Abstract
Developing novel lanthanide metal-organic frameworks (Ln-MOFs) to rapidly and reliably differentiate both metal ions in solution and volatile organic compounds (VOCs) in vapor is highly challenging. Here, we describe versatile Eu3+/Tb3+-MOFs based on a flexible ligand. It is noteworthy that the film fabricated using bimetallic Eu0.47Tb0.63-MOF and polyvinyl alcohol could serve as an easy and convenient luminescent platform for distinguishing different metal ions and VOCs. The luminescent film exhibits notable fingerprint correlation between the metal ions/VOCs and the emission intensity ratio of Eu3+/Tb3+ ions in Ln-MOFs. As a result, the bimetallic Ln-MOFs show fast recognition of Fe3+ ion with a response time of <10 s, and can effectively probe styrene vapor within 4 min. Since the developed Ln-MOF film is stable and reliable, this work presents a promising strategy to explore luminescent platforms capable of effectively sensing different metal ions and VOCs.
Similar content being viewed by others
Introduction
The detection of different metal ions has received great attention due to the increasing content of metal ions in soil and wastewater, which is relevant for environmental and industrial monitoring, human health, food safety, etc1,2,3,4. Unlike organic contaminants, metal ions cannot be easily biodegraded. More seriously, many heavy metal ions are known to be toxic or carcinogenic5,6,7,8. Therefore, it is important to develop efficient and economical approaches to sense and distinguish different metal ions. It is well known that iron is ubiquitous in our living environment, and Fe3+ cation has extensive applications including playing an important role in biological systems. However, concentrations of Fe3+ ions over or below the normal level can seriously affect human health9,10,11,12. In the past few years, various kinds of analytical methods have been explored for the detection of different metal ions. As compared with other detection approaches, fluorescent sensors possess many advantages, such as high sensitivity, good reliability, and real-time detection13,14,15,16,17. While some fluorescent metal-organic frameworks (MOFs) showed a promising potential in probing metal ions18,19,20,21,22, MOF-based fluorescent platforms for effective and easy detection of different metal ions in aqueous solution have not been well investigated so far.
On the other hand, volatile organic compounds (VOCs) are toxic and harmful substances that are widely present in indoor and outdoor air. These are mainly derived from the emissions of chemical industries, automobile exhausts, building blocks, etc23, 24. Therefore, an effective probe of VOCs is another important task for both environment protection and human health. While some porous materials exhibit promising results in sensing VOCs in solution, fluorescence sensing of trace VOCs in vapor is still challenging on account of their low vapor pressure at room temperature25, 26. Styrene is a common VOC that is widely used as a precursor for the synthesis of rubber, plastic products, resins, and polyester in synthetic industries27,28,29. As potentially carcinogenic species, styrene vapor can cause harm, such as the irritation of skin, eyes, and respiratory system30,31,32. Among all styrene vapor detection methods reported to date, gas chromatography, surface acoustic wave, and mass spectrometry are most commonly used33, 34. However, these techniques are expensive, complicated, bulky, and unportable. Thus, a portable and easy to operate technology is needed. Fluorescence sensing has attracted much attention due to its high sensitivity, low cost, portability, etc. However, fluorescent sensing of styrene with high sensitivity and easy operation has not been well explored so far.
As a promising class of porous materials, lanthanide MOFs (Ln-MOFs) have attracted wide interest for their luminescent properties derived from essential features of lanthanide elements35,36,37,38,39,40. The introduction of suitable organic chromophoric ligands into Ln-MOFs may enhance their light absorption ability to increase the fluorescent emission of Ln3+ ions by the so-called “antennae effect”41. Therefore, sensing metal ions/VOCs can be realized by tuning the energy transfer efficiency from “antennae” to Ln3+ ions through host–guest interactions. A few luminescent Ln-MOFs with optical properties that are responsive to the guest molecules have been developed42,43,44. However, their luminescent signals only respond to specific molecules. Increasing the capability of distinguishing multiple metal ions or organic molecules by luminescent bimetallic Ln-MOFs has not been well investigated.
In this work, we report a series of versatile bimetallic Ln-MOFs, i.e., [Ln·(CTP-COOH)(H2O)3] (Ln-CTP-COOH, Ln = Eu3+ and Tb3+), constructed from a flexible ligand hexa-(4-carboxyl-phenoxy)-cyclotriphosphazene (CTP-COOH). These differ from prior literature studies26, 45, since the characteristic emission intensity of Eu3+ (616 nm) and Tb3+ (547 nm) ions in bimetallic Eu0.47Tb0.63-CTP-COOH highly depends on various guests (metal ions and VOCs). Polyvinyl alcohol (PVA) films containing bimetallic Eu0.47Tb0.63-CTP-COOH are fabricated to serve as a convenient platform for selective recognition of different metal ions in aqueous solution and VOCs in vapor. It is notable that the host–guest interactions not only tune the energy transfer efficiency from organic ligand “antenna” to Ln3+ ions, but also are able to modulate the energy assignation between Tb3+ and Eu3+ (Tb3+ → Eu3+) in the Ln-MOFs. The luminescent properties of the Ln-MOFs highly depend on the guest metal ions and VOCs. Therefore, excellent Ln-MOF materials can effectively sense different metal ions and VOCs through two-dimensional (2D) readouts by combining different ratiometric emission intensity of Eu3+ and Tb3+ ions. As compared with conventional single Ln3+ emission intensity-based methods for chemical sensing, the present dual-readout orthogonal identification scheme is expected to be more reliable, accurate, and powerful. More importantly, the Ln-MOFs could achieve recognition of Fe3+ ion in aqueous solution with a response time of <10 s and 60% fluorescence quenching, and recognize styrene in vapor within 4 min and 97% quenching efficiency.
Results
Synthesis and structure description
The organic ligand CTP-COOH was prepared in a high yield (94%) using phosphonitrilic chloride trimer and methyl 4-hydroxybenzoate as starting materials (Supplementary Fig. 1), and characterized (Supplementary Figs. 2–6) by Fourier transform infrared (FT-IR) spectroscopy, 1H/13C nuclear magnetic resonance (NMR) spectroscopy, and electrospray ionization mass spectrometry (ESI-MS). Colorless crystals of Ln-CTP-COOH (Ln = Eu and Tb) were successfully synthesized by the solvothermal reaction of ligand CTP-COOH with lanthanide chloride in a mixed solvent of cyclohexanol and deionized water (v/v = 1:1) heated at 100 °C for 5 days. Due to similar coordination behavior of different lanthanide ions, different kinds of Ln3+ ions could be introduced into Ln-MOF simultaneously with Ln3+ ions distributed randomly over the metal sites. Therefore, seven bimetallic Ln-MOFs, i.e., Eu x Tb1-x-CTP-COOH (x = 0.84, 0.69, 0.47, 0.38, 0.19, 0.12, and 0.06), were prepared by using mixed lanthanide chloride according to above-mentioned method. The contents of Eu3+ and Tb3+ ions in desired MOFs were determined by inductively coupled plasma mass spectrometry (ICP-MS) and energy-dispersive X-ray (EDX) spectroscopy (Supplementary Table 1 and Supplementary Figs. 7 and 8). The structures of all Ln-MOFs are isostructural to each other, as confirmed by single-crystal X-ray diffraction (Fig. 1 and Supplementary Tables 2 and 3) and FT-IR (Supplementary Fig. 2). The morphology of all Ln-MOFs was studied by field emission scanning electron microscopy (Supplementary Fig. 9). The Ln-MOFs were also characterized by powder X-ray diffraction (XRD) (Supplementary Fig. 10a), and thermogravimetric analysis (TGA, Supplementary Fig. 10b). More importantly, powder XRD curves (Supplementary Fig. 11) and quantum yields (Φ, Supplementary Table 5) of all post-heating Ln-MOFs (150 °C for 10 h) did not show obvious changes as compared with that of pre-heating Ln-MOFs, demonstrating that the structures of all Ln-MOFs in this work were relatively stable. See Supplementary Discussion for further details.
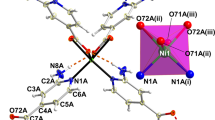
Crystal structure of Eu-CTP-COOH. a Coordination environment around Eu3+ ion (50% ellipsoids) with hydrogen atoms being omitted for clarity. b Tri-capped triangular prismatic and bi-capped triangular prismatic geometry of Eu1 and Eu2 in Eu-CTP-COOH, respectively. c Packing framework of Eu-CTP-COOH, where Eu3+ is represented as a polyhedron
By taking Eu-CTP-COOH as a representative system, Eu-CTP-COOH crystallizes in triclinic space group P-1. The asymmetric unit contains two crystallographic Eu3+ ions (Eu1 and Eu2), one independent hexa-carboxylate anion CTP-COO6−, and five coordinated aqua molecules (Fig. 1a). The Eu1 ion is non-coordinated by nine oxygen atoms: seven from four different CTP-COO6− ligands, and two from coordinated water molecules, representing a tri-capped triangular prismatic geometry (Fig. 1b). The Eu2 ion is coordinated by eight oxygen atoms: five from three different COO6− ligands, and three from coordinated water molecules. The local coordination geometry of the Eu2 ion is bi-capped triangular prismatic (Fig. 1b). The bond length of Eu–O ranges from 2.3238(19) to 2.5932(19) Å, typical values in Eu(III)–carboxylate complexes46. Every CTP-COOH6− ligand is coordinated to eight Eu3+ ion centers. The CTP-COOH6− ligand acts as a μ8-bridge, and four different carboxylate groups adopt μ2-η2: η1, μ2-η1: η1, μ1-η1: η1, and μ1-η1:η0-bridging modes, respectively, (Supplementary Figs. 12 and 13). Eu1 ion and Eu2 ion are connected via four bridging carboxylate acids to form metallic dimers, which are further linked by CTP-COO6− to generate a three-dimensional framework (Fig. 1c). In the crystal structure of Eu-CTP-COOH, the central P3N3 ring is nearly planar, the average P–N distance is 1.584 Å, and the average N–P–N and P–N–P angles are 116.38o and 120.98o, respectively. These values are very similar to those of previously reported phosphonitrilic chloride trimer derivatives47, 48.
Photophysical properties
In powder, film, or THF solution, all Ln-MOFs exhibited characteristic emission with sharp and well-separated emission bands (Fig. 2). The photophysical data of all Ln-MOFs were shown in Supplementary Figs. 14–17 and Supplementary Table 4. In addition, the calculated energy transfer efficiency of the Tb3+ → Eu3+ transition in Eu x Tb1-x-CTP-COOH was over 13% (Supplementary Figs. 18–22), suggesting relatively efficient energy transfer. More importantly, the luminescent colors of these bimetallic Ln-MOFs could be fine-tuned by modulating the ratios of Eu3+/Tb3+ during the synthesis procedures (Fig. 2). These luminescent colors also indicate that the organic linker CTP-COOH in MOF structures is an excellent antenna chromosphere to sensitize both Eu3+ and Tb3+ ions. These samples of Eu x Tb1−x-CTP-COOH (x = 0.84, 0.69, 0.47, 0.38, 0.19, 0.12, 0.06) exhibit tunable emission colors controlled through enhancing and weakening the corresponding emission intensities of Eu3+ and Tb3+ ions under 280 nm irradiation, respectively. The observed emission colors of Eu x Tb1−x-CTP-COOH match well with the calculated chromaticity coordinates according to the 1931 Commission Internationale deL’Eclairage (CIE) chromaticity diagram. Upon the increase in the ratio of Tb3+ in powder, film, and THF solution (Supplementary Fig. 23), the emission colors of these bimetallic Ln-MOFs gradually shift from red region to green region. When the ratio of Eu3+ ions is 0.19 (Eu0.19Tb0.81-CTP-COOH), near white light emission could be achieved, and the CIE values are (0.37, 0.35) in powder, (0.38, 0.36) in film, and (0.41, 0.34) in THF solution. Especially, a near white light LED was successfully fabricated through coating the phosphor Eu0.19Tb0.81-CTP-COOH on the surface of the 280 nm ultraviolet (UV) LED chip. The emission spectrum of white light LED exhibits the characteristic emission of Eu3+ and Tb3+ ions under a drive voltage of 3 V, and its CIE value is (0.28, 0.30), belonging to near white light (Supplementary Fig. 23d).
Emission spectra and corresponding photograph of Ln-MOFs. a In powder. b In film. c In THF solution. d Photograph of powder samples under 254 nm UV lamp. e Photograph of film samples under 254 nm UV lamp. f Photograph of liquid samples under 254 nm UV lamp. Ln-MOFs: Eu x Tb1-x-CTP-COOH (x = 1, 0.84, 0.69, 0.47, 0.38, 0.19, 0.12, 0.06, and 0)
Metal ion decoding in aqueous solution
It is understood that single emission intensity of luminescent materials varies on many uncontrollable factors, such as the preparation process of materials, instrument parameters, and concentration of probes23, 49, 50. Therefore, the sensing of cations/anions/small molecules through relative emission intensity of luminescent materials is more reliable and accurate than the method using single emission intensity. Because the emission intensity of Eu3+ and Tb3+ ions in the fluorescent spectrum of Eu0.47Tb0.53−CTP-COOH excited at 280 nm was comparable, we chose Eu0.47Tb0.53−CTP-COOH as a phosphor to study the sensing properties for different metal ions in aqueous solution. In order to develop a sensor that could be easily reused, a film sensor (Eu0.47Tb0.53-CTP-COOH PVA film) was successfully fabricated through doping Eu0.47Tb0.53-CTP-COOH into PVA, followed by coating them on the surface of polyethylene glycol terephthalate sheet. As compared with conventional powder sensors based on Ln-MOFs as phosphors11, 23, 50, the film sensor in this work did not need complicated and time-consuming centrifugal treatment during the cycle experiments. As shown in Fig. 3a, the emission spectra exhibit obvious metal ion-dependent effect: various metal ions induce markedly different effects on the luminescence of Eu0.47Tb0.53-CTP-COOH PVA film, particularly for Fe3+ ion. The incorporation of Fe3+ ion into the Eu0.47Tb0.53-CTP-COOH PVA film can drastically quench the characteristic emission of Eu3+ and Tb3+ ions. However, other metal ions only gave a little (Al3+, Ba2+, Cd2+, Co2+, Cr3+, Fe2+, K+, Na+, Ni2+, Li+, Pb2+, and Zn2+) or mild (Ca2+, Cu2+ Mg2+, and Hg2+) effect on the characteristic emission intensity of the Eu0.47Tb0.53-CTP-COOH PVA film. Thus, this unique feature could be used for the detection of Fe3+ ion in aqueous solution.
Metal ion decoding and Fe3+ sensing in aqueous solution. a Fluorescence response of the Eu0.47Tb0.53-CTP-COOH PVA film having different metal ions in aqueous solution with concentration of 1 × 10−2 M for 30 min (λex: 280 nm, slit: 5 nm). Inset shows photographs of films having different metal ions under 256 nm UV irradiation. b Decoded map for different metal ions based on the ratios of I547/I491 and I616/I592. c Emission spectra of Eu0.47Tb0.53-CTP-COOH PVA film in aqueous solution containing different concentrations of Fe3+ ion. Inset shows color changes of Eu0.47Tb0.53-CTP-COOH PVA film having different concentrations of Fe3+ ion. d Stern-Volmer plot of Eu0.47Tb0.53-CTP-COOH PVA film quenched by FeCl3 aqueous solution
It was noted that the film exhibited strong metal ion-dependent luminescence. For example, Cr3+, Ca2+, Na+, and Ni2+ ions gave a significant quenching effect for the emissions of both Eu3+ and Tb3+ ions, while Mg2+, K+, Fe2+, Cu2+, and Co2+ ions mainly quenched the emission of Tb3+ ion, and Ag+, Al3+, Ba2+, Cd2+, Hg2+, Pb2+, and Zn2+ ions showed a quenching effect for the emission of Eu3+ ion. These obvious changes in the emission intensity of the Eu3+/Tb3+ ions enable the Eu0.47Tb0.53-CTP-COOH PVA film to emit metal ion-dependent colors that were clearly distinguishable by the naked eye (inset of Fig. 3a). The above results clearly demonstrate that the metal ions can effectively tune the energy transfer efficiency from “antenna” organic species to Eu3+/Tb3+ ions, and also modulate the energy allocation between Eu3+ and Tb3+ ions in the luminescent spectra of bimetallic Ln-MOFs11, 50. Therefore, the tunable energy allocation between Eu3+ and Tb3+ ions in the luminescent spectra of bimetallic Ln-MOFs could enhance or weaken the emission of Ln3+ ion in Eu0.47Tb0.53-CTP-COOH PVA film. According to these interesting properties toward different metal ions, a reasonable dual−readout 2D decoded map for different metal ions can be fabricated based on the emission intensity ratios of Tb3+ ions (I547/I491) and Eu3+ ions (I616/I592) (Fig. 3b). In this decoded map, the information of the metal ions could be obtained by the ratiometric emission intensity of I547/I491 and I616/I592 from metal ions included Eu0.47Tb0.53-CTP-COOH PVA film, i.e., each of the metal ions relates to one unique 2D readout (I547/I491, I616/I592) in this decoded map. Thus, different metal ions in aqueous solution could be easily recognized and differentiated from this decoded map.
For example, we randomly selected one among these analyte metal ions for the encapsulation into the Eu0.47Tb0.53-CTP-COOH PVA film, and obtained the coordinate value of (I547/I491, I616/I592) as (2.16, 1.87). The metal ion could then be recognized as Zn2+ ion according to its location in the decoded map. In order to study the influence of counterions on the coordinate values (I547/I491, I616/I592) of metal ions in different metal salts, the coordinate values (I547/I491, I616/I592) of Zn2+ ions in ZnCl2, Zn(NO3)2 and Zn(CH3COO)2 in aqueous solution were measured, and they are (2.16, 1.87), (2.34, 1.90) and (2.24, 1.82), respectively. As shown in Supplementary Fig. 24, these coordination values of Zn2+ ions with different counterions are relatively close in this decoded map, and the exclusive area of Zn2+ ion has no overlap with other metal ions’ areas.
Detection of Fe3+ ion in aqueous solution
To confirm the high sensitivity of the Eu0.47Tb0.53-CTP-COOH PVA film for the detection of Fe3+ ion, we measured the fluorescence responses of Eu0.47Tb0.53-CTP-COOH PVA film in the presence of different concentrations of Fe3+ ion. The characteristic emission intensity of the Eu0.47Tb0.53-CTP-COOH PVA film gradually decreased upon increasing Fe3+ concentration from 0 to 2.5 × 10−4 M, and was completely quenched after the addition of 2.5 × 10−4 M (Fig. 3c). The luminescent colors of the test film samples changed from light red to gray when soaked in different concentrations of FeCl3 aqueous solution. To the naked eye, we can easily distinguish different colors in different Fe3+ ion concentrations. Furthermore, the response rate of emission intensity at 547 and 616 nm were also studied. As shown in Fig. 4c, the time-dependent emission intensity of the Eu0.47Tb0.53-CTP-COOH PVA film at 547 and at 616 nm was rapidly reduced as soon as 1 × 10−4 M of Fe3+ aqueous solution was incorporated, and 60% fluorescence quenching at 547 nm was observed within 10 s and 69% fluorescence quenching at 616 nm within 25 s. These results implied that the response of this film sensor for Fe3+ ion is much quicker than that some reported MOFs for Fe3+ ion sensing, which could range from minutes to hour51, 52. In order to utilize the Ln-MOFs practically, competition experiments were carried out to evaluate the selective ability of Eu0.47Tb0.53-CTP-COOH PVA film for the recognition of Fe3+ ion through recording its emission intensity in the presence of one, five, ten, or more interference metal ions. As shown in Fig. 4a, b, in the presence of one, five, ten, or more interference metal ions, the Eu0.47Tb0.53-CTP-COOH PVA film still showed excellent recognition and sensing properties for Fe3+ in aqueous solution.
Anti-interference and reversibility of Eu0.47Tb0.53-CTP-COOH PVA film for the detection of Fe3+ ion. a Competition experiments of the Eu0.47Tb0.53-CTP-COOH PVA film for the detection of Fe3+ in the presence of other metal ions. b Competition experiments for the detection of Fe3+ in the presence of five, ten, or more interference metal ions. 1: Cu2+ + Al3+ + Co2+ + Ba2 + + Zn2+; 2: 1 + Fe3+; 3: K+ + Hg2+ + Pb2+ + Ca2+ + Ni2+; 4: 3 + Fe3+; 5: Li+ + Ag+ + Cd2+ + Mn2+ + Mg2+; 6: 5 + Fe3+; 7: Zn2+ + Na+ + Cr3+ + Fe2+ + Cu2+; 8: 7 + Fe3+; 9: Cu2+ + Fe2+ + Al3+ + Ni2+ + Ca2+ + Zn2+ + Pb2+ + Co2+ + Ba2+ + Ca2+; 10: 9 + Fe3+; 11: Li+ + Ag+ + Cd2+ + Mn2+ + Mg2+ + Zn2+ + Na+ + Cr3+ + Hg2+ + K+; 12: 11 + Fe3+; 13: all metal ions used except for Fe3+; 14: 13 + Fe3+. c Time-dependent emission intensity of Eu0.47Tb0.53-CTP-COOH PVA film incorporated with 1 × 10−4 M Fe3+ ion in aqueous solution. d Powder XRD patters of Eu0.47Tb0.53-CTP-COOH PVA film incorporated with Fe3+ ion and washed by deionized water
To further assess the quenching ability of Fe3+ ion in aqueous solution, the quenching constant (KSV) was calculated using the Stern-Volmer (SV) equation: I0/I = 1 + KSV [C]. The I0 and I are the luminescent intensity at 547 nm in the absence and presence of Fe3+ ions, and [C] is the concentration of Fe3+ ion. The Stern-Volmer plot for the Fe3+ ion showed a good linear correlation, and the value of KSV was estimated to be 8.55 × 104 M−1, revealing a strong quenching effect on the luminescence of Eu0.47Tb0.53-CTP-COOH PVA film (Fig. 3d)53. In addition, the limitation of detection (LOD) of the film sensor for Fe3+ is 3.86 μM (Supplementary Fig. 25). According to the reported results, the LOD of many MOFs for the Fe3+ ion ranges from 10−3 to 10−6 M, meaning that the present film sensor possesses highly sensitive properties for the Fe3+ ion in aqueous solution54. To examine the recyclability of the film for the Fe3+ ion sensing, the film sample was washed with deionized water for 3–5 times after the Fe3+ detection, and then the luminescent intensity was recorded again (Supplementary Fig. 26). The characteristic emission of Eu3+ and Tb3+ ions can be effectively recovered. Although the emission intensity gradually reduced upon the recycling, the Eu0.47Tb0.53-CTP-COOH PVA film can still be used for more than ten times. Facile reusability of the Eu0.47Tb0.53-CTP-COOH PVA film could be further certified by powder XRD patterns of the film sensor that was incorporated with Fe3+ and washed with water (Fig. 4d). In addition, the common anion NO3− exhibited a slight effect on the Fe3+ detection (Supplementary Figs. 27 and 28), excluding the disturbance from counterions. Therefore, all above-mentioned results confirm that the Eu0.47Tb0.53-CTP-COOH PVA film as a fluorescent probe has high sensitivity and very quick response time for the Fe3+ ion detection in aqueous solution. In addition, control experiment results (Supplementary Discussion; Supplementary Figs. 29 and 30) demonstrate that the influence of counterions (such as NO3− and Cl−) on the sensing of Fe3+ was not obvious, and Fe3+ ion plays a key role for enabling luminescence changes of the Eu0.47Tb0.53-CTP-COOH PVA film in aqueous solution even when there was a small number of colloids in the Fe(NO3)3 and FeCl3 aqueous solution.
Recognition mechanism of Fe3+ ion
The underlying mechanism of luminescent quenching by Fe3+ ion might be due to following three aspects. The first, the powder XRD pattern of the Eu0.47Tb0.53-CTP-COOH PVA film after incorporating Fe3+ ion was consistent with that of the as-prepared sample, and thus the collapse of the framework structure could be ruled out (Fig. 4d). The second, very fast response time and recoverable ability indicate that the luminescent quenching cannot be ascribed to the cations exchange, which were further confirmed by the powder XRD (Supplementary Fig. 31). The powder XRD peaks of the Eu0.47Tb0.53-CTP-COOH PVA film incorporated with different metal ions have no obvious changes as compared with the original one, indicating that the framework of the Ln-MOFs was not changed during the sensing process. The last, the competitive energy absorption between Fe3+ ion and organic ligand CTP-COOH was another possible reason for this phenomenon55, 56. There might be a competitive energy absorption process between ligand CTP-COOH and Fe3+ ion in aqueous solution when the excitation spectra between the donor and receptor have a certain degree of overlap. Thus, the UV–visible (Vis) spectra of Fe3+ ion and other tested metal ions were examined, and the corresponding spectra are shown in Supplementary Fig. 32. The obvious spectral overlap between the absorption spectrum of Fe3+ ion and the excitation spectrum of Eu0.47Tb0.53-CTP-COOH was observed, suggesting that the excited energy can be effectively absorbed by Fe3+ ion to decrease the energy transfer efficiency from ligand CTP-COOH to the Eu3+/Tb3+ ions for the luminescent quenching57. The UV–Vis spectra of other metal ions in aqueous solution showed only slight overlap with the excitation spectrum of Eu0.47Tb0.53-CTP-COOH. Moreover, the fluorescence lifetime of Eu0.47Tb0.53-CTP-COOH before and after the addition of Fe3+ ion presented no obvious change, demonstrating that the luminescent quenching process belongs to a static mechanism, and there is no energy transfer within the Eu0.47Tb0.53-CTP-COOH-Fe3+ system (Supplementary Fig. 33).
VOC decoding in vapor
The outstanding sensing performance of the Eu0.47Tb0.53-CTP-COOH PVA film incorporated with different metal ions in aqueous solution inspired our enormous interest in sensing trace VOCs in the vapor state, especially for some carcinogenic and toxic VOCs. In order to test the sensitivity and response rate of the Eu0.47Tb0.53-CTP-COOH PVA film as a vapor sensor, a set-up was fabricated by placing a Eu0.47Tb0.53-CTP-COOH PVA film into a cell, followed by adding one droplet (ca. 5 μL) of VOCs (including styrene, nitrobenzene, o-xylene, m-xylene, p-xylene, benzene, toluene, ethylbenzene, chlorobenzene, 1,2-dichlorobenzene, nitromethane, 1,4-dioxane, acetonitrile, acetone, DMF, MeOH, ethanol, THF, formaldehyde, and acetaldehyde) into the cell (Fig. 5a). The cell was then capped to form a hermetic environment, and the luminescent response of the Eu0.47Tb0.53-CTP-COOH PVA film for VOCs was recorded after 5 min. As shown in Fig. 5b, styrene exhibited a significant quenching effect on the characteristic emission of the Eu0.47Tb0.53-CTP-COOH PVA film, and the emission intensity at 547 nm for Tb3+ ion and 616 nm for Eu3+ was almost completely quenched within 5 min. However, other VOCs only showed slight enhancement effect (DMF, MeOH, ethanol, acetone, acetonitrile, and acetaldehyde) or mild quenching effect (nitrobenzene) on the luminescence intensity of the Eu0.47Tb0.53-CTP-COOH PVA film (Fig. 5b and Supplementary Fig. 34). Therefore, styrene vapor could be effectively detected based on the Eu0.47Tb0.53-CTP-COOH PVA film as a sensor.
VOC decoding in vapor. a In situ solid-state luminescent sensor set-up. b Emission spectra of Eu0.47Tb0.53-CTP-COOH PVA film exposed to different VOCs for 5 min. The inset shows the photographs of color changes for the sensor incorporated with different VOCs. c Two-dimensional decoded map of VOCs based on the emission intensity ratio (I616/I592) of Eu3+ ion as x-axis and the ratio (I547/I491) of Tb3+ as y-axis
As compared with the sensing effect of the Eu0.47Tb0.53-CTP-COOH PVA film for metal ions, the characteristic emission intensity of Eu3+ or Tb3+ ion showed nonspecific enhancement or quenching to VOCs. However, these VOCs could still be easily distinguished based on characteristic emission intensity ratios of Tb3+ ion (I547/I491) and Eu3+ ion (I616/I592). These results clearly indicate that the Eu0.47Tb0.53-CTP-COOH PVA film is an excellent sensor for probing different VOCs. This kind of luminescent sensor for detecting different VOCs is remarkable, because it does not need any additional calibration. The interesting VOC-dependent emission of the Eu0.47Tb0.53-CTP-COOH PVA film can be ascribed to guest-dependent energy transfer from ligand CTP-COOH to Eu3+/Tb3+ ions. Therefore, the characteristic emission intensity of the Eu0.47Tb0.53-CTP-COOH PVA film is relatively sensitive to the included VOCs by tuning the energy transfer from CTP-COOH to Eu3+/Tb3+ ions. The emission intensity ratios of I547/I491 for Tb3+ ion and I616/I592 for Eu3+ ion is almost constant, and unique for each VOC guest in the luminescent spectrum of the Eu0.47Tb0.53-CTP-COOH PVA film. Such characteristic relationship between VOCs and the film sensor could be used to draw an emission-fingerprint map of VOCs based on the ratio of I616/I592 as x-axis and the ratio of I547/I491 as y-axis (Fig. 5c). This internal reference strategy is an excellent approach to solve the problem of variable emission intensity encountered when detecting VOCs by a single emissive transition. As shown in Fig. 5c, even for those VOCs having very similar structural motifs such as isomers of o-xylene, m-xylene, and p-xylene, as well as homologs of benzene, toluene, and ethylbenzene, they could still be unambiguously distinguishable.
Styrene sensing in vapor
As a potential human carcinogen, styrene vapor may be harmful to skin, eyes, and respiratory system. At high levels (10,000 ppm) of styrene vapor, rats and pigs become comatose after several minutes, even died after 30 to 60 min of exposure58. Therefore, it is necessary to develop convenient and reliable sensors for the styrene detection. However, fluorescent sensors for styrene have not been fully developed until now.
The obtained results indicate that the Eu0.47Tb0.53-CTP-COOH PVA film is a promising candidate for the styrene detection. Supplementary Movies 1 and 2 show that yellow emission of the Eu0.47Tb0.53-CTP-COOH PVA film under UV 254 nm became faded gradually until completely disappeared in the styrene environment. As shown in Fig. 6a, time-dependent emission spectra of the Eu0.47Tb0.53-CTP-COOH PVA film in styrene vapor (ca. 5 μL) show remarkable quenching effect, i.e., >50% and 47% fluorescent quenching at 547 nm for Tb3+ ion and at 616 nm for Eu3+ ion within 1 min, respectively. When the response time was up to 4 min, the characteristic emission of Eu3+ and Tb3+ ions were almost completely quenched. Furthermore, time-dependent fluorescent decay curves of the Eu0.47Tb0.53-CTP-COOH PVA film at 547 and 616 nm upon the addition of styrene (ca. 5 μL) can be well fitted using single exponential functions (Fig. 6c, d), and the observed rate constants (Kobs = 1/t1/2, t1/2 is observed half-life) and t1/2 are 0.0118 s−1, 58.55 s for Tb3+ ion at 547 nm, and 0.0097 s−1, 71.27 s for Eu3+ at 616 nm, respectively. These results (high Kobs, short t1/2, and short response time) indicate that the response of the Eu0.47Tb0.53-CTP-COOH PVA film for styrene vapor is extremely quick. In addition, the Eu0.47Tb0.53-CTP-COOH PVA film exhibits (Supplementary Figs. 35 and 36) similar response behavior in lower concentration environment of styrene vapor (2 μL). Single exponential fitted results of time-dependent fluorescent decay curves for the Eu0.47Tb0.53-CTP-COOH PVA film also present that this styrene sensor can effectively recognize and detect styrene molecule in the presence of other interference VOCs (Fig. 6e–j). For instance, the Kobs and t1/2 values of the sensor in nitrobenzene vapor environment are 0.0026 s−1 and 266.29 s, respectively. But, higher Kobs (0.0219 s−1) and shorter t1/2 (31.81 s) were observed in the mixture environment of nitrobenzene and styrene vapors. Under more interference VOCs (nitrobenzene, toluene, ethylbenzene, and 1,2-dichlorobenzene), when inducing styrene into the sensing system, the value of Kobs increased from 0.0018 to 0.0672 s−1, i.e., 37 times of enhancement.
Sensing properties of styrene in vapor. a Time-dependent luminescent spectra of the Eu0.47Tb0.53-CTP-COOH PVA film responded to 5 μL styrene vapor. b Quenching and recovery tests of the Eu0.47Tb0.53-CTP-COOH PVA film for styrene vapor. c, d Time-dependent fluorescent decay curves of the Eu0.47Tb0.53-CTP-COOH PVA film at 547 and 616 nm upon the addition of styrene vapor (2.27 mg/cm3, 5 μL). e–j Time-dependent fluorescent decay curves of the Eu0.47Tb0.53-CTP-COOH PVA film at 547 nm and 616 nm in the presence of one or more interference VOCs
The repeatability for a sensory material is a crucial parameter to assess the sensor practicability. After the detection of styrene, the sample of Eu0.47Tb0.53-CTP-COOH PVA film@styrene could be recovered by simply heating at 100 °C for 3 h. The powder XRD pattern (Supplementary Fig. 37) of the recovered Eu0.47Tb0.53-CTP-COOH PVA film matched well with the as-prepared Eu0.47Tb0.53-CTP-COOH PVA film. As shown in Fig. 6b, the characteristic emission intensity at 547 nm after ten cycles remained unchanged, and its powder XRD pattern still matched well with the as-prepared one (Supplementary Fig. 37), confirming its good stability and reusability.
Sensing mechanism of styrene
The mechanism for the detection of styrene by the Eu0.47Tb0.53-CTP-COOH PVA film was further investigated in detail. In this study, there are three reasons that can cause the fluorescence quenching of the Eu0.47Tb0.53-CTP-COOH PVA film in the presence of styrene vapor: (1) styrene induced framework collapse of the Eu0.47Tb0.53-CTP-COOH PVA film, (2) direct interaction of styrene with the emission centers of Ln3+, and (3) styrene interaction with the ligand of the Eu0.47Tb0.53-CTP-COOH PVA film. As shown in Supplementary Figs. 38 and 39, the film sensor treated by styrene presents very similar powder XRD pattern to the original film sensor, indicating that the framework remains intact in the presence of styrene. Furthermore, styrene has no available functional groups to coordinate with Ln3+, and thus has no direct interaction with Ln3+. Therefore, the luminescence quenching of the Eu0.47Tb0.53-CTP-COOH PVA film under styrene is most likely due to the interaction between styrene and CTP-COOH ligand. The luminescence quenching is typically attributed to photoinduced electron transfer (PET), Förster resonance energy transfer (FRET), or Dexter electron transfer (ET) mechanism59, 60. In general, the main driving force for PET is the energy gap between the lowest unoccupied molecular orbitals (LUMO) of donor and acceptor, and the donor should have higher energy level of LUMO than that of acceptor if PET is the luminescence quenching mechanism.59, 61, 62 However, as calculated by the density functional theory (DFT) at the B3LYP/6-31 G* level (Fig. 7a), the LUMO energy level of the ligand CTP-COOH (donor) is obviously lower than that of styrene (acceptor). This result eliminates the possibility of the PET process on the luminescence quenching of the Eu0.47Tb0.53-CTP-COOH PVA film by styrene. In addition, the absorption spectrum of styrene has no obvious overlap with the emission spectrum of ligand CTP-COOH, and thus the FRET mechanism could also be ruled out. On the other hand, mild quenching effect of nitrobenzene for the fluorescence of the Eu0.47Tb0.53-CTP-COOH PVA film could be ascribed to the PET mechanism.
Sensing mechanism of styrene. a Schematic representation of electronic structures of ligand CTP-COOH, styrene and nitrobenzene. b Simplified schematic diagram of LMET and Dexter-ET process in Eu0.47Tb0.53-CTP-COOH with styrene. c Schematic illustration of the mechanism of styrene sensing by Eu0.47Tb0.53-CTP-COOH
The emission lifetime of the Eu0.47Tb0.53-CTP-COOH PVA film in the absence and presence of styrene vapor is 0.47 and 0.38 ms, respectively (Supplementary Figs. 39 and 40), suggesting that the most possible quenching mechanism of styrene should be attributed to the Dexter-ET from triplet level (T1) of ligand CTP-COOH to the triplet of styrene63. In this work, bright yellow emission of the Eu0.47Tb0.53-CTP-COOH PVA film is based on the ligand-to-metal energy transfer (LMET) process: the ligand strongly absorbs UV light, and then transfers the absorbed energy to 5D0 level of Eu3+ ion and 5D4 level of Tb3+ ion, thus promoting the characteristic emission of Eu3+ and Tb3+ ions (Fig. 7). In the presence of styrene, since the calculated triplet energy level (T1) of styrene lies between triplet T1 of ligand CTP-COOH and 5D0 level of Eu3+, the ligand CTP-COOH and styrene compete with the LMET process through suppressing or blocking the energy transfer from the ligand to Ln3+ ions. As a result, the emission of the Eu0.47Tb0.53-CTP-COOH PVA film is restricted or quenched, and thus styrene is successfully detected (Fig. 7b, c).
Discussion
In summary, we have developed a new strategy using bimetallic Eu3+/Tb3+ MOFs as self-calibrating luminescent sensors for distinguishing different metal ions in aqueous solution and VOCs in vapor. The bimetallic Ln-MOFs serve as a luminescent platform owing to excellent fingerprint correlation between metal ions/VOCs and characteristic emission intensity ratios of I547/I491 for Tb3+ ion and I616/I592 for Eu3+ ion. We have successfully demonstrated the first decoded map that can effectively and reliably distinguish different metal ions in aqueous solution based on the emission intensity ratios of I616/I592 for Eu3+ ion as the x-axis and I547I491 for Tb3+ ion as the y-axis. According to this decoded map, different metal ions in aqueous solution could be easily recognized. Based on a similar principle, a decoded map for sensing different VOCs in vapor has also been established. Such versatile bimetallic Ln-MOF sensor exhibits high sensitivity and selectivity for Fe3+ ion in aqueous solution and styrene in vapor. The capability to unambiguously recognize metal ions/VOCs makes bimetallic Ln-MOFs a promising luminescent platform with potentially valuable applications.
Methods
Materials and methods
All reagents and chemicals were purchased commercially (Sigma-Aldrich and Alfa) and used without further purifications unless otherwise specified. FT-IR spectra were carried out using a Nicolet Is-10 (Nicolet) Fourier Transform Infrared Spectrometer. TGA was made with TA Q50 at a heating rate of 15 °C/min under N2 atmosphere and over a temperature range from room temperature to 800 °C. All the ESI-MS spectra were recorded in LCQ DECA XP mass spectrometer. NMR spectra were taken on a DRX-400 MHz (Bruker) superconducting-magnet NMR spectrometer with TMS as an internal standard. UV–Vis absorption spectrum was determined on a Shimadzu UV-3600 UV–Vis–NIR spectrophotometer. The photoluminescence measurements in the solid state and THF solution were conducted in a Shimadzu RF-5301PC fluorescence spectrophotometer. Fluorescent lifetimes were obtained with the FLS980 steady state spectrometer with a pulsed xenon lamp. ICP-MS data were obtained on an ICP-9000 (N + M) inductively coupled plasma emission spectrometer. Scanning electron microscope images were performed on a JSM-6700F (JEOL) equipped with an energy-dispersive X-ray spectroscopy (EDS) instrument. Powder XRD measurements were recorded on a Rigaku D/Max-2500 X-ray diffractometer using Cu Kα radiation with 2θ range of 2–50°, 40 KeV, and 30 mA having a scanning rate of 0.01° s−1 (2θ) at room temperature.
Synthesis of methyl 4-hydroxybenzoate
4-Hydroxy benzoic acid (35 mmol, 5.3 g) and MeOH (80 mL) were taken in a 150 mL round-bottom flask fitted with a reflux condenser. Concentrated sulfuric acid (2.5 mL) was added as a catalyst and the contents were refluxed for 12 h. The reaction mixture was cooled and excess MeOH was removed using a rotary evaporator. The residue obtained was dissolved in chloroform and washed thoroughly with 10% sodium bicarbonate solution. The organic layer was dried over anhydrous MgSO4 and concentrated to give the product in 95% yield. 1H NMR (400 Hz, CDCl3, ppm): 7.973–7.951 (d, 2 H, Ar–H), 6.887–6.865 (d, 2 H, Ar–OH), 5.8 (s, 1 H, Ar–OH), 3.896 (s, 3 H, COOCH3).
Synthesis of hexa-(4-carbomethoxy-phenoxy)-cyclotriphosphazene (CTP-COOCH3)
Potassium carbonate (11.38 g, 0.082 mol) and methyl 4-hydroxybenzoate (6.54 g, 0.043 mol) were added into a 250 mL round-bottom flask, and then methyl 4-hydroxybenzoate was dissolved by dry THF (120 mL). The mixture solution was heated to 70 °C under the N2, and P3N3Cl6 (2.49 g, 0.007 mol) was added. The mixture was stirred at this temperature overnight. The solvent was removed by evaporation under a reduced pressure, and the obtained solid was re-dissolved in dichloromethane (70 mL). The organic phase was washed with deionized water and then dried over anhydrous MgSO4. Dichloromethane was removed by evaporation under a reduced pressure, the white powder was heated at 60 °C by vacuum oven overnight, and the desired product was obtained (6.1 g, yield 83.7%). 1H NMR (400 MHz, DMSO-d6, ppm): 7.77–7.79 (d, 12 H, Ar–H), 7.03–7.06 (d, 12 H, Ar–H), 3.87 (s, 18 H, –COOCH3). 13C NMR (CDCl3, 100 MHz, ppm):165.98, 153.62, 131.35, 127.36, 120.57, 52.30.
Synthesis of ligand hexa-(4-carboxyl-phenoxy)-cyclotriphosphazene (CTP-COOH)
Sodium hydroxide (1.8 g, 45 mmol) was dissolved in deionized water (80 mL), CTP-COOCH3 (5 g, 4.8 mmol) was dissolved by THF (100 mL), and two solutions were mixed. The mixture was stirred for 1.5 h at 70 °C. After the reaction, THF was removed by rotatory evaporator, and the residual solution was poured into deionized water (300 mL) with dilute hydrochloric acid acidified to pH ca. 2–3. The product as a precipitate was obtained after stirring. The precipitate was collected by filtration, washed with deionized water and dried at 60 °C overnight by vacuum oven. The desired ligand CTP-COOH was obtained as white powder (4.3 g, yield 94%). FT-IR (KBr): v/cm−1 3002.38, 2668.64, 2538.03, 1697.86, 1602.81, 1507.77, 1281.41, 1150.08, 947.66, 774.98, 548.61. 1H NMR (400 MHz, DMSO-d6, ppm) 13.05 (s, 6 H, –COOH), 7.86–7.84 (d, 12 H, Ar–H), 7.00–7.03 (d, 12 H, Ar–H). 13C NMR (300 MHz, DMSO-d6, ppm) 166.69, 153.23, 131.82, 128.82. ESI-MS: 980.09 (CTP-COOH + Na+).
Typical synthesis of [Eu x Tb1-x-CTP-COOH·(H2O)3] (x = 1, 0.84, 0.69, 0.47, 0.38, 0.19, 0.12, 0.06, 0)
These Eu/Tb-MOFs were synthesized according to the following procedure3. According to the pre-designed ratio, Eu x Tb y Cl3•H2O (x + y = 0.5 mmol) and ligand (0.5 mmol) were placed in a 50 mL vial. Deionized water (6 mL) and NaOH solution (0.5 mL, 2 M) were added, and the mixture was stirred at room temperature. After 10 min, warm cyclohexanol (7 mL) was added and the biphasic mixture was stirred for 15 min. The vial was heated to 100 °C and kept at this temperature for 5 days. The colorless crystal was obtained after washing several times with ethanol. The Eu3+/Tb3+ ratio in reaction as well as the lanthanide concentration in the final product is listed in Supplementary Table 1.
Preparation of luminescent platform
The luminescent platform Eu0.47Tb0.53-CTP-COOH PVA film was prepared as follows. First, PVA (1 g, Mw 85,000–124,000, 99 + % hydrolyzed) was dissolved in deionized water (100 mL) at 85 °C for 1.5 h to afford PVA solution (10 g/L). Next, the ground Eu0.47Tb0.53-CTP-COOH powder (10 mg) was dispersed in PVA solution (10 mL), and the sample was treated under ultrasonicaton for 0.5 h to achieve uniformly dispersed Eu0.47Tb0.53-CTP-COOH PVA suspension (Ln-MOF concentration: 1 mg/mL). Finally, dropping the Eu0.47Tb0.53-CTP-COOH PVA suspension onto the polyethylene glycol terephthalate sheet (1 × 2.5 cm) and drying at 60 °C for 3 h afforded the desired luminescent platform.
X-ray crystal structure determination
Diffraction intensity data for single crystal of Ln-MOFs Eu-CTP-COOH (Supplementary Data 1) and Tb-CTP-COOH (Supplementary Data 2) were collected at 100 K on a Bruker Apex II CCD diffractometer equipped with graphite-monochromated Mo-Kα radiation (λ = 0.71073 Å). Data processing was accomplished with the Bruker SAINT software package. Absorption corrections were applied using the Numerical Mu from Formula method (SADABS). The structure was solved by direct methods using SHELXS-97 program of the SHELXTL package and refined by using the full-matrix least squares method on F2 with SHELXTL-97. Anisotropic displacement parameters were applied to all non-hydrogen atoms. Hydrogen atoms were located geometrically and were added to the structure factor calculation.
Quantum yield (Φ) measurements
The quantum yield (Φ) was determined by employing the method reported in literature64, and Φ of a given material could be calculated by a direct comparison with standard phosphors having known Φ values. The quantum yield could be calculated from the following equation:
where RST and Rx apply for these exciting radiation reflected by the standard and the sample, respectively, and ΦST is the quantum yield of the standard phosphor. The IST and Ix terms correspond to the integrated photon flux (photon s−1) for the sample and standard phosphors. Sodium salicylate was chosen as the standard and the ΦST reported is 55% at room temperature65.
Metal ion decoding and Fe3+ ion sensing in aqueous solution
Decoding metal ions: The luminescent platform Eu0.47Tb0.53-CTP-COOH PVA film was immersed into different metal ions (1 × 10−2 M) for 30 min, then the emission spectra of Eu0.47Tb0.53-CTP-COOH PVA film@Metal ions were recorded (λex = 280 nm, slit: 5 nm).
Sensing for Fe3+ ion: The Eu0.47Tb0.53-CTP-COOH PVA film was immersed in deionized water in a quartz cell (2 mL). Then, the Fe3+ aqueous solution was gradually added by a micro syringe, and luminescent spectra were recorded after 3 min equilibration.
VOC decoding and styrene probing in vapors
Decoding VOCs: A home-made set-up was fabricated by placing a Eu0.47Tb0.53-CTP-COOH PVA film into the cell, followed by adding one drop (5 μL) of VOCs (styrene, nitrobenzene, o-xylene, m-xylene, p-xylene, benzene, toluene, ethylbenzene, chlorobenzene, 1,2-dichlorobenzene, nitromethane, 1,4-dioxane, acetonitrile, acetone, DMF, MeOH, ethanol, THF, formaldehyde, and acetaldehyde) into the cell. The cell was then capped to form a hermetic environment, and then luminescent response of Eu0.47Tb0.53-CTP-COOH PVA film towards VOCs was recorded after 5 min.
Probing for styrene in vapors: The luminescent platform Eu0.47Tb0.53-CTP-COOH PVA film was fixed into the cell, followed by adding one droplet styrene (2 μL OR 5 μL) into the cell. The cell was then capped, and time-dependent luminescent spectra were recorded.
Data availability
The data that support this study are available from the authors on reasonable request. See author contributions for specific data sets. The X-ray crystallographic data for the structures reported in this article have been deposited at the Cambridge Crystallographic Data Center (CCDC) under deposition numbers CCDC 1553768 and 1553769. These data can be obtained free of charge from The Cambridge Crystallographic Data Center via www.ccdc.cam.ac.uk/data_request/cif.
References
Carter, K. P., Young, A. M. & Palmer, A. E. Fluorescent sensors for measuring metal ions in living systems. Chem. Rev. 114, 4564–4601 (2014).
Song, X. Z. et al. Single-crystal-to-single-crystal transformation of a europium(III) metal-organic framework producing a multi-responsive luminescent sensor. Adv. Funct. Mater. 24, 4034–4041 (2014).
Shenashen, M. A., El-Safty, S. A. & Elshehy, E. A. Architecture of optical sensor for recognition of multiple toxic metal ions from water. J. Hazard. Mater. 260, 833–843 (2013).
Hwang, K. et al. Photocaged DNAzymes as a general method for sensing metal ions in living cells. Angew. Chem. Int. Ed. 53, 13798–13802 (2014).
Ding, S. Y. et al. Thioether-based fluorescent covalent organic framework for selective detection and facile removal of mercury(II). J. Am. Chem. Soc. 138, 3031–3037 (2016).
Weishaupt, R. et al. A protein-nanocellulose paper for sensing copper ions at the nano- to micromolar level. Adv. Funct. Mater. 27, 1604291 (2017).
Yang, G. H., Wan, X. J., Su, Y. K., Zeng, X. R. & Tang, J. N. Acidophilic S-doped carbon quantum dots derived from cellulose fibers and their fluorescence sensing performance for metal ions in an extremely strong acid environment. J. Mater. Chem. A 4, 12841–12849 (2016).
Hoang, M., Huang, P. J. & Liu, J. W. G-Quadruplex DNA for fluorescent and colorimetric detection of thallium(I). ACS Sens. 1, 137–143 (2016).
Dang, S., Ma, E., Sun, Z. M. & Zhang, H. A layer-structured Eu-MOF as a highly selective fluorescent probe for Fe3+ detection through a cation-exchange approach. J. Mater. Chem. 22, 16920–16926 (2012).
Zheng, M. et al. Fast response and high sensitivity europium metal organic framework fluorescent probe with chelating terpyridine sites for Fe3+. ACS Appl. Mater. Interfaces 5, 1078–1083 (2013).
Chen, D. M., Zhang, N. N., Liu, C. S. & Du, M. Template-directed synthesis of a luminescent Tb-MOF material for highly selective Fe3+ and Al3+ ion detection and VOC vapor sensing. J. Mater. Chem. C. 5, 2311–2317 (2017).
Xu, H., Hu, H. C., Cao, C. S. & Zhao, B. Lanthanide organic framework as a regenerable luminescent probe for Fe3+. Inorg. Chem. 54, 4585–4587 (2015).
Bai, G. X., Tsang, M. K. & Hao, J. H. Luminescent ions in advanced composite materials for multifunctional applications. Adv. Funct. Mater. 26, 6330–6350 (2016).
Adinarayana, B., Thomas, A. P., Yadav, P., Kumar, A. & Srinivasan, A. Bipyricorrole: a corrole homologue with a monoanionic core as a fluorescence ZnII sensor. Angew. Chem. Int. Ed. 55, 969–973 (2016).
Li, Y. T. et al. Core–shell upconversion nanoparticle@metal–organic framework nanoprobes for luminescent/magnetic dual-mode targeted imaging. Adv. Mater. 27, 4075–4080 (2015).
Hosomi, T., Masai, H., Fujihara, T., Tsuji, Y. & Terao, J. A typical metal-ion-responsive color-tunable emitting insulated π-conjugated polymer film. Angew. Chem. Int. Ed. 55, 13427–13431 (2016).
Li, H., Zhang, P., Smaga, L. P., Hoffman, R. A. & Chan, J. Photoacoustic probes for ratiometric imaging of copper(II). J. Am. Chem. Soc. 137, 15628–15631 (2015).
Beer, P. D. & Gale, P. A. Anion recognition and sensing: the state of the art and future perspectives. Angew. Chem. Int. Ed. 40, 486–516 (2001).
Li, Y. T. et al. Coordination-responsive drug release inside gold nanorod@metal-organic framework core–shell nanostructures for near-infrared-induced synergistic chemo-photothermal therapy. Nano Res. https://doi.org/10.1007/s12274-017-1874-y (2017).
He, L. C. et al. Core–shell noble-metal@metal-organic-framework nanoparticles with highly selective sensing property. Angew. Chem. Int. Ed. 52, 3741–3745 (2013).
Zhou, Y. & Yan, B. A responsive MOF nanocomposite for decoding volatile organic compounds. Chem. Commun. 52, 2265–2268 (2016).
Liu, Y. L. & Tang, Z. Y. Multifunctional nanoparticle@MOF core–shell nanostructures. Adv. Mater. 25, 5819–5825 (2013).
Dong, M. J., Zhao, M., Ou, S., Zou, C. & Wu, C. D. A luminescent dye@MOF platform: emission fingerprint relationships of volatile organic molecules. Angew. Chem. Int. Ed. 53, 1575–1579 (2014).
Yang, X. L. et al. A multiresponsive metal–organic framework: direct chemiluminescence, photoluminescence, and dual tunable sensing applications. Adv. Funct. Mater. 26, 393–398 (2016).
Small, L. J. & Neno, T. M. Direct electrical detection of iodine gas by a novel metal-organic-framework-based sensor. ACS Appl. Mater. Interfaces 9, 44649–44655 (2017).
Zhou, Z. C. et al. Internanofiber spacing adjustment in the bundled nanofibers for sensitive fluorescence detection of volatile organic compounds. Anal. Chem. 89, 3814–3818 (2017).
Wang, Y. et al. Real-time detection of styrene using SAW sensors based on hexafluoroisopropanol group functionalized hydrogen-bond acidic polymers. Sens. Actuators B 206, 252–257 (2015).
Van Rooij, J. G. M. et al. Trends in occupational exposure to styrene in the European glass fibre-reinforced plastics industry. Ann. Occup. Hyg. 52, 337–349 (2008).
Babaee, R., Bonakdarpour, B., Nasernejad, B. & Fallah, N. Kinetics of styrene biodegradation in synthetic wastewaters using an industrial activated sludge. J. Hazard. Mater. 184, 111–117 (2010).
Johnson, A. C. Relationship between styrene exposure and hearing loss: review of human studies. Int. J. Occup. Med. Environ. Health 20, 15–325 (2007).
Lawton, B. W., Hoffmann, J. & Triebig, G. The ototoxicity of styrene: a review of occupational investigations. Int. Arch. Occup. Environ. Health 79, 93–102 (2006).
Rueff, J., Teixeira, J. P. L., Santos, S. & Gaspar, J. F. Genetic effects and biotoxicity monitoring of occupational styrene exposure. Clin. Chim. Acta 399, 8–23 (2009).
Prado, C., Ibarra, I. & Periago, J. F. Evaluation of styrene in air by thermal desorption-gas chromatography. J. Chromatogr. A. 778, 255–262 (1997).
Chen, Q. F., Milburn, R. K. & Karellas, N. S. Real time monitoring of hazardous airborne chemicals: a styrene investigation. J. Hazard. Mater. 132, 261–268 (2006).
Cui, Y. J. et al. A luminescent mixed-lanthanide metal-organic framework thermometer. J. Am. Chem. Soc. 134, 3979–3982 (2012).
Sava, D. F., Rohwer, L. E. S., Rodriguez, M. A. & Nenoff, T. M. Intrinsic broad-band white-light emission by a tuned, corrugated metal−organic framework. J. Am. Chem. Soc. 134, 3983–3986 (2012).
Sava, D. F., Rohwer, L. E. S., Rodriguez, M. A. & Nenoff, T. M. Efficient photoluminescence via metal—ligand alteration in a new MOFs family. Chem. Mater. 26, 2943–2951 (2014).
Sava, D. F. et al. Multifunctional, tunable metal–organic framework materials platform for bioimaging applications. ACS Appl. Mater. Interfaces 9, 22268–22277 (2017).
Betard, A. & Fischer, R. A. Metal-organic framework thin films: from fundamentals to applications. Chem. Rev. 112, 1055–1083 (2012).
Achmann, S. et al. Metal-organic frameworks for sensing applications in the gas phase. Sensors 9, 1574–1589 (2009).
Li, L. et al. Visible-light excited luminescent thermometer based on single lanthanide organic frameworks. Adv. Funct. Mater. 26, 8677–8684 (2016).
Cui, Y. et al. Dual-emitting MOF ⊃ dye composite for ratiometric temperature sensing. Adv. Mater. 27, 1420–1425 (2015).
Hao, J. N. & Yan, B. Amino-decorated lanthanide(III) organic extended frameworks for multi-color luminescence and fluorescence sensing. J. Mater. Chem. C. 2, 6758–6764 (2014).
Asha, K. S., Bhattacharjee, R. & Mandal, S. Complete transmetalation in a metal–organic framework by metal ion metathesis in a single crystal for selective sensing of phosphate ions in aqueous media. Angew. Chem. Int. Ed. 55, 11528–11532 (2016).
Gai, Y. L. et al. Mixed-lanthanide metal–organic frameworks with tunable color and white light emission. Cryst. Growth Des. 17, 940–944 (2017).
Feng, R. et al. Novel luminescent three-dimensional heterometallic complexes with 2-fold interpenetrating (3,6)-connected nets. Cryst. Growth Des. 11, 1705–1712 (2011).
Li, B. et al. Temperature-controlled synthesis and luminescent properties of two novel coordination polymers modeled by hexa-carboxylate ligand derived from cyclotriphosphazene. Dalton Trans. 42, 2588–2593 (2013).
Yu, W. J. et al. Lanthanide coordination polymers with hexa-carboxylate ligands derived from cyclotriphosphazene as bridging linkers: synthesis, thermal and luminescent properties. CrystEngComm 15, 7732–7739 (2013).
Zhou, J. M. et al. A bimetallic lanthanide metal–organic material as a self-calibrating color-gradient luminescent sensor. Adv. Mater. 27, 7072–7077 (2015).
Zhan, C., Ou, S., Zou, C., Zhao, M. & Wu, C. D. A luminescent mixed-lanthanide-organic framework sensor for decoding different volatile organic molecules. Anal. Chem. 86, 6648–6653 (2014).
Zhou, Y., Chen, H. H. & Yan, B. An Eu3+ post-functionalized nanosized metal–organic framework for cation exchange-based Fe3+-sensing in an aqueous environment. J. Mater. Chem. A 2, 13691–13697 (2014).
Xiang, Z., Fang, C., Leng, S. & Cao, D. An amino group functionalized metal–organic framework as a luminescent probe for highly selective sensing of Fe3+ ions. J. Mater. Chem. A 2, 7662–7665 (2014).
Xu, X. Y. & Yan, B. Eu(III)-functionalized MIL-124 as fluorescent probe for highly selectively sensing ions and organic small molecules especially for Fe(III) and Fe(II). ACS Appl. Mater. Interfaces 7, 721–729 (2015).
Hou, G. G., Liu, Y., Liu, Q. K., Ma, J. P. & Dong, Y. B. NbO lattice MOFs based on octahedral M(II) and ditopic pyridyl substituted diketonate ligands: structure, encapsulation and guest-driven luminescent property. Chem. Commun. 47, 10731–10733 (2011).
Zhou, J. M., Shi, W., Li, H. M., Li, H. & Cheng, P. Experimental studies and mechanism analysis of high-sensitivity luminescent sensing of pollutional small molecules and ions in Ln4O4 cluster based microporous metal–organic frameworks. J. Phys. Chem. C. 118, 416–426 (2014).
Cheung, W. et al. π-Plasmon absorption of carbon nanotubes for the selective and sensitive detection of Fe3+ ions. Chem. Sci. 7, 5192–5199 (2016).
Li, H., Wang, H. & Wang, L. Synthesis and sensing application of highly luminescent and water stable polyaspartate functionalized LaF3 nanocrystals. J. Mater. Chem. C. 1, 1105–1110 (2013).
Bearman, K. R. et al. A novel piezo-optical styrene sensor incorporating polymer-supported tribromide ion. Chem. Commun. 980–981 (2002).
Nagarkar, S. S., Joarder, B., Chaudhari, A. K., Mukherjee, S. & Ghosh, S. K. Highly selective detection of nitro explosives by a luminescent metal–organic framework. Angew. Chem. Int. Ed. 52, 2881–2885 (2013).
Ramachandra, S. et al. Förster resonance energy transfer in quantum dot-dye-loaded zeolite L nanoassemblies. Small 7, 1488–1494 (2011).
Germain, M. E. & Knapp, M. J. Optical explosives detection: from color changes to fluorescence turn-on. Chem. Soc. Rev. 38, 2543–2555 (2009).
Xu, B. W., Wu, X. F., Li, H. B., Tong, H. & Wang, L. X. Selective detection of TNT and picric acid by conjugated polymer film sensors with donor–acceptor architecture. Macromolecules 44, 5089–5092 (2011).
Qi, Z. W., You, Q. & Chen, Y. Nucleotide/Tb3+ coordination polymer nanoparticles as luminescent sensor and scavenger for nitrite ion. Anal. Chim. Acta 902, 168–173 (2016).
Bril, A. & Jagerveenis, A. W. D. Some methods of luminescence efficiency measurements. J. Res. Natl. Bur. Stand. Sect. A-Phys. Chem. 80, 401–407 (1976).
Malta, O. L. et al. Experimental and theoretical emission quantum yield in the compound Eu(thenoyltrifluoroacetonate)3·2(dibenzyl sulfoxide). Chem. Phys. Lett. 282, 233–238 (1998).
Acknowledgements
This work is financially supported by the National Natural Science Foundation of China (No. 21404017), the special program of Chongqing Science & Technology Commission (No: cstc2016shmszx80052 and cstc2017zdcy-zdyfX0007), the Science and Technology Research Program of Chongqing Municipal Education Commission (No. KJ1709223), the Program for Innovation Team Building at Institutions of Higher Education in Chongqing (No. CXTDX201601030), NTU start-up grant (M408040000), the Singapore Academic Research Fund (No. RG19/16, RG121/16, and RG11/17), and the Singapore Agency for Science, Technology and Research (A*STAR) AME IRG grant (No. A1783c0007). C. L. Yang acknowledges the scholarship support from CSC (No. 201508505177).
Author information
Authors and Affiliations
Contributions
Y.S., J.H.Y., C.L.Y., C.D., and Y.L.Z.: conceived the project. Y.S., J.H.Y., and C.L.Y.: designed and performed most of experiments. Y.S., X.Z.Y., Y.B.L., C.L.Y., and Y.L.Z.: analysis of the data. R.G., S.Z.F.P., W.Q.L., and G.F.L.: performed the single crystal, FT-IR, TGA, and XRD experiments. Y.S. and C.L.Y.: carried out the syntheses. Y.S. and C.L.Y.: prepared the manuscript. S.Z.F.P., J.H.Y., W.Q.L., C.D., and Y.L.Z.: revised the manuscript. All the authors discussed the results and commented on the manuscript. Y.S. and J.H.Y. contributed equally to this work. C.D., C.L.Y., and Y.L.Z. supervised the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Su, Y., Yu, J., Li, Y. et al. Versatile bimetallic lanthanide metal-organic frameworks for tunable emission and efficient fluorescence sensing. Commun Chem 1, 12 (2018). https://doi.org/10.1038/s42004-018-0016-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42004-018-0016-0
This article is cited by
-
AIE-based UiO-66/TiO2:fast response toluene detection and photocatalytic degradation
Journal of Materials Science (2024)
-
Luminescent sensor materials based on rare-earth element complexes for detecting cations, anions, and small molecules
Russian Chemical Bulletin (2022)
-
Controlled Assembly of Luminescent Lanthanide-Organic Frameworks via Post-Treatment of 3D-Printed Objects
Nano-Micro Letters (2021)
-
The Fluorescence Property of Zirconium-Based MOFs Adsorbed Sulforhodamine B
Journal of Fluorescence (2020)
-
Photoluminescent organisms: how to make fungi glow through biointegration with lanthanide metal-organic frameworks
Scientific Reports (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.