Abstract
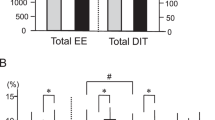
Brown adipose tissue (BAT) thermogenesis is activated by feeding. Recently, we revealed a secretin-mediated gut–BAT–brain axis, which stimulates satiation in mice, but the purpose of meal-induced BAT activation in humans has been unclear. In this placebo-controlled, randomized crossover study, we investigated the effects of intravenous secretin on BAT metabolism (measured with [18F]FDG and [15O]H2O positron emission tomography) and appetite (measured with functional magnetic resonance imaging) in healthy, normal weight men (GUTBAT trial no. NCT03290846). Participants were blinded to the intervention. Secretin increased BAT glucose uptake (primary endpoint) compared to placebo by 57% (median (interquartile range, IQR), 0.82 (0.77) versus 0.59 (0.53) μmol per 100 g per min, 95% confidence interval (CI) (0.09, 0.89), P = 0.002, effect size r = 0.570), while BAT perfusion remained unchanged (mean (s.d.) 4.73 (1.82) versus 6.14 (3.05) ml per 100 g per min, 95%CI (−2.91, 0.07), P = 0.063, effect size d = −0.549) (n = 15). Whole body energy expenditure increased by 2% (P = 0.011) (n = 15). Secretin attenuated blood-oxygen level-dependent activity (primary endpoint) in brain reward circuits during food cue tasks (significance level false discovery rate corrected at P = 0.05) (n = 14). Caloric intake did not significantly change, but motivation to refeed after a meal was delayed by 39 min (P = 0.039) (n = 14). No adverse effects were detected. Here we show in humans that secretin activates BAT, reduces central responses to appetizing food and delays the motivation to refeed after a meal. This suggests that meal-induced, secretin-mediated BAT activation is relevant in the control of food intake in humans. As obesity is increasing worldwide, this appetite regulating axis offers new possibilities for clinical research in treating obesity.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All statistical parametric images of the fMRI study (including the effect size maps) can be found from NeuroVault at https://neurovault.org/collections/ECURNRON/. The accession number for the RNA-Seq data presented in this article is GEO GSE113764. SCTR expression in human tissues is available in the Online Biology Gene Portal System (BioGPS) at http://ds.biogps.org/?dataset=GSE1133&gene=6344. Datasets that support the findings of this study are available in the Supplementary Information. Source data are provided with this paper. The data that support the plots within this paper, as well as other findings of this study, are available from the corresponding author (P.N.) upon reasonable request.
References
Virtanen, K. A. et al. Functional brown adipose tissue in healthy adults. N. Engl. J. Med. 360, 1518–1525 (2009).
Cypess, A. M. et al. Identification and importance of brown adipose tissue in adult humans. N. Engl. J. Med. 360, 1509–1517 (2009).
van Marken Lichtenbelt, W. D. et al. Cold-activated brown adipose tissue in healthy men. N. Engl. J. Med. 360, 1500–1508 (2009).
Blondin, D. P. et al. Contributions of white and brown adipose tissues and skeletal muscles to acute cold-induced metabolic responses in healthy men. J. Physiol. 593, 701–714 (2015).
Villarroya, F., Cereijo, R., Villarroya, J. & Giralt, M. Brown adipose tissue as a secretory organ. Nat. Rev. Endocrinol. 13, 26–35 (2017).
Kajimura, S., Spiegelman, B. M. & Seale, P. Brown and beige fat: physiological roles beyond heat generation. Cell Metab. 22, 546–559 (2015).
Rothwell, N. J. & Stock, M. J. A role for brown adipose tissue in diet-induced thermogenesis. Nature 281, 31–35 (1979).
Glick, Z., Teague, R. J. & Bray, G. A. Brown adipose tissue: thermic response increased by a single low protein, high carbohydrate meal. Science 213, 1125–1127 (1981).
U Din, M. et al. Postprandial oxidative metabolism of human brown fat indicates thermogenesis. Cell Metab. 28, 207–216.e3 (2018).
Chondronikola, M., Porter, C., Malagaris, I., Nella, A. A. & Sidossis, L. S. Brown adipose tissue is associated with systemic concentrations of peptides secreted from the gastrointestinal system and involved in appetite regulation. Eur. J. Endocrinol. 177, 33–40 (2017).
Crovetti, R., Porrini, M., Santangelo, A. & Testolin, G. The influence of thermic effect of food on satiety. Eur. J. Clin. Nutr. 52, 482–488 (1998).
Li, Y. et al. Secretin-activated brown fat mediates prandial thermogenesis to induce satiation. Cell 175, 1561–1574.e12 (2018).
Blondin, D. et al. Inhibition of intracellular triglyceride lipolysis suppresses cold-induced brown adipose tissue metabolism and increases shivering in humans. Cell Metab. 25, 438–447 (2017).
Wu, C., Jin, X., Tsueng, G., Afrasiabi, C. & Su, A. I. BioGPS: building your own mash-up of gene annotations and expression profiles. Nucleic Acids Res. 44, D313–D316 (2016).
Su, A. I. et al. A gene atlas of the mouse and human protein-encoding transcriptomes. Proc. Natl Acad. Sci. USA. 101, 6062–6067 (2004).
Nummenmaa, L. et al. μ-opioid receptor system mediates reward processing in humans. Nat. Commun. 9, 1500 (2018).
Gilbert, J.-A. et al. The effect of tesofensine on appetite sensations. Obesity 20, 553–561 (2012).
Balaz, M. et al. Inhibition of mevalonate pathway prevents adipocyte browning in mice and men by affecting protein prenylation. Cell Metab. 29, 901–916.e8 (2019).
Sekar, R. & Chow, B. K. C. Lipolytic actions of secretin in mouse adipocytes. J. Lipid Res. 55, 190–200 (2014).
Butcher, R. W. & Carlson, L. A. Effects of secretin on fat mobilizing lipolysis and cyclic AMP levels in rat adipose tissue. Acta Physiol. Scand. 79, 559–563 (1970).
Raiko, J. et al. Brown adipose tissue triglyceride content is associated with decreased insulin sensitivity, independently of age and obesity. Diabetes, Obes. Metab. 17, 516–519 (2015).
U Din, M. et al. Human brown fat radiodensity indicates underlying tissue composition and systemic metabolic health. J. Clin. Endocrinol. Metab. 102, 2258–2267 (2017).
Yaney, G. C. & Corkey, B. E. Fatty acid metabolism and insulin secretion in pancreatic beta cells. Diabetologia 46, 1297–1312 (2003).
Stein, D. T. et al. The insulinotropic potency of fatty acids is influenced profoundly by their chain length and degree of saturation. J. Clin. Invest. 100, 398–403 (1997).
Afroze, S. et al. The physiological roles of secretin and its receptor. Ann. Transl. Med. 1, 29 (2013).
Broeders, E. P. M. et al. The bile acid chenodeoxycholic acid increases human brown adipose tissue activity. Cell Metab. 22, 418–426 (2015).
Amin, A., Dhillo, W. S. & Murphy, K. G. The central effects of thyroid hormones on appetite. J. Thyroid Res. 2011, 306510 (2011).
Bianco, A. C. & Silva, J. E. Intracellular conversion of thyroxine to triiodothyronine is required for the optimal thermogenic function of brown adipose tissue. J. Clin. Invest. 79, 295–300 (1987).
Lahesmaa, M. et al. Hyperthyroidism increases brown fat metabolism in humans. J. Clin. Endocrinol. Metab. 99, E28–E35 (2014).
Bessesen, D. H. & Van Gaal, L. F. Progress and challenges in anti-obesity pharmacotherapy. Lancet Diabetes Endocrinol. 6, 237–248 (2018).
van Ooijen, A. M. J., van Marken Lichtenbelt, W. D., van Steenhoven, A. A. & Westerterp, K. R. Seasonal changes in metabolic and temperature responses to cold air in humans. Physiol. Behav. 82, 545–553 (2004).
Yoneshiro, T. et al. Recruited brown adipose tissue as an antiobesity agent in humans. J. Clin. Invest. 123, 3404–3408 (2013).
U.-Din, M. et al. Human brown adipose tissue [15O]O2 PET imaging in the presence and absence of cold stimulus. Eur. J. Nucl. Med. Mol. Imag. https://doi.org/10.1007/s00259-016-3364-y (2016).
Orava, J. et al. Different metabolic responses of human brown adipose tissue to activation by cold and insulin. Cell Metab. 14, 272–279 (2011).
Patlak, C. S. & Blasberg, R. G. Graphical evaluation of blood-to-brain transfer constants from multiple-time uptake data. Generalizations. J. Cereb. Blood Flow. Metab. 5, 584–590 (1985).
Virtanen, K. A. et al. Human adipose tissue glucose uptake determined using [18 F]-fluoro-deoxy-glucose ([18 F]FDG) and PET in combination with microdialysis. Diabetologia 44, 2171–2179 (2001).
Peltoniemi, P. et al. Lumped constant for [18 F]fluorodeoxyglucose in skeletal muscles of obese and nonobese humans. Am. J. Physiol. Metab. 279, E1122–E1130 (2000).
Weir, J. B. & de, V. New methods for calculating metabolic rate with special reference to protein metabolism. J. Physiol. 109, 1–9 (1949).
Meriläinen, P. T. Metabolic monitor. Int. J. Clin. Monit. Comput. 4, 167–177 (1987).
Nummenmaa, L. et al. Dorsal striatum and its limbic connectivity mediate abnormal anticipatory reward processing in obesity. PLoS ONE 7, e31089 (2012).
Esteban, O. et al. fMRIPrep: a robust preprocessing pipeline for functional MRI. Nat. Methods https://doi.org/10.1038/s41592-018-0235-4 (2019).
Gorgolewski, K. et al. Nipype: A flexible, lightweight and extensible neuroimaging data processing framework in Python. Front. Neuroinform. https://doi.org/10.3389/fninf.2011.00013 (2011).
Tustison, N. J. et al. N4ITK: Improved N3 bias correction. IEEE Trans. Med. Imag. https://doi.org/10.1109/TMI.2010.2046908 (2010).
Dale, A. M., Fischl, B. & Sereno, M. I. Cortical surface-based analysis: I. Segmentation and surface reconstruction. Neuroimage https://doi.org/10.1006/nimg.1998.0395 (1999).
Klein, A. et al. Mindboggling morphometry of human brains. PLoS Comput. Biol. https://doi.org/10.1371/journal.pcbi.1005350 (2017).
Fonov, V., Evans, A., McKinstry, R., Almli, C. & Collins, D. Unbiased nonlinear average age-appropriate brain templates from birth to adulthood. Neuroimage https://doi.org/10.1016/s1053-8119(09)70884-5 (2009).
Avants, B. B., Epstein, C. L., Grossman, M. & Gee, J. C. Symmetric diffeomorphic image registration with cross-correlation: evaluating automated labeling of elderly and neurodegenerative brain. Med. Image Anal. https://doi.org/10.1016/j.media.2007.06.004 (2008).
Zhang, Y., Brady, M. & Smith, S. Segmentation of brain MR images through a hidden Markov random field model and the expectation-maximization algorithm. IEEE Trans. Med. Imag. https://doi.org/10.1109/42.906424 (2001).
Cox, R. W. AFNI: Software for analysis and visualization of functional magnetic resonance neuroimages. Comput. Biomed. Res. https://doi.org/10.1006/cbmr.1996.0014 (1996).
Jenkinson, M., Bannister, P., Brady, M. & Smith, S. Improved optimization for the robust and accurate linear registration and motion correction of brain images. Neuroimage https://doi.org/10.1016/S1053-8119(02)91132-8 (2002).
Greve, D. N. & Fischl, B. Accurate and robust brain image alignment using boundary-based registration. Neuroimage https://doi.org/10.1016/j.neuroimage.2009.06.060 (2009).
Behzadi, Y., Restom, K., Liau, J. & Liu, T. T. A component based noise correction method (CompCor) for BOLD and perfusion based fMRI. Neuroimage https://doi.org/10.1016/j.neuroimage.2007.04.042 (2007).
Power, J. D. et al. Methods to detect, characterize, and remove motion artifact in resting state fMRI. Neuroimage https://doi.org/10.1016/j.neuroimage.2013.08.048 (2014).
Pruim, R. H. R. et al. ICA-AROMA: a robust ICA-based strategy for removing motion artifacts from fMRI data. Neuroimage https://doi.org/10.1016/j.neuroimage.2015.02.064 (2015).
Flint, A., Raben, A., Blundell, J. E. & Astrup, A. Reproducibility, power and validity of visual analogue scales in assessment of appetite sensations in single test meal studies. Int. J. Obes. Relat. Metab. Disord. 24, 38–48 (2000).
Soininen, P. et al. High-throughput serum NMR metabonomics for cost-effective holistic studies on systemic metabolism. Analyst 134, 1781 (2009).
Li, Y., Fromme, T., Schweizer, S., Schöttl, T. & Klingenspor, M. Taking control over intracellular fatty acid levels is essential for the analysis of thermogenic function in cultured primary brown and brite/beige adipocytes. EMBO Rep. 15, 1069–1076 (2014).
Rosenthal, R. in The Handbook of Research Synthesis (eds Cooper, H. et al.) 231–244 (Russell Sage Foundation, 1994).
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate—a practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B-Methodol. 57, 289–300 (1995).
Acknowledgements
The study was conducted within the Centre of Excellence into Cardiovascular and Metabolic Diseases supported by the Academy of Finland (grant no. 307402), University of Turku, Åbo Akademi University; and funded by the Instrumentarium Science Foundation (grant no. 190014) (S.L.), The Paulo Foundation (S.L.), Turku University Hospital Foundation (S.L.) and The Finnish Medical Foundation (grant no. 2985) (S.L.). TUM researchers were supported by the Else Kröner-Fresenius Stiftung (M.K.) and the German Research Foundation (grant no. DFG-CRC 1371: P13 (M.K.), Z02 (K. Steiger)).
Author information
Authors and Affiliations
Contributions
M.K., P.N., K.A.V. and S.L. conceived and designed the study. S.L., L.S. and M.L. performed the human experiments. K. Schnabl conceived the study design for the SCTR expression and regulation study and carried out animal and cell culture experiments. S.L., L.S., K. Schnabl, K.L., M.U.-D. and R.K analysed data. L.N., L.L.E. and T.V. helped with statistical analysis. O.E. and A.K.K. contributed to PET/CT data collection. K. Steiger conducted the immunohistochemical studies. L.N. conceived the fMRI study design. T.N. and M.T. provided BAT biopsies from study participants. M.B. and C.W. provided the analysis of fasted human BAT biopsies. K. Schnabl, Y.L and M.K edited the paper and contributed physiological aspects of secretin-activated brown fat in mouse studies complementing the human study. S.L. wrote the paper with input from authors. All authors read and approved the paper.
Corresponding author
Ethics declarations
Competing interests
M.K. and Y.L. are inventors on a patent application from the Technical University of Munich (publication no. WO/2017/20285; international application no. PCT/EP2017/062420) addressing the role of SCTR agonists and modulators in the regulation of energy homeostasis. This patent is based on the initial discovery that meal-induced secretin inhibits food intake, and this anorexigenic action of secretin depends on the activation of brown fat12. The remaining authors declare no competing interests.
Additional information
Peer review information Nature Metabolism thanks Andrew Gray, Marc Tittgemeyer and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Christoph Schmitt.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
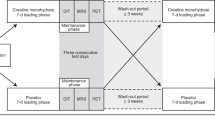
Extended Data Fig. 1 Enrolment and analysis.
CONSORT flowchart on the enrolment and analysis of study participants.
Extended Data Fig. 2 Visual analogue scale –questions.
Composite satiety score calculated with scale 1–100. Composite satiety score = satiety + fullness + (100 − prospective food consumption) + (100 − hunger)).
Extended Data Fig. 3 PET results and secretin receptor expression.
a. Muscle glucose uptake (GU) is increased by secretin compared to placebo. Data were analyzed with two-sided paired samples t-test and included n = 15 subjects. b. Secretin receptor expression in 79 human tissues, retrieved from http://ds.biogps.org/?dataset=GSE1133&gene=6344. Data are expressed as fold over the median (M), error bars show standard error. c. There is no significant change in perfusion after secretin infusion as compared to placebo. Data were analyzed with two-sided paired samples t-test and included n = 15 subjects.
Extended Data Fig. 4 Additional functional magnetic resonance and composite satiety score results.
a, Down-regulated brain reward anticipation response due to injection of secretin. This separate analysis (n = 14) including subjects with high movement artifacts was originally done. In the placebo condition, palatable food led to increased BOLD activity in brain reward circuits. This effect was diminished in the secretin condition. Interaction effect between food categories and conditions were found in the reward circuits. Significance level was FDR-corrected at p value 0.05. MFC = medial frontal cortex, CC = cingulate cortex, OCC = occipital cortex, Cau = caudate, MTG = middle temporal gyrus, Ins = insula, PCC = posterior cingulate cortex. b, composite satiety score area under the curve (AUC) was not increased postprandially, c nor for the entire study day (pre-prandial, prandial and postprandial phases together). Values are normalized, dividing by the value of the first time point. Data were analyzed by two-sided paired samples t-test, n = 14.
Extended Data Fig. 5 Additional positron emission tomography and secretin receptor gene expression results.
a, Brown adipose tissue (BAT) glucose uptake (GU) in cold and after secretin. There is no correlation between BAT GU in cold exposure and after secretin infusion. Data were analyzed by Spearman’s rank correlation in order to avoid assumptions around linearity of associations (n = 15). b, human secretin receptor (SCTR) expression on mRNA level, assessed by qPCR, in supraclavicular BAT is weakly associated with body-mass-index (BMI) in n = 14 fasted participants. Data were analyzed by Pearson’s correlation. c, SCTR expression analyzed by RNASeq does not correlate with BMI in n = 14 non-fasted participants. Data were analyzed by Pearson’s correlation.
Extended Data Fig. 6 Changes in circulating bile acids, hormones and carbohydrates.
a. Secretin did not increase serum bile acid levels compared to placebo. Values are normalized, dividing by the value of the first time point. Each timepoint was analyzed by two-sided, paired Wilcoxon signed-rank test. Median values and standard errors are shown on graph, n = 14. b. Serum hormone and c. carbohydrate heatmap secretin vs. placebo. Values are normalized, dividing by the value of the first time point. Each timepoint was analyzed by two-sided paired Wilcoxon signed-rank test, n = 12. * = p < 0.05, ** = p < 0.01, *** p < 0.001.
Supplementary information
Supplementary Information
Supplementary Tables 1–4
Source data
Source Data Fig. 2
Statistical source data.
Source Data Fig. 4
Statistical source data.
Source Data Fig. 5
Statistical source data.
Source Data Fig. 6
Statistical source data.
Source Data Extended Data Fig. 3
Statistical source data.
Source Data Extended Data Fig. 4
Statistical source data.
Source Data Extended Data Fig. 5
Statistical source data.
Source Data Extended Data Fig. 6
Statistical source data.
Rights and permissions
About this article
Cite this article
Laurila, S., Sun, L., Lahesmaa, M. et al. Secretin activates brown fat and induces satiation. Nat Metab 3, 798–809 (2021). https://doi.org/10.1038/s42255-021-00409-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s42255-021-00409-4
This article is cited by
-
Secretin-dependent signals in the ventromedial hypothalamus regulate energy metabolism and bone homeostasis in mice
Nature Communications (2024)
-
Hormone-induced body-brain interaction and the impact on cognition
European Journal of Nuclear Medicine and Molecular Imaging (2024)
-
Secretin: a hormone for HCO3− homeostasis
Pflügers Archiv - European Journal of Physiology (2024)
-
The intestine as an endocrine organ and the role of gut hormones in metabolic regulation
Nature Reviews Gastroenterology & Hepatology (2023)
-
Secretin modulates appetite via brown adipose tissue-brain axis
European Journal of Nuclear Medicine and Molecular Imaging (2023)