Abstract
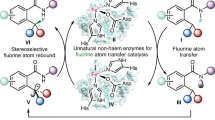
Bioorthogonal chemistry provides a new avenue for disease treatment by generating therapeutic agents in situ. However, two crucial issues have to be considered for future practical applications. One is the prevention of metabolic inactivation of the in situ-synthesized drug molecules. The other is enhancing the biocompatibility and tumour cell selectivity of the bioorthogonal catalyst. Here, to tackle the above issues, we design a biocompatible hydrogen-bonded organic framework-based dual prodrugs activation platform (namely Apt@E-F@PHOF-1). The ferric porphyrin ligands of hydrogen-bonded organic framework-based bioorthogonal pre-catalyst are reduced to ferrous porphyrin by the abundant glutathione in tumour, which then catalyses the cleavage reaction to synthesize 5-fluorouracil (5FU) and 5-ethynyluracil. The 5FU catabolic enzyme inhibitor 5-ethynyluracil prevents 5FU metabolic inactivation. As an example of hydrogen-bonded organic framework-based bioorthogonal prodrug activation, this work provides insights into prevention of drug inactivation by using bioorthogonal chemistry, thus enhancing tumour inhibition and reducing therapeutic side effects demonstrated by both in vitro and orthotopic metastatic mouse model experiments.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
Crystallographic data for the structures reported in this paper have been deposited at the Cambridge Crystallographic Data Centre, under deposition numbers CCDC 2231664. Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/. The data that support the findings of this study are presented in the text and Supplementary Information. Source data are provided with this paper. All other data that support the findings reported herein are available on reasonable request from the corresponding author.
References
Li, S. et al. SuFExable polymers with helical structures derived from thionyl tetrafluoride. Nat. Chem. 13, 858–867 (2021).
Cheng, Y. et al. Diversity oriented clicking delivers beta-substituted alkenyl sulfonyl fluorides as covalent human neutrophil elastase inhibitors. Proc. Natl Acad. Sci. USA 119, e2208540119 (2022).
Schumann, B. et al. Bump-and-hole engineering identifies specific substrates of glycosyltransferases in living cells. Mol. Cell 78, 824–834.e815 (2020).
Cambier, C. J., Banik, S. M., Buonomo, J. A. & Bertozzi, C. R. Spreading of a mycobacterial cell-surface lipid into host epithelial membranes promotes infectivity. eLife 9, e60648 (2020).
Ngai, W. S. C. et al. Bioorthogonally activatable base editing for on-demand pyroptosis. J. Am. Chem. Soc. 144, 5411–5417 (2022).
Liu, Z. et al. Cell membrane-camouflaged liposomes for tumor cell-selective glycans engineering and imaging in vivo. Proc. Natl Acad. Sci. USA 118, e2022769118 (2021).
Weiss, J. T. et al. Extracellular palladium-catalysed dealkylation of 5-fluoro-1-propargyl-uracil as a bioorthogonally activated prodrug approach. Nat. Commun. 5, 3277 (2014).
Clavadetscher, J., Indrigo, E., Chankeshwara, S. V., Lilienkampf, A. & Bradley, M. In-cell dual drug synthesis by cancer-targeting palladium catalysts. Angew. Chem. Int. Ed. 56, 6864–6868 (2017).
Zheng, Y. et al. Enrichment-triggered prodrug activation demonstrated through mitochondria-targeted delivery of doxorubicin and carbon monoxide. Nat. Chem. 10, 787–794 (2018).
Sancho-Albero, M. et al. Cancer-derived exosomes loaded with ultrathin palladium nanosheets for targeted bioorthogonal catalysis. Nat. Catal. 2, 864–872 (2019).
Wang, F. et al. A biocompatible heterogeneous MOF–Cu catalyst for in vivo drug synthesis in targeted subcellular organelles. Angew. Chem. Int. Ed. 58, 6987–6992 (2019).
Wang, Q. et al. A bioorthogonal system reveals antitumour immune function of pyroptosis. Nature 579, 421–426 (2020).
Wang, W. et al. In situ activation of therapeutics through bioorthogonal catalysis. Adv. Drug Deliv. Rev. 176, 113893 (2021).
Wang, Z. et al. A bimetallic metal–organic framework encapsulated with DNAzyme for intracellular drug synthesis and self-sufficient gene therapy. Angew. Chem. Int. Ed. 60, 12431–12437 (2021).
Geng, J. et al. Switching on prodrugs using radiotherapy. Nat. Chem. 13, 805–810 (2021).
Zhang, X. et al. Degradable ZnS-supported bioorthogonal nanozymes with enhanced catalytic activity for intracellular activation of therapeutics. J. Am. Chem. Soc. 144, 12893–12900 (2022).
You, Y. et al. DNA-based platform for efficient and precisely targeted bioorthogonal catalysis in living systems. Nat. Commun. 13, 1459 (2022).
Zhang, Y. H. et al. SphK2 confers 5-fluorouracil resistance to colorectal cancer via upregulating H3K56ac-mediated DPD expression. Oncogene 39, 5214–5227 (2020).
Glorieux, C. et al. Overexpression of NAD(P)H:quinone oxidoreductase 1 (NQO1) and genomic gain of the NQO1 locus modulates breast cancer cell sensitivity to quinones. Life Sci. 145, 57–65 (2016).
Florianczyk, B. & Grzybowska, L. Metallothionein levels in cell fractions from breast cancer tissues. Acta Oncol. 39, 141–143 (2000).
Zhong, L., Shen, H., Huang, C., Jing, H. & Cao, D. AKR1B10 induces cell resistance to daunorubicin and idarubicin by reducing C13 ketonic group. Toxicol. Appl. Pharmacol. 255, 40–47 (2011).
Rustum, Y. M. et al. Thymidylate synthase inhibitors in cancer therapy: direct and indirect inhibitors. J. Clin. Oncol. 15, 389–400 (1997).
Church, D. et al. ‘Toxgnostics’: an unmet need in cancer medicine. Nat. Rev. Cancer 14, 440–445 (2014).
Sasmal, P. K. et al. Catalytic azide reduction in biological environments. ChemBioChem 13, 1116–1120 (2012).
Cao-Milan, R. et al. Thermally gated bioorthogonal nanozymes with supramolecularly confined porphyrin catalysts for antimicrobial uses. Chem 6, 1113–1124 (2020).
Huang, R. et al. Polymer-based bioorthogonal nanocatalysts for the treatment of bacterial biofilms. J. Am. Chem. Soc. 142, 10723–10729 (2020).
Balendiran, G. K., Dabur, R. & Fraser, D. The role of glutathione in cancer. Cell Biochem. Funct. 22, 343–352 (2004).
Lin, R. B. et al. Multifunctional porous hydrogen-bonded organic framework materials. Chem. Soc. Rev. 48, 1362–1389 (2019).
Wang, B., Lin, R. B., Zhang, Z., Xiang, S. & Chen, B. Hydrogen-bonded organic frameworks as a tunable platform for functional materials. J. Am. Chem. Soc. 142, 14399–14416 (2020).
Song, X. et al. Design rules of hydrogen-bonded organic frameworks with high chemical and thermal stabilities. J. Am. Chem. Soc. 144, 10663–10687 (2022).
Liang, W. et al. Enzyme encapsulation in a porous hydrogen-bonded organic framework. J. Am. Chem. Soc. 141, 14298–14305 (2019).
Chen, G. et al. Protein-directed, hydrogen-bonded biohybrid framework. Chem 7, 2722–2742 (2021).
Tang, Z. et al. A biocatalytic cascade in an ultrastable mesoporous hydrogen-bonded organic framework for point-of-care biosensing. Angew. Chem. Int. Ed. 60, 23608–23613 (2021).
Tang, J. et al. In situ encapsulation of protein into nanoscale hydrogen-bonded organic frameworks for intracellular biocatalysis. Angew. Chem. Int. Ed. 60, 22315–22321 (2021).
Wied, P. et al. Combining a genetically engineered oxidase with hydrogen-bonded organic frameworks (HOFs) for highly efficient biocomposites. Angew. Chem. Int. Ed. 61, e202117345 (2022).
Chen, G. et al. Hydrogen-bonded organic framework biomimetic entrapment allowing non-native biocatalytic activity in enzyme. Nat. Commun. 13, 4816 (2022).
Yu, D. et al. Hydrogen-bonded organic framework (HOF)-based single-neural stem cell encapsulation and transplantation to remodel impaired neural networks. Angew. Chem. Int. Ed. 61, e202201485 (2022).
Yin, Q. et al. An ultra-robust and crystalline redeemable hydrogen-bonded organic framework for synergistic chemo-photodynamic therapy. Angew. Chem. Int. Ed. 57, 7691–7696 (2018).
Wang, B. et al. Microporous hydrogen-bonded organic framework for highly efficient turn-up fluorescent sensing of aniline. J. Am. Chem. Soc. 142, 12478–12485 (2020).
Liu, B. T. et al. Ionic hydrogen-bonded organic frameworks for ion-responsive antimicrobial membranes. Adv. Mater. 32, e2005912 (2020).
Liu, B. T. et al. Construction of function-oriented core-shell nanostructures in hydrogen-bonded organic frameworks for near-infrared-responsive bacterial inhibition. Angew. Chem. Int. Ed. 60, 25701–25707 (2021).
Wang, Y. et al. Chemically engineered porous molecular coatings as reactive oxygen species generators and reservoirs for long-lasting self-cleaning textiles. Angew. Chem. Int. Ed. 61, e202115956 (2022).
Zhang, H. et al. NIR-II hydrogen-bonded organic frameworks (HOFs) used for target-specific amyloid-beta photooxygenation in an Alzheimer’s disease model. Angew. Chem. Int. Ed. 61, e202109068 (2022).
Xuan, W. et al. Molecular self-assembly of bioorthogonal aptamer-prodrug conjugate micelles for hydrogen peroxide and pH-independent cancer chemodynamic therapy. J. Am. Chem. Soc. 142, 937–944 (2020).
Lu, T. & Chen, F. Multiwfn: a multifunctional wavefunction analyzer. J. Comput. Chem. 33, 580–592 (2012).
Yu, C. et al. Copper azide nanoparticle-encapsulating MOF-derived porous carbon: electrochemical preparation for high-performance primary explosive film. Small 18, e2107364 (2022).
Giovani, S., Singh, R. & Fasan, R. Efficient conversion of primary azides to aldehydes catalyzed by active site variants of myoglobin. Chem. Sci. 7, 234–239 (2016).
Dubey, M., Bernasek, S. L. & Schwartz, J. Highly sensitive nitric oxide detection using X-ray photoelectron spectroscopy. J. Am. Chem. Soc. 129, 6980–6981 (2007).
Harriman, A. Luminescence of porphyrins and metalloporphyrins. J. Chem. Soc. Faraday Trans. 1 77, 369–377 (1981).
Zhang, Y. H., Luo, D. D., Wan, S. B. & Qu, X. J. S1PR2 inhibitors potently reverse 5-FU resistance by downregulating DPD expression in colorectal cancer. Pharmacol. Res. 155, 104717 (2020).
Wang, Y. et al. Cancer-cell-activated in situ synthesis of mitochondria-targeting AIE photosensitizer for precise photodynamic therapy. Angew. Chem. Int. Ed. 60, 14945–14953 (2021).
Acknowledgements
This work was supported by the National Key R&D Program of China (2022YFA1205804; 2021YFF1200700, J.R.), the National Nature Science Foundation of China (21820102009, X.Q.; 91856205, X.Q.; 22237006, J.R.; 22122704, C.Z.), the Key Program of Frontier of Sciences (CAS QYZDJ-SSW-SLH052, X.Q.) and the Jilin Innovation Project (2023DJ02). We thank Figdraw for providing materials for the graphics.
Author information
Authors and Affiliations
Contributions
X.Q. and J.R. designed research; C.H., C.Z. and Q.D. performed research and analysed data; H.Z. and D.Y. analysed data; C.H., C.Z. and X.Q. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Catalysis thanks Yao Chen and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Methods, Tables 1–6, Note 1 and Figs. 1–46.
Supplementary Data 1
The single-crystal structure of PHOF-1.
Supplementary Data 2
Statistical source data for supplementary figures.
Source data
Source Data Fig. 3
Statistical source data.
Source Data Fig. 4
Statistical source data.
Source Data Fig. 5
Statistical source data.
Source Data Fig. 6
Statistical source data.
Source Data Fig. 7
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, C., Zhao, C., Deng, Q. et al. Hydrogen-bonded organic framework-based bioorthogonal catalysis prevents drug metabolic inactivation. Nat Catal 6, 729–739 (2023). https://doi.org/10.1038/s41929-023-00999-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-023-00999-0