Abstract
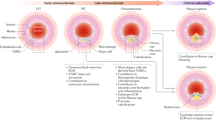
Stable atherosclerotic plaques are characterized by a thick, extracellular matrix-rich fibrous cap populated by protective ACTA2+ myofibroblast (MF)-like cells, assumed to be almost exclusively derived from smooth muscle cells (SMCs). Herein, we show that in murine and human lesions, 20% to 40% of ACTA2+ fibrous cap cells, respectively, are derived from non-SMC sources, including endothelial cells (ECs) or macrophages that have undergone an endothelial-to-mesenchymal transition (EndoMT) or a macrophage-to-mesenchymal transition (MMT). In addition, we show that SMC-specific knockout of the Pdgfrb gene, which encodes platelet-derived growth factor receptor beta (PDGFRβ), in Apoe−/− mice fed a Western diet for 18 weeks resulted in brachiocephalic artery lesions nearly devoid of SMCs but with no changes in lesion size, remodelling or indices of stability, including the percentage of ACTA2+ fibrous cap cells. However, prolonged Western diet feeding of SMC Pdgfrb-knockout mice resulted in reduced indices of stability, indicating that EndoMT- and MMT-derived MFs cannot compensate indefinitely for loss of SMC-derived MFs. Using single-cell and bulk RNA-sequencing analyses of the brachiocephalic artery region and in vitro models, we provide evidence that SMC-to-MF transitions are induced by PDGF and transforming growth factor-β and dependent on aerobic glycolysis, while EndoMT is induced by interleukin-1β and transforming growth factor-β. Together, we provide evidence that the ACTA2+ fibrous cap originates from a tapestry of cell types, which transition to an MF-like state through distinct signalling pathways that are either dependent on or associated with extensive metabolic reprogramming.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon request. Additional data and code are available at GSE163519. Source data are provided with this paper.
References
Virmani, R, Kolodgie, F. D., Burke, A. P., Farb, A & Schwartz, S. M. Lessons from sudden coronary death: a comprehensive morphological classification scheme for atherosclerotic lesions. Arterioscler. Thromb. Vasc. Biol. 20, 1262–1275 (2000).
Davies, M. J., Richardson, P. D., Woolf, N., Katz, D. R. & Mann, J. Risk of thrombosis in human atherosclerotic plaques: role of extracellular lipid, macrophage and smooth muscle cell content. Heart 69, 377–381 (1993).
Libby, P., Ridker, P. M. & Hansson, G. K. Progress and challenges in translating the biology of atherosclerosis. Nature 473, 317–325 (2011).
Feil, S. et al. Transdifferentiation of vascular smooth muscle cells to macrophage-like cells during atherogenesis. Circ. Res. 115, 662–667 (2014).
Shankman, L. S. et al. KLF4-dependent phenotypic modulation of smooth muscle cells has a key role in atherosclerotic plaque pathogenesis. Nat. Med. 21, 628–637 (2015).
Chen, P. -Y. et al. Endothelial-to-mesenchymal transition drives atherosclerosis progression. J. Clin. Invest. 125, 4514–4528 (2015).
Evrard, S. M. et al. Endothelial-to-mesenchymal transition is common in atherosclerotic lesions and is associated with plaque instability. Nat. Commun. 7, 11853 (2016).
Caplice, N. M. et al. Smooth muscle cells in human coronary atherosclerosis can originate from cells administered at marrow transplantation. Proc. Natl Acad. Sci. USA 100, 4754–4759 (2003).
Albarrán-Juárez, J., Kaur, H., Grimm, M., Offermanns, S. & Wettschureck, N. Lineage tracing of cells involved in atherosclerosis. Atherosclerosis 251, 445–453 (2016).
Gomez, D. et al. Interleukin-1β has atheroprotective effects in advanced atherosclerotic lesions of mice. Nat. Med. 24, 1418–1429 (2018).
Durgin, B. G. et al. Smooth muscle cell-specific deletion of Col15a1 unexpectedly leads to impaired development of advanced atherosclerotic lesions. Am. J. Physiol. Heart Circ. Physiol. 312, H943–H958 (2017).
Cherepanova, O. A. et al. Activation of the pluripotency factor OCT4 in smooth muscle cells is atheroprotective. Nat. Med. 22, 657–665 (2016).
Newman, A. A. C. et al. Irradiation abolishes smooth muscle investment into vascular lesions in specific vascular beds. JCI Insight 3, e121017 (2018).
Betsholtz, C. Insight into the physiological functions of PDGF through genetic studies in mice. Cytokine Growth Factor Rev. 15, 215–228 (2004).
Hideto, S et al. Functional blockade of platelet-derived growth factor receptor-β but not of receptor-α prevents vascular smooth muscle cell accumulation in fibrous cap lesions in apolipoprotein E–deficient mice. Circulation 103, 2955–2960 (2001).
Kozaki, K. et al. Blockade of platelet-derived growth factor or its receptors transiently delays but does not prevent fibrous cap formation in ApoE null mice. Am. J. Pathol. 161, 1395–1407 (2002).
Tomas, L. et al. Altered metabolism distinguishes high-risk from stable carotid atherosclerotic plaques. Eur. Heart J. 39, 2301–2310 (2018).
Alencar, G. F. et al. The stem cell pluripotency genes Klf4 and Oct4 regulate complex SMC phenotypic changes critical in late-stage atherosclerotic lesion pathogenesis. Circulation 142, 2045–2059 (2020)
Wirka, R. C. et al. Atheroprotective roles of smooth muscle cell phenotypic modulation and the TCF21 disease gene as revealed by single-cell analysis. Nat. Med. https://doi.org/10.1038/s41591-019-0512-5 (2019).
Winkels, H. et al. Atlas of the immune cell repertoire in mouse atherosclerosis defined by single-cell RNA sequencing and mass cytometry. Circ. Res. 122, 1675–1688 (2018).
Kadl, A. et al. Identification of a novel macrophage phenotype that develops in response to atherogenic phospholipids via Nrf2. Circ. Res 107, 737–746 (2010).
Serbulea, V. et al. Macrophage phenotype and bioenergetics are controlled by oxidized phospholipids identified in lean and obese adipose tissue. Proc. Natl Acad. Sci. USA 115, E6254–E6263 (2018).
Gomez, D. & Owens, G. K. Smooth muscle cell phenotypic switching in atherosclerosis. Cardiovasc. Res. 95, 156–164 (2012).
Allahverdian, S., Pannu, P. S. & Francis, G. A. Contribution of monocyte-derived macrophages and smooth muscle cells to arterial foam cell formation. Cardiovasc. Res. 95, 165–172 (2012).
Gomez, D., Shankman, L. S., Nguyen, A. T. & Owens, G. K. Detection of histone modifications at specific gene loci in single cells in histological sections. Nat. Methods 10, 171–177 (2013).
McDonald, O. G., Wamhoff, B. R., Hoofnagle, M. H. & Owens, G. K. Control of SRF binding to CArG box chromatin regulates smooth muscle gene expression in vivo. J. Clin. Invest. 116, 36–48 (2006).
McDonald, O. G. & Owens, G. K. Programming smooth muscle plasticity with chromatin dynamics. Circ. Res. 100, 1428–1441 (2007).
Carroll, M. et al. CGP 57148, a tyrosine kinase inhibitor, inhibits the growth of cells expressing BCR–ABL, TEL–ABL and TEL–PDGFR fusion proteins. Blood 90, 4947–4952 (1997).
Kerkelä, R. et al. Cardiotoxicity of the cancer therapeutic agent imatinib mesylate. Nat. Med. 12, 908–916 (2006).
Sarrazy, V. et al. Integrins αvβ5 and αvβ3 promote latent TGF-β1 activation by human cardiac fibroblast contraction. Cardiovasc. Res. 102, 407–417 (2014).
Fiume, L. et al. Galloflavin prevents the binding of lactate dehydrogenase A to single-stranded DNA and inhibits RNA synthesis in cultured cells. Biochem. Biophys. Res. Commun. 430, 466–469 (2013).
Manerba, M. et al. Galloflavin (CAS 568-80-9): a novel inhibitor of lactate dehydrogenase. ChemMedChem 7, 311–317 (2012).
Gibson, F. S., Gupta, D., Shorr, R. & Rodriguez, R. An efficient, economical synthesis of the novel anti-tumor agent CPI-613. Org. Process Res. Dev. 15, 855–857 (2011).
Egawa, Y., Saigo, C., Kito, Y., Moriki, T. & Takeuchi, T. Therapeutic potential of CPI-613 for targeting tumorous mitochondrial energy metabolism and inhibiting autophagy in clear cell sarcoma. PLoS ONE 13, e0198940 (2018).
Alistar, A. et al. Safety and tolerability of the first-in-class agent CPI-613 in combination with modified FOLFIRINOX in patients with metastatic pancreatic cancer: a single-centre, open-label, dose-escalation, phase 1 trial. Lancet Oncol. 18, 770–778 (2017).
Steitz, S. A. et al. Osteopontin inhibits mineral deposition and promotes regression of ectopic calcification. Am. J. Pathol. 161, 2035–2046 (2002).
Speer, M. Y. et al. Inactivation of the osteopontin gene enhances vascular calcification of matrix gla protein–deficient mice: evidence for osteopontin as an inducible inhibitor of vascular calcification in vivo. J. Exp. Med. 196, 1047–1055 (2002).
Bevilacqua, M. P., Pober, J. S., Wheeler, M. E., Cotran, R. S. & Gimbrone, M. A. Interleukin-1 activation of vascular endothelium. Effects on procoagulant activity and leukocyte adhesion. Am. J. Pathol. 121, 394–403 (1985).
Maleszewska, M. et al. IL-1β and TGFβ2 synergistically induce endothelial-to-mesenchymal transition in an NFκB-dependent manner. Immunobiology 218, 443–454 (2013).
Bennett, M. R., Sinha, S. & Owens, G. K. Vascular smooth muscle cells in atherosclerosis. Circ. Res. 118, 692–702 (2016).
Levéen, P. et al. Mice deficient for PDGFβ show renal, cardiovascular and hematological abnormalities. Genes Dev. 8, 1875–1887 (1994).
Lindahl, P., Johansson, B. R., Levéen, P. & Betsholtz, C. Pericyte loss and microaneurysm formation in PDGFB-deficient mice. Science 277, 242–245 (1997).
Wang, S. et al. TGF-β/Smad3 signalling regulates the transition of bone marrow-derived macrophages into myofibroblasts during tissue fibrosis. Oncotarget 7, 8809–8822 (2015).
Chen, P.-Y. et al. Endothelial TGF-β signalling drives vascular inflammation and atherosclerosis. Nat. Metab. https://doi.org/10.1038/s42255-019-0102-3 (2019).
Baylis, R. A., Gomez, D., Mallat, Z., Pasterkamp, G. & Owens, G. K. The CANTOS trial: one important step for clinical cardiology but a giant leap for vascular biology. Arterioscler. Thromb. Vasc. Biol. 37, e174–e177 (2017).
He, C. et al. PDGFRβ signalling regulates local inflammation and synergizes with hypercholesterolaemia to promote atherosclerosis. Nat. Commun. 6, 7770 (2015).
Giles, F. J. et al. Nilotinib in imatinib-resistant or imatinib-intolerant patients with chronic myeloid leukemia in chronic phase: 48-month follow-up results of a phase II study. Leukemia 27, 107–112 (2013).
Hochhaus, A. et al. Long-term benefits and risks of frontline nilotinib versus imatinib for chronic myeloid leukemia in chronic phase: 5-year update of the randomized ENESTnd trial. Leukemia 30, 1044–1054 (2016).
Kim, T. D. et al. Peripheral artery occlusive disease in chronic phase chronic myeloid leukemia patients treated with nilotinib or imatinib. Leukemia 27, 1316–1321 (2013).
Larson, R. A. et al. Efficacy and safety of nilotinib versus imatinib in patients with newly diagnosed chronic myeloid leukemia in chronic phase (CML-CP): long-term follow-up of ENESTnd. Blood 124, 4541–4541 (2014).
Mayr, M et al. Proteomic and metabolomic analyses of atherosclerotic vessels from apolipoprotein E-deficient mice reveal alterations in inflammation, oxidative stress and energy metabolism. Arterioscler. Thromb. Vasc. Biol. 25, 2135–2142 (2005).
Bories, G. F. P. & Leitinger, N. Macrophage metabolism in atherosclerosis. FEBS Lett. 591, 3042–3060 (2017).
Theodorou, K. & Boon, R. A. Endothelial cell metabolism in atherosclerosis. Front. Cell Dev. Biol. 6, 82 (2018).
Bjørklund, M. M. et al. Induction of atherosclerosis in mice and hamsters without germline genetic engineering. Circ. Res. 114, 1684–1689 (2014).
Iwata, H. et al. Bone marrow-derived cells contribute to vascular inflammation but do not differentiate into smooth muscle cell lineages. Circulation 122, 2048–2057 (2010).
Pouwer, M. G. et al. The BCR-ABL1 inhibitors imatinib and ponatinib decrease plasma cholesterol and atherosclerosis, and nilotinib and ponatinib activate coagulation in a translational mouse model. Front. Cardiovasc. Med. 5, 55 (2018).
Rossi, F. et al. Imatinib upregulates compensatory integrin signaling in a mouse model of gastrointestinal stromal tumor and is more effective when combined with dasatinib. Mol. Cancer Res. 8, 1271–1283 (2010).
Schindelin, J. et al. Fiji: an open-source platform for biological-image analysis. Nat. Methods 9, 676–682 (2012).
Acknowledgements
The authors acknowledge R. Tripathi for help isolating and maintaining the lineage-tagged murine SMC line; W. B. Evans for help in image preprocessing for the automated counting pipeline; R. Adams and R. Klein (MPI) for providing the PdgfrbFL/FL mouse and R. Adams for providing the Cdh5-CreERT2 mouse (MPI); E. Pinteaux (University of Manchester) for generating and sharing the Il1r1FL/FL mouse; and S. Francis (University of Sheffield) for advice on the in vivo murine EC experiments; V. N. Albert for aid using Adobe Illustrator. Parts of this work were performed with the assistance of University of Virginia Core Facilities, including the Flow Cytometry Facility, Advanced Microscopy Facility, Genome Technology and Analysis Core and Bioinformatics Core. This work was supported by NIH R01 grants HL132904, HL136314 and HL141425, as well as a Leducq Foundation Transatlantic Network Grant to G.K.O. A.A.C.N. and V.S. were supported by Basic and Translational Cardiovascular Training Grants 5 T32 HL 007284-41 and 5 T32 HL007284-43, respectively. R.A.B. was supported by NIH Fellowship F30 HL 136188-04 and the Basic and Translational Cardiovascular Training Grant 5 T32 HL 007284-40. M.S.R. was supported by a Small Research and Travel Grant, an Ingrassia Family Echols Scholars Research Grant and a Harrison Undergraduate Research Award.
Author information
Authors and Affiliations
Contributions
A.A.C.N., V.S. and R.A.B. conceptualized and performed the bulk of the experiments, validation, data collection, analysis and interpretation, and contributed significantly to development of methodology, manuscript writing and editing. X.G.B. contributed to data collection, validation, analysis and editing of the manuscript. L.S.S. aided in data collection and analysis, and developed unbiased pipelines to measure immunofluorescence staining and quantify subluminal cell location in human and murine samples. G.F.A. and K.O. contributed to collection, processing and analysis of scRNA-seq studies. R.A.D., S.K. and S.S. contributed to data collection and analysis. M.S.R. and A.S. contributed to conceptualization, validation, data collection, analysis and interpretation of in vitro cell analyses. L.G. characterized human lesions. R.V. and A.F. contributed human coronary artery lesion specimens, funding and methodology for analysis of human coronary artery lesions. O.A.C. provided methodological help and supervision throughout the duration of this project. G.K.O. supervised the entire project and had a major role in conceptualization, experimental design, data interpretation, funding and manuscript writing and editing.
Corresponding author
Ethics declarations
Competing interests
A.F. declares one competing interest with Amgen. All other authors declare no competing interests.
Additional information
Peer review information Primary Handling Editor: George Caputa.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Generation and validation of SMC-specific conditional PDGFRB deletion in SMC lineage tracing mice.
a, Myh11-CreERT2/ROSA-STOPFL/FL/eYFP; Apoe−/− (Myh11-CreERT2) mice were crossed with a PdgfrbFL/FL mouse to generate PdgfrbSMC−FL/FL and -WT/WT animals. Heterozygote animals were bred (PdgfrbFL/WT; Myh11-CreERT2/ROSA STOPFL/FL/eYFP; Apoe-/−) to generate littermate FL/FL and WT/WT controls, which were used in all experiments. b, Analyses were conducted on the BCA, at multiple locations past the aortic arch in order to assess patterns across the entire region, or within the aortic root. c, Quantification of IF staining at 18 weeks of WD in the media of BCA lesions showed efficient KO of PDGFRB in >94% of SMC. ***p-value = 0.0006. d, PDGFRB KO in SMC is not associated with decreases in the total number of DAPI+ or Myh11-eYFP+ cells (SMC) in the media, but there was a significant reduction in the total number of PDGFRB+ cells and Myh11-eYFP+ PDGFRB+ cells. ***p-values = 0.0006. e, Validation of genetic deletion of Pdgfrb gene was confirmed for each experimental mouse. f, Representative images showing PDGFRB loss within the SMC-rich media in PdgfrbSMC−Δ/Δ mice. Scale bar: 20 μm. Graphs were analyzed using two-tailed Mann-Whitney U test, biologically independent animals are indicated as individual dots in (c,d), error bars represent mean ± SEM.
Extended Data Fig. 2 SMC investment is significantly impaired in the aortic root of PdgfrbSMC-Δ/Δ mice and SMC-PDGFRB KO did not show reduced dedifferentiation, proliferation, or apoptosis of medial SMC following 18 weeks of WD.
a, Representative images of aortic root lesions from PdgfrbSMC-WT/WT and (b) PdgfrbSMC-Δ/Δ mice. c, Quantification of Myh11-eYFP+ cells content in the fibrous caps of aortic root lesions from PdgfrbSMC-WT/WT and PdgfrbSMC−Δ/Δ mice, demonstrated a significant reduction in SMC-derived cells in aortic root lesions. ***p-value = 0.0002. d, Representative image of the media from the BCA of PdgfrbSMC-WT/WT and PdgfrbSMC−Δ/Δ mice show ACTA2 downregulation in the innermost layer of the media. e, Quantification of Myh11-eYFP+ACTA2+ cells in the media. f, Representative image of Ki67 staining in Myh11-eYFP+ SMC in the media. g, Quantification of Ki67 expression in the media, representing proliferation. Ki67+: **p-value = 0.0017, Myh11-eYFP+ Ki67+ *p-value = 0.0130, Myh11-eYFP- Ki67+ *p-value = 0.0337. h, Representative image of cleaved caspase 3 staining in Myh11-eYFP+ SMC in the media. Myh11-eYFP− Casp3+ p-value = 0.0476. i, Quantification of Casp3 expression in the media, representing apoptosis. *p-value = 0.0476. A: Adventitia, M: Media, L: Lesion. Scale bar: 100 μm (whole lesion) or 20 μm (zoom). Graphs were analyzed using Graphs were analyzed using two-way ANOVA with Sidak correction and multiple comparisons or two-tailed Mann-Whitney U test, biologically independent animals are indicated as individual dots in (c,h,i), n = 8 (WT and KO) in (g), error bars represent mean ± SEM.
Extended Data Fig. 3 PdgfrbSMC−Δ/Δ did not result in differences in BCA lesion size or remodeling indices after 18 weeks of WD and ACTA2+ cells co-localize with LGALS3 or CD31 in lesions lacking SMC investment.
a, MOVAT representation of PdgfrbSMC-WT/WT and PdgfrbSMC-Δ/Δ mice after 18 weeks of WD. b, Lesion, (c) External elastic lamina (EEL), d, or lumen area were not significantly changed at three locations. e, Necrotic core area was also unchanged. ACTA2 staining co-localizes with LGALS3 (f) or CD31 (g) in the fibrous cap of PdgfrbSMC-Δ/Δ mice. In PdgfrbSMC-WT/WT mice, ACTA2+ eYFP+ cells co-stain with LGALS3 (white) or CD31 (yellow). Scale bar: 100 μm (a) and 20 μm (f, g). X-axis values represent distance past the aortic arch. Graphs were analyzed using two-way ANOVA with Sidak correction and multiple comparisons, biologically independent animals are indicated as individual dots in (b,c,d,e), error bars represent mean ± SEM.
Extended Data Fig. 4 UMAP presentation of scRNA-seq data showing gene expression of the eYFP transgene, Pdgfrb, and Col15a1 in PdgfrbSMC-WT/WT and PdgfrbSMC-Δ/Δ mice.
a, UMAP representations of each scRNA-seq library from cells isolated from BCAs from PdgfrbSMC-WT/WT and PdgfrbSMC-Δ/Δ mice fed 18 weeks of WD. b, Percentage of cells in each cluster from each respective group described in A, unsorted media and underlying adventitia cells (left), unsorted lesion cells (middle), and eYFP+ sorted cells from media and underlying adventitia cells (right). c–h, UMAP representations of expression levels of target genes from scRNA-seq analysis of cells isolated from BCAs of mice fed 18 weeks of WD (top). Violin plots showing expression of each target gene by cluster (bottom). c, Demonstrates presence of eYFP transgene, identifying the SMC-derived cell populations. d, Demonstrates loss of Pdgfrb gene expression in PdgfrbSMC-Δ/Δ mice only in SMC-derived cells. e, Col15a1 gene expression is highly enriched and specific for cluster 6, which is reduced in the PdgfrbSMC-Δ/Δ mice. f–h, Osteochondrogenic genes represented in SMC clusters.
Extended Data Fig. 5 Analysis of BCA lesions from gain of function mutant (m) PCSK9-AAV8 and Ldlr−/− models of murine atherosclerosis.
a, Schematic of experimental design for experiments using Myh11-CreERT2/ROSA-STOPFL/FL-eYFP mice infected once with mPCSK9-AAV8 (Myh11-CreERT2 mPCSK9). Representative images depicting (b) EndoMT (eYFP− ACTA2+ CD31+ /ACTA2+) and (c) MMT (eYFP− ACTA2+ LGALS3+ /ACTA2+). d, Quantification of Myh11-eYFP+ cells in the 30 μm fibrous cap area. e, Quantification of the percentage of ACTA2+ cells derived from SMC (Myh11-eYFP+ ACTA2+) and from non-SMC sources (Myh11-eYFP−ACTA2+) in Myh11-CreERT2 mPCSK9 mice. f, Quantification of MMT and EndoMT from (b & c). g, Schematic of Ldlr−/− Myh11-CreERT2/ROSA-STOPFL/FL-eYFP (Myh11-CreERT2 Ldlr−/−) experimental design. Representative images depicting (h) EndoMT (Myh11-eYFP− ACTA2+ CD31+ /ACTA2+) and (i) MMT (Myh11-eYFP− ACTA2+ LGALS3+ /ACTA2+). j, Quantification of Myh11-eYFP+ cells in the 30µm fibrous cap area. k, Quantification of the percentage of ACTA2+ cells derived from SMC (Myh11-eYFP+ ACTA2+) and from non-SMC sources (Myh11-eYFP− ACTA2+) in Myh11-CreERT2 Ldlr−/− mice. l, Quantification of MMT and EndoMT from (h) & (i). Scale bar: 100 μm (b,c,h,i top) or 20 μm (b,c,h,i bottom). Biologically independent animals are indicated as individual dots in (d,f,j,l), (E) n = 7, 6 (WT and KO: 330, 480 μm), (k) n=5 (WT and KO), error bars represent mean ± SEM.
Extended Data Fig. 6 Generation and validation of EC-lineage tracing mice and EC-specific deletion of Il1r1.
a, Cdh5-CreERT2/ROSA-STOPFL/FL-eYFP/Apoe−/− (Cdh5-CreERT2) mice were generated to lineage trace EC and their progeny within the BCA. Mice were then crossed to an Il1r1-Flox mouse to generate Il1r1EC−fl/fl and −WT/WT animals. Heterozygote animals were bred (Il1r1Fl/WT; Cdh5-CreERT2/ROSA-STOPFL/FL-eYFP/Apoe−/−) to generate littermate FL/FL and WT/WT controls, which were used in all experiments. b, Image denoting the endothelial monolayer with Cdh5-eYFP+ staining only in this layer. c, Validation of genetic deletion of Il1r1 in organs using isolated DNA. Lanes A, C, E contain the FL and WT genotyping reactions while lanes B, D, F contain the excision reaction. Representative images of Cdh5-eYFP+ staining in (d) Liver, (e) Lung, and (f) Aorta. g, Gating strategy for isolation of Cdh5-eYFP+ endothelial cells. h, Genotyping and recombination analysis of Cdh5-eYFP+ cells sorted from Il1r1EC-WT/WT (lanes A, B) and Il1r1EC-FL/FL (lanes C, D) mice. Lanes A, C contain FL and WT genotyping reaction while B, D contain the excision reaction. Scale bar: 100 μm (d,e,f).
Extended Data Fig. 7 Lethal radiation and BMT significantly increases the percentage of EC in the lesion and fibrous cap.
a, Representative images of BCA lesions in Cdh5-CreERT2 mice fed 18 weeks of WD with or without 1200cGy of radiation, which is known to ablate SMC accumulation within the lesion and fibrous cap. b, Quantification of Cdh5-eYFP+ cells in the fibrous cap showed significantly increased EC-derived cells after radiation. ***p-values < 0.0001. c, The increased percentage of EC-derived cells within the fibrous cap was not associated with changes in overall ACTA2+ cells within the fibrous cap. d, Zoom in of highlighted area in (a) showing EC-derived cells express ACTA2. Single cell breakdown and quantification of Cdh5-eYFP+ ACTA2+ cells per total Cdh5-eYFP+ (e) or per total ACTA2+ cells, **p-value = 0.0023, (f) within the fibrous cap, ***p-values < 0.0001. g, Representative image of bone marrow cells (BMC) expressing ACTA2 within the fibrous cap of Cdh5-CreERT2 mice after radiation. h, Quantification and comparison of MMT by co-incident staining of ACTA2 and marker protein staining for LGALS3 or for the BMC lineage tag, tdTomato. Scale bar: 100 μm (whole lesion) or 50 μm (zoom). Graphs were analyzed using two-tailed Mann-Whitney U test, error bars represent mean ± SEM.
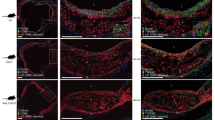
Extended Data Fig. 8 A subset of ACTA2+ cells in human coronary lesions are derived from a non-SMC source.
Representative images of stable human lesions of (a) ISH-PLA staining or (b) CD31 staining, showing the fibrous cap outlined using serially stained H&E and MOVAT slides. c, Analysis following ISH-PLA, where PLA dots correspond to cells with H3K4me2 on the MYH11 promoter. d, Graph representing the portion of ACTA2+ cells that are SMC (ACTA2+ PLA+) or non-SMC derived in human lesions (ACTA2+ PLA−). e, Analysis of the percent of ACTA2+ cells that are CD31+ within the fibrous cap of human coronary lesions. f, Graph representing a portion of ACTA2+ cells that co-stain with CD31 within the fibrous cap. Data were obtained by assessing co-localization of DAPI, ACTA2, and PLA or DAPI, ACTA2, and CD31 (see Methods). g, Total calculated efficiency of ISH-PLA positivity (ACTA2+ ISHPLA+) calculated based on total # of ACTA2+ cells within media, with the assumption that 100% of ACTA2+ cells in the media will be PLA+. Each Sample ID is a distinct patient and this total efficiency includes error introduced by the semi-automated quantification pipeline, calculated separately in (h–j). Comparison of single-cell counting by manual human input and the semi-automated pipeline of DAPI (h), PLA+ (i), and PLA+ ACTA2+ /ACTA2+ (j) using 6 distinct subsections of human coronary artery vessels, that were chosen to keep the manual counter blinded. Statistical analysis of Manual versus semi-auto detection comparison was performed with paired two-tailed t-tests. Individual patients are indicated as individual dots in (d,f-j). Scale bar: 1000 μm (a,b) or 100 μm (c,e). Graphs shown as mean ± SEM.
Extended Data Fig. 9 Bioenergetic and transcriptional changes of SMC in response to PDGF and inhibitors of pyruvate metabolism.
Murine SMC were serum starved for 24–72 hours and then treated with Vehicle, PDGF-BB or -DD (50ng/mL), and/or Galloflavin (10 μM or 50 μM) or CPI613 (20 μM). a, Glycolytic stress test (GST) measuring extracellular acidification rate (ECAR) of treated SMC, representing glycolytic ability. b, ECAR was measured initially in the absence of glucose, after injection of 20mM D-glucose (basal glycolysis), 1 μM oligomycin (glycolytic capacity), and 80mM 2-deoxy-D-glucose (non-glycolytic; used for normalization). Glycolytic reserve was determined by subtracting post-glucose ECAR from post-oligomycin ECAR. [Aerobic glycolysis p = 0.0206; Glycolytic capacity p = 0.0441 and p = 0.0234]. c, Mitochondrial stress test (MST) measuring the oxygen consumption rate (OCR) of cells, representing mitochondrial respiratory ability. d, OCR was measured initially (basal respiration), and after injection of 1 μM oligomycin, 2 μM BAM15 (respiratory capacity), and 10 μM antimycin A & 1 μM rotenone (non-mitochondrial; used for normalization). Spare respiratory capacity was determined by subtracting the initial OCR from the post-BAM15 OCR. [Respiratory capacity p = 0.0013 and p = 0.0248; Spare respiratory capacity p = 0.0007 and p = 0.0165]. e, Energy capacity map representing the bioenergetic potential of SMC (x-axis, maximal ECAR, units are mpH/min; y-axis, maximal OCR, units are pmol O2/min). f, mRNA expression of Col15a1, measured by qPCR in SMC treated for 24 hours. [Vehicle p < 0.0001, PDGF-BB p < 0.0001, PDGF-DD p = 0.0004, and Galloflavin 10 μM p = 0.0007]. g, mRNA expression of Col15a1, Mmp3, and Acta2, measured by qPCR in SMC treated with Vehicle, PDGF-BB, and/or Galloflavin 10 μM for 24 hours [Col15a1 p <0.0001, p = 0.003, and p = 0.0457; Mmp3 p = 0.0354 and p = 0.0437; Acta2 p = 0.0685 and p = 0.0192]. h, Energy capacity map representing the bioenergetic potential of SMC treated with Vehicle, PDGF-BB, and/or CPI613 for 24 hours. i, mRNA expression of Col15a1, and Mmp3, measured by qPCR. [Col15a1 p = 0.0226 and p = 0.0007; Mmp3 p = 0.0002 and p < 0.0001]. Graphs were analyzed using one-way ANOVA with Tukey’s correction for post-hoc analysis, error bars represent mean ± SEM.
Extended Data Fig. 10 Cultured SMC stimulated to myofibroblast-like state with PDGF and TGFβ, require aerobic glycolysis to maintain myofibroblast-associated gene expression.
a, Murine aortic SMC were serum starved for 24–72 hours. After serum starvation, SMCs were treated with vehicle control or PDGF-DD (10ng/mL) and TGFβ1 (10ng/mL), Galloflavin (10µM), and/or CPI613 (20µM) for 24 hours. Next, cells were treated a second time, with PDGF-DD (10ng/mL) and TGFβ1 (10ng/mL), this time in combination with Galloflavin (10µM) or CPI613 (20µM) for another 24 hours. b, Pyruvate metabolism pathway map, highlighting lactate dehydrogenase (LDH) as the target of Galloflavin and pyruvate dehydrogenase (PDH) as the target of CPI613. c, Mitochondrial stress test (MST) measuring the oxygen consumption rate (OCR). OCR was measured initially (basal respiration), and after injection of 1 μM oligomycin, 2 μM BAM15 (respiratory capacity), and 10 μM antimycin A & 1 μM rotenone (non-mitochondrial; used for normalization). Spare respiratory capacity was determined by subtracting the initial OCR from the post-BAM15 OCR. [Basal respiration p = 0.0003, p = 0.009, and p = 0.0026; Respiratory Capacity p < 0.0001, p < 0.0001, and p = 0.0008] (d) Glycolytic stress test (GST) measuring extracellular acidification rate (ECAR). ECAR was measured initially in the absence of glucose, after injection of 20mM D-glucose (basal glycolysis), 1μM oligomycin (glycolytic capacity), and 80mM 2-deoxy-D-glucose (non-glycolytic; used for normalization). [Glycolysis p < 0.0001, p = 0.0002, and p < 0.0001; Glycolytic Capacity p = 0.0006, p = 0.0052, and p = 0.0006]. e, Energy capacity map representing the bioenergetic potential of SMC (x-axis, maximal ECAR, units are mpH/min; y-axis, maximal OCR, units are pmol O2/min). f, mRNA expression of ECM and ECM-interacting genes as measured by qPCR. [Col1a1 p = 0.0009, p = 0.0310, p = 0.0647; Col15a1 p = 0.0049 and p = 0.0003; Acta2 p = 0.0009 and p = 0.0003; Spp1 p = 0.0004, p = 0.0008, and p = 0.0006]. Graphs were analyzed using one-way ANOVA with Tukey’s correction for post-hoc analysis with n ≥ 3, error bars represent mean ± SEM.
Supplementary information
Supplementary Information
Supplementary Figs. 1–12.
Source data
Source Data Extended Data Fig. 1
Unprocessed gels.
Source Data Extended Data Fig. 6
Unprocessed gels.
Rights and permissions
About this article
Cite this article
Newman, A.A.C., Serbulea, V., Baylis, R.A. et al. Multiple cell types contribute to the atherosclerotic lesion fibrous cap by PDGFRβ and bioenergetic mechanisms. Nat Metab 3, 166–181 (2021). https://doi.org/10.1038/s42255-020-00338-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s42255-020-00338-8
This article is cited by
-
miR-449a disturbs atherosclerotic plaque stability in streptozotocin and high-fat diet-induced diabetic mice by targeting CEACAM1
Diabetology & Metabolic Syndrome (2024)
-
Dysregulated cellular metabolism in atherosclerosis: mediators and therapeutic opportunities
Nature Metabolism (2024)
-
Cholesterol lowering depletes atherosclerotic lesions of smooth muscle cell-derived fibromyocytes and chondromyocytes
Nature Cardiovascular Research (2024)
-
Cardiovascular Mechano-Epigenetics: Force-Dependent Regulation of Histone Modifications and Gene Regulation
Cardiovascular Drugs and Therapy (2024)
-
Targeting deubiquitinase OTUB1 protects vascular smooth muscle cells in atherosclerosis by modulating PDGFRβ
Frontiers of Medicine (2024)