may be affected by uncontrolled confounding or information bias caused by use of self-reported screen use10,12, another major limitation is the possibility of reverse causation—that is, that recreational screen use is increased as a consequence of mental health issues8. Recently, a few experimental studies have investigated the short-term effects of reducing social media use (not overall recreational screen use) on mental health13,14,15,16,17. However, evidence is still inconclusive primarily due to methodological limitations such as lack of objective assessment of intervention compliance or non-compliance to the intervention.
In addition to the impact on self-reported mental health, the use of screen media may also influence biomarkers of stress e.g., daily levels of cortisol and cortisone, which display a distinct diurnal pattern18,19. Cortisol secretion is regulated by the hypothalamus-pituitary-adrenal axis (HPA-axis) in response to stress20,21,22. A flatter diurnal cortisol slope has been associated with several physical and mental health outcomes e.g., obesity, depression, and externalizing symptoms21, while a higher cortisol awakening response has been associated with higher general life stress and prior-day feelings of sadness and being overwhelmed23,24. Few studies have investigated the relationship between the recreational use of screen media and biomarkers of stress25,26,27,28,29, but experimental studies are warranted.
Considering the limitations of previous observational and experimental studies, we aimed to investigate the causal relationship between recreational digital screen use (all digital screen use unrelated to work (or study) and measured outside self-reported working hours) and multiple measures of mental health in adults using data from a recent cluster randomized controlled trial (the SCREENS trial)30,31. Specifically, we investigated the efficacy of limiting recreational digital screen use on self-reported overall mental well-being, mood, and daily rhythms of biomarkers of stress (salivary cortisol and cortisone) in adults.
Results
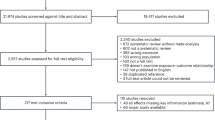
A total of 1420 families indicated an interest in the study and were screened for eligibility. Of these, 95 were eligible and all provided their consent to participate. A total of 92 families (including 171 adults) completed baseline measurements but three families withdrew prior to randomization. Thus, a total of 89 families (164 adults) were randomly allocated to the intervention group (45 families) or the control group (44 families) between June 2019 and March 2021 (Fig. 1).
Baseline characteristics of participants are presented for each group in Table 1. Personal factors and levels of screen media use were evenly distributed between the two groups, but participants allocated to the control group were about two years older than participants in the intervention group (Table 1).
Recreational digital screen use during the experiment
Adults in the intervention group had a median of 2.7 h/week (IQR: 1.7 to 3.6) of recreational digital screen use, while in contrast, the control group had a median of 15.5 h/week (IQR: 9.6 to 22.8). The proportion of participants who were compliant with the intervention has been reported previously31.
Well-being and mood states
All adults (control = 82, intervention = 82) were included in the analyses presented in Table 2. Participants in the intervention group significantly increased their self-reported mental well-being, whereas those in the control group did not perceive any changes in either direction. The intervention effect (mean between-group difference) for the WHO-5 Well-Being Index was 8.48 points, 95% CI: 4.90 to 12.07 (Cohen’s d: 0.72, 95% CI: 0.39 to 1.05) in favor of the screen use reduction intervention (Table 2).
Both groups significantly improved their total mood disturbance scores, but participants allocated to screen reduction had a significantly greater improvement of −6.83 points 95% CI: −12.68 to −0.97 (Cohen’s d: −0.38, 95% CI: −0.70 to −0.05) compared to control. Analyses of the mood subscales revealed significant intervention effects for tension, fatigue, and vigor in favor of the intervention (Table 2).
Individual participant change scores for WHO-5 Well-Being Index and total mood disturbance score are shown in Fig. 2. Raw mean scores with standard deviations of well-being and mood scores at baseline and follow-up for each group are provided in Supplementary Table 1.
Cortisol and cortisone
The median number of saliva samples per participant at baseline was 11 (IQR: 9 to 12) for both control and intervention, while it was 11 (IQR: 9 to 12) for participants allocated to control and 12 (IQR: 10 to 12) for participants allocated to the intervention at follow-up. Measured cortisol and cortisone concentrations are displayed in Figs. 3, 4.
Figure 3 shows the 3-day median and interquartile ranges for cortisol concentration by group allocation for each time point. The figure also displays the exact cortisol concentration for each saliva sample included in the analyses after the removal of extreme outliers (points are randomly spread at each time point solely for illustration purposes).
Figure 4 shows the 3-day median and interquartile ranges for cortisone concentration by group allocation for each time point. The figure also displays the exact cortisone concentration for each saliva sample included in the analyses after the removal of extreme outliers (points are randomly spread at each time point solely for illustration purposes).
No significant within-group changes were found in any measures of daily cortisol or cortisone levels except for awakening cortisone among adults allocated to the control group (mean increase of 1.70 nmol/L (95% CI: 0.33 to 3.07)). We found no significant intervention effect on change in any of the cortisol and cortisone measures except for the awakening cortisone level (mean difference in change −1.97 nmol/L (95% CI: −3.91 to −0.03) comparing intervention vs. control) (Table 3). The standardized mean difference (Cohen’s d) for the cortisol and cortisone awakening sample were −0.08 (95%CI −0.40 to 0.22) and −0.09 (95% CI: −0.41 to 0.21). Raw mean scores with standard deviations of cortisol and cortisone measures at baseline and follow-up for each group are provided in Supplementary Table 2.
Discussion
In this cluster randomized controlled trial, we investigated the efficacy of reducing household recreational digital screen use on overall mental well-being, mood, and daily biomarkers of stress in adults. We found significant improvements in overall self-reported mental well-being and mood in favor of recreational screen reduction. No consistent intervention effects were found for measures of daily cortisol and cortisone levels.
Our study provides novel experimental evidence that restricting recreational digital screen use increases self-reported overall mental well-being and mood. These findings are in line with results from observational studies reported in systematic reviews10,11. To the best of our knowledge, no other trials have investigated the impact of limiting overall recreational digital screen use on perceived mental well-being and mood. Some trials have investigated the short-term effects of limiting social media engagement on mental well-being and shown mixed results13,14,15,16,17. A key difference between previous trials and our trial is the high level of intervention compliance observed in our trial. All previous trials except for Wezel et al. used self-reported measures of compliance, which may be prone to bias12. Wezel et al. used an objective measure to document compliance to change in the amount of social media use and they reported no effect on well-being. However, their attempt to reduce social media use to 50 and 10% among participants in the two respective intervention groups was unsuccessful, which could explain the absence of an effect on the mental well-being outcomes. In addition, interventions in previous trials were aimed at individual participants rather than families. There are pros and cons of the individual-centered intervention design, but a substantial proportion of screen media use in families occur as part of a wider social context. Our family-based design could be an important factor for the observed effect on mental well-being and mood in our trial because it may increase intervention compliance through social change processes that can initiate and consolidate personal change32.
The intervention effect of 8.48 points (95% CI: 4.90 to 12.07) on the WHO-5 Well-Being Index corresponds to a standardized mean difference of 0.72 (Cohen’s d). The moderate to large standardized mean difference is noteworthy given that the population under study were healthy adults. In addition, the confidence interval overlaps 10, which is considered to be the minimal clinically important difference33,34. The intervention effect for the total mood disturbance score was low to moderate with a standardized mean difference of 0.38 (Cohen’s d). In relation to the results for mental well-being, it suggests that screen use has a smaller influence on mood states than on overall mental well-being. The use of screen media devices may affect well-being and mood through several pathways. One pathway may be that most adults always carry a smartphone around. The constant availability has been suggested to induce a perceived obligation to be accessible at all times, a notion that has been suggested to induce feelings of stress, depression, and guilt35. Another pathway may be that passive social media use affects well-being negatively through social comparison36. Finally, Afifi et al. suggest that engagement with screen media devices may possibly disrupt and displace fundamental behaviors such as eating, exercising, and sleeping, which may induce stress29.
This is the first trial exploring the effect of limiting screen media use on daily levels of cortisol and cortisone. Our study provided no consistent evidence for a causal relationship between restricting screen media use and change in biomarkers for stress, at least in the short term of 2 weeks. The absence of an effect on cortisol and cortisone may be due to a lack of statistical power to detect small effect sizes. An alternative explanation could be that our sample consisted of healthy adults. We cannot rule out that limiting screen use in adults with symptoms of stress or depression, who often display altered diurnal cortisol slopes and cortisol awakening responses37,38, could have a positive effect on their cortisol and cortisone profiles.
A key strength of our study is the experimental design, which increases the confidence that the observed effects of limiting screen media use on well-being and mood are causal because known and unknown confounding factors are expected to be equally distributed between the groups being compared. Another strength of our study is the implementation of objective assessments of intervention compliance31. Furthermore, the included participants and non-eligible participants had similar background characteristics i.e., age, sex, educational attainment, and recreational screen media use31. Also, results were robust after stratification for whether participants were enrolled before or after COVID-19 i.e., March 2020 (data not shown). However, the results should be interpreted with the following limitations in mind. First, due to the behavioral nature of the intervention, blinding was not possible, which may have introduced bias in the self-reported outcomes because participants may potentially have been influenced by knowledge of being in the screen use reduction group. We expect that this possible bias, to some degree, could have exaggerated the effect of screen use reduction on subjective mental well-being and mood as some participants may have responded in the direction of a hypothesis of benefit or that they think the researcher's desire (the concept of demand characteristics)39. Second, although participants in the intervention group successfully decreased their screen media use, we also observed a modest decrease in screen media use in the control group. This was likely because some families were motivated to try decreasing their screen media engagement prior to randomization. Third, although our assessment followed standard recommendations for assessment of salivary cortisol21,22,40, objective assessment of the timing of samples may have further strengthened our results. Finally, findings may not be generalizable beyond healthy adults who live in households with children.
Collectively, our study provides experimental evidence that limiting recreational digital screen use positively affects mental well-being and mood in adults. Our findings highlight the importance of awareness of the amount of time adults spend using recreational digital screen media devices. Future experimental studies should explore if the observed relationships depend on specific types of screen media content or different motivations for digital screen use. Furthermore, studies examining the impact of long-term reductions in screen use are warranted.
Methods
Study design
This study is a secondary analysis of the SCREENS trial (a parallel cluster randomized controlled trial), and it is reported in compliance with the CONSORT statement41. The trial design is described in detail in the study protocol30. The SCREENS trial was designed to investigate the efficacy of reducing household screen media use on several outcomes, and not to evaluate the pragmatic effectiveness of the intervention. We randomly allocated families (cluster unit) to reduce recreational screen use for a period of 2 weeks (intervention) or continue using screen media as usual (control). The cluster design was chosen to enhance compliance with the screen reduction intervention. The first family was enrolled on June 6, 2019 and the last family completed a follow-up on March 30, 2021.
We obtained ethical approval from the Ethical Committee of Southern Denmark (S-20170213). All participants gave written informed consent before baseline. The trial was registered at ClinicalTrials.gov (NCT04098913).
Study participants
Families residing in the Region of Southern Denmark with at least one child aged 6–10 years were recruited through a population-based survey42. A digital survey invitation was sent to a randomly chosen adult in each family via a mandatory digital mailbox (e-Boks). The Danish Health Data Authority performed the random selection of survey invitees using data from the Danish Civil Registration System43. The survey included questions on the families’ screen media habits44. Respondents were asked to answer a question (yes/no) on whether they were interested in participating in the SCREENS trial. We assessed whether the families were eligible based on survey data using the following criteria: The responding parent had self-reported recreational screen use >40th percentile (2.4 h/day) based on the first 1000 survey responses, all children in the household had to be >4 years of age and adults had to be full-time students or employed full-time (with no regular night shifts). The responding parent from families fulfilling these criteria were telephoned to confirm that at least one adult and one child in the household would be willing to participate in the trial, and that at least one participating adult and all participating children would be able to handover their smartphone(s) and tablet(s) for a period of 2 weeks.
We excluded individual participants from eligible families if the participants were not able to engage in everyday physical activities, if they had been diagnosed with a sleep disorder, a neuropsychiatric disorder, or a developmental disorder, or if they had been on stress-related sick leave within the last 3 months.
Randomization
The Odense Patient data Explorative Network (OPEN) generated the block randomization sequence using permuted blocks of two to four families. OPEN had no role in the delivery of the intervention or collection of data. The randomization was performed by a member of the research team via an online randomization website within the families’ households after completion of the baseline assessments. The member of the research team had no knowledge of the group allocation until after the randomization was performed. The study was open-label because blinding of participants was not achievable due to the behavioral nature of the intervention.
Interventions
Families allocated to the control group were instructed to carry on with their usual screen media habits. Families allocated to the screen reduction intervention were instructed to change their screen media habits substantially. First, members of families assigned to the intervention group were instructed to reduce their individual recreational digital screen use to a maximum of 3 h/week during the 2-week intervention period. Second, all participants were instructed to handover their tablet(s) and/or smartphone(s) in exchange for a non-smart cell phone (Nokia 130). Some adults were not able to because they had mixed work and leisure smartphones, but at least one adult had to if the family wanted to participate. Third, families were encouraged to talk about their expected challenges of reducing recreational screen use for 2 weeks and list potential solutions. Fourth, adults were allowed up to 30 min/day of so-called necessary screen media use (e.g., arranging appointments, checking online banking, etc.). Children who had to use screens for homework were allowed to do this to the extend necessary. During the intervention, all screen media use had to be registered in simple daily diaries. Also, three to five intervention reminders were positioned in places where families gather and in rooms where family members typically use screen media. Families who completed the SCREENS experiment received a financial reimbursement of 70 Euros. The intervention components are described in more detail in the study protocol30.
Assessment of intervention compliance
Recreational use of smartphones, tablets, and computers was objectively assessed using non-commercial Device Tracker apps and television use was assessed using a monitor developed in-house31,45. A total of 78 (95%) adults allocated to the intervention group were considered compliant with the intervention because they had less than 7 hours/week of recreational screen use during the experiment31. In this paper, we report descriptive statistics (median and interquartile ranges) of recreational digital screen use during the experiment period to allow for a comparison of the contrast in screen exposure.
Outcomes
All outcomes of the SCREENS trial are described in the study protocol30. The following sections describe the outcomes of the current paper.
Well-being
A digital version of the WHO-5 Well-Being Index was used to assess overall mental well-being before the baseline protocol started and immediately after the completion of the experiment period. The five-item questionnaire is widely used as an outcome measure of mental well-being in clinical trials and has acceptable validity, and is psychometrically sound34,46. The WHO-5 Well-Being Index consists of five statements about how a person has felt (e.g., “I have felt cheerful in good spirits”) over the previous 2 weeks. Participants had to provide one of the following answers: All of the time (5), most of the time (4), more than half of the time (3), less than half of the time (2), some of the time (1), at no time (0). The final score was calculated for each adult by adding the scores from the five items and multiplying the raw sum by 434. Higher scores correspond to better mental well-being.
Mood states
Overall mood states were assessed using the Profile of Mood States questionnaire, which is a validated and widely used scale to assess mood states in healthy populations47,48. The questionnaire consists of 65 words (e.g., friendly, tense, angry, etc.) and participants had to answer one of the following: Not at all (0), A little (1), Moderately (2), Quite a lot (3), Extremely (4). For two of the questions, the scoring was reversed (from 4 to 0). For participants who had missing data on less than 5 single items (baseline n = 14, follow-up n = 15), we imputed the observed median for each item from participants with complete data for the item. Scores were summed in six distinct subscales (tension, depression, anger, fatigue, confusion, and vigor). Total mood disturbance was calculated by adding tension, depression, anger, fatigue, and confusion scores and subtracting the vigor score47. Lower scores correspond to a better mood for all subscales (except for the vigor score) and total mood disturbance.
Cortisol and cortisone
We assessed salivary biomarkers of stress (cortisol/cortisone). We used an ambulatory assessment of cortisol and cortisone to provide a more ecologically valid picture of the potential changes in daily rhythms of cortisol/cortisone secretion in response to the intervention40. Participants received face-to-face instructions from a member of the research team on how to complete the saliva sampling using Salivettes and synthetic swaps (Starstedt, Nümbrecht, Germany). We emphasized the importance of reporting the actual time of each sample in the daily sampling diary. Participants were also provided with a written sampling manual, including pictures. Salivary samples had to be collected on the three days leading up to randomization and during the last three days of the experiment (Supplementary Fig. 1) to increase the reliability of the measurements49. Participants were instructed to complete three samples in the morning (upon awakening, +30 min, +45 min) which is the minimum protocol to assess the cortisol/cortisone awakening response suggested in previous expert consensus guidelines22. After collecting the awakening sample, participants were instructed to start a pre-programmed alarm clock (Dual Digital Timer, S. Brannan Sons Ltd., England) for reminders for the +30 min, +45 min sample collections. Participants were also instructed to collect one sample prior to bedtime. The research team carefully instructed participants to refrain from smoking, exercising, toothbrushing, eating, or drinking anything but water during the morning sampling routine and 30 min prior to collection of the bedtime sample. Participants were instructed to place the samples in a sample rack in their freezer during the course of the study. All samples were transported in refrigerated boxes and stored in a freezer at the University of Southern Denmark. Finally, we transported the saliva samples to the clinical biochemistry department at Slagelse Hospital, Region Zealand, where the samples were analyzed. Cortisol and cortisone levels were determined using isotope dilution liquid chromatography-tandem mass spectrometry (LC-MS/MS).
We calculated daily diurnal cortisol and cortisone slopes by subtracting the bedtime concentration from the awakening concentration and dividing it by the time elapsed between the two samples50. The following daily cortisol and cortisone awakening response metrics were calculated: CAR30 (30 min concentration–awakening concentration), CAR45 (45 min concentration–awakening concentration), CARpeak (peak concentration at 30 or 45 min—awakening concentration), area under the curve ground (CARaucG), and area under the curve with respect to increase (CARaucI) was calculated using the two formulas provided by Pruessner et al.51.
Cortisol and cortisone data were positively skewed at each time point. Thus, data were log-transformed at each time point to identify extreme outliers (>±3 SD) for the cortisol or cortisone concentrations at each time point. After a case-by-case evaluation, 36 cortisol and 38 cortisone samples of the total 3636 and 3642 samples were identified as extreme outliers, respectively, and these were deleted40. Log-transformed variables were only used to identify extreme outliers and were not included in any analyses. Morning saliva samples and evening saliva samples deviating more than 15 min (n = 261) and 60 min (n = 118) from the planned sampling protocol, respectively, were excluded from the analyses.
Sample size
The determination of the sample size of the SCREENS trial was based on results from our pilot study52, and the SCREENS trial aimed to have 80% power to detect a 24 min/day group-mean difference in children’s non-sedentary time (primary outcome in the SCREENS trial). Thus, the sample size was not powered to detect a specific change in overall well-being, mood, or daily cortisol and cortisone measures. However, results from our pilot study suggest that our total sample size of 164 adults has the power to detect standardized effect sizes >0.35 (Cohen’s d) for the cortisol and cortisone area under the curve measures49.
Statistical approach
Data were analyzed using linear mixed-effects models, including an interaction term between group allocation and the baseline/follow-up variable. Mixed-effects tobit regression was used for the moods subscales to account for potential floor and ceiling effects53, and values were censored at 0 (lower limit) for the tension, depression, anger, fatigue, and confusion subscales, and at 32 (upper limit) for the vigor subscale. All analyses included family-level and participant-level random intercepts to account for potential correlation due to clustering. Distinct models were run with the following variables as outcomes: WHO-5 Well-Being Index, total mood disturbance score, each of the Profile of mood states subscales, cortisol and cortisone awakening sample, diurnal cortisol and cortisone slopes, and five different measures of the cortisol and cortisone awakening response (CAR30, CAR45, CARpeak, CARaucG, and CARaucI). All models were adjusted for age because there was a significant difference between the two groups at baseline. In addition to family-level and participant-level random intercepts, models for cortisol and cortisone also included a day of assessment level random intercept, because days were clustered within participants, and participants were clustered within families. We report mean baseline levels of outcomes, estimated within-group mean change in outcomes, and the intervention effect, which is the interaction between the group and the time variable from each of the linear or tobit mixed-effects models. All estimates were provided with 95% confidence intervals and analyses were conducted according to the intention-to-treat principles. We calculated Cohen’s d using the changes scores and standard deviations, and the number of participants in each group.
We found no violations of the assumptions of linear or tobit mixed-effects models. Statistical analyses were performed using STATA 17 using an α-level of 0.05 (two-sided).
Data availability
Deidentified data can be made available upon request for research purposes after a data handling agreement is made in accordance with the General Data Protection Regulation and the Danish Data Protection Act. Contact the corresponding author in case of data inquiries.
References
Heidi Amalie Rosendahl Jensen, M. D., Sofie Rossen Møller, Julie Ellegaard Ibáñez Román, Kamilla Kragelund, Anne Illemann Christensen og Ola Ekholm. Danskernes Sundhed - Den Nationale Sundhedsprofil 2021. (https://www.sst.dk/da/Udgivelser/2022/Danskernes-sundhed, 2022).
Goodwin, R. D., Weinberger, A. H., Kim, J. H., Wu, M. & Galea, S. Trends in anxiety among adults in the United States, 2008–2018: rapid increases among young adults. J. Psychiatr. Res. 130, 441–446 (2020).
Moreno-Agostino, D. et al. Global trends in the prevalence and incidence of depression:a systematic review and meta-analysis. J. Affect. Disord. 281, 235–243 (2021).
Yang, X. et al. Global, regional and national burden of anxiety disorders from 1990 to 2019: results from the Global Burden of Disease Study 2019. Epidemiol. Psychiatr. Sci. 30, e36 (2021).
Eurostat. 7.2% of people in the EU suffer from chronic depression. https://ec.europa.eu/eurostat/web/products-eurostat-news/-/edn-20210910-1 (2019).
Public Health Agency of Sweden. Statistik om ungas psykiska hälsa. https://www.folkhalsomyndigheten.se/livsvillkor-levnadsvanor/psykisk-halsa-och-suicidprevention/statistik-psykisk-halsa/statistik-om-ungas-psykiska-halsa/ (2022).
Makin, S. Searching for digital technology’s effects on well-being. Nature 563, S138–s140 (2018).
Orben, A. & Przybylski, A. K. The association between adolescent well-being and digital technology use. Nat. Hum. Behav. 3, 173–182 (2019).
Twenge, J. M., Haidt, J., Joiner, T. E. & Campbell, W. K. Underestimating digital media harm. Nat. Hum. Behav. 4, 346–348 (2020).
Wang, X., Li, Y. & Fan, H. The associations between screen time-based sedentary behavior and depression: a systematic review and meta-analysis. BMC Public Health 19, 1524 (2019).
Vahedi, Z. & Saiphoo, A. The association between smartphone use, stress, and anxiety: a meta-analytic review. Stress Health 34, 347–358 (2018).
Parry, D. A. et al. A systematic review and meta-analysis of discrepancies between logged and self-reported digital media use. Nat. Hum. Behav. 5, 1535–1547 (2021).
van Wezel, M. M. C., Abrahamse, E. L. & Vanden Abeele, M. M. P. Does a 7-day restriction on the use of social media improve cognitive functioning and emotional well-being? Results from a randomized controlled trial. Addict. Behav. Rep. 14, 100365 (2021).
Przybylski, A. K., Nguyen, T.-V. T., Law, W. & Weinstein, N. Does taking a short break from social media have a positive effect on well-being? Evidence from three preregistered field experiments. J. Technol. Behav. Sci. https://doi.org/10.1007/s41347-020-00189-w (2021).
Brailovskaia, J., Ströse, F., Schillack, H. & Margraf, J. Less Facebook use – More well-being and a healthier lifestyle? An experimental intervention study. Comput. Hum. Behav. 108, 106332 (2020).
Hall, J. A., Xing, C., Ross, E. M. & Johnson, R. M. Experimentally manipulating social media abstinence: results of a four-week diary study. Media Psychol. 24, 259–275 (2021).
Tromholt, M. The Facebook experiment: quitting Facebook leads to higher levels of well-being. Cyberpsychol. Behav. Soc. Netw. 19, 661–666 (2016).
Hellhammer, D. H., Wüst, S. & Kudielka, B. M. Salivary cortisol as a biomarker in stress research. Psychoneuroendocrinology 34, 163–171 (2009).
Bae, Y. J. et al. Salivary cortisone, as a biomarker for psychosocial stress, is associated with state anxiety and heart rate. Psychoneuroendocrinology 101, 35–41 (2019).
Chrousos, G. P. Stress and disorders of the stress system. Nat. Rev. Endocrinol. 5, 374–381 (2009).
Adam, E. K. et al. Diurnal cortisol slopes and mental and physical health outcomes: a systematic review and meta-analysis. Psychoneuroendocrinology 83, 25–41 (2017).
Stalder, T. et al. Assessment of the cortisol awakening response: expert consensus guidelines. Psychoneuroendocrinology 63, 414–432 (2016).
Chida, Y. & Steptoe, A. Cortisol awakening response and psychosocial factors: a systematic review and meta-analysis. Biol. Psychol. 80, 265–278 (2009).
Adam, E. K., Hawkley, L. C., Kudielka, B. M. & Cacioppo, J. T. Day-to-day dynamics of experience-cortisol associations in a population-based sample of older adults. Proc. Natl Acad. Sci. USA 103, 17058–17063 (2006).
Nabi, R. L., Prestin, A. & So, J. Could watching TV be good for you? Examining how media consumption patterns relate to salivary cortisol. Health Commun. 31, 1345–1355 (2016).
Teychenne, M. et al. The association between sedentary behaviour and indicators of stress: a systematic review. BMC Public Health 19, 1357 (2019).
Jackson, S. E. et al. Sedentary behaviour and chronic stress in old age: a cross-sectional analysis of TV viewing and hair cortisol concentrations. Psychoneuroendocrinology 109, 104375 (2019).
Teychenne, M., Olstad, D. L., Turner, A. I., Costigan, S. A. & Ball, K. Sedentary behaviour and hair cortisol amongst women living in socioeconomically disadvantaged neighbourhoods: a cross-sectional study. Int. J. Environ. Res. Public Health 15, 586 (2018).
Afifi, T. D., Zamanzadeh, N., Harrison, K. & Acevedo Callejas, M. WIRED: the impact of media and technology use on stress (cortisol) and inflammation (interleukin IL-6) in fast paced families. Comput. Hum. Behav. 81, 265–273 (2018).
Rasmussen, M. G. B. et al. Short-term efficacy of reducing screen media use on physical activity, sleep, and physiological stress in families with children aged 4-14: study protocol for the SCREENS randomized controlled trial. BMC Public Health 20, 380 (2020).
Pedersen, J. et al. Effects of limiting recreational screen media use on physical activity and sleep in families with children: a cluster randomized clinical trial. JAMA Pediatr. 176, 741–749 (2022).
Borek, A. J. & Abraham, C. How do small groups promote behaviour change? An integrative conceptual review of explanatory mechanisms. Appl. Psychol. Health Well Being 10, 30–61 (2018).
Hoffman, C. J. et al. Effectiveness of mindfulness-based stress reduction in mood, breast- and endocrine-related quality of life, and well-being in stage 0 to III breast cancer: a randomized, controlled trial. J. Clin. Oncol. 30, 1335–1342 (2012).
Topp, C. W., Ostergaard, S. D., Sondergaard, S. & Bech, P. The WHO-5 Well-Being Index: a systematic review of the literature. Psychother. Psychosom. 84, 167–176 (2015).
Thomée, S., Dellve, L., Härenstam, A. & Hagberg, M. Perceived connections between information and communication technology use and mental symptoms among young adults - a qualitative study. BMC Public Health 10, 66 (2010).
Verduyn, P., Ybarra, O., Résibois, M., Jonides, J. & Kross, E. Do social network sites enhance or undermine subjective well-being? A critical review. Soc. Issues Policy Rev. 11, 274–302 (2017).
Hinkelmann, K. et al. Association between cortisol awakening response and memory function in major depression. Psychol. Med. 43, 2255–2263 (2013).
Vreeburg, S. A. et al. Major depressive disorder and hypothalamic-pituitary-adrenal axis activity: results from a large cohort study. Arch. Gen. Psychiatry 66, 617–626 (2009).
Orne, M. T. On the social-psychology of the psychological experiment - with particular reference to demand characteristics and their implications. Am. Psychol. 17, 776–783 (1962).
Stoffel, M., Neubauer, A. B. & Ditzen, B. How to assess and interpret everyday life salivary cortisol measures: a tutorial on practical and statistical considerations. Psychoneuroendocrinology 133, 105391 (2021).
Campbell, M. K., Piaggio, G., Elbourne, D. R. & Altman, D. G. Consort 2010 statement: extension to cluster randomised trials. BMJ 345, e5661 (2012).
Pedersen, J. et al. Recreational screen media use in Danish school-aged children and the role of parental education, family structures, and household screen media rules. Prev. Med. 155, 106908 (2022).
Schmidt, M., Pedersen, L. & Sørensen, H. T. The Danish Civil Registration System as a tool in epidemiology. Eur. J. Epidemiol. 29, 541–549 (2014).
Klakk, H. et al. The development of a questionnaire to assess leisure time screen-based media use and its proximal correlates in children (SCREENS-Q). BMC Public Health 20, 664 (2020).
Kristensen, P. L. et al. Criterion validity of a research-based application for tracking screen time on android and iOS smartphones and tablets. Comput. Hum. Behav. Rep. 5, 100164 (2022).
Sischka, P. E., Costa, A. P., Steffgen, G. & Schmidt, A. F. The WHO-5 well-being index – validation based on item response theory and the analysis of measurement invariance across 35 countries. J. Affect. Disord. Rep. 1, 100020 (2020).
McNair, D. M., Lorr, M. & Droppleman, L. F. Profile of mood states. (1971).
Nyenhuis, D. L., Yamamoto, C., Luchetta, T., Terrien, A. & Parmentier, A. Adult and geriatric normative data and validation of the profile of mood states. J. Clin. Psychol. 55, 79–86 (1999).
Sorensen, S. O., Pedersen, J., Rasmussen, M. G., Kristensen, P. L. & Grontved, A. Feasibility of home-based sampling of salivary cortisol and cortisone in healthy adults. BMC Res. Notes 14, 406 (2021).
Adam, E. K. & Kumari, M. Assessing salivary cortisol in large-scale, epidemiological research. Psychoneuroendocrinology 34, 1423–1436 (2009).
Pruessner, J. C., Kirschbaum, C., Meinlschmid, G. & Hellhammer, D. H. Two formulas for computation of the area under the curve represent measures of total hormone concentration versus time-dependent change. Psychoneuroendocrinology 28, 916–931 (2003).
Rasmussen, M. G. B. et al. Feasibility of two screen media reduction interventions: Results from the SCREENS pilot trial. PLoS ONE 16, e0259657 (2021).
Twisk, J. & Rijmen, F. Longitudinal tobit regression: a new approach to analyze outcome variables with floor or ceiling effects. J. Clin. Epidemiol. 62, 953–958 (2009).
Acknowledgements
We would like to thank all families for their participation, and the Open Patient data Explorative Network for assisting with trial management.
Author information
Authors and Affiliations
Contributions
J.P. helped design the study, recruit participants, collect data, manage data flow, prepare data for analysis, conduct the statistical analyses, interpret data, and led the writing of the paper. M.G.B.R. helped design the study, recruit participants, collect data, and manage data flow. S.O.S. helped recruit participants, collect data, and manage data flow. S.R.M. helped collect data and manage data flow. L.G.O. helped design the study and achieve ethical approval. S.B. conceptualized and designed the study. P.L.K. conceptualized and designed the study and managed data flow. E.P. supervised the preparation of data for analysis, data analysis, and helped interpret data. A.G. conceptualized and designed the study, received funding, supervised data analysis, and helped interpret data. All authors reviewed and commented on the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pedersen, J., Rasmussen, M.G.B., Sørensen, S.O. et al. Effects of limiting digital screen use on well-being, mood, and biomarkers of stress in adults. npj Mental Health Res 1, 14 (2022). https://doi.org/10.1038/s44184-022-00015-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s44184-022-00015-6