Abstract
Land-applied municipal biosolids, produced from municipal wastewater treatment sludge, contributes to microplastics contamination in agroecosystems. The impacts of biosolids on microplastic concentrations in agricultural soil have been previously investigated, however, the potential for microplastics transport from biosolid-amended croplands has not been previously quantified. In this study, manure and biosolids were applied to field plots, runoff was collected following natural precipitation events and the potential of bacterial biofilm to grow on different microplastic morphologies was investigated. Higher concentrations of microplastics were detected in runoff from plots with land-applied biosolid in comparison with manure-amended and control plots. Fibers and fragments were the most frequently detected plastic morphologies in runoff, correlated with their decreased surface roughness. The potential of biosolids to contribute to microplastic contamination to U.S. surface waters was quantified which is among the first to quantify the potential for nonpoint source microplastic contamination of surface waters adjacent to agricultural production areas.
Similar content being viewed by others
Introduction
Municipal biosolids are often land applied as a fertilizer and soil conditioner1 with the rate of biosolid application depending on the annual crop nitrogen requirement2. Approximately 0.1% of agricultural land in the United States, or 99,000 hectares, is under biosolids application annually3. Although biosolids contain valuable nutrients, contaminants such as trace organic compounds, heavy metals, and pathogens may also be present and have the potential to impact downstream water quality4,5. More recently, biosolids have been demonstrated to contain microplastics (MPs) at concentrations of up to 286 particles/g of sludge (dry weight)6,7,8.
MPs, defined as polymer particles smaller than 5 mm, have been detected in air, soil, drinking water, as well as human and animal feces9,10,11,12,13. Environmental and health concerns related to MPs include their ability to sorb contaminants such as heavy metals14, and their susceptibility to biofilm growth15. For example, nickel and Cr (VI) which are among the most harmful and carcinogenic pollutants16, have shown the potential to be absorbed by MPs14. Pathogenic bacteria can attach to polyethylene MPs17. MPs also have the potential to be ingested by aquatic organisms in freshwater and marine environments18, decrease the growth rate of beneficial soil microorganisms19, and affect terrestrial organisms such as fungi20 by changing their community structure and functions21.
MPs have been shown to be transported through stormwater runoff in urban and suburban areas21,22 In a study conducted by Liu et al.23, the runoff was identified as the major source of MP in retention ponds. Stormwater is also a pathway for MPs to wastewater treatment plants (WWTP) in areas with combined sewer and stormwater collection systems24,25. Other routes for MPs to WWTPs include the use of personal care products and textile fibers from domestic wastewater26. In municipal WWTPs, over 95% of MPs are eventually removed and accumulated in biosolids27,28.
There are a limited number of studies that investigated the fate and transport of MPs in agricultural soils. Li et al.29, investigated the vertical transport of MPs in soil with vegetation and found that crop roots move MPs upwards, maintaining their presence in upper soil layers. Han et al.30, studied the effect of rain-induced runoff on the horizontal transport of plastic particles and found that smaller plastics with lower densities exhibited higher mobility. A less well-understood transport route for MPs to surface water is via land application of municipal biosolids. Although biosolids have the potential to act as a major source of MP pollution, to the best of our knowledge there are only a few studies that have assessed MPs accumulation in soil from land-applied biosolids and WWTP sludges31,32,33,34. Crossman et al.32, studied the effect of biosolids application on MP occurrence in soil and reported concentrations of MPs of 8.7 and 14 particles/g soil. They collected soil samples from areas with and without a biosolid application history and reported that biosolid applications resulted in MP export to the soil at all sampling sites. Schell et al.34 evaluated the horizontal transport of MPs in runoff from agricultural fields where biosolids were applied as fertilizer and demonstrated that MPs will be transported in runoff from biosolids-amended plots, however, this observation was based on a single field plot.
Further studies are needed to quantify the expected MP concentrations in runoff, as well as investigate how different MP morphologies are transported from croplands. Currently, there are no estimates about the potential release of MP from US cropland from land-applied biosolids. The objectives of this study were to determine the effect of land application of biosolids on the MP concentration in agricultural runoff, investigate preferential transport of different MP morphologies, compare biosolids and manure as two soil amendments that could serve as sources of MPs, and calculate the potential for MP transport from corn and soybean production areas within the United States.
Results
Concentration and morphology of MP in manure, biosolids, soil, and runoff samples
The MPs detected in cattle manure had an average concentration of 1.5 ± 0.2 particle/g (dry weight), 56% were fragments and 44% were fibers. No other morphologies were found. The average MP concentration in biosolids was 9.1 ± 1.7 particle/g (dry weight). The total number of MPs applied to each of the biosolid and manure-amended plots were estimated as 1.8 × 106 and 2.9 × 105 particles, respectively The distribution of the MPs identified in biosolids based on morphology was identified (Fig. 1) and more than 80% of the particles were either fibers or fragments.
Cylindrical MPs with a high length to diameter ratio were identified as fibers. Sheet-like MPs with low thickness and high surface area were identified as films. Spherical and granular MPs were identified as beads. Sponge-like mass structures were identified as foams. Any other MPs with irregular shapes were identified as fragments.
On average, the concentration of MPs found in the topsoil layer of the control, manure-amended, and biosolid-amended plots were 0.9 ± 0.1 particle/g (dry weight), 1.1 ± 0.3 particle/g (dry weight) and 2.6 ± 0.6 particle/g (dry weight), respectively. Table S1 shows the average MP concentrations for the top (0–5 cm) and bottom layers (5–15 cm) of the soil. The MPs concentration in the soil before biosolid/manure application was similar to what was found after the conclusion of the experiment in the control plots. An image of the MPs detected in amended biosolids and runoff samples are shown in Fig. S1.
MP concentrations in runoff were determined following five natural rainfall events (Fig. 2). The MP concentrations in runoff ranged from 10−14 particles/L (control plots), 16−31 particles/L (biosolid plots), and 8−20 particles/L (manure plots). The pH of runoff samples ranged from 6.5 to 7.5. The volume of runoff collected during each sampling event (Table S2) was used with the MP concentrations to estimate the total number of MPs transported in runoff (Supplementary Data 1). An estimated 7000 MPs, which represented 0.4% of the total MPs applied to the biosolid-amended plots, were accounted for in the runoff. The percent transport was 0.3% for manure-amended plots. Details of these calculations are provided in Supplementary Data 1. The amounts of precipitation in the sampling events ranged from 1.7 to 5.1 cm (Table S3), and were higher than the average annual rainfall from 2010 to 2020 (Table S4) as shown in Fig. 2, there is no clear relationship between rainfall intensity and MP concentration. No MPs were detected in rainfall samples collected on July 21st and 27th indicating that wet deposition did not contribute appreciable MPs to the field plots.
Statistical analysis of data
When examining the time by treatment component for MP concentrations in plot runoffs, a p value of 0.0562 was observed, prompting an investigation of the comparison of the treatment means at each time point (Table S5). Additionally, the plot of the treatment means by time which is presented in Fig. S2 shows that there is a clear interaction over time, and therefore, simple effects must be examined.
Although Fig. 2 shows that the MP concentrations in runoff from biosolids-amended plots is higher than from the control and manure plots at all the sampling points, when examining the comparison of means at each time point, there are no significant differences among the treatments on July 21, July 27, or August 17 sampling points. There are no significant differences between manure and control at any of the time points. However, on July 30, a significant difference between biosolids and manure was observed. On September 10, a significant difference between biosolids and manure was observed, as well as a significant difference between biosolids and control. The 95% confidence intervals and p values are presented in Table S6. The mean MP concentration in runoff from biosolids-amended plots is significantly higher than the manure-amended plots on July 30 and September 10, and the control plots on September 10. For the soil data, the p value for the test of the difference in means (standard F test) was 0.0966 (Table S7) which indicates that there is marginal evidence of a difference among the treatment means. However, the post hoc pairwise comparisons of the means did not yield significant differences amongst any of the treatments (at the 0.05 level).
The effect of morphology on MP transport
The MPs morphology distribution detected in runoff samples from all plots was on average 33% fibers, 63% fragments, 3% films, and 2% foam. No beads were found in the runoff samples (Fig. 3). The SEM images from MPs found in biosolids (Fig. 4a), showed the beads have a rough surface, facilitating the attachment and growth of biofilm on their surfaces, consistent with results from ref. 35, who demonstrated biofilm growth on MPs beads detected in surface water. The surface of films and foams are also rough (Fig. 4b), although the growth of bacterial biofilm on their surface was less than what was observed on beads. In contrast, the surface roughness of fibers and fragments was considerably lower than the other MPs morphologies, likely leading to their greater transport by runoff. In order to confirm the theory of bacterial biofilm on the surface of beads, which had increased surface roughness, MPs were extracted from biosolids and were stained with propidium iodide (Figs. S3, S4). The areas highlighted in red were suspected to have bacterial biofilm and confirmatory SEM images were taken from the same area of the stained MPs (Figs. S5,S6). The confocal and SEM images demonstrate the growth of bacterial biofilm on beads with rough surfaces, in comparison to fibers and fragments with relatively smooth surfaces.
Plastic characterization
Only two out of 27 particles (7%) were identified as non-plastic organics, which validates the efficiency of the oxidation and extraction method used for biosolids samples. Of the particles identified as plastics (including fragments, films, beads, and foams), polyethylene (PE) was the most common plastic type found in the biosolids at 34% (Fig. 5). Polyethylene terephthalate (PET) and polypropylene (PP) were the second and third dominant plastics type identified in biosolid samples at 17 and 13%, respectively. Other plastic types identified in biosolids were acrylic (PC), polystyrene (PS), polyvinyl chloride (PVC), polyester (PES), nylon (NY), and phenol resin (PR). The spectra for the four most commonly detected MPs can be found in Fig. S7.
The pie chart shows a total of nine different polymeric structures that were identified as MPs extracted from dry biosolid samples, namely polyethylene terephthalate (PET), polypropylene (PP), acrylic (PC), polystyrene (PS), polyvinyl chloride (PVC), nylon (NY), polyester (PES), phenol resin (PR), and polyethylene (PE).
Estimation of released MPs by runoff from corn and soybean production areas
Corn and soybean were selected as the model crops to estimate the quantity of MPs released by runoff from land-applied biosolids (Fig. 6). Based on our calculations a total of 64 billion MPs particles are potentially transported to surface water via runoff from the biosolid applied to corn and soybean fields in the United States each year. Details for calculating the number of MPs at risk of being transported via runoff from biosolid-amended corn and soybean fields in the United States are presented in Supplementary Data 2. As expected, Iowa, Illinois, Minnesota, and Nebraska were the top states with the potential for MP transport from corn and soybean fields, with 8.0, 7.7, 5.7, and 5.2 billion MP particles, respectively. The same approach was separately applied to the state of Nebraska one of the major producers of corn and soybean in the United States with almost 15.4 million acres of corn and soybean fields. To have a more detailed analysis, the data was separated by the county to investigate the release of 5.2 billion MPs particles with increased local precision (Fig. 7). Further details for the calculations for Nebraska counties can be found in Supplementary Data 3.
The applied biosolids was calculated by assuming 7.5 and 12.5 dmt/acre of biosolids were applied on 0.1% of all the corn and soybean fields, respectively. By using the average concentration of 9.1 particle/g, the total number of MPs in all applied biosolids was estimated. Since 0.4% of all the MP particles was found to be transferred via runoff in this study, we used this number to estimate the number of transported MPs per state.
Discussion
The average concentration of MPs in biosolids is within the range of 4.1 to 286 particle/g (dry weight) reported in previous studies8,32. The MP concentrations detected in cattle manure were also consistent with several other studies36,37; which found MP concentrations in sheep, swine, and cattle manure ranging from 0.9 to 1.5 particle/g (dry weight). The lower concentrations of MP in manure indicate that prioritizing the use of animal manures over biosolids as alternatives to chemical fertilizers could lower the MP loading to agricultural fields.
The MP concentration in soils from the biosolids-amended plots and control plots are within the reported range of MP concentrations previously reported from soils with and without sludge amendment38,39. The concentration of MPs in the runoff from biosolids-amended plots ranged from 16–31 particle/L. When compared with MP concentrations in urban and suburban stormwater runoff (water phase) reported by other studies (Table S8), it appears that MP loading to surface waters from biosolids-amended cropland is a considerable source of terrestrial MPs to surface waters as the reported concentration of MP in the current study exceeds the MP concentration in stormwater runoff reported by several other studies. A discussion on the extraction methods used in this study for runoff, biosolid, manure, and soil samples is available in Supplementary Note 1.
Fragments and fibers were the most abundant MP shape types in biosolids observed in this study. Other studies investigating the occurrence of MPs in biosolids or WWTP sludges have found various morphologies. Nyang et al.8, reported the majority of the MPs detected in biosolids were either fragments or fibers and only one bead particle was identified, whereas Xu et al.40, found microbeads/pellets to be the dominant MP type in sludge. The Microbead-Free Waters Act in 2015 restricted plastic microbeads in all cosmetic and non-prescription products41, which may result in fewer detections of microbeads after this date.
Fibers and fragments, which were the primary morphologies detected in biosolids, were also found more frequently in a runoff. Foams, beads, and films were detected in the biosolids at 2, 5, and 10%, respectively, but were rarely detected in the runoff, which may be attributed to the entrapment of beads and films in biosolid organic matter due to their increased surface roughness. Fibers and fragments were also the most abundant morphologies detected in the runoff from control and manure plots. The presence of MPs detected in control samples may have originated from airborne MPs transported via wind or wet/dry deposition (although MPs were not detected in two rainfall samples analyzed), MPs used in fertilizers, or MPs from other agricultural practices. Bullard et al.42, demonstrated fibers were highly susceptible to transport via wind and several studies previously confirmed the occurrence of MPs in agricultural land43,44.
The three polymer types found in the greatest percentages in biosolids were PE, PET, and PP (Fig. 5). Crossman et al.32, reported PE and PP as the most abundant MPs detected in biosolids from two different providers. Nyang et al.8, reported that the most commonly detected MPs from WWTP sludge and biosolids was PES followed by polyvinyl acetate and PE. In another study, Li et al.45, revealed that the majority of the particles extracted from WWTP sludge and identified as MPs; were polyolefin, acrylic fibers, PE, and polyamide. PE MPs which are frequently observed in terrestrial and aqueous environments with various morphologies46,47, are among the most abundant polymer types found in biosolids8,32. Stormwater, a contributor to WWTP influent, also contains high concentrations of PE particles48. PET and PP are also shown to enter WWTP influent and partition into sludge that is later transformed into biosolids49,50. PS is another polymer widely used in packaging51 and has also been detected in stormwater runoff52,53 and marine and terrestrial environments54,55. Because of the high PS production volume, relatively low density, and demonstrated occurrence in the environment, we expected to detect PS in biosolids at a higher frequency than 8%. PS was also not frequently detected in WWTP sludges in prior studies8,32,56. The additional sources for PS MPs in the environment merits further investigation.
Corn and soybean are the most commonly planted crops across the United States57 and these two crops comprise almost 70% of harvested cropland acres in the United States58. Therefore, choosing corn and soybean as model crops could provide a realistic baseline estimate of MPs at risk of transport from biosolid-applied agricultural land. The Midwestern US corn belt may be a major contributor to MP contamination due to land application of biosolids in intense agricultural production regions. The total number of MPs transported to the environment from biosolids could be even higher considering other biosolid end uses in addition to corn and soybean production59. It is important to note that our estimates of MPs in runoff are generalized across the US based on runoff concentrations obtained from a specific field location. Environmental factors such as soil texture, vegetation, slope, and rainfall frequency and intensity, all affect contaminant transport by runoff from the fertilized croplands60,61,62 and are expected to also influence MP transport in the runoff. Rainfall intensity has been shown to considerably increase the transport of nutrients and sediments, while vegetation coverage decreases transport60,62. It has also been demonstrated that soil moisture, clay content, and slope are in direct proportion with the risk of pesticide and nutrient transport from amended farmlands61,63. In the future, estimates of MP transport from corn and soybean production areas should be refined to include these variables.
The transport of MPs to agricultural freshwater streams may be a notable contributor to MP occurrence in marine systems. Moreover, as shown by ref. 64, MPs can also accumulate in river sediments over time and serve as a continuous source of MPs. Although in this study no vertical transport of MPs through the soil was observed over the study period, other studies have observed MP transport through soil layers and showed that depending on soil characteristics, crop type, and rainfall duration, MPs have the ability to migrate into deeper soil layers29,32,65. Therefore, a portion of the smaller MPs may be transported through soil horizons resulting in groundwater contamination66.
This study provides evidence that runoff from fields with land-applied biosolids transports MPs to surface water in agricultural regions, which may ultimately influence the health of aquatic organisms67,68 and impact the quality of drinking water systems using surface water as a water supply source. Best management practices commonly used in agriculture for removing dissolved and particulate contaminants from agricultural runoff, such as constructed wetlands or grass buffer strips, should be evaluated for their potential to remove MPs. Additional research should focus on the origin and fate of plastics in other agricultural production systems (e.g., irrigation or mulch film)44 and to better quantify further impacts to surface water and soil quality in agroecosystems as well as the potential contributions of understudied agricultural production processes to global MP contamination”.
Methods
Plot construction
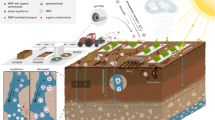
Six plots with dimensions of 3.6 m × 10 m were constructed at the University of Nebraska’s Rogers Memorial Farm located 19 km east of Lincoln, NE (Fig. 8). The soils in the area are classified as Aksarben silty clay loam (fine, smectitic, mesic Typic Argiudolls) that are deep, moderately well-drained, and formed in loess material. Soil characteristics of the six plots are presented in Tables S9–S14. Each plot was surrounded on three sides by galvanized sheet metal strips which extended to a depth of 10 cm into the soil. The plot slope was 4%. Runoff was collected in a metal trough located at the downslope end of each plot which conveyed runoff into a metal collection tank.
Panel (a) is the illustration of the constructed plots that were located at Rogers Memorial Farm, Lincoln, Nebraska. Panel (b) shows the schematic of the constructed plots. Galvanized aluminum collection buckets were placed inside the collection tanks below the pipe, and grab samples were taken from the buckets. The contents of the buckets were added to each tank and the total collected runoff volume from each plot was measured.
Biosolids (Table S15) obtained from a wastewater treatment facility in the area were applied to meet the typical 1-year nitrogen requirement for corn on two of the six plots (177 kg biosolids per plot which equals 4 dmt/acres). The facility with secondary treatment and ~27.6 × 106 m3 of wastewater per year, produced biosolid from sludges from the primary and secondary treatment processes. The solids were processed by anaerobic digestion and then dewatered prior to land application.
The same mass of manure (Table S16) obtained from an operating cattle production facility in Nebraska was applied to two other plots, while two additional plots were maintained as controls and did not receive any biosolids or manure, although they did receive chemical fertilizer previously. The applied materials (biosolid and manure) were manually added to the plots and were evenly distributed using rakes. The plots were arranged one meter apart in a random fashion. Sorghum was planted in the plots following biosolid and manure application.
Sample collection
Grab samples of well-mixed runoff were collected from each plot in 250 mL glass jars following natural precipitation events of more than 18 mm rainfall that occurred between July and September 2020. Sampling occurred immediately after the precipitation events and in order to homogenize the collected runoff, the contents of each tank was vigorously mixed using a wooden broom for at least 2 min and during mixing, grab samples were taken from the middle layer of the runoff suspension. After sampling, the contents of each tank were drained and the tanks were cleaned after each precipitation event. The volume and pH of each sample were recorded. Jars were covered with aluminum foil and stored at 4 °C until analysis. Biosolids and manure were also collected at the time of application in a sterilized glass jar, covered with aluminum foil, and dried in the oven at 75 °C. Soil cores were collected in triplicate from all the plots using metal liners before biosolids and manure application, and at the end of the experiments. The cores were then divided between 0–5 cm and 5–15 cm depths, stored in clean glass jars covered with aluminum, and dried in an oven at 75 °C. Composite soil samples for each plot were created by mixing the dried triplicate samples from the same depth, which resulted in two samples for each plot at two depths.
Microplastics extraction methods
Runoff samples
Runoff samples were dried at 70 °C until ~10 mL volume remained. To oxidize the samples, 15 mL of 0.05 M iron (Fe(II)) solution, and 15 mL H2O2 were added to each sample and held at 70 °C for 4 h. Samples were mixed using a glass rod every 15 min and then sonicated for 1 h. The container walls were rinsed every 15 min using 5 mL of deionized (DI) water. Samples were held at room temperature for 24 h and density separated. The supernatant was filtered through 12-μm glass fiber filter paper. To separate any of the remaining MPs in the runoff solution, 80 mL of saturated sodium chloride (NaCl) solution (ρ = 1.17 g/cm3) was added and the jars were vigorously shaken after being vortexed. Jars were kept for 24 h, density separated, and filtered. The procedure was repeated using saturated zinc bromide (ZnBr2) solution (ρ = 1.72 g/cm3). MPs were counted on the filters using a Nikon SMZ25 Stereo Dissecting Microscope (Nikon Corporation, Japan). The recovery rate for this separation method was evaluated. Briefly, MP particles from different polymer types (incl. Teflon, low-density PE, PS foam, PP, polyurethane, NY, PVC), shapes (film, fragment, and fiber), and densities (0.05–2 g/cm3) were mechanically generated. Particles were sieved and the MPs passing a 1 mm mesh size were evaluated. DI water (200 mL) was added into 250 mL sterilized glass jars and spiked in triplicate with 20 MP particles. Dried soil (1 g) was added to each jar to simulate suspended solids and the contents were mixed using a glass rod. The average recovery for MPs using this method was 88 ± 1.6%. This experiment to evaluate recovery rate was repeated with particles passing a 500 μm sieve, and found to be 88 ± 3.3%.
Biosolid, manure, and soil samples
The method used for analyzing biosolids was a modification of the procedure described by ref. 31, with an oxidation step added to the procedure because matrices such as soil, manure, and biosolids with high amounts of organics could entrap plastic particles, and therefore it is crucial to add an oxidation step to remove organics and release all the plastic particles. Dried biosolid (5 g) or soil samples were placed in 50 mL glass centrifuge tubes. To increase the recovery rate, the procedure was conducted inside a single tube minimizing the transfer of contents between different glassware. To oxidize, 4 mL of 0.05 M Fe(II) solution was added followed by 4 mL of H2O2, the contents were vigorously mixed using a glass rod to homogenize. The tubes were left at room temperature under a fume hood for 30 min and held at 70 °C for 3 h, mixing every 15 min. To minimize the risk of damaging MPs due to continued heat exposure, the temperature inside the tube was checked approximately every 5 min for the first 30 min, and every 15–20 min for the next 2.5 h. If the tube contents exceeded 70 °C, the tubes were placed inside a cold-water bath for 2 min. After oxidation, 30 mL of DI water was added and tubes were sonicated for 30 min to release any particles attached to the tubes during drying. The tubes were vortexed, vigorously shaken, and then centrifuged for 15 min at 2000 rpm or 672 rcf (Sorvall Legend RT, Thermo Electron Corp., Germany). To extract the separated MPs, the supernatant was filtered through a 12-μm glass fiber filter paper. The walls of the tubes were rinsed three times using DI water and filtered through the same filter. Then, the filter was placed in a clean petri dish. To extract the remaining MPs in the tube, 30 mL of NaCl solution was added to the precipitate and the tubes were vortexed, mixed using a glass rod, and vigorously shaken prior to a second centrifugation step and filtered through a new filter paper. For the third and final step of density separation, 30 mL of NaCl and ZnBr2 solutions in a ratio of 15:4 was used. All the filters were observed with the microscope and the number of collected MPs were counted.
The recovery rate of the method used for analyzing solid samples was also evaluated. Dried soil (5 g) was added to centrifuge tubes in triplicates. Each tube was then spiked with 10 MP particles generated as described above yielding an average efficiency of 90%. The procedure to measure the recovery rate was repeated with MP passing a 500-μm sieve, and found to be 93 ± 4.7%.
Statistical analysis
All data analysis were performed in SAS’ PROC GLIMMIX. The concentration of MP was presented as the number of plastic particles per gram of dry weight (for soil, manure, and biosolid) and the number of particles per liter for runoff samples. As there were five measurements per plot (one measurement after each of the five precipitation events), the study was analyzed as repeated measures. The covariance structure used was a first-order autoregressive structure (AR1) which was selected based on Akaike’s information criteria (Akaike 1974), with correction for small sample size fit statistics in comparison to a variety of other covariance structures. The degrees of freedom were adjusted using the Kenward Rogers adjustment to provide accurate standard errors. A comparison of treatment means at each time point was also performed for each plot to see if there is any significant difference between the concentration of MP in runoff of each plot in different time points. Lastly, an analysis of variance using a standard F test with a linear mixed model was conducted on the data related to MP concentration in the first soil layers of the plots to see if there is any difference between the MP concentrations in the first soil layers of biosolid, control, and manure plots. A post hoc pairwise comparison of means with Tukey’s adjustment was also conducted for multiple comparisons of the means.
Scanning electron microscopy and confocal laser scanning microscope system
To investigate the surface characteristics of the MPs, previously dried particles were examined using scanning electron microscopy (SEM). To prepare the samples, MPs that were previously extracted from biosolids were placed onto a SEM stub, air-dried overnight, and sputter-coated with a layer of chromium (2–3 nm thick) using a Desk V sputter (Denton Vacuum Inc.). All the images were collected under a Hitachi S4700 SEM.
In another round of imaging, separately extracted MPs from biosolids were first imaged under Confocal Laser Scanning Microscope System (Nikon A1-NiE upright confocal system). To prepare the samples for the confocal system, MPs were stained in the dark for 7–10 min with Propidium Iodide, which is a red fluorescent nuclear and chromosome counterstain and is not permeant to live bacteria, at 1:1000 in DI water. Later, the stained MPs were imaged by confocal microscope using the 488 laser line for green autofluorescence and the 560 laser line for the red Propidium Iodide in the sequence mode, using a 20x lens with a 3x digital zoom. As a confirmation of what was observed in confocal, the surface of the same particles needed to be imaged under SEM. To do so, the samples were dried in an oven at 40 °C for three days and later were mounted on the SEM stub for surface imaging. Wet peroxide oxidation was omitted for extraction of MPs that were observed with SEM due to the impact this process may have on the surface characteristics of the particles. Instead, 25 g of biosolids was placed in a clean beaker, DI water was added, the beaker was covered with aluminum and the content was stirred overnight followed by centrifugation, density separation using NaCl and ZnBr2 solutions, and filtration as described above.
Attenuated total reflectance-Fourier transform infrared spectroscopy (ATR-FTIR)
Particles (27) extracted from the biosolids larger than 300 µm, which accounts for 20% of the total MPs extracted from biosolids, were removed from the filters using micro tweezers. ATR-FTIR analysis was conducted using a Thermo Fisher iS-50 with a diamond ATR crystal. Measurements consisted of 128 scans at a resolution of 4 cm−1 in the range of 400 to 4000 cm−1. The data was baseline-corrected and the Thermo polymer library were used. The results of the spectra were later compared with the standard spectra of each polymer to confirm the identified types.
Quality assurance
Wet and dry deposition were monitored in the field and in the lab, respectively. A rain gauge was placed in the field to sample the rainfall and three empty glass petridishes with and without filter papers were placed in the lab area for microscopic analysis and extraction processes, respectively. Soil and runoff samples were always covered with aluminum foil immediately after sampling and when drying. All the glassware used to sample, extract, and store the filters were rinsed with DI water three times prior to use. In order to minimize the errors and the possibility of MP loss due to transfer, for solid samples, all the extraction steps were conducted inside a single glass centrifuge tube, and for runoff samples, all the steps were performed inside the same glass jar that was used for grab sampling.
Microplastic transport estimation
According to the US EPA’s Biosolids Technology Fact Sheet on land application of biosolids3 the typical amount of biosolids applied to corn and soybean fields per year is 5–10 and 5–20 dry metric tons (dmt) per acre, respectively. Therefore, we considered the average (7.5 and 12.5 dmt/acre) to be applied on corn and soybean fields, respectively. The US EPA also estimates biosolids are applied to approximately 0.1% of available agricultural lands in the United States annually69,70. Thus, by integrating these values with our findings on MPs abundance in biosolids and MPs transfer from land application of biosolids, we estimated the MPs at risk of release by runoff from corn and soybean fields in the United States and Nebraska as a model state with considerable corn and soybean production. The acreage of planted corn and soybean per state was acquired from the Farm Service Agency (FSA) of the USDA on January 3, 2022.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All data that were used for generating the charts in the main manuscript and supplementary information can be downloaded from the University of Nebraska-Lincoln data repository (https://doi.org/10.32873/unl.dr.20230111).
References
Roychand, R. et al. Recycling biosolids as cement composites in raw, pyrolyzed and ashed forms: a waste utilisation approach to support circular economy. J. Build. Eng. 38, 102199 (2021).
EPA. Process Design Manual: Land Application of Sewage Sludge and Domestic Septage (US EPA, 1995).
EPA. Biosolids Technology Fact Sheet, Land Application of Biosolids (US EPA, 2000).
Giudice, B. D., Young, T. M. & Bibb, J. P. Modification of GLEAMS for modeling movement of organic contaminants from land-applied biosolids. J. Environ. Manage. 234, 484–493 (2019).
Li, S., Li, R., Tang, Y. & Chen, G. Microwave-induced heavy metal removal from dewatered biosolids for cost-effective composting. J. Clean. Prod. 241, 118342 (2019).
Mohajerani, A. & Karabatak, B. Microplastics and pollutants in biosolids have contaminated agricultural soils: An analytical study and a proposal to cease the use of biosolids in farmlands and utilise them in sustainable bricks. Waste Manag. 107, 252–265 (2020).
Lares, M., Ncibi, M. C., Sillanpää, M. & Sillanpää, M. Occurrence, identification and removal of microplastic particles and fibers in conventional activated sludge process and advanced MBR technology. Water Res. 133, 236–246 (2018).
Harley-Nyang, D., Memon, F. A., Jones, N. & Galloway, T. Investigation and analysis of microplastics in sewage sludge and biosolids: a case study from one wastewater treatment works in the UK. Sci. Total Environ. 823, 153735 (2022).
Kosuth, M., Mason, S. A. & Wattenberg, E. V. Anthropogenic contamination of tap water, beer, and sea salt. PLoS ONE 13, e0194970 (2018).
Fahrenfeld, N. L., Arbuckle-Keil, G., Naderi Beni, N. & Bartelt-Hunt, S. L. Source tracking microplastics in the freshwater environment. Trends Anal. Chem. 112, 248–254 (2019).
Dong, S. et al. Transport characteristics of fragmental polyethylene glycol terephthalate (PET) microplastics in porous media under various chemical conditions. Chemosphere 276, 130214 (2021).
Luqman, A. et al. Microplastic contamination in human stools, foods, and drinking water associated with Indonesian coastal population. Environments 8, 138 (2021).
Yan, Z. et al. An efficient method for extracting microplastics from feces of different species. J. Hazard. Mater. 384, 121489 (2020).
Brennecke, D., Duarte, B., Paiva, F., Caçador, I. & Canning-Clode, J. Microplastics as vector for heavy metal contamination from the marine environment. Estuar. Coast. Shelf Sci. 178, 189–195 (2016).
Qi, K. et al. Uptake of Pb(II) onto microplastic-associated biofilms in freshwater: adsorption and combined toxicity in comparison to natural solid substrates. J. Hazard. Mater. 411, 125115 (2021).
Xu, R. et al. Removal of microplastics and attached heavy metals from secondary effluent of wastewater treatment plant using interpenetrating bipolar plate electrocoagulation. Sep. Purif. Technol. 290, 120905 (2022).
Lai, K. P., Tsang, C. F., Li, L., Yu, R. M. K. & Kong, R. Y. C. Microplastics act as a carrier for wastewater-borne pathogenic bacteria in sewage. Chemosphere 301, 134692 (2022).
Barboza, L. G. A. et al. Microplastics in wild fish from North East Atlantic Ocean and its potential for causing neurotoxic effects, lipid oxidative damage, and human health risks associated with ingestion exposure. Sci. Total Environ. 717, 134625 (2020).
Zhu, D. et al. Exposure of soil collembolans to microplastics perturbs their gut microbiota and alters their isotopic composition. Soil Biol. Biochem. 116, 302–310 (2018).
Souza Machado, A. A., Kloas, W., Zarfl, C., Hempel, S. & Rillig, M. C. Microplastics as an emerging threat to terrestrial ecosystems. Glob. Chang. Biol. 24, 1405–1416 (2018).
Fan, P., Tan, W. & Yu, H. Effects of different concentrations and types of microplastics on bacteria and fungi in alkaline soil. Ecotoxicol. Environ. Saf. 229, 113045 (2022).
Mak, C. W., Tsang, Y. Y., Leung, M. M.-L., Fang, J. K.-H. & Chan, K. M. Microplastics from effluents of sewage treatment works and stormwater discharging into the Victoria Harbor, Hong Kong. Mar. Pollut. Bull. 157, 111181 (2020).
Liu, F., Olesen, K. B., Borregaard, A. R. & Vollertsen, J. Microplastics in urban and highway stormwater retention ponds. Sci. Total Environ. 671, 992–1000 (2019).
Galafassi, S., Nizzetto, L. & Volta, P. Plastic sources: a survey across scientific and grey literature for their inventory and relative contribution to microplastics pollution in natural environments, with an emphasis on surface water. Sci. Total Environ. 693, 133499 (2019).
Simon, M., van Alst, N. & Vollertsen, J. Quantification of microplastic mass and removal rates at wastewater treatment plants applying focal plane array (FPA)-based Fourier transform infrared (FT-IR) imaging. Water Res. 142, 1–9 (2018).
Yang, L., Zhang, Y., Kang, S., Wang, Z. & Wu, C. Microplastics in freshwater sediment: a review on methods, occurrence, and sources. Sci. Total Environ. 754, 141948 (2021).
Lv, X. et al. Microplastics in a municipal wastewater treatment plant: fate, dynamic distribution, removal efficiencies, and control strategies. J. Clean. Prod. 225, 579–586 (2019).
Koutnik, V. S. et al. Unaccounted microplastics in wastewater sludge: where do they go? ACS ES&T Water 1, 1086–1097 (2021).
Li, H., Lu, X., Wang, S., Zheng, B. & Xu, Y. Vertical migration of microplastics along soil profile under different crop root systems. Environ. Pollut. 278, 116833 (2021).
Han, N., Zhao, Q., Ao, H., Hu, H. & Wu, C. Horizontal transport of macro- and microplastics on soil surface by rainfall induced surface runoff as affected by vegetations. Sci. Total Environ. 831, 154989 (2022).
Corradini, F. et al. Evidence of microplastic accumulation in agricultural soils from sewage sludge disposal. Sci. Total Environ. 671, 411–420 (2019).
Crossman, J., Hurley, R. R., Futter, M. & Nizzetto, L. Transfer and transport of microplastics from biosolids to agricultural soils and the wider environment. Sci. Total Environ. 724, 138334 (2020).
Wang, W., Ndungu, A. W., Li, Z. & Wang, J. Microplastics pollution in inland freshwaters of China: a case study in urban surface waters of Wuhan, China. Sci. Total Environ. 575, 1369–1374 (2017).
Schell, T. et al. Fate of microplastics in agricultural soils amended with sewage sludge: is surface water runoff a relevant environmental pathway? Environ. Pollut. 293, 118520 (2022).
Eriksen, M. et al. Microplastic pollution in the surface waters of the Laurentian Great Lakes. Mar. Pollut. Bull. 77, 177–182 (2013).
Beriot, N., Peek, J., Zornoza, R., Geissen, V. & Huerta Lwanga, E. Low density-microplastics detected in sheep faeces and soil: a case study from the intensive vegetable farming in Southeast Spain. Sci. Total Environ. 755, 142653 (2021).
Yang, J. et al. Abundance and morphology of microplastics in an agricultural soil following long-term repeated application of pig manure. Environ. Pollut. 272, 116028 (2021).
van den Berg, P., Huerta-Lwanga, E., Corradini, F. & Geissen, V. Sewage sludge application as a vehicle for microplastics in eastern Spanish agricultural soils. Environ. Pollut. 261, 114198 (2020).
Collivignarelli, M. C., Carnevale Miino, M., Caccamo, F. M. & Milanese, C. Microplastics in sewage sludge: a known but underrated pathway in wastewater treatment plants. Sustainability 13, 12591 (2021).
Xu, Q. et al. Investigation of the microplastics profile in sludge from China’s largest water reclamation plant using a feasible isolation device. J. Hazard. Mater. 388, 122067 (2020).
U.S. Food and Drug Administration (FDA). The microbead-free waters act of 2015: FAQs. https://www.fda.gov/drugs/laws-acts-and-rules/microbead-free-waters-act-2015-faqs (2022).
Bullard, J. E., Ockelford, A., O’Brien, P. & McKenna Neuman, C. Preferential transport of microplastics by wind. Atmos. Environ. 245, 118038 (2021).
Yu, L. et al. Distribution characteristics of microplastics in agricultural soils from the largest vegetable production base in China. Sci. Total Environ. 756, 143860 (2021).
Tian, L., Jinjin, C., Ji, R., Ma, Y. & Yu, X. Microplastics in agricultural soils: sources, effects, and their fate. Curr. Opin. Environ. Sci. Heal. 25, 100311 (2022).
Li, X. et al. Microplastics in sewage sludge from the wastewater treatment plants in China. Water Res. 142, 75–85 (2018).
You, H. et al. Adsorption–desorption behavior of methylene blue onto aged polyethylene microplastics in aqueous environments. Mar. Pollut. Bull. 167, 112287 (2021).
Duis, K. & Coors, A. Microplastics in the aquatic and terrestrial environment: sources (with a specific focus on personal care products), fate and effects. Environ. Sci. Eur. 28, 2 (2016).
Lutz, N., Fogarty, J. & Rate, A. Accumulation and potential for transport of microplastics in stormwater drains into marine environments, Perth region, Western Australia. Mar. Pollut. Bull. 168, 112362 (2021).
Edo, C., González-Pleiter, M., Leganés, F., Fernández-Piñas, F. & Rosal, R. Fate of microplastics in wastewater treatment plants and their environmental dispersion with effluent and sludge. Environ. Pollut. 259, 113837 (2020).
Horton, A. A. et al. Semi-automated analysis of microplastics in complex wastewater samples. Environ. Pollut. 268, 115841 (2021).
Black, J. E., Kopke, K. & O’Mahony, C. Towards a circular economy: using stakeholder subjectivity to identify priorities, consensus, and conflict in the Irish EPS/XPS market. Sustainability 11, 6834 (2019).
Piñon-Colin, T. et al. Microplastics in stormwater runoff in a semiarid region, Tijuana, Mexico. Sci. Total Environ. 704, 135411 (2020).
Ziajahromi, S., Drapper, D., Hornbuckle, A., Rintoul, L. & Leusch, F. D. L. Microplastic pollution in a stormwater floating treatment wetland: detection of tyre particles in sediment. Sci. Total Environ. 713, 136356 (2020).
Turner, A. Polystyrene foam as a source and sink of chemicals in the marine environment: an XRF study. Chemosphere 263, 128087 (2021).
Campanale, C. et al. Microplastics pollution in the terrestrial environments: poorly known diffuse sources and implications for plants. Sci. Total Environ. 805, 150431 (2022).
Ziajahromi, S. & Leusch, F. D. L. Systematic assessment of data quality and quality assurance/quality control (QA/QC) of current research on microplastics in biosolids and agricultural soils. Environ. Pollut. 294, 118629 (2022).
American Soybean Association. 2017 Soystats: A Reference Guide to Important Soybean Facts and Figures (American Soybean Association, 2017).
USDS Farm Service Agency (FSA). Crop acreage data. https://www.fsa.usda.gov/news-room/efoia/electronic-reading-room/frequently-requested-information/crop-acreage-data/index (2020).
Okoffo, E. D. et al. Release of plastics to Australian land from biosolids end-use. Environ. Sci. Technol. 54, 15132–15141 (2020).
Yao, Y., Dai, Q., Gao, R., Gan, Y. & Yi, X. Effects of rainfall intensity on runoff and nutrient loss of gently sloping farmland in a karst area of SW China. PLoS ONE 16, e0246505 (2021).
An, J., Geng, J., Yang, H., Song, H. & Wang, B. Effect of ridge height, row grade, and field slope on nutrient losses in runoff in contour ridge systems under seepage with rainfall condition. Int J Environ Res Public Health 18, 2022 (2021).
Fong, C. R., Gaynus, C. J. & Carpenter, R. C. Extreme rainfall events pulse substantial nutrients and sediments from terrestrial to nearshore coastal communities: a case study from French Polynesia. Sci. Rep. 10, 2955 (2020).
Tang, X., Zhu, B. & Katou, H. A review of rapid transport of pesticides from sloping farmland to surface waters: Processes and mitigation strategies. J. Environ. Sci. 24, 351–361 (2012).
Drummond, J. D. et al. Microplastic accumulation in riverbed sediment via hyporheic exchange from headwaters to mainstems. Sci. Adv. 8, eabi9305 (2022).
Liu, M. et al. Microplastic and mesoplastic pollution in farmland soils in suburbs of Shanghai, China. Environ. Pollut. 242, 855–862 (2018).
Samandra, S. et al. Microplastic contamination of an unconfined groundwater aquifer in Victoria, Australia. Sci. Total Environ. 802, 149727 (2022).
Karami, A., Romano, N., Galloway, T. & Hamzah, H. Virgin microplastics cause toxicity and modulate the impacts of phenanthrene on biomarker responses in African catfish (Clarias gariepinus). Environ. Res. 151, 58–70 (2016).
Warrack, S., Challis, J. K., Hanson, M. L. & Rennie, M. D. Microplastics flowing into Lake Winnipeg: densities, sources, flux, and fish exposures. Proc. Manitoba’s Undergrad. Sci. Eng. Res. 3, 5–15 (2017).
Lu, Q., He, Z. L. & Stoffella, P. J. Land application of biosolids in the USA: a review. Appl. Environ. Soil Sci. 2012, 1–11 (2012).
National Research Council. Biosolids Applied to Land: Advancing Standards and Practices (National Academies Press, 2002).
Acknowledgements
The authors gratefully acknowledge the assistance of Dr. You (Joe) Zhou, Beadle Center, University of Nebraska-Lincoln for assistance with SEM and confocal microscopy imaging.
Author information
Authors and Affiliations
Contributions
N.N.B. wrote the manuscript, performed sampling, analyzed the samples, performed Confocal and SEM imaging and ATR-FTIR, created the figures, and incorporated the edits and comments all other authors had during internal revisions. S.K. performed sampling, contributed to field work, created GIS maps, wrote the sections related to the estimation of MPs release from land-applied biosolids across US and Nebraska, and edited and reviewed the paper. T.M. conceived the study design, assisted with the plot construction, and edited and reviewed the paper. J.G. conceived of the study design, assisted with the plot construction, and edited and reviewed the paper. A.S. assisted with the plot construction, and edited and reviewed the paper. S.B.-H. initiated the research concept, contributed to the experimental planning, supervised the project, edited the paper drafts, and finalized the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Naderi Beni, N., Karimifard, S., Gilley, J. et al. Higher concentrations of microplastics in runoff from biosolid-amended croplands than manure-amended croplands. Commun Earth Environ 4, 42 (2023). https://doi.org/10.1038/s43247-023-00691-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s43247-023-00691-y
This article is cited by
-
Characterization and source apportionment of microplastics in Indian composts
Environmental Monitoring and Assessment (2024)
-
Effect of conventional and biodegradable microplastics on earthworms during vermicomposting process
Environmental Geochemistry and Health (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.