Abstract
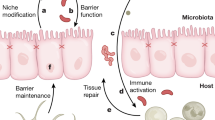
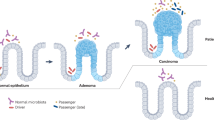
Colorectal cancer (CRC) is highly prevalent in Western society, and increasing evidence indicates strong contributions of environmental factors and the intestinal microbiota to CRC initiation, progression and even metastasis. We have identified a synergistic inflammatory tumor-promoting mechanism through which the resident intestinal microbiota boosts invasive CRC development in an epithelial-to-mesenchymal transition-prone tissue environment. Intestinal epithelial cell (IEC)-specific transgenic expression of the epithelial-to-mesenchymal transition regulator Zeb2 in mice (Zeb2IEC-Tg/+) leads to increased intestinal permeability, myeloid cell-driven inflammation and spontaneous invasive CRC development. Zeb2IEC-Tg/+ mice develop a dysplastic colonic epithelium, which progresses to severely inflamed neoplastic lesions while the small intestinal epithelium remains normal. Zeb2IEC-Tg/+ mice are characterized by intestinal dysbiosis, and microbiota depletion with broad-spectrum antibiotics or germ-free rederivation completely prevents cancer development. Zeb2IEC-Tg/+ mice represent the first mouse model of spontaneous microbiota-dependent invasive CRC and will help us to better understand host–microbiome interactions driving CRC development in humans.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
Statistical source data for Figs. 1–7 and Extended Data Figs. 1, 3 and 5–10 are provided with the paper. RNA sequencing data on ZEB2-overexpressing DLD-1 cells have been deposited in the Gene Expression Omnibus under accession code GSE148823. Sequencing data from the shallow whole-genome sequencing on Zeb2IEC-Tg/+ and control colon tumor tissue have been deposited in the Sequence Read Archive database under accession code PRJNA626401. All other data supporting the findings of this study are available from the corresponding author on reasonable request.
Change history
10 July 2020
A Correction to this paper has been published: https://doi.org/10.1038/s43018-020-0097-4
References
Brenner, H., Kloor, M. & Pox, C. P. Colorectal cancer. Lancet 383, 1490–1502 (2014).
Bray, F. et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 68, 394–424 (2018).
Vogelstein, B. et al. Cancer genome landscapes. Science 339, 1546–1558 (2013).
O’Keefe, S. J. Diet, microorganisms and their metabolites, and colon cancer. Nat. Rev. Gastroenterol. Hepatol. 13, 691–706 (2016).
Bashir, A., Miskeen, A. Y., Hazari, Y. M., Asrafuzzaman, S. & Fazili, K. M. Fusobacterium nucleatum, inflammation, and immunity: the fire within human gut. Tumour Biol. 37, 2805–2810 (2016).
Brennan, C. A. & Garrett, W. S. Fusobacterium nucleatum—symbiont, opportunist and oncobacterium. Nat. Rev. Microbiol. 17, 156–166 (2019).
West, N. R., McCuaig, S., Franchini, F. & Powrie, F. Emerging cytokine networks in colorectal cancer. Nat. Rev. Immunol. 15, 615–629 (2015).
Jin, C. et al. Commensal microbiota promote lung cancer development via γδ T cells. Cell 176, 998–1013.e16 (2019).
De Craene, B. & Berx, G. Regulatory networks defining EMT during cancer initiation and progression. Nat. Rev. Cancer 13, 97–110 (2013).
Thiery, J. P., Acloque, H., Huang, R. Y. & Nieto, M. A. Epithelial–mesenchymal transitions in development and disease. Cell 139, 871–890 (2009).
Nieto, M. A., Huang, R. Y., Jackson, R. A. & Thiery, J. P. EMT: 2016. Cell 166, 21–45 (2016).
Stemmler, M. P., Eccles, R. L., Brabletz, S. & Brabletz, T. Non-redundant functions of EMT transcription factors. Nat. Cell Biol. 21, 102–112 (2019).
Goossens, S., Vandamme, N., Van Vlierberghe, P. & Berx, G. EMT transcription factors in cancer development re-evaluated: beyond EMT and MET. Biochim. Biophys. Acta Rev. Cancer 1868, 584–591 (2017).
Skrypek, N., Goossens, S., De Smedt, E., Vandamme, N. & Berx, G. Epithelial-to-mesenchymal transition: epigenetic reprogramming driving cellular plasticity. Trends Genet. 33, 943–959 (2017).
Kahlert, C. et al. Overexpression of ZEB2 at the invasion front of colorectal cancer is an independent prognostic marker and regulates tumor invasion in vitro. Clin Cancer Res. 17, 7654–7663 (2011).
Li, M. Z. et al. ZEB2 promotes tumor metastasis and correlates with poor prognosis of human colorectal cancer. Am. J. Transl. Res. 9, 2838–2851 (2017).
Rokavec, M. et al. IL-6R/STAT3/miR-34a feedback loop promotes EMT-mediated colorectal cancer invasion and metastasis. J. Clin. Invest. 124, 1853–1867 (2014).
Sreekumar, R. et al. Assessment of nuclear ZEB2 as a biomarker for colorectal cancer outcome and TNM risk stratification. JAMA Netw. Open 1, e183115 (2018).
Tatari, M. N. et al. ZEB2-transgene expression in the epidermis compromises the integrity of the epidermal barrier through the repression of different tight junction proteins. Cell. Mol. Life Sci. 71, 3599–3609 (2014).
Goossens, S. et al. ZEB2 drives immature T-cell lymphoblastic leukaemia development via enhanced tumour-initiating potential and IL-7 receptor signalling. Nat. Commun. 6, 5794 (2015).
Madison, B. B. et al. Cis elements of the villin gene control expression in restricted domains of the vertical (crypt) and horizontal (duodenum, cecum) axes of the intestine. J. Biol. Chem. 277, 33275–33283 (2002).
Barker, N., van de Wetering, M. & Clevers, H. The intestinal stem cell. Genes Dev. 22, 1856–1864 (2008).
Bollrath, J. & Greten, F. R. IKK/NF-κB and STAT3 pathways: central signalling hubs in inflammation-mediated tumour promotion and metastasis. EMBO Rep. 10, 1314–1319 (2009).
Zeilstra, J. et al. CD44 expression in intestinal epithelium and colorectal cancer is independent of p53 status. PLoS ONE 8, e72849 (2013).
Dalerba, P. et al. Phenotypic characterization of human colorectal cancer stem cells. Proc. Natl Acad. Sci. USA 104, 10158–10163 (2007).
Merlos-Suarez, A. et al. The intestinal stem cell signature identifies colorectal cancer stem cells and predicts disease relapse. Cell Stem Cell 8, 511–524 (2011).
Clevers, H. At the crossroads of inflammation and cancer. Cell 118, 671–674 (2004).
Taniguchi, K. & Karin, M. NF-κB, inflammation, immunity and cancer: coming of age. Nat. Rev. Immunol. 18, 309–324 (2018).
Asquith, M. & Powrie, F. An innately dangerous balancing act: intestinal homeostasis, inflammation, and colitis-associated cancer. J. Exp. Med. 207, 1573–1577 (2010).
Johansson, M. E. et al. The inner of the two Muc2 mucin-dependent mucus layers in colon is devoid of bacteria. Proc. Natl Acad. Sci. USA 105, 15064–15069 (2008).
De Filippo, K. et al. Mast cell and macrophage chemokines CXCL1/CXCL2 control the early stage of neutrophil recruitment during tissue inflammation. Blood 121, 4930–4937 (2013).
Germann, M. et al. Neutrophils suppress tumor-infiltrating T cells in colon cancer via matrix metalloproteinase-mediated activation of TGFβ. EMBO Mol. Med. 12, e10681 (2020).
Jackstadt, R. et al. Epithelial NOTCH signaling rewires the tumor microenvironment of colorectal cancer to drive poor-prognosis subtypes and metastasis. Cancer Cell 36, 319–336.e7 (2019).
Grivennikov, S. I. et al. Adenoma-linked barrier defects and microbial products drive IL-23/IL-17-mediated tumour growth. Nature 491, 254–258 (2012).
Guinney, J. et al. The consensus molecular subtypes of colorectal cancer. Nat. Med. 21, 1350–1356 (2015).
Marisa, L. et al. Gene expression classification of colon cancer into molecular subtypes: characterization, validation, and prognostic value. PLoS Med. 10, e1001453 (2013).
Laibe, S. et al. A seven-gene signature aggregates a subgroup of stage II colon cancers with stage III. OMICS 16, 560–565 (2012).
De Sousa, E. M. F. et al. Methylation of cancer-stem-cell-associated Wnt target genes predicts poor prognosis in colorectal cancer patients. Cell Stem Cell 9, 476–485 (2011).
Smith, J. J. et al. Experimentally derived metastasis gene expression profile predicts recurrence and death in patients with colon cancer. Gastroenterology 138, 958–968 (2010).
Budinska, E. et al. Gene expression patterns unveil a new level of molecular heterogeneity in colorectal cancer. J. Pathol. 231, 63–76 (2013).
Li, H. et al. Reference component analysis of single-cell transcriptomes elucidates cellular heterogeneity in human colorectal tumors. Nat. Genet. 49, 708–718 (2017).
Calon, A. et al. Stromal gene expression defines poor-prognosis subtypes in colorectal cancer. Nat. Genet. 47, 320–329 (2015).
Isella, C. et al. Stromal contribution to the colorectal cancer transcriptome. Nat. Genet. 47, 312–319 (2015).
Irrazabal, T., Belcheva, A., Girardin, S. E., Martin, A. & Philpott, D. J. The multifaceted role of the intestinal microbiota in colon cancer. Mol. Cell 54, 309–320 (2014).
Nakatsu, G. et al. Gut mucosal microbiome across stages of colorectal carcinogenesis. Nat. Commun. 6, 8727 (2015).
Bullman, S. et al. Analysis of Fusobacterium persistence and antibiotic response in colorectal cancer. Science 358, 1443–1448 (2017).
Gopalakrishnan, V. et al. Gut microbiome modulates response to anti-PD-1 immunotherapy in melanoma patients. Science 359, 97–103 (2018).
Routy, B. et al. Gut microbiome influences efficacy of PD-1-based immunotherapy against epithelial tumors. Science 359, 91–97 (2018).
Vetizou, M. et al. Anticancer immunotherapy by CTLA-4 blockade relies on the gut microbiota. Science 350, 1079–1084 (2015).
Viaud, S. et al. The intestinal microbiota modulates the anticancer immune effects of cyclophosphamide. Science 342, 971–976 (2013).
Matson, V. et al. The commensal microbiome is associated with anti-PD-1 efficacy in metastatic melanoma patients. Science 359, 104–108 (2018).
Iida, N. et al. Commensal bacteria control cancer response to therapy by modulating the tumor microenvironment. Science 342, 967–970 (2013).
Nenci, A. et al. Epithelial NEMO links innate immunity to chronic intestinal inflammation. Nature 446, 557–561 (2007).
Welz, P. S. et al. FADD prevents RIP3-mediated epithelial cell necrosis and chronic intestinal inflammation. Nature 477, 330–334 (2011).
Takahashi, N. et al. RIPK1 ensures intestinal homeostasis by protecting the epithelium against apoptosis. Nature 513, 95–99 (2014).
Kaser, A. et al. XBP1 links ER stress to intestinal inflammation and confers genetic risk for human inflammatory bowel disease. Cell 134, 743–756 (2008).
Dannappel, M. et al. RIPK1 maintains epithelial homeostasis by inhibiting apoptosis and necroptosis. Nature 513, 90–94 (2014).
Gunther, C. et al. Caspase-8 regulates TNF-α-induced epithelial necroptosis and terminal ileitis. Nature 477, 335–339 (2011).
Becker, C. et al. In vivo imaging of colitis and colon cancer development in mice using high resolution chromoendoscopy. Gut 54, 950–954 (2005).
Johansson, M. E. & Hansson, G. C. Preservation of mucus in histological sections, immunostaining of mucins in fixed tissue, and localization of bacteria with FISH. Methods Mol. Biol. 842, 229–235 (2012).
Scott, C. L. et al. The transcription factor ZEB2 is required to maintain the tissue-specific identities of macrophages. Immunity 49, 312–325.e5 (2018).
Muyzer, G., de Waal, E. C. & Uitterlinden, A. G. Profiling of complex microbial populations by denaturing gradient gel electrophoresis analysis of polymerase chain reaction-amplified genes coding for 16S rRNA. Appl. Environ. Microbiol. 59, 695–700 (1993).
Herlemann, D. P. et al. Transitions in bacterial communities along the 2000 km salinity gradient of the Baltic Sea. ISME J. 5, 1571–1579 (2011).
Ihrmark, K. et al. New primers to amplify the fungal ITS2 region—evaluation by 454-sequencing of artificial and natural communities. FEMS Microbiol. Ecol. 82, 666–677 (2012).
Hildebrand, F., Tadeo, R., Voigt, A. Y., Bork, P. & Raes, J. LotuS: an efficient and user-friendly OTU processing pipeline. Microbiome 2, 30 (2014).
McMurdie, P. J. & Holmes, S. Phyloseq: a bioconductor package for handling and analysis of high-throughput phylogenetic sequence data. Pac. Symp. Biocomput. 235–246 (2012).
Sante, T. et al. ViVar: a comprehensive platform for the analysis and visualization of structural genomic variation. PLoS ONE 9, e113800 (2014).
Gagnon-Bartsch, J. A. & Speed, T. P. Using control genes to correct for unwanted variation in microarray data. Biostatistics 13, 539–552 (2012).
Sayan, A. E. et al. SIP1 protein protects cells from DNA damage-induced apoptosis and has independent prognostic value in bladder cancer. Proc. Natl Acad. Sci. USA 106, 14884–14889 (2009).
Acknowledgements
We thank M. Gomez, S. Ganal, J. Kirundi, K. McCoy and A. MacPherson for generating axenic Zeb2IEC-Tg/+ mice at the University of Bern, Switzerland. We thank L. Bellen and K. Barbry for animal care and the VIB Flow Core for training, support and access to the instrument park. K.S. was a predoctoral fellow with the Institute for the Promotion of Innovation by Science and Technology (IWT) and was supported by a ‘Kom op tegen Kanker’ (Stand Up To Cancer) grant from the Flemish Cancer Society. Work in the G.v.L. laboratory is supported by research grants from the FWO, Strategic Basic Research (SBO) program, Geneeskundige Stichting Koningin Elisabeth (GSKE), CBC Banque Prize, Charcot Foundation, Stichting Tegen Kanker, Cancer Research Institute Ghent and Concerted Research Actions (GOA) of Ghent University. The laboratory of G.B. is supported by grants from the FWO, SBO, GOA, Vlaamse Liga Tegen Kanker and Stichting Tegen Kanker. The laboratory of L.V. is supported by grants from the FWO, SBO and Cancer Research Institute Ghent.
Author information
Authors and Affiliations
Contributions
K.S., I.P., G. Blancke, E.H., E.D., M.S., H.V., S.J., J.T., N.V. and D.N. performed the experiments. K.S., I.P., E.H., E.D., E.R., J.J.H., A.W., D.N., P.V.V., G.D.H., P.B., E.E., P.W., B.N.L., C.C., S.T., S.G., G. Berx, L.V. and G.v.L. analyzed the data. E.T. and L.B. provided reagents. G. Berx, L.V. and G.v.L provided ideas and coordinated the project. K.S., G. Berx, L.V. and G.v.L. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Expression of Zeb2 in the mouse intestinal epithelium.
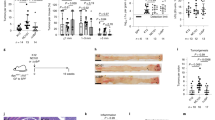
a, Schematic representation of the conditional ROSA26-based Zeb2 overexpression mouse model. The mouse Zeb2 open reading frame was targeted to the ROSA26 locus preceded by a floxed (fl) transcriptional stop (PGK-Neo-3XpA) cassette and followed by IRES-EGFP reporter sequence. b, R26-Zeb2Tg/Tg mice were crossed with a Cre transgenic line under the control of Villin promoter, generating R26-Zeb2Tg/+ villin-creTg/+ transgenic (Tg/+) mice and R26-Zeb2Tg/+ villin-cre+/+ (WT) controls. c-d, Kaplan-Meier survival curve (c) and clinical stool score (d) of male (n=12 mice) and female (n=10 mice) Zeb2IEC-Tg/+ mice based on Hemoccult assay. Data are presented as mean ± SEM.
Extended Data Fig. 2 Histopathology of Zeb2IEC-Tg/+ mice.
a, Representative H&E stained colon section from wild-type control mouse showing normal proximal colon (representative for n=10 mice). Left panel, magnification of boxed area. b, Representative H&E stained sections of Zeb2IEC-Tg/+ mouse (representative for n=7 mice) demonstrating multicentric invasive adenocarcinoma in a background of atypical mucosal hyperplasia with multifocal intraepithelial neoplasia and intramucosal carcinoma. Also sparse inflammatory cell infiltrates, predominantly lymphocytes and plasma cells, can be observed in the lamina propria and submucosa. Left panel, higher magnification showing severe atypical hyperplasia/intramucosal carcinoma characterized by tumultuous proliferation of irregular glandular units (upper), and of the invading adenocarcinomatous component characterized by irregular glandular profiles with abundant mucus collection, transmural infiltration of the intestinal wall and spread along the mesenteric ligament (lower). c, PAS staining of section of distal colon and rectum of Zeb2IEC-Tg/+ mouse (representative for n=7 mice). Abundant collection of PAS-positive mucus is evident in the irregular neoplastic glandular units transmurally invading the intestinal wall and infiltrating along the mesenteric ligament. d, Macroscopic view on proximal colon tissue dissected in 44-week old WT and Zeb2IEC-Tg/+ mice. e, Macroscopic dissected proximal colon in 31-week old Zeb2IEC-Tg/+ mouse displaying transmural cancerous outgrowth.
Extended Data Fig. 3 Zeb2IEC-Tg/+ mice develop severe pathology in colon but not in small intestine.
a, Quantification of Ki67-, CD44-, P-Stat3- and P-Smad3-positive cells in different regions of the colon (mucosa, submucosa, muscle or serosa) of 25-30-week old wild-type (WT) and Zeb2IEC-Tg/+ mice (Tg) (Ki67 Tg/+, n=9 mice and WT, n=6 mice; CD44 Tg/+, n=5 mice and WT, n=5 mice; P-Stat3 Tg/+, n=7 mice and WT, n=5 mice; P-Smad3 Tg/+, n=6 mice and WT, n=3 mice). For each tissue region and mouse, 3 separate areas were analyzed and averages per mouse per region were plotted in GraphPad Prism. Data are presented as mean ± SEM. ** p<0.01, *** p<0.001, **** p<0.0001. Two way ANOVA, corrected for multiple comparisons using Sidak test in GraphPad Prism. b, Representative FACS plots of Ki67+EPCAM+ cells in colon (left panel) and small intestine (right panel) in 5 week old wild-type (WT) and Zeb2IEC-Tg/+ mice. The experiment was performed twice with similar results. c, Quantitative PCR analysis for Zeb2 and eGFP in lysates from the small intestine of Zeb2IEC-Tg/+ (Tg/+, n=6 mice) and wild-type littermate controls (WT, n=5 mice). Data are presented as mean ± SEM and were analyzed with two-tailed Mann Whitney test, p-value = 0.0043 for both. d, Quantification of Ki67-positive cells in different regions of the small intestine of 25-30-week old wild-type (WT, n=4 mice) and Zeb2IEC-Tg/+ mice (Tg, n=5 mice). For each tissue region and mouse, 3 separate areas were analyzed and averages per mouse per region were plotted in GraphPad Prism. Data are presented as mean ± SEM and were analyzed with two-way ANOVA with Sidak correction. e, Quantitative PCR analysis for inflammatory markers (TNF, IL-1β, Cox2, Cxcl5 and A20), CD44, and Paneth cell markers (Crypt-1 and LysP) in lysates from the small intestine of Zeb2IEC-Tg/+ (Tg/+, n=6 mice) and wild-type littermate controls (WT, n=5 mice). Data are presented as mean ± SEM and were analyzed with two-tailed Mann Whitney test, p-value = 0.0087 for IL-1β and 0.0043 for Cox2; ns, non-significant.
Extended Data Fig. 4 Tumorigenic potential and copy number profiling of Zeb2 transgenic tissue.
a, Orthotopic transplantation of intestinal tumor cells from Zeb2 transgenic mice into the cecum of NSG mice. Zeb2 transgenic cells could be identified by immunohistochemical staining for the GFP transgene. Scale bar: 200µm; boxed area Scale bar: 100µm. Data representative for one experiment on 5 NSG mice. b, Copy number analysis by shallow whole genome sequencing of Zeb2IEC-Tg/+ colon tumor tissue (n=4 mice, lanes 3-6), normal lung tissue (n=2 mice, lanes 1-2) and normal colon tissue from a wild-type littermate control mouse (lane 7). Genomic aberrations are positioned along the murine chromosomes and visualized in red for deletions and blue for amplifications.
Extended Data Fig. 5 Induction of ZEB2 expression leads to a loss of cell adhesion complexes such as tight and gap junctions.
RNA-Seq analysis on Doxycycline (Dox)-inducible ZEB2-overexpressing DLD-1 colorectal cancer cell line clones. Data represents the mean centered rlog-transformed counts (n=3 biological repeats).
Extended Data Fig. 6 Zeb2 expression and intestinal barrier integrity in the small intestine.
Quantitative PCR analysis for expression of tight junction proteins (Claudin-4, -6 and -7, Occludin, Tjp-1, F11R/JAM-1) and desmosomes (Desmocollin-1 and -3, Desmoglein-2, Plakophillin-1) in lysates from the small intestine of Zeb2IEC-Tg/+ (Tg/+, n=6 mice) and wild-type littermate controls (WT, n=5 mice). Data are presented as mean ± SEM and were analyzed with two-tailed Mann Whitney test. *, p-value = 0.0173; ** p-value = 0.0043; ns, non-significant.
Extended Data Fig. 7 Microbiota dysbiosis in Zeb2IEC-Tg/+ mice.
Abundance of significantly different taxa between wild-type (WT, n=21 mice) and Zeb2IEC-Tg/+ (Tg/+, n=34 mice) fecal samples based on MaAslin and t-test FDR correction analysis. The horizontal straight line indicates the group means and the dotted line indicates the group medians.
Extended Data Fig. 8 Microbiota dysbiosis in Zeb2IEC-Tg/+ mice.
a, b, Altered bacterial diversity/community structure in mucosal samples from 25 week-old wild-type (WT, in green, n=28 mice) and Zeb2IEC-Tg/+ (Tg/+, in red, n=17 mice) mice. α-diversity measured by the Shannon Diversity Index at the genus level (p= 0.1439, Welch two Sample t-test) (a). β-diversity represented by a PCoA of Bray-Curtis dissimilarity distances of bacterial composition (b). c, d, Differentially abundant taxa between wild-type (WT) and Zeb2IEC-Tg/+ (Tg/+). Linear Discriminant Analysis Effect Size (LEfSe) cladogram of differentially abundant taxa in mucosal samples from WT (in green, n=21 mice) and Tg/+ (in red, n=34 mice) mice (c). Linear Discriminant Analyis (LDA) scores (d) of differentially abundant taxa combined with multiple testing corrections (analysis associated with Extended Data Fig. 8c): t-test with FDR correction (*: padj <0.05) (no significant differences were identified based on MaAslin analysis). [phylum (p), class (c), order (o), family (f), genera (g), species (s)]. e, Abundance of significantly different taxa between wild-type (WT) and Zeb2IEC-Tg/+ (Tg/+) mucosal samples based on t-test FDR correction analysis. The horizontal straight line indicates the group means and the dotted line indicates the group medians. f, g, Mycobiota profiling in Zeb2IEC-Tg/+ mice. Fungal diversity/community (WT n=33 mice, in green, and Tg/+ n=22 mice, in red). α-diversity measured by the Shannon Diversity Index at the genus level (p= 0.0717, Welch two Sample t-test) (f). β-diversity represented by a PCoA of Bray-Curtis dissimilarity distances of fungal composition (g). Each box represents the third (upper) and first (lower) quartiles. Lines inside the boxes represent the median.
Extended Data Fig. 9 ABX treated Zeb2IEC-Tg/+ mice are protected from CRC development.
a, Clinical stool score of untreated (n=6 mice), vancomycin-treated (n=6 mice) and aztreonam-treated (n=4 mice) Zeb2IEC-Tg/+ mice based on Hemoccult assay. Data are presented as mean ± SEM. b, Colon pathology detected by endoscopy and represented as MEICS (murine endoscopic index of colitis severity) score. Each symbol represents one mouse. Data are presented as mean ± SEM. c, Colon histology of Aztreonam (left panel) and Vancomycin (right panel) treated Zeb2IEC-Tg/+ mice by H&E staining. Scale bar: 200µm. Representative images from one experiment with n=4 mice.
Extended Data Fig. 10 Axenic Zeb2IEC-Tg/+ mice are protected from CRC development.
a, Quantification of Ki67-, CD44-, P-Stat3- and P-Smad3-positive cells in different regions of the colon (mucosa, submucosa, muscle or serosa) of 20 week-old germfree wild-type (WT) and Zeb2IEC-Tg/+ mice (Tg). For each tissue region and mouse, 3 separate areas were analyzed and averages per mouse per region were plotted in GraphPad Prism. Data are presented as mean ± SEM and were analyzed with two-way ANOVA with Sidak correction (Ki67 WT, n=4 mice and Tg/+, n=5 mice; CD44 WT, n=4 mice and Tg/+, n=4 mice; P-Smad3 WT, n=3 mice and Tg/+, n=6 mice). b, Flow cytometry analysis of colonic lamina propria immune cells in 12 week-old germfree WT (n=6 mice) and Zeb2IEC-Tg/+ (n=6 mice). Data are presented as mean ± SEM, two-sided student’s t test. Each symbol represents one mouse.
Supplementary information
Source data
Source Data Fig. 1
Statistical source data Fig. 1
Source Data Fig. 2
Statistical source data Fig. 2
Source Data Fig. 3
Statistical source data Fig. 3
Source Data Fig. 4
Statistical source data Fig. 4
Source Data Fig. 5
Statistical source data Fig. 5
Source Data Fig. 6
Statistical source data Fig. 6
Source Data Fig. 7
Statistical source data Fig. 7
Source Data Extended Data Fig. 1
Statistical source data Extended Data Fig. 1
Source Data Extended Data Fig. 3
Statistical source data Extended Data Fig. 3
Source Data Extended Data Fig. 5
Statistical source data Extended Data Fig. 5
Source Data Extended Data Fig. 6
Statistical source data Extended Data Fig. 6
Source Data Extended Data Fig. 7
Statistical source data Extended Data Fig. 7
Source Data Extended Data Fig. 8
Statistical source data Extended Data Fig. 8
Source Data Extended Data Fig. 9
Statistical source data Extended Data Fig. 9
Source Data Extended Data Fig. 10
Statistical source data Extended Data Fig. 10
Rights and permissions
About this article
Cite this article
Slowicka, K., Petta, I., Blancke, G. et al. Zeb2 drives invasive and microbiota-dependent colon carcinoma. Nat Cancer 1, 620–634 (2020). https://doi.org/10.1038/s43018-020-0070-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s43018-020-0070-2
This article is cited by
-
Microbiota-derived tryptophan catabolites mediate the chemopreventive effects of statins on colorectal cancer
Nature Microbiology (2023)
-
Ileal immune tonus is a prognosis marker of proximal colon cancer in mice and patients
Cell Death & Differentiation (2021)