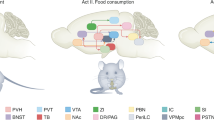
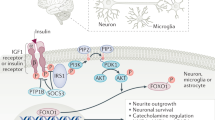
Abstract
Growing evidence implicates the brain in the regulation of both immediate fuel availability (for example, circulating glucose) and long-term energy stores (that is, adipose tissue mass). Rather than viewing the adipose tissue and glucose control systems separately, we suggest that the brain systems that control them are components of a larger, highly integrated, ‘fuel homeostasis’ control system. This conceptual framework, along with new insights into the organization and function of distinct neuronal systems, provides a context within which to understand how metabolic homeostasis is achieved in both basal and postprandial states. We also review evidence that dysfunction of the central fuel homeostasis system contributes to the close association between obesity and type 2 diabetes, with the goal of identifying more effective treatment options for these common metabolic disorders.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Change history
29 June 2021
A Correction to this paper has been published: https://doi.org/10.1038/s42255-021-00431-6
References
Beck, B. & Richy, S. Dietary modulation of ghrelin and leptin and gorging behavior after weight loss in the obese Zucker rat. J. Endocrinol. 202, 29–34 (2009).
Dulloo, A. G. & Calokatisa, R. Adaptation to low calorie intake in obese mice: contribution of a metabolic component to diminished energy expenditures during and after weight loss. Int. J. Obes. 15, 7–16 (1991).
MacLean, P. S. et al. Peripheral metabolic responses to prolonged weight reduction that promote rapid, efficient regain in obesity-prone rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 290, R1577–R1588 (2006).
Melby, C. L., Paris, H. L., Foright, R. M. & Peth, J. Attenuating the biologic drive for weight regain following weight loss: must what goes down always go back up? Nutrients 9, 468 (2017).
Morton, G. J., Cummings, D. E., Baskin, D. G., Barsh, G. S. & Schwartz, M. W. Central nervous system control of food intake and body weight. Nature 443, 289–295 (2006).
Williams, G. et al. The hypothalamus and the control of energy homeostasis different circuits, different purposes. Physiol. Behav. 74, 683–701 (2001).
Alonge, K. M., D’Alessio, D. A. & Schwartz, M. W. Brain control of blood glucose levels: implications for the pathogenesis of type 2 diabetes. Diabetologia https://doi.org/10.1007/s00125-020-05293-3 (2020).
Rickels, M. R. & Robertson, R. P. Pancreatic islet transplantation in humans: recent progress and future directions. Endocr. Rev. 40, 631–668 (2019).
Brown, J. M., Scarlett, J. M. & Schwartz, M. W. Rethinking the role of the brain in glucose homeostasis and diabetes pathogenesis. J. Clin. Invest. 129, 3035–3037 (2019).
Faber, C. L., Deem, J. D., Campos, C. A., Taborsky, G. J. & Morton, G. J. CNS control of the endocrine pancreas. Diabetologia 63, 2086–2094 (2020).
Smyth, S. & Heron, A. Diabetes and obesity: the twin epidemics. Nat. Med. 12, 75–80 (2006).
Considine, R. V. et al. Serum immunoreactive-leptin concentrations in normal-weight and obese humans. N. Engl. J. Med. 334, 292–295 (1996).
Frederich, R. C. et al. Leptin levels reflect body lipid content in mice: evidence for diet-induced resistance to leptin action. Nat. Med. 1, 1311–1314 (1995).
Hummel, K. P., Dickie, M. M. & Coleman, D. L. Diabetes, a new mutation in the mouse. Science 153, 1127–1128 (1966).
Tartaglia, L. A. et al. Identification and expression cloning of a leptin receptor, OB-R. Cell 83, 1263–1271 (1995).
Ingalls, A. M., Dickie, M. M. & Snell, G. D. Obese, a new mutation in the house mouse. J. Hered. 41, 317–318 (1950).
Kennedy, G. C. The role of depot fat in the hypothalamic control of food intake in the rat. Proc. R. Soc. Lond. B Biol. Sci. 140, 578–596 (1953).
Butler, P. C. et al. Effects of meal ingestion on plasma amylin concentration in NIDDM and nondiabetic humans. Diabetes 39, 752–756 (1990).
Elliott, R. M. et al. Glucagon-like peptide-1(7–36)amide and glucose-dependent insulinotropic polypeptide secretion in response to nutrient ingestion in man: acute post-prandial and 24-h secretion patterns. J. Endocrinol. 138, 159–166 (1993).
GIBBS, J., YOUNG, R. C. & SMITH, G. P. Cholecystokinin elicits satiety in rats with open gastric fistulas. Nature 245, 323–325 (1973).
Strubbe, J. & Steffens, A. Rapid insulin release after ingestion of a meal in the unanesthetized rat. Am. J. Physiol. 229, 1019–1022 (1975).
Berthoud, H.-R., Lynn, P. A. & Blackshaw, L. A. Vagal and spinal mechanosensors in the rat stomach and colon have multiple receptive fields. Am. J. Physiol. 280, R1371–R1381 (2001).
Phillips, R. J. & Powley, T. L. Tension and stretch receptors in gastrointestinal smooth muscle: re-evaluating vagal mechanoreceptor electrophysiology. Brain Res. Rev. 34, 1–26 (2000).
Frecka, J. M. & Mattes, R. D. Possible entrainment of ghrelin to habitual meal patterns in humans. Am. J. Physiol. 294, G699–G707 (2008).
Wren, A. M. et al. Ghrelin enhances appetite and increases food intake in humans. J. Clin. Endocrinol. Metab. 86, 5992 (2001).
Johnen, H. et al. Tumor-induced anorexia and weight loss are mediated by the TGF-β superfamily cytokine MIC-1. Nat. Med. 13, 1333–1340 (2007).
Tsai, V. W. W., Husaini, Y., Sainsbury, A., Brown, D. A. & Breit, S. N. The MIC-1/GDF15-GFRAL pathway in energy homeostasis: implications for obesity, cachexia and other associated diseases. Cell Metab. 28, 353–368 (2018).
Borner, T. et al. GDF15 induces anorexia through nausea and emesis. Cell Metab. https://doi.org/10.1016/j.cmet.2019.12.004 (2020).
Kleinert, M. et al. Exercise increases circulating GDF15 in humans. Mol. Metab. 9, 187–191 (2018).
Patel, S. et al. GDF15 provides an endocrine signal of nutritional stress in mice and humans. Cell Metab. 29, 707–718 (2019).
Emmerson, P. J. et al. The metabolic effects of GDF15 are mediated by the orphan receptor GFRAL. Nat. Med. 23, 1215–1219 (2017).
Hsu, J.-Y. et al. Non-homeostatic body weight regulation through a brainstem-restricted receptor for GDF15. Nature 550, 255–259 (2017).
Mullican, S. E. et al. GFRAL is the receptor for GDF15 and the ligand promotes weight loss in mice and nonhuman primates. Nat. Med. 23, 1150–1157 (2017).
Yang, L. et al. GFRAL is the receptor for GDF15 and is required for the anti-obesity effects of the ligand. Nat. Med. 23, 1158–1166 (2017).
Wischhusen, J., Melero, I. & Fridman, W. H. Growth/differentiation factor-15 (GDF-15): from biomarker to novel targetable immune checkpoint. Front. Immunol. 11, 951 (2020).
Celi, F. S. et al. Minimal changes in environmental temperature result in a significant increase in energy expenditure and changes in the hormonal homeostasis in healthy adults. Eur. J. Endocrinol. 163, 863–872 (2010).
Langeveld, M. et al. Mild cold effects on hunger, food intake, satiety and skin temperature in humans. Endocr. Connect. 5, 65–73 (2016).
Macari, M., Dauncey, M. J. & Ingram, D. L. Changes in food intake in response to alterations in the ambient temperature: modifications by previous thermal and nutritional experience. Pflügers Arch. 396, 231–237 (1983).
Seeley, R. J. et al. Behavioral, endocrine, and hypothalamic responses to involuntary overfeeding. Am. J. Physiol. 271, R819–R823 (1996).
Ravussin, Y. et al. Evidence for a non-leptin system that defends against weight gain in overfeeding. Cell Metab. 28, 289–299 (2018).
White, C. L., Purpera, M. N., Ballard, K. & Morrison, C. D. Decreased food intake following overfeeding involves leptin-dependent and leptin-independent mechanisms. Physiol. Behav. 100, 408–416 (2010).
Coleman, D. L. Effects of parabiosis of obese with diabetes and normal mice. Diabetologia 9, 294–298 (1973).
Coleman, D. L. Obese and diabetes: two mutant genes causing diabetes–obesity syndromes in mice. Diabetologia 14, 141–148 (1978).
D’Alessio, D. A., Kieffer, T. J., Taborsky, G. J. & Havel, P. J. Activation of the parasympathetic nervous system is necessary for normal meal-induced insulin secretion in rhesus macaques. J. Clin. Endocrinol. Metab. 86, 1253–1259 (2001).
Havel, P. J., Veith, R. C., Dunning, B. E. & Taborsky, G. J. Pancreatic noradrenergic nerves are activated by neuroglucopenia but not by hypotension or hypoxia in the dog. Evidence for stress-specific and regionally selective activation of the sympathetic nervous system. J. Clin. Invest. 82, 1538–1545 (1988).
Lerner, R. L. & Porte, D. Studies of secretin-stimulated insulin responses in man. J. Clin. Invest. 51, 2205–2210 (1972).
Rosario, W. et al. The brain-to-pancreatic islet neuronal map reveals differential glucose regulation from distinct hypothalamic regions. Diabetes 65, 2711–2723 (2016).
Thorens, B. Neural regulation of pancreatic islet cell mass and function. Diabetes Obes. Metab. 16, 87–95 (2014).
Berthoud, H. R., Bereiter, D. A., Trimble, E. R., Siegel, E. G. & Jeanrenaud, B. Cephalic phase, reflex insulin secretion neuroanatomical and physiological characterization. Diabetologia 20, 393–401 (1981).
Teff, K. L. How neural mediation of anticipatory and compensatory insulin release helps us tolerate food. Physiol. Behav. 103, 44–50 (2011).
Ahrén, B. Autonomic regulation of islet hormone secretion–implications for health and disease. Diabetologia 43, 393–410 (2000).
Brochu, M. et al. Visceral adipose tissue is an independent correlate of glucose disposal in older obese postmenopausal women. J. Clin. Endocrinol. Metab. 85, 2378–2384 (2000).
Kim, K. & Park, S. M. Association of muscle mass and fat mass with insulin resistance and the prevalence of metabolic syndrome in Korean adults: a cross-sectional study. Sci. Rep. 8, 2703 (2018).
Faerch, K. et al. Natural history of insulin sensitivity and insulin secretion in the progression from normal glucose tolerance to impaired fasting glycemia and impaired glucose tolerance: the Inter99 study. Diabetes Care 32, 439–444 (2009).
Retnakaran, R. et al. Hyperbolic relationship between insulin secretion and sensitivity on oral glucose tolerance test. Obesity 16, 1901–1907 (2008).
Walton, C., Godsland, I. F., Proudler, A. J., Felton, C. V. & Wynn, V. Effect of body mass index and fat distribution on insulin sensitivity, secretion, and clearance in nonobese healthy men. J. Clin. Endocrinol. Metab. 75, 170–175 (1992).
Morton, G. J. et al. Evidence that the sympathetic nervous system elicits rapid, coordinated, and reciprocal adjustments of insulin secretion and insulin sensitivity during cold exposure. Diabetes 66, 823–834 (2017).
Muta, K. et al. Glucoregulatory responses to hypothalamic preoptic area cooling. Brain Res. 1710, 136–145 (2019).
Dhillon, H. et al. Leptin directly activates SF1 neurons in the VMH, and this action by leptin is required for normal body-weight homeostasis. Neuron 49, 191–203 (2006).
Flak, J. N. et al. Ventromedial hypothalamic nucleus neuronal subset regulates blood glucose independently of insulin. J. Clin. Invest. 130, 2943–2952 (2020).
German, J. P. et al. Leptin activates a novel CNS mechanism for insulin-independent normalization of severe diabetic hyperglycemia. Endocrinology 152, 394–404 (2011).
Meek, T. H. et al. Leptin action in the ventromedial hypothalamic nucleus is sufficient, but not necessary, to normalize diabetic hyperglycemia. Endocrinology 154, 3067–3076 (2013).
Balland, E. et al. Leptin signaling in the arcuate nucleus reduces insulin’s capacity to suppress hepatic glucose production in obese Mice. Cell Rep. 26, 346–355 (2019).
Hashikawa, K. et al. Esr1+ cells in the ventromedial hypothalamus control female aggression. Nat. Neurosci. 20, 1580–1590 (2017).
Krause, W. C. & Ingraham, H. A. Origins and functions of the ventrolateral VMH: a complex neuronal cluster orchestrating sex differences in metabolism and behavior. in Sex and Gender Factors Affecting Metabolic Homeostasis, Diabetes and Obesity (ed. Mauvais-Jarvis, F.) 1043, 199–213 (Springer International Publishing, 2017).
Vander Tuig, J. G., Knehans, A. W. & Romsos, D. R. Reduced sympathetic nervous system activity in rats with ventromedial hypothalamic lesions. Life Sci. 30, 913–920 (1982).
Kunwar, P. S. et al. Ventromedial hypothalamic neurons control a defensive emotion state. eLife 4, e06633 (2015).
Minokoshi, Y., Haque, M. S. & Shimazu, T. Microinjection of leptin into the ventromedial hypothalamus increases glucose uptake in peripheral tissues in rats. Diabetes 48, 287–291 (1999).
Toda, C. et al. Extracellular signal-regulated kinase in the ventromedial hypothalamus mediates leptin-induced glucose uptake in red-type skeletal muscle. Diabetes 62, 2295–2307 (2013).
Noble, E. E., Billington, C. J., Kotz, C. M. & Wang, C. Oxytocin in the ventromedial hypothalamic nucleus reduces feeding and acutely increases energy expenditure. Am. J. Physiol. 307, R737–R745 (2014).
Gavini, C. K., Jones, W. C. & Novak, C. M. Ventromedial hypothalamic melanocortin receptor activation: regulation of activity energy expenditure and skeletal muscle thermogenesis: VMH and skeletal muscle thermogenesis. J. Physiol. 594, 5285–5301 (2016).
Meek, T. H. et al. Functional identification of a neurocircuit regulating blood glucose. Proc. Natl Acad. Sci. USA 113, E2073–E2082 (2016).
Borg, W. P., Sherwin, R. S., During, M. J., Borg, M. A. & Shulman, G. I. Local ventromedial hypothalamus glucopenia triggers counterregulatory hormone release. Diabetes 44, 180–184 (1995).
Furigo, I. C. et al. Growth hormone enhances the recovery of hypoglycemia via ventromedial hypothalamic neurons. FASEB J. https://doi.org/10.1096/fj.201901315R (2019).
Garfield, A. S. et al. A parabrachial-hypothalamic cholecystokinin neurocircuit controls counterregulatory responses to hypoglycemia. Cell Metab. 20, 1030–1037 (2014).
Sabatini, P. V. et al. tTARGIT AAVs mediate the sensitive and flexible manipulation of intersectional neuronal populations in mice. eLife 10, e66835 (2021).
Burdakov, D. & González, J. A. Physiological functions of glucose-inhibited neurones. Acta Physiol. 195, 71–78 (2009).
Shimazu, T. & Minokoshi, Y. Systemic glucoregulation by glucose-sensing neurons in the ventromedial hypothalamic nucleus (VMH). J. Endocr. Soc. 1, 449–459 (2017).
Hirschberg, P. R., Sarkar, P., Teegala, S. B. & Routh, V. H. Ventromedial hypothalamus glucose‐inhibited neurones: A role in glucose and energy homeostasis? J. Neuroendocrinol. 32, e12773 (2020).
Flak, J. N. et al. Leptin-inhibited PBN neurons enhance responses to hypoglycemia in negative energy balance. Nat. Neurosci. 17, 1744–1750 (2014).
Dunn-Meynell, A. A. et al. Relationship among brain and blood glucose levels and spontaneous and glucoprivic feeding. J. Neurosci. 29, 7015–7022 (2009).
Fioramonti, X., Chrétien, C., Leloup, C. & Pénicaud, L. Recent advances in the cellular and molecular mechanisms of hypothalamic neuronal glucose detection. Front. Physiol. 8, 875 (2017).
Hwang, J. J. et al. Blunted rise in brain glucose levels during hyperglycemia in adults with obesity and T2DM. JCI Insight 2, e95913 (2017).
Williams, E. K. et al. Sensory neurons that detect stretch and nutrients in the digestive system. Cell 166, 209–221 (2016).
Donovan, C. M. Portal vein glucose sensing. Diabetes Nutr. Metab. 15, 308–312 (2002).
Chan, O. & Sherwin, R. S. Is there cross talk between portal and hypothalamic glucose-sensing circuits? Diabetes 63, 2617–2619 (2014).
Jokiaho, A. J., Donovan, C. M. & Watts, A. G. The rate of fall of blood glucose determines the necessity of forebrain-projecting catecholaminergic neurons for male rat sympathoadrenal responses. Diabetes 63, 2854–2865 (2014).
Campfield, L. A., Smith, F. J., Guisez, Y., Devos, R. & Burn, P. Recombinant mouse OB protein: evidence for a peripheral signal linking adiposity and central neural networks. Science 269, 546–549 (1995).
Schwartz, M. W. et al. Specificity of leptin action on elevated blood glucose levels and hypothalamic neuropeptide Y gene expression in ob/ob mice. Diabetes 45, 531–535 (1996).
van den Hoek, A. M. et al. Leptin deficiency per se dictates body composition and insulin action in ob/ob mice. J. Neuroendocrinol. 20, 120–127 (2008).
Ahima, R. S. et al. Role of leptin in the neuroendocrine response to fasting. Nature 382, 250–252 (1996).
Schwartz, M. W., Seeley, R. J., Campfield, L. A., Burn, P. & Baskin, D. G. Identification of targets of leptin action in rat hypothalamus. J. Clin. Invest. 98, 1101–1106 (1996).
Ahrén, B., Månsson, S., Gingerich, R. L. & Havel, P. J. Regulation of plasma leptin in mice: influence of age, high-fat diet, and fasting. Am. J. Physiol. 273, R113–R120 (1997).
Clément, K. et al. A mutation in the human leptin receptor gene causes obesity and pituitary dysfunction. Nature 392, 398–401 (1998).
Montague, C. T. et al. Congenital leptin deficiency is associated with severe early-onset obesity in humans. Nature 387, 903–908 (1997).
Zhang, Y. et al. Positional cloning of the mouse obese gene and its human homologue. Nature 372, 425–432 (1994).
D’souza, A. M., Asadi, A., Johnson, J. D., Covey, S. D. & Kieffer, T. J. Leptin deficiency in rats results in hyperinsulinemia and impaired glucose homeostasis. Endocrinology 155, 1268–1279 (2014).
Pelleymounter, M. A. et al. Effects of the obese gene product on body weight regulation in ob/ob mice. Science 269, 540–543 (1995).
Oral, E. A. et al. Leptin-replacement therapy for lipodystrophy. N. Engl. J. Med. 346, 570–578 (2002).
Petersen, K. F. et al. Leptin reverses insulin resistance and hepatic steatosis in patients with severe lipodystrophy. J. Clin. Invest. 109, 1345–1350 (2002).
Shimomura, I., Hammer, R. E., Ikemoto, S., Brown, M. S. & Goldstein, J. L. Leptin reverses insulin resistance and diabetes mellitus in mice with congenital lipodystrophy. Nature 401, 73–76 (1999).
Havel, P. J. et al. Marked and rapid decreases of circulating leptin in streptozotocin diabetic rats: reversal by insulin. Am. J. Physiol. 274, R1482–R1491 (1998).
Chinookoswong, N., Wang, J. L. & Shi, Z. Q. Leptin restores euglycemia and normalizes glucose turnover in insulin-deficient diabetes in the rat. Diabetes 48, 1487–1492 (1999).
German, J. P. et al. Leptin deficiency causes insulin resistance induced by uncontrolled diabetes. Diabetes 59, 1626–1634 (2010).
Hidaka, S. et al. Chronic central leptin infusion restores hyperglycemia independent of food intake and insulin level in streptozotocin-induced diabetic rats. FASEB J. 16, 509–518 (2002).
Fujikawa, T. et al. Leptin engages a hypothalamic neurocircuitry to permit survival in the absence of insulin. Cell Metab. 18, 431–444 (2013).
Fujikawa, T., Chuang, J.-C., Sakata, I., Ramadori, G. & Coppari, R. Leptin therapy improves insulin-deficient type 1 diabetes by CNS-dependent mechanisms in mice. Proc. Natl Acad. Sci. USA 107, 17391–17396 (2010).
Morton, G. J., Meek, T. H., Matsen, M. E. & Schwartz, M. W. Evidence against hypothalamic–pituitary–adrenal axis suppression in the antidiabetic action of leptin. J. Clin. Invest. 125, 4587–4591 (2015).
Perry, R. J. et al. Leptin reverses diabetes by suppression of the hypothalamic–pituitary–adrenal axis. Nat. Med. 20, 759–763 (2014).
Alix, P. M., Guebre-Egziabher, F. & Soulage, C. O. Leptin as an uremic toxin: deleterious role of leptin in chronic kidney disease. Biochimie 105, 12–21 (2014).
Mittendorfer, B. et al. Recombinant human leptin treatment does not improve insulin action in obese subjects with type 2 diabetes. Diabetes 60, 1474–1477 (2011).
Moon, H.-S. et al. Efficacy of metreleptin in obese patients with type 2 diabetes: cellular and molecular pathways underlying leptin tolerance. Diabetes 60, 1647–1656 (2011).
Hanai, K. et al. Central action of acidic fibroblast growth factor in feeding regulation. Am. J. Physiol. 256, R217–R223 (1989).
Oomura, Y. et al. A new brain glucosensor and its physiological significance. Am. J. Clin. Nutr. 55, 278S–282S (1992).
Sasaki, K. et al. Effects of fibroblast growth factors and related peptides on food intake by rats. Physiol. Behav. 56, 211–218 (1994).
Suh, J. M. et al. Endocrinization of FGF1 produces a neomorphic and potent insulin sensitizer. Nature 513, 436–439 (2014).
Tennant, K. G., Lindsley, S. R., Kirigiti, M. A., True, C. & Kievit, P. Central and peripheral administration of fibroblast growth factor 1 improves pancreatic islet insulin secretion in diabetic mouse models. Diabetes 68, 1462–1472 (2019).
Scarlett, J. M. et al. Central injection of fibroblast growth factor 1 induces sustained remission of diabetic hyperglycemia in rodents. Nat. Med. 22, 800–806 (2016).
Brown, J. M. et al. The hypothalamic arcuate nucleus–median eminence is a target for sustained diabetes remission induced by fibroblast growth factor 1. Diabetes 68, 1054–1061 (2019).
Scarlett, J. M. et al. Peripheral mechanisms mediating the sustained antidiabetic action of FGF1 in the brain. Diabetes 68, 654–664 (2019).
Coppari, R. et al. The hypothalamic arcuate nucleus: a key site for mediating leptin’s effects on glucose homeostasis and locomotor activity. Cell Metab. 1, 63–72 (2005).
Berglund, E. D. et al. Direct leptin action on POMC neurons regulates glucose homeostasis and hepatic insulin sensitivity in mice. J. Clin. Invest. 122, 1000–1009 (2012).
Gonçalves, G. H. M., Li, W., Garcia, A. V. C. -G., Figueiredo, M. S. & Bjørbæk, C. Hypothalamic agouti-related peptide neurons and the central melanocortin system are crucial mediators of leptin’s antidiabetic actions. Cell Rep. 7, 1093–1103 (2014).
Huo, L. et al. Leptin-dependent control of glucose balance and locomotor activity by POMC neurons. Cell Metab. 9, 537–547 (2009).
Bentsen, M. A. et al. Transcriptomic analysis links diverse hypothalamic cell types to fibroblast growth factor 1-induced sustained diabetes remission. Nat. Commun. 11, 4458 (2020).
Alonge, K. M. et al. Hypothalamic perineuronal net assembly is required for sustained diabetes remission induced by fibroblast growth factor 1 in rats. Nat. Metab. 2, 1025–1033 (2020).
Claret, M. et al. Deletion of Lkb1 in pro-opiomelanocortin neurons impairs peripheral glucose homeostasis in mice. Diabetes 60, 735–745 (2011).
Smith, M. A. et al. Ribosomal S6K1 in POMC and AgRP neurons regulates glucose homeostasis but not feeding behavior in mice. Cell Rep. 11, 335–343 (2015).
Chhabra, K. H. et al. Hypothalamic POMC deficiency improves glucose tolerance despite insulin resistance by increasing glycosuria. Diabetes 65, 660–672 (2016).
Chhabra, K. H. et al. Reduced renal sympathetic nerve activity contributes to elevated glycosuria and improved glucose tolerance in hypothalamus-specific Pomc knockout mice. Mol. Metab. 6, 1274–1285 (2017).
Shin, A. C. et al. Insulin receptor signaling in POMC, but Not AgRP, neurons controls adipose tissue insulin action. Diabetes 66, 1560–1571 (2017).
Ren, H. et al. FoxO1 target Gpr17 activates AgRP neurons to regulate food intake. Cell 149, 1314–1326 (2012).
Xu, A. W. et al. PI3K integrates the action of insulin and leptin on hypothalamic neurons. J. Clin. Invest. 115, 951–958 (2005).
Könner, A. C. et al. Insulin action in AgRP-expressing neurons is required for suppression of hepatic glucose production. Cell Metab. 5, 438–449 (2007).
Aponte, Y., Atasoy, D. & Sternson, S. M. AGRP neurons are sufficient to orchestrate feeding behavior rapidly and without training. Nat. Neurosci. 14, 351–355 (2011).
Krashes, M. J. et al. Rapid, reversible activation of AgRP neurons drives feeding behavior in mice. J. Clin. Invest. 121, 1424–1428 (2011).
Engström Ruud, L., Pereira, M. M. A., de Solis, A. J., Fenselau, H. & Brüning, J. C. NPY mediates the rapid feeding and glucose metabolism regulatory functions of AgRP neurons. Nat. Commun. 11, 442 (2020).
van de Wall, E. et al. Collective and individual functions of leptin receptor modulated neurons controlling metabolism and ingestion. Endocrinology 149, 1773–1785 (2008).
Deem, J. D. et al. Cold-induced hyperphagia requires AgRP neuron activation in mice. eLife 9, e58764 (2020).
Liu, T. et al. Fasting activation of AgRP neurons requires NMDA receptors and involves spinogenesis and increased excitatory tone. Neuron 73, 511–522 (2012).
Mizuno, T. M. et al. Hypothalamic pro-opiomelanocortin mRNA is reduced by fasting in ob/ob and db/db mice, but is stimulated by leptin. Diabetes 47, 294–297 (1998).
Takahashi, K. A. & Cone, R. D. Fasting induces a large, leptin-dependent increase in the intrinsic action potential frequency of orexigenic arcuate nucleus neuropeptide Y/agouti-related protein neurons. Endocrinology 146, 1043–1047 (2005).
Kolaczynski, J. W., Ohannesian, J. P., Considine, R. V., Marco, C. C. & Caro, J. F. Response of leptin to short-term and prolonged overfeeding in humans. J. Clin. Endocrinol. Metab. 81, 4162–4165 (1996).
Colao, A. et al. Managing hyperglycemia in patients with Cushing’s disease treated with pasireotide: medical expert recommendations. Pituitary 17, 180–186 (2014).
Hsia, D. S., Grove, O. & Cefalu, W. T. An update on sodium-glucose co-transporter-2 inhibitors for the treatment of diabetes mellitus. Curr. Opin. Endocrinol. Diabetes Obes. 24, 73–79 (2017).
Scaroni, C., Zilio, M., Foti, M. & Boscaro, M. Glucose metabolism abnormalities in Cushing syndrome: from molecular basis to clinical management. Endocr. Rev. 38, 189–219 (2017).
Shi, J. et al. Review: traumatic brain injury and hyperglycemia, a potentially modifiable risk factor. Oncotarget 7, 71052–71061 (2016).
Farooqi, I. S. et al. Partial leptin deficiency and human adiposity. Nature 414, 34–35 (2001).
Flak, J. N. & Myers, M. G. Minireview: CNS mechanisms of leptin action. Mol. Endocrinol. 30, 3–12 (2016).
Bal, N. C. et al. Both brown adipose tissue and skeletal muscle thermogenesis processes are activated during mild to severe cold adaptation in mice. J. Biol. Chem. 292, 16616–16625 (2017).
Morrison, S. F., Madden, C. J. & Tupone, D. Central neural regulation of brown adipose tissue thermogenesis and energy expenditure. Cell Metab. 19, 741–756 (2014).
Carey, M. et al. Central K ATP channels modulate glucose effectiveness in humans and rodents. Diabetes 69, 1140–1148 (2020).
Timper, K. & Brüning, J. C. Hypothalamic circuits regulating appetite and energy homeostasis: pathways to obesity. Dis. Model. Mech. 10, 679–689 (2017).
Barrett, P., Mercer, J. G. & Morgan, P. J. Preclinical models for obesity research. Dis. Model. Mech. 9, 1245–1255 (2016).
Deem, J. D., Muta, K., Scarlett, J. M., Morton, G. J. & Schwartz, M. W. How should we think about the role of the brain in glucose homeostasis and diabetes? Diabetes 66, 1758–1765 (2017).
Xu, J. et al. Genetic identification of leptin neural circuits in energy and glucose homeostases. Nature 556, 505–509 (2018).
Halban, P. A. et al. β-cell failure in type 2 diabetes: postulated mechanisms and prospects for prevention and treatment. Diabetes Care 37, 1751–1758 (2014).
Hermann, G. M. et al. Neonatal catch-up growth increases diabetes susceptibility but improves behavioral and cardiovascular outcomes of low birth weight male mice. Pediatr. Res 66, 53–58 (2009).
Perng, W., Oken, E. & Dabelea, D. Developmental overnutrition and obesity and type 2 diabetes in offspring. Diabetologia 62, 1779–1788 (2019).
Schellong, K. et al. Sex-specific epigenetic alterations of the hypothalamic Agrp–Pomc system do not explain ‘diabesity’ in the offspring of high-fat diet (HFD) overfed maternal rats. J. Nutr. Biochem. 75, 108257 (2020).
de Kloet, A. D. & Herman, J. P. Fat–brain connections: adipocyte glucocorticoid control of stress and metabolism. Front. Neuroendocrinol. 48, 50–57 (2018).
Magomedova, L. & Cummins, C. L. Glucocorticoids and metabolic control. Handb. Exp. Pharmacol. 233, 73–93 (2016).
Warner, A. & Mittag, J. Thyroid hormone and the central control of homeostasis. J. Mol. Endocrinol. 49, R29–R35 (2012).
Carrageta, D. F., Oliveira, P. F., Alves, M. G. & Monteiro, M. P. Obesity and male hypogonadism: tales of a vicious cycle. Obes. Rev. 20, 1148–1158 (2019).
López, M. & Tena-Sempere, M. Estradiol effects on hypothalamic AMPK and BAT thermogenesis: a gateway for obesity treatment? Pharmacol. Ther. 178, 109–122 (2017).
Mauvais-Jarvis, F., Clegg, D. J. & Hevener, A. L. The role of estrogens in control of energy balance and glucose homeostasis. Endocr. Rev. 34, 309–338 (2013).
Kooijman, S., van den Heuvel, J. K. & Rensen, P. C. N. Neuronal control of brown fat activity. Trends Endocrinol. Metab. 26, 657–668 (2015).
Bartness, T. J., Vaughan, C. H. & Song, C. K. Sympathetic and sensory innervation of brown adipose tissue. Int. J. Obes. 34, S36–S42 (2010).
McNeill, B. T., Morton, N. M. & Stimson, R. H. Substrate utilization by brown adipose tissue: what’s hot and what’s not? Front. Endocrinol. 11, 571659 (2020).
Morrison, S. F. & Nakamura, K. Central mechanisms for thermoregulation. Annu. Rev. Physiol. 81, 285–308 (2019).
Rossi, M. A. & Stuber, G. D. Overlapping brain circuits for homeostatic and hedonic feeding. Cell Metab. 27, 42–56 (2018).
Cone, R. D. Anatomy and regulation of the central melanocortin system. Nat. Neurosci. 8, 571–578 (2005).
Sutton, A. K., Myers, M. G. & Olson, D. P. The role of PVH circuits in leptin action and energy balance. Annu Rev. Physiol. 78, 207–221 (2016).
Fry, M. & Ferguson, A. V. The sensory circumventricular organs: brain targets for circulating signals controlling ingestive behavior. Physiol. Behav. 91, 413–423 (2007).
Ollmann, M. M. et al. Antagonism of central melanocortin receptors in vitro and in vivo by agouti-related protein. Science 278, 135–138 (1997).
Schwartz, M. W. et al. Leptin increases hypothalamic pro-opiomelanocortin mRNA expression in the rostral arcuate nucleus. Diabetes 46, 2119–2123 (1997).
Zhan, C. et al. Acute and long-term suppression of feeding behavior by POMC neurons in the brainstem and hypothalamus, respectively. J. Neurosci. 33, 3624–3632 (2013).
Baskin, D. G., Breininger, J. F. & Schwartz, M. W. Leptin receptor mRNA identifies a subpopulation of neuropeptide Y neurons activated by fasting in rat hypothalamus. Diabetes 48, 828–833 (1999).
Erickson, J. C., Hollopeter, G. & Palmiter, R. D. Attenuation of the obesity syndrome of ob/ob mice by the loss of neuropeptide Y. Science 274, 1704–1707 (1996).
Winkler, Z. et al. Hypoglycemia-activated hypothalamic microglia impairs glucose counterregulatory responses. Sci. Rep. 9, 6224 (2019).
Luquet, S. NPY/AgRP neurons are essential for feeding in adult mice but can be ablated in neonates. Science 310, 683–685 (2005).
Gropp, E. et al. Agouti-related peptide-expressing neurons are mandatory for feeding. Nat. Neurosci. 8, 1289–1291 (2005).
Zhang, X. & van den Pol, A. N. Dopamine/tyrosine hydroxylase neurons of the hypothalamic arcuate nucleus release GABA, communicate with dopaminergic and other arcuate neurons, and respond to dynorphin, met-enkephalin, and oxytocin. J. Neurosci. 35, 14966–14982 (2015).
Zhang, X. & van den Pol, A. N. Hypothalamic arcuate nucleus tyrosine hydroxylase neurons play orexigenic role in energy homeostasis. Nat. Neurosci. 19, 1341–1347 (2016).
Atasoy, D., Betley, J. N., Su, H. H. & Sternson, S. M. Deconstruction of a neural circuit for hunger. Nature 488, 172–177 (2012).
Garfield, A. S. et al. A neural basis for melanocortin-4 receptor-regulated appetite. Nat. Neurosci. 18, 863–871 (2015).
Li, M. M. et al. The paraventricular hypothalamus regulates satiety and prevents obesity via two genetically distinct circuits. Neuron 102, 653–667 (2019).
Leinninger, G. M. et al. Leptin action via neurotensin neurons controls orexin, the mesolimbic dopamine system and energy balance. Cell Metab. 14, 313–323 (2011).
Hawke, Z. et al. PACAP neurons in the hypothalamic ventromedial nucleus are targets of central leptin signaling. J. Neurosci. 29, 14828–14835 (2009).
Patterson, C. M., Leshan, R. L., Jones, J. C. & Myers, M. G. Molecular mapping of mouse brain regions innervated by leptin receptor-expressing cells. Brain Res. 1378, 18–28 (2011).
Rezai-Zadeh, K. et al. Leptin receptor neurons in the dorsomedial hypothalamus are key regulators of energy expenditure and body weight, but not food intake. Mol. Metab. 3, 681–693 (2014).
Chou, T. C. et al. Critical role of dorsomedial hypothalamic nucleus in a wide range of behavioral circadian rhythms. J. Neurosci. 23, 10691–10702 (2003).
Grill, H. J. & Hayes, M. R. Hindbrain neurons as an essential hub in the neuroanatomically distributed control of energy balance. Cell Metab. 16, 296–309 (2012).
Zhang, C. et al. Area postrema cell types that mediate nausea-associated behaviors. Neuron https://doi.org/10.1016/j.neuron.2020.11.010 (2020).
Ludwig, M. Q. et al. A genetic map of the mouse dorsal vagal complex and its role in obesity. Nat. Metab. https://doi.org/10.1038/s42255-021-00363-1 (2021).
Sabatini, P. V. et al. GFRAL-expressing neurons suppress food intake via aversive pathways. Proc. Natl Acad. Sci. USA 118, e2021357118 (2021).
Bai, L. et al. Genetic identification of vagal sensory neurons that control feeding. Cell 179, 1129–1143 (2019).
Kaelberer, M. M., Rupprecht, L. E., Liu, W. W., Weng, P. & Bohórquez, D. V. Neuropod cells: the emerging biology of gut–brain sensory transduction. Annu Rev. Neurosci. 43, 337–353 (2020).
Huo, L., Maeng, L., Bjørbaek, C. & Grill, H. J. Leptin and the control of food intake: neurons in the nucleus of the solitary tract are activated by both gastric distension and leptin. Endocrinology 148, 2189–2197 (2007).
Huo, L., Gamber, K. M., Grill, H. J. & Bjørbaek, C. Divergent leptin signaling in proglucagon neurons of the nucleus of the solitary tract in mice and rats. Endocrinology 149, 492–497 (2008).
Tsai, V. W.-W. et al. GDF15 mediates adiposity resistance through actions on GFRAL neurons in the hindbrain AP/NTS. Int J. Obes. 43, 2370–2380 (2019).
Alhadeff, A. L. et al. Endogenous glucagon-like peptide-1 receptor signaling in the nucleus tractus solitarius is required for food intake control. Neuropsychopharmacology 42, 1471–1479 (2017).
Hayes, M. R. et al. Endogenous leptin signaling in the caudal nucleus tractus solitarius and area postrema is required for energy balance regulation. Cell Metab. 23, 744 (2016).
Cheng, W. et al. Calcitonin receptor neurons in the mouse nucleus tractus solitarius control energy balance via the non-aversive suppression of feeding. Cell Metab. 31, 301–312 (2020).
Cheng, W. et al. Leptin receptor–expressing nucleus tractus solitarius neurons suppress food intake independently of GLP1 in mice. JCI Insight 5, e134359 (2020).
Wadden, T. A. et al. Effect of subcutaneous semaglutide vs placebo as an adjunct to intensive behavioral therapy on body weight in adults with overweight or obesity: the STEP 3 randomized clinical trial. JAMA https://doi.org/10.1001/jama.2021.1831 (2021).
Palmiter, R. D. The parabrachial nucleus: CGRP neurons function as a general alarm. Trends Neurosci. 41, 280–293 (2018).
Chen, J. Y., Campos, C. A., Jarvie, B. C. & Palmiter, R. D. Parabrachial CGRP neurons establish and sustain aversive taste memories. Neuron 100, 891–899 (2018).
Campos, C. A., Bowen, A. J., Schwartz, M. W. & Palmiter, R. D. Parabrachial CGRP neurons control meal termination. Cell Metab. 23, 811–820 (2016).
D’Agostino, G. et al. Appetite controlled by a cholecystokinin nucleus of the solitary tract to hypothalamus neurocircuit. eLife 5, e12225 (2016).
Wu, Q., Boyle, M. P. & Palmiter, R. D. Loss of GABAergic signaling by AgRP neurons to the parabrachial nucleus leads to starvation. Cell 137, 1225–1234 (2009).
Shah, B. P. et al. MC4R-expressing glutamatergic neurons in the paraventricular hypothalamus regulate feeding and are synaptically connected to the parabrachial nucleus. Proc. Natl Acad. Sci. USA 111, 13193–13198 (2014).
Li, A.-J., Wang, Q. & Ritter, S. Selective pharmacogenetic activation of catecholamine subgroups in the ventrolateral medulla elicits key glucoregulatory responses. Endocrinology 159, 341–355 (2018).
Hudson, B. & Ritter, S. Hindbrain catecholamine neurons mediate consummatory responses to glucoprivation. Physiol. Behav. 82, 241–250 (2004).
Cryer, P. E. Glucose counterregulation: prevention and correction of hypoglycemia in humans. Am. J. Physiol. 264, E149–E155 (1993).
Li, A.-J., Wang, Q., Elsarelli, M. M., Brown, R. L. & Ritter, S. Hindbrain catecholamine neurons activate orexin neurons during systemic glucoprivation in male rats. Endocrinology 156, 2807–2820 (2015).
Aklan, I. et al. NTS catecholamine neurons mediate hypoglycemic hunger via medial hypothalamic feeding pathways. Cell Metab. 31, 313–326 (2020).
Hensch, T. K. Critical period plasticity in local cortical circuits. Nat. Rev. Neurosci. 6, 877–888 (2005).
Bouret, S. G. Trophic action of leptin on hypothalamic neurons that regulate feeding. Science 304, 108–110 (2004).
Kamitakahara, A., Bouyer, K., Wang, C.-H. & Simerly, R. A critical period for the trophic actions of leptin on AgRP neurons in the arcuate nucleus of the hypothalamus. J. Comp. Neurol. 526, 133–145 (2018).
Bouret, S. G., Draper, S. J. & Simerly, R. B. Formation of projection pathways from the arcuate nucleus of the hypothalamus to hypothalamic regions implicated in the neural control of feeding behavior in mice. J. Neurosci. 24, 2797–2805 (2004).
Rupp, A. C. et al. Specific subpopulations of hypothalamic leptin receptor-expressing neurons mediate the effects of early developmental leptin receptor deletion on energy balance. Mol. Metab. 14, 130–138 (2018).
Ahima, R. S., Prabakaran, D. & Flier, J. S. Postnatal leptin surge and regulation of circadian rhythm of leptin by feeding. Implications for energy homeostasis and neuroendocrine function. J. Clin. Invest. 101, 1020–1027 (1998).
Biddinger, J. E., Lazarenko, R. M., Scott, M. M. & Simerly, R. Leptin suppresses development of GLP-1 inputs to the paraventricular nucleus of the hypothalamus. eLife 9, e59857 (2020).
Rinaman, L. Postnatal development of catecholamine inputs to the paraventricular nucleus of the hypothalamus in rats. J. Comp. Neurol. 438, 411–422 (2001).
Mirzadeh, Z. et al. Perineuronal net formation during the critical period for neuronal maturation in the hypothalamic arcuate nucleus. Nat. Metab. 1, 212–221 (2019).
Carulli, D. et al. Animals lacking link protein have attenuated perineuronal nets and persistent plasticity. Brain 133, 2331–2347 (2010).
Pizzorusso, T. Reactivation of ocular dominance plasticity in the adult visual cortex. Science 298, 1248–1251 (2002).
Gogolla, N., Caroni, P., Luthi, A. & Herry, C. Perineuronal nets protect fear memories from erasure. Science 325, 1258–1261 (2009).
Tamashiro, K. L. K., Terrillion, C. E., Hyun, J., Koenig, J. I. & Moran, T. H. Prenatal stress or high-fat diet increases susceptibility to diet-induced obesity in rat offspring. Diabetes 58, 1116–1125 (2009).
Glavas, M. M. et al. Early overnutrition results in early-onset arcuate leptin resistance and increased sensitivity to high-fat diet. Endocrinology 151, 1598–1610 (2010).
Acknowledgements
M.G.M. is supported by grants from the National Institutes of Health (NIH) National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK; DK056731, DK104999 and DK020572) and the Marilyn H. Vincent Foundation. A.H.A. is supported by NIH NIDDK (F32 DK122660). N.R. is supported by the NIDDK-funded University of Washington Diabetes, Obesity and Metabolism Fellowship Training Grant (T32 DK007247). M.W.S. is supported by NIDDK grants DK083042 and DK089056.
Author information
Authors and Affiliations
Contributions
M.G.M. and M.W.S. wrote the initial draft of the manuscript. All authors contributed to the writing of the manuscript, drafting of figures and editing of the manuscript and figures.
Corresponding author
Ethics declarations
Competing interests
M.G.M. receives research funding from AstraZeneca, Novo Nordisk and Ionis, and consults for LG Chem. M.W.S. receives research funding from Novo Nordisk. The authors declare no other competing interests.
Additional information
Peer review information Nature Metabolism thanks Young-Hwan Jo, Miguel López and Giles Yeo for their contribution to the peer review of this work. Primary Handling Editor: Christoph Schmitt.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Myers, M.G., Affinati, A.H., Richardson, N. et al. Central nervous system regulation of organismal energy and glucose homeostasis. Nat Metab 3, 737–750 (2021). https://doi.org/10.1038/s42255-021-00408-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s42255-021-00408-5
This article is cited by
-
Microbiota–gut–brain axis and its therapeutic applications in neurodegenerative diseases
Signal Transduction and Targeted Therapy (2024)
-
An RNA-seq atlas of mouse brain areas during fasting and diet-induced obesity
Scientific Data (2024)
-
Advanced neurobiological tools to interrogate metabolism
Nature Reviews Endocrinology (2023)
-
The ventromedial hypothalamic nucleus: watchdog of whole-body glucose homeostasis
Cell & Bioscience (2022)
-
Hindbrain circuits in the control of eating behaviour and energy balance
Nature Metabolism (2022)