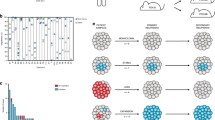
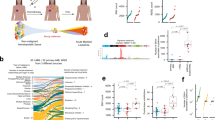
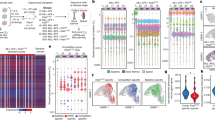
Abstract
Comparison of intratumor genetic heterogeneity in cancer at diagnosis and relapse suggests that chemotherapy induces bottleneck selection of subclonal genotypes. However, evolutionary events subsequent to chemotherapy could also explain changes in clonal dominance seen at relapse. We therefore investigated the mechanisms of selection in childhood B-cell precursor acute lymphoblastic leukemia (BCP-ALL) during induction chemotherapy where maximal cytoreduction occurs. To distinguish stochastic versus deterministic events, individual leukemias were transplanted into multiple xenografts and chemotherapy administered. Analyses of the immediate post-treatment leukemic residuum at single-cell resolution revealed that chemotherapy has little impact on genetic heterogeneity. Rather, it acts on extensive, previously unappreciated, transcriptional and epigenetic heterogeneity in BCP-ALL, dramatically reducing the spectrum of cell states represented, leaving a genetically polyclonal but phenotypically uniform population, with hallmark signatures relating to developmental stage, cell cycle and metabolism. Hence, canalization of the cell state accounts for a significant component of bottleneck selection during induction chemotherapy.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
Bulk and single-cell DNA- and RNA-seq and methylation array data (Supplementary Table 9) that support the findings of the present study have been deposited in the European Genome–phenome Archive (EGA), which is hosted by the European Bioinformatics Institute and the Centre for Genomic Regulation, under accession no. EGAS00001004407. Previously published WGS data from refs. 25,29 are available under accession identifiers EGAD00001000076 and EGAD00001000636. Source data are provided with this paper.
Code availability
Code used for this analysis is available at https://github.com/UCL-BLIC-analysis/Turati_NatCancer_2021.
References
Almendro, V., Marusyk, A. & Polyak, K. Cellular heterogeneity and molecular evolution in cancer. Annu. Rev. Pathol. Mech. Dis. 8, 277–302 (2013).
Anderson, K. et al. Genetic variegation of clonal architecture and propagating cells in leukaemia. Nature 469, 356–361 (2011).
Gerlinger, M. et al. Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N. Engl. J. Med. 366, 883–892 (2012).
Jamal-Hanjani, M. et al. Tracking the evolution of non-small-cell lung cancer. N. Engl. J. Med. 376, 2109–2121 (2017).
McGranahan, N. & Swanton, C. Clonal heterogeneity and tumor evolution: past, present, and the future. Cell 168, 613–628 (2017).
Notta, F. et al. Evolution of human BCR-ABL1 lymphoblastic leukaemia-initiating cells. Nature 469, 362–367 (2011).
Mullighan, C. G. et al. Genomic analysis of the clonal origins of relapsed acute lymphoblastic leukemia. Science 322, 1377–1380 (2008).
Dobson, S. M. et al. Relapse-fated latent diagnosis subclones in acute B lineage leukemia are drug tolerant and possess distinct metabolic programs. Cancer Discov. https://doi.org/10.1158/2159-8290.cd-19-1059 (2020).
Ding, L. et al. Clonal evolution in relapsed acute myeloid leukaemia revealed by whole-genome sequencing. Nature 481, 506–510 (2012).
Landau, D. A. et al. Evolution and impact of subclonal mutations in chronic lymphocytic leukemia. Cell 152, 714–726 (2013).
Clevers, H. The cancer stem cell: premises, promises and challenges. Nat. Med. 17, 313–319 (2011).
Good, Z. et al. Single-cell developmental classification of B cell precursor acute lymphoblastic leukemia at diagnosis reveals predictors of relapse. Nat. Publ. Gr. 24, https://doi.org/10.1038/nm.4505 (2018).
Nguyen, A., Yoshida, M., Goodarzi, H. & Tavazoie, S. F. Highly variable cancer subpopulations that exhibit enhanced transcriptome variability and metastatic fitness. Nat. Commun. 7, 11246 (2016).
Ramirez, M. et al. Diverse drug-resistance mechanisms can emerge from drug-tolerant cancer persister cells. Nat. Commun. 7, 10690 (2016).
Vander Velde, R. et al. Resistance to targeted therapies as a multifactorial, gradual adaptation to inhibitor specific selective pressures. Nat. Commun. 11, 2393 (2020).
Schuh, A. et al. Monitoring chronic lymphocytic leukemia progression by whole genome sequencing reveals heterogeneous clonal evolution patterns. Blood 120, 4191–4196 (2012).
Almendro, V. et al. Inference of tumor evolution during chemotherapy by computational modeling and in situ analysis of genetic and phenotypic cellular diversity. Cell Rep. 6, 514–527 (2014).
Greaves, M. Nothing in cancer makes sense except …. BMC Biol. 16, 22 (2018).
Greaves, M. Leukaemia ‘firsts’ in cancer research and treatment. Nat. Rev. Cancer 16, 163–172 (2016).
Cooper, S. L. & Brown, P. A. Treatment of pediatric acute lymphoblastic leukemia. Pediatr. Clin. North Am. 62, 61–73 (2015).
Panzer-Grumayer, E. R. et al. Rapid molecular response during early induction chemotherapy predicts a good outcome in childhood acute lymphoblastic leukemia. Blood 95, 790–794 (2000).
Theunissen, P. M. J. Normal and Malignant B-cells in Acute Lymphoblastic Leukemia: Insight into Clonal Distribution, Minimal Residual Disease and Normal B-cell Recovery. PhD thesis, Erasmus Univ. Rotterdam (2016).
Deleye, L. et al. Performance of four modern whole genome amplification methods for copy number variant detection in single cells. Sci. Rep. https://doi.org/10.1038/s41598-017-03711-y (2017).
Mullighan, C. G. et al. Genome-wide analysis of genetic alterations in acute lymphoblastic leukaemia. Nature 446, 758–764 (2007).
Papaemmanuil, E. et al. RAG-mediated recombination is the predominant driver of oncogenic rearrangement in ETV6-RUNX1 acute lymphoblastic leukemia. Nat. Genet. 46, 116–125 (2014).
Swaminathan, S. et al. Mechanisms of clonal evolution in childhood acute lymphoblastic leukemia. Nat. Immunol. 16, 766–774 (2015).
Hong, D. et al. Initiating and cancer-propogating cells in TEL-AML1-associated childhood leukemia. Science 319, 336–339 (2008).
Lutz, C. et al. Quiescent leukaemic cells account for minimal residual disease in childhood lymphoblastic leukaemia. Leukemia 27, 1204–1207 (2013).
Russell, L. J. et al. Characterisation of the genomic landscape of CRLF2-rearranged acute lymphoblastic leukemia. Genes Chromosom. Cancer 56, 363–372 (2017).
Ebinger, S. et al. Characterization of rare, dormant, and therapy-resistant cells in acute lymphoblastic leukemia. Cancer Cell 30, 849–862 (2016).
Jiao, Y., Widschwendter, M. & Teschendorff, A. E. Systems biology: a systems-level integrative framework for genome-wide DNA methylation and gene expression data identifies differential gene expression modules under epigenetic control. Bioinformatics 30, 2360–2366 (2014).
Laurenti, E. et al. The transcriptional architecture of early human hematopoiesis identifies multilevel control of lymphoid commitment. Nat. Immunol. 14, 756–763 (2013).
Vazquez-Santillan, K., Melendez-Zajgla, J., Jimenez-Hernandez, L., Martínez-Ruiz, G. & Maldonado, V. NF-κB signaling in cancer stem cells: a promising therapeutic target? Cell. Oncol. (Dordr.) 38, 327–339 (2015).
Tirosh, I. et al. Single-cell RNA-seq supports a developmental hierarchy in human oligodendroglioma. Nat. Publ. Gr. 539, 309–313 (2016).
Niyaz, M., Khan, M. S. & Mudassar, S. Hedgehog signaling: an Achilles’ heel in cancer. Transl. Oncol. 12, 1334–1344 (2019).
Li, L. & Bhatia, R. Stem cell quiescence. Clin. Cancer Res. 17, 4936–4941 (2011).
Kwon, J. S. et al. Controlling depth of cellular quiescence by an Rb-E2F network switch. Cell Rep. 20, 3223–3235 (2017).
Fujimaki, K. & Yao, G. Crack the state of silence: tune the depth of cellular quiescence for cancer therapy. Mol. Cell. Oncol. 5, e1403531 (2018).
Ji, Z. & Ji, H. TSCAN: pseudo-time reconstruction and evaluation in single-cell RNA-seq analysis. Nucleic Acids Res. 44, e117 (2016).
Rehman, S. K. et al. Colorectal cancer cells enter a diapause-like DTP state to survive chemotherapy. Cell, 284, 226–242 (2021).
Baersch, G. et al. Good engraftment of B-cell precursor ALL in NOD-SCID mice. Klin. Padiatr. 209, 178–185 (1997).
Kang, M. H. et al. Activity of vincristine, l-ASP, and dexamethasone against acute lymphoblastic leukemia is enhanced by the BH3-mimetic ABT-737 in vitro and in vivo. Blood 110, 2057–2066 (2007).
Samuels, A. L. et al. A pre-clinical model of resistance to induction therapy in pediatric acute lymphoblastic leukemia. Blood Cancer J. 4, e232–e232 (2014).
Szymanska, B. et al. Pharmacokinetic modeling of an induction regimen for in vivo combined testing of novel drugs against pediatric acute lymphoblastic leukemia xenografts. PLoS ONE 7, e33894 (2012).
Laurenti, E. & Göttgens, B. From haematopoietic stem cells to complex differentiation landscapes. Nat. Publ. Gr. 553, 418–426 (2018).
Quek, L. et al. Clonal heterogeneity of acute myeloid leukemia treated with the IDH2 inhibitor enasidenib. Nat. Med. 24, 1167–1177 (2018).
Böiers, C. et al. A human IPS model implicates embryonic B-myeloid fate restriction as developmental susceptibility to B acute lymphoblastic leukemia-associated ETV6-RUNX1. Dev. Cell 44, 362–377.e7 (2018).
Potter, N. E. et al. Single-cell mutational profiling and clonal phylogeny in cancer. Genome Res. 23, 2115–2125 (2013).
Moignard, V. et al. Decoding the regulatory network of early blood development from single-cell gene expression measurements. Nat. Biotechnol. 33, 269–276 (2015).
Ghorani, E. et al. The T cell differentiation landscape is shaped by tumour mutations in lung cancer. Nat. Cancer 1, 546–561 (2020).
Kowalczyk, M. S. et al. Single-cell RNA-seq reveals changes in cell cycle and differentiation programs upon aging of hematopoietic stem cells. Genome Res. 25, 1860–1872 (2015).
Sergushichev, A. A. An algorithm for fast preranked gene set enrichment analysis using cumulative statistic calculation. Preprint at bioRxiv https://doi.org/10.1101/060012 (2016).
Zhao, X. et al. Single-cell RNA-seq reveals the differentiation hierarchy of normal human bone marrow and a distinct transcriptome signature of monosomy 7 cells. Blood 128, 574 (2016).
Aryee, M. J. et al. Minfi: a flexible and comprehensive Bioconductor package for the analysis of Infinium DNA methylation microarrays. Bioinformatics 30, 1363–1369 (2014).
Fortin, J. P., Triche, T. J. & Hansen, K. D. Preprocessing, normalization and integration of the Illumina HumanMethylationEPIC array with minfi. Bioinformatics 33, 558–560 (2017).
Tian, Y. et al. ChAMP: updated methylation analysis pipeline for Illumina BeadChips. Bioinformatics 33, 3982–3984 (2017).
Zhou, W., Laird, P. W. & Shen, H. Comprehensive characterization, annotation and innovative use of Infinium DNA methylation BeadChip probes. Nucleic Acids Res. 45, e22 (2017).
Dedeurwaerder, S. et al. Evaluation of the Infinium Methylation 450K technology. Epigenomics 3, 771–784 (2011).
Leek, J. T., Johnson, W. E., Parker, H. S., Jaffe, A. E. & Storey, J. D. Relapse fated latent diagnosis subclones in acute B lineage leukaemia are drug tolerant and possess distinct metabolic programs. Bioinformatics 28, 882–883 (2012).
Alemu, E. Y., Carl, J. W., Bravo, H. C. & Hannenhalli, S. Determinants of expression variability. Nucleic Acids Res. 42, 3503–3514 (2014).
Ecker, S. et al. Genome-wide analysis of differential transcriptional and epigenetic variability across human immune cell types. Genome Biol. https://doi.org/10.1186/s13059-017-1156-8 (2017).
Smyth, G. K. limma: linear models for microarray data. in Bioinformatics and Computational Biology Solutions Using R and Bioconductor (eds Gentleman, R. et al.) 397–420 (Springer, 2005).
Ritchie, M. E. et al. Limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 43, e47 (2015).
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B 57, 289–300 (1995).
Phipson, B. & Oshlack, A. DiffVar: a new method for detecting differential variability with application to methylation in cancer and aging. Genome Biol. 15, 465 (2014).
Peters, T. J. et al. De novo identification of differentially methylated regions in the human genome. Epigenet. Chromatin 8, 2–16 (2015).
McLean, C. Y. et al. GREAT improves functional interpretation of cis-regulatory regions. Nat. Biotechnol. 28, 495–501 (2010).
Liberzon, A. et al. The molecular signatures database hallmark gene set collection. Cell Syst. 1, 417–425 (2015).
Hu, Y. & Smyth, G. K. ELDA: extreme limiting dilution analysis for comparing depleted and enriched populations in stem cell and other assays. J. Immunol. Methods 347, 70–78 (2009).
Lun, A. T. L., McCarthy, D. J. & Marioni, J. C. A step-by-step workflow for low-level analysis of single-cell RNA-seq data with Bioconductor. F1000Research 5, 2122 (2016).
Laurenti, E. et al. Cell stem cell hematopoietic stem cell function and survival depend on c-Myc and N-Myc activity. Stem Cell 3, 611–624.
Satija, R., Farrell, J. A., Gennert, D., Schier, A. F. & Regev, A. Spatial reconstruction of single-cell gene expression data. Nat. Biotechnol. 33, 495–502 (2015).
Acknowledgements
We thank all clinicians and the Blood Cancer UK Leukaemia Cell Bank for samples, Y. Guo for the assistance with the in vivo work, B. Gaal for sharing her MATLAB expertise, I. Titley and A. Ford for cell sorting and Ki67 analysis of single quiescent cells, and Fluidigm for technical support. We also thank the UCL-CI CRUK-funded flow cytometry, microscopy and genomics facility for access to instrumentation and for practical help with FACS analysis (W. Day, Y. Guo, B. Wilbourne and G. Morrow). We thank the UCL Legion and Myriad High Performance Computing Facility (Legion@UCL; Myriad@UCL), and associated support services, for the use of their services in completion of this work. The present study was supported by Blood Cancer UK project grants (to T.E., M.G. and S.E.W.J.) and partially supported by the Francis Crick Institute, which receives its core funding from Cancer Research UK (grant no. FC001202), the UK Medical Research Council (grant no. FC001202) and the Wellcome Trust (grant no. FC001202). V.A.T. was funded by grants from Children with Cancer UK and Blood Cancer UK, N.P was founded by Gabriel’s Angels, and J.A.G.-A. and L.C. were funded by the Cancer Research UK–UCL Centre Award. M.T. is a postdoctoral fellow supported by the European Union’s Horizon 2020 research and innovation program (Marie Skłodowska-Curie grant agreement no. 747852-SIOMICS), P.V.L. is a Winton Group Leader in recognition of the Winton Charitable Foundation’s support toward the establishment of the Francis Crick Institute and M.G. was supported by the Wellcome Trust. L.J.R was funded by the Kay Kendall Leukaemia Fund and Leuka, G.C. was funded by the Italian Association for Cancer Research grants (IG2017 no. 20564 to A.B. and IG2015 no. 17593 to G.C.). A.B. and G.C. were funded by the Comitato Maria Letizia Verga and S.E.W.J. by the UK Medical Research Council (grant no. MC_UU_12009/5).
Author information
Authors and Affiliations
Contributions
V.A.T., T.E., M.G. and S.E.J. conceived the study. V.A.T., M.L., J.B., A.W. and M.H. conducted the methodology. V.A.T., N.P., I.M. and A.D. did the investigations. J.A.G.-A., M.T., P.V.L., S.B. and J.H. carried out the formal analysis. J.A.G-A., A.W., S.E., C.D., C.J., C.L. and J.H. curated the data. S.E.J., G.W.H., A.B., L.R., S.I., P.A., G.C. and L.R. provided the resources. T.E., S.E.J and M.G. supervised the project. V.A.T., R.G., G.M., T.E. and M.G. wrote the manuscript. T.E. and M.G. acquired the funding.
Corresponding authors
Ethics declarations
Competing interests
M.L. was an employee of rgw Fluidigm Corporation at the time of the present study. The other authors declare no competing interests.
Additional information
Peer review information Nature Cancer thanks the anonymous reviewers for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Single cell whole genome sequencing analysis.
a, Representative traces of two whole-genome sequenced cells. Mapped reads are plotted across all chromosomes to highlight variations in copy number status as changes in the ploidy of the cells (n = 2). b, Diagram explaining the data processing pipeline. c, Scatterplots comparing the average normalized, GC-corrected and ploidy-adjusted counts per bins for pairs of copy-number events, where each dot represents an individual cell (n = 420 cells) harvested from Pt1 PDXs (across all treatment conditions and timepoints). Red lines divide the plots in 4 quadrants based on the nominal thresholds for calling each CNA.
Extended Data Fig. 2 Single cell gene expression analysis and mFISH analysis.
a, Representative plots describing the flow cytometry sorting approach used to collect CD34 + /CD38-/CD19 + cells. DAPI and forward scatter were used to identify viable cells (gate p1). To ensure that no dying cells were sorted, the p1 gate was further refined based on cells staining with the cell viability dye carboxyfluorescein succinimidyl ester (CFSE - FITC channel) (gate p2 – used to sort bulk cells). Positive vs negative combinations of specific antibodies were used to identify the populations of interest: CD34 + /CD38-/CD19 + were collected from gate p4. b, Representative heat map from a FLEXsix single-cell Q-PCR transcription panel showing the expression level of selected relevant transcripts in 11 single-cells sorted from the same patient as negative for CD38 expression. UBC is polyubiquitin-C used as a housekeeping gene control, CB8 is a single normal cord blood control cell. c and d, Phylogenetic trees showing the subclonal architectures of Pt10 and Pt11 bulk and CD34+/CD19+/CD38-/low sorted cells.
Extended Data Fig. 3 mFISH analysis.
a, Flow-sorting strategy used to assess engraftment and isolate human leukemic cells (hCD45+) from mouse cells (mCD45+). b, Representative Multicolor FISH image. The subclonal variegation within each sample was assessed using fluorochrome-labelled custom- made probes detecting AML1 (RUNX1), TEL (ETV6), p16 and PAX5 genes. c and d, Tables showing the copy number status associated with each of the subclones identified within Pt2 and Pt3 engrafted leukemias. e-f, BM engraftment data before and after chemotherapy for Pt2 (n = 11 control mice and n = 8 treated mice) and Pt3 (n = 10 control mice and n = 13 treated mice) at d0 and d28 as assessed by flow cytometry. Note engraftment is expressed here in the conventional way as hCD45 + /(hCD45+ +mCD45+; the >10 log cytoreduction achieved by chemotherapy is obscured in this representation due to the accompanying decrease in mouse CD45+ cells. Data are presented as mean values +/− SD. The statistical significance of each comparison (Paired t-test, two-tailed) is shown (p-value). g, Absolute numbers of human white blood cells (WBC) retrieved at d28 from both control (n = 5 mice) and treated mice (n = 8 mice) as quantified by the XP-300TM Automated Haematology Analyser (SYSMEX Co., Japan). Data are presented as mean values +/− SD. h, Dot plot showing treatment responses across different metastatic sites (brain, liver, spleen), with representative images of the reduction in spleen size following treatment (n = 2 control mice and n = 5 treated mice). Data are presented as mean values +/− SD. Error bars are shown for treated samples where n≥3. i, Treatment response kinetics of xenografts derived from high and low risk patients. Response was assessed by flow cytometry comparing human chimerism at d7 and d28 to d0 engraftment. In the plot each line represents a diffent patient (including some of the patients used for downstream analysis (pt1, pt2, pt3)). Data are presented as mean values+/− SD (n = 3 mice). j, Graph tracking the size of individual subclones from Pt2 xenografted leukemias before (d0 BM aspiration) and after treatment (d28 BM and d28 spleen). Multiple lines of the same color highlight the kinetic of the same subclone across different treated xenografts.
Extended Data Fig. 4 scWGS analysis.
a-b, TimeScape plots visualizing the clonal composition of two untreated (Ctrl) and two treated mice at 3 timepoints: i) in the primary diagnostic leukemia ii) at d0 in the xenograft (BM aspirate) and ii) at d28 in the xenograft (total BM harvest). c, Hierarchical classification of the main clones identified across all analyzed Pt1 specimens (primary diagnosis and xenografts d0-d28); provides a legend for the color coding used for the TimeScape plots in panels a and b (left) as well as Fig. 4d-e.
Extended Data Fig. 5 Population RNAseq analysis.
a-b, Graphical representations of the standard and multi-arm treatment models and resulting PCA analysis. In the standard model (a) matching d0 and d28 cells harvested from untreated (n = 37) and treated (n = 17) mice were analyzed. Untreated samples, shown in light blue, represent d0 cells from untreated mice and treated mice as well as d28 samples from untreated mice. In the multi-arm model (b), two consecutive rounds of transplantation and treatment (7 days) allowed the analysis of ‘acutely treated’ (n = 8 samples; green), ‘treatment withdrawn’ (n = 6 samples; dark yellow), ‘chronically treated’ (n = 5 samples; red), and ‘untreated control’ (n = 5 samples; dark blue) cells harvested from n = 3 mice each (see main text). ‘Relapse-like’ cells were also obtained through a separate arm of the experiment, whereby n = 3 mice were treated for 28 days and then let to relapse in situ in the absence of further treatment (6–8 weeks). c, Untreated and treated cells from primary recipients of Pt2 cells were transplanted into secondary recipients at three different limiting doses (100, 1*103, 1*104 cells). Engraftment (%) of BM, spleen and brain was assessed by flow cytometry and is shown in the dot plot (n = 4 mice for control; n = 5 mice for treated 100 and 1*103 cells; n = 4 mice for treated 1*104 cells). d, Estimated LIC frequencies for treated and untreated cell populations from each analyzed tissue (n = 4 mice for control; n = 5 mice for treated 100 and 1*103 cells; n = 4 mice for treated 1*104 cells) as calculated using the ELDA software69. In the top plot the number of initially injected cells (x-axis) is plotted against the log fraction of non-responders, corresponding to mice with no engraftment (y-axis).
Extended Data Fig. 6 Single cell RNA sequencing analysis of fresh diagnostic samples.
a-b, Analysis of the biological states, processes and lineages underpinning the intercellular transcriptional heterogeneity amongst diagnostic cells from either Pt1 or Pt2 using the bioinformatics tool scran70 to decompose gene expression heterogeneity into technical noise and a biologically relevant component. A list of genes ranked by their biologically meaningful variance was then used as input for enrichment analysis with the fGSEA R-package using the Hallmark pathway gene sets (MSigDB) and lineage markers defined in71. The importance of each given signature in explaining variation amongst untreated cells is reflected in the normalized enrichment score (NES) reported next to the signature itself. P-values are obtained using 2-sided permutation test52 with 10,000 permutations. All shown processes have a Benjamini-Hochberg FDR-ajusted p-value <0.05. c, Heatmap showing the expression of gene sets associated with different lineages and differentiation stages of the hematopoietic hierarchy. Each line of the map represents an individual diagnostic cell from either Pt1 or Pt2. d, Bar plots visualizing the percentage of individual diagnostic cells from either Pt1 or Pt2 assigned to their closest normal hematopoietic cell lineage genes (signature with the highest Z-score average). The analysis is based on the expression of known markers32,53.
Extended Data Fig. 7 Single cell RNA sequencing lineage analysis.
Series of t-SNE plots of n = 448 untreated and n = 163 treated cells showing the positive (TRUE) or negative (FALSE) assignment of each lineage across different regions of the plot. Each cell is colored in red if it expresses genes associated with the named lineage (>0.75 average z-score over genes in the signature) or in grey if it does not express the signature or expresses it below the cut-off level. The plots do not show relative expression levels but aim to highlight how the same cell can have concomitantly high expression of gene markers characteristic of different lineages.
Extended Data Fig. 8 Single cell RNA sequencing clustering analysis.
a, UMAP clustering of the single cell RNA-seq xenograft data. The dimensionality reduction tool identifies 4 main clusters of cells. Clusters 0, 2 and 3 correspond to different states of untreated cells, while cluster 1 mostly comprises chemotherapy-treated cells. b, Heatmap showing the top positively differentially expressed features that characterize each of the UMAP clusters. c, UMAP plots visualizing the expression of selected biomarkers for each of the clusters. Images produced using the Seurat analysis package72.
Extended Data Fig. 9 Boxplots of scRNAseq gene expression signatures.
Plot showing the expression of relevant MSigDB 50 Hallmark gene sets across single cells grouped by treatment status (untreated (n = 448 cells) and treated (n = 163 cells)). The median expression per class is shown as a solid black line within each box. The box covers the 25th–75th percentiles range; whiskers cover all data points within a 1.5x the inter-quantile ranges from the bounds of the box; remaining data points are plotted as outliers.
Extended Data Fig. 10 Single cell RNA sequencing cell cycle, lineage and pseudotime analysis.
a, Violin plots showing the expression of E2F1 and MYC genes in n = 448 untreated and n = 163 treated cells. Both genes are on average 10 log2 folds downregulated in treated cells. b, Stacked bar plot comparing the inferred lineage of each n = 448 untreated and n = 163 treated single cell with its cell cycle status as determined by the expression of G1/S, G2/M, MYC targets, and E2F targets signatures. c, TSCAN analysis displaying a PCA analysis of the data (n = 611 cells) and inferred pseudotime. The TSCAN package uses a cluster-based minimum spanning tree (MST) approach to order cells based on the gradual transition of their transcriptomes39. A line on the PCA highlights the constructed pseudo-temporal path. The table shows the number of treated and untreated cells within each cluster. The numbers of treated and untreated cells analysed are shown in the histograms at the top of the figure. d, Plot showing the distribution of deep, shallow quiescent and cycling cells (n = 611 cells), and lineage scores, across the TSCAN pseudotime clusters. e, Plots displaying the dynamic expression along the inferred pseudo-temporal path of representative genes with a known role in B-cell differentiation. Each cell (n = 611 cells) is shown as a circle (treated) or a triangle (untreated), the color of which reflects the cluster it belongs to (see panel c). f, Heatmap showing the top variable genes that contribute to clustering of n = 448 untreated and n = 163 chemotherapy-treated cells. PRS cells (n = 10 cells) as defined by this approach are evidenced by black arrows. g, Violin plots showing the expression of mitochondrial genes in treated, PRS and untreated cells as defined by either pseudotime (n = 163, 30 and 418 cells respectively; left plot) or t-SNE analysis (n = 163, 10 and 428 cells respectively; right plot).
Supplementary information
Supplementary Tables
Supplementary Tables 1–9.
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 2
Statistical source data.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 4
Statistical source data.
Source Data Fig. 5
Statistical source data.
Source Data Fig. 6
Statistical source data.
Source Data Fig. 7
Statistical source data.
Source Data Fig. 8
Statistical source data.
Source Data Extended Data Fig. 2
Statistical source data.
Source Data Extended Data Fig. 3
Statistical source data.
Source Data Extended Data Fig. 4
Statistical source data.
Source Data Extended Data Fig. 5
Statistical Source Data
Source Data Extended Data Fig. 6
Statistical source data.
Source Data Extended Data Fig. 7
Statistical source data.
Source Data Extended Data Fig. 8
Statistical source data.
Source Data Extended Data Fig. 9
Statistical source data.
Source Data Extended Data Fig. 10
Statistical source data.
Rights and permissions
About this article
Cite this article
Turati, V.A., Guerra-Assunção, J.A., Potter, N.E. et al. Chemotherapy induces canalization of cell state in childhood B-cell precursor acute lymphoblastic leukemia. Nat Cancer 2, 835–852 (2021). https://doi.org/10.1038/s43018-021-00219-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s43018-021-00219-3
This article is cited by
-
Genomic hallmarks and therapeutic implications of G0 cell cycle arrest in cancer
Genome Biology (2023)
-
miR-126 identifies a quiescent and chemo-resistant human B-ALL cell subset that correlates with minimal residual disease
Leukemia (2023)
-
State-dependent evolutionary models reveal modes of solid tumour growth
Nature Ecology & Evolution (2023)
-
In vivo PDX CRISPR/Cas9 screens reveal mutual therapeutic targets to overcome heterogeneous acquired chemo-resistance
Leukemia (2022)