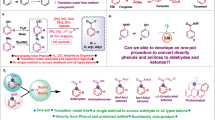
Abstract
Although molecular dinitrogen (N2) is widely used as a carrier or inert gas for many catalytic reactions, it is rarely considered as a catalytic promoter. Here, we report that N2 could be used to reduce the activation energy for catalytic hydrodeoxygenation over ruthenium-based catalysts. Specifically, we report a 4.3-fold activity increase in the catalytic hydrodeoxygenation of p-cresol to toluene over a titanium oxide supported ruthenium catalyst (Ru/TiO2) by simply introducing 6 bar N2 under batch conditions at 160 °C and 1 bar hydrogen. Detailed investigations indicate that N2 can be adsorbed and activated on the metallic ruthenium surface to form hydrogenated nitrogen species, which offer protic hydrogen to lower the activation energy of direct carbonaromatic–oxygen bond scission and the hydrogenation of hydroxy groups. Thus, by employing different ruthenium catalysts, including Ru/TiO2, Ru/Al2O3, Ru/ZrO2 and Ru/C, we demonstrate that N2 promotion of hydrodeoxygenation can be regarded as a general strategy.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data Availability
The data that support the plots within this paper and other findings of this study are available from the corresponding author uon reasonable request.
References
Shaver, M. P. & Fryzuk, M. D. Activation of molecular nitrogen: coordination, cleavage and functionalization of N2 mediated by metal complexes. Adv. Synth. Catal. 345, 1061–1076 (2003).
Erisman, J. W., Sutton, M. A., Galloway, J., Klimont, Z. & Winiwarter, W. How a century of ammonia synthesis changed the world. Nat. Geosci. 1, 636–639 (2008).
Hori, M. & Mori, M. Synthesis of heterocycles utilizing N2-TiCI4-Li-TMSCI. J. Org. Chem. 60, 1480–1481 (1995).
Vol’pin, M. E., Shur, V. B. & Berkovich, E. G. Transformations of molecular nitrogen into aromatic amines under the action of titanium compounds. Inorg. Chim. Acta 280, 264–274 (1998).
Kitano, M. et al. Ammonia synthesis using a stable electride as an electron donor and reversible hydrogen store. Nat. Chem. 4, 934–940 (2012).
Aika, K., Hori, H. & Ozaki, A. Activation of nitrogen by alkali metal promoted transition metal I. ammonia synthesis over ruthenium promoted by alkali metal. J. Catal. 27, 424–431 (1972).
Schulz, H. Short history and present trends of Fischer–Tropsch synthesis. Appl. Catal. A 186, 3–12 (1999).
Kunkes, E. L. et al. Catalytic conversion of biomass to monofunctional hydrocarbons and targeted liquid-fuel classes. Science 322, 417–421 (2008).
Mellmer, M. A. et al. Solvent-enabled control of reactivity for liquid-phase reactions of biomass-derived compounds. Nat. Catal. 1, 199–207 (2018).
Vispute, T. P., Zhang, H., Sanna, A., Xiao, R. & Huber, G. W. Renewable chemical commodity feedstocks from integrated catalytic processing of pyrolysis oils. Science 330, 1222–1227 (2010).
Xu, C., Arancon, R. A., Labidi, J. & Luque, R. Lignin depolymerisation strategies: towards valuable chemicals and fuels. Chem. Soc. Rev. 43, 7485–7500 (2014).
Li, C., Zhao, X., Wang, A., Huber, G. W. & Zhang, T. Catalytic transformation of lignin for the production of chemicals and fuels. Chem. Rev. 115, 11559–11624 (2015).
Luo, Z., Wang, Y., He, M. & Zhao, C. Precise oxygen scission of lignin derived aryl ethers to quantitatively produce aromatic hydrocarbons in water. Green. Chem. 18, 433–441 (2016).
Shao, Y. et al. Selective production of arenes via direct lignin upgrading over a niobium-based catalyst. Nat. Commun. 8, 16104 (2017).
Omotoso, T., Boonyasuwat, S. & Crossley, S. P. Understanding the role of TiO2 crystal structure on the enhanced activity and stability of Ru/TiO2 catalysts for the conversion of lignin-derived oxygenates. Green. Chem. 16, 645–652 (2014).
Liu, G. et al. MoS2 monolayer catalyst doped with isolated Co atoms for the hydrodeoxygenation reaction. Nat. Chem. 9, 810–816 (2017).
Huang, Y.-B., Yan, L., Chen, M.-Y., Guo, Q.-X. & Fu, Y. Selective hydrogenolysis of phenols and phenyl ethers to arenes through direct C–O cleavage over ruthenium–tungsten bifunctional catalysts. Green. Chem. 17, 3010–3017 (2015).
Newman, C. et al. Effects of support identity and metal dispersion in supported ruthenium hydrodeoxygenation catalysts. Appl. Catal. A 477, 64–74 (2014).
Cui, X. et al. Highly selective hydrogenation of arenes using nanostructured ruthenium catalysts modified with a carbon-nitrogen matrix. Nat. Commun. 7, 11326 (2016).
Li, Z., Assary, R. S., Atesin, A. C., Curtiss, L. A. & Marks, T. J. Rapid ether and alcohol C-O bond hydrogenolysis catalyzed by tandem high-valent metal triflate + supported Pd catalysts. J. Am. Chem. Soc. 136, 104–107 (2014).
Bergem, H., Xu, R., Brown, R. C. & Huber, G. W. Low temperature aqueous phase hydrogenation of the light oxygenate fraction of bio-oil over supported ruthenium catalysts. Green. Chem. 19, 3252–3262 (2017).
Gilkey, M. J., Brady, C., Vlachos, D. G. & Xu, B. Characterization of oxidation states in metal/metal oxide catalysts in liquid-phase hydrodeoxygenation reactions with a trickle bed reactor. Ind. Eng. Chem. Res. 57, 5591–5598 (2018).
Omotoso, T. O., Baek, B., Grabow, L. C. & Crossley, S. P. Experimental and first-principles evidence for interfacial activity of Ru/TiO2 to toluene. ChemCatChem 9, 2642–2651 (2017).
Nelson, R. C. et al. Experimental and theoretical insights into the hydrogen-efficient direct hydrodeoxygenation mechanism of phenol over Ru/TiO2. ACS Catal. 5, 6509–6523 (2015).
Jiménez, I., Gago, R. & Albella, J. M. Spectroscopy of π bonding in hard graphitic carbon nitride films: superstructure of basal planes and hardening mechanisms. Phys. Rev. B 62, 4262–4264 (2000).
Kalescky, R., Kraka, E. & Cremer, D. Identification of the strongest bonds in chemistry. J. Phys. Chem. A 117, 8981–8995 (2013).
Chen, G. et al. Interfacial electronic effects control the reaction selectivity of platinum catalysts. Nat. Mater. 15, 564–569 (2016).
Luksirikul, P., Tedsree, K., Moloney, M. G., Green, M. L. & Tsang, S. C. Electron promotion by surface functional groups of single wall carbon nanotubes to overlying metal particles in a fuel-cell catalyst. Angew. Chem. Int. Ed. 51, 6998–7001 (2012).
Elmasides, C., Kondarides, D. I., Grünert, W. & Verykios, X. E. XPS and FTIR study of Ru/Al2O3 and Ru/TiO2 catalysts: reduction characteristics and interaction with a methane-oxygen mixture. J. Phys. Chem. B 103, 5227–5239 (1999).
Li, H.-J., Yeh, C.-H. & Ho, J.-J. The catalytic adsorption and dissociation of carbon dioxide on a double icosahedral Ru19 nanocluster—A theoretical study. Chem. Phys. Lett. 585, 149–152 (2013).
Zhang, S.-T. et al. Density functional theory study on the metal–support interaction between Ru cluster and anatase TiO2(101) surface. J. Phys. Chem. C. 118, 3514–3522 (2014).
Honkala, K. et al. Ammonia synthesis from first-principles calculations. Science 307, 555–558 (2005).
Jacobsen, C. J. H. et al. Structure sensitivity of supported ruthenium catalysts for ammonia synthesis. J. Mol. Catal. A 163, 19–26 (2000).
Ma, X. L., Liu, J. C., Xiao, H. & Li, J. Surface single-cluster catalyst for N2-to-NH3 thermal conversion. J. Am. Chem. Soc. 140, 46–49 (2018).
Liu, J. C. et al. Heterogeneous Fe3 single-cluster catalyst for ammonia synthesis via an associative mechanism. Nat. Commun. 9, 1610 (2018).
Chang, C.-R., Wang, Y.-G. & Li, J. Theoretical investigations of the catalytic role of water in propene epoxidation on gold nanoclusters: A hydroperoxyl-mediated pathway. Nano Res 4, 131–142 (2010).
Chang, C.-R., Huang, Z.-Q. & Li, J. The promotional role of water in heterogeneous catalysis: mechanism insights from computational modeling. WiREs Comput. Mol. Sci. 6, 679–693 (2016).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1991).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Anisimov, V. I., Aryasetiawan, F. & Lichtenstein, A. First-principles calculations of the electronic structure and spectra of strongly correlated systems: dynamical mean-field theory. J. Phys. Condens. Matter 9, 7359–7367 (1997).
Tang, Y., Zhao, S., Long, B., Liu, J.-C. & Li, J. On the nature of support effects of metal dioxides MO2 (M = Ti, Zr, Hf, Ce, Th) in single-atom gold catalysts: importance of quantum primogenic effect. J. Phys. Chem. C. 120, 17514–17526 (2016).
Henkelman, G., Uberuaga, B. P. & Jónsson, H. A climbing image nudged elastic band method for finding saddle points and minimum energy paths. J. Chem. Phys. 113, 9901–9904 (2000).
Acknowledgements
H.D. thanks SCG Chemicals, SCG Packaging (Thailand) and the National Natural Science Foundation of China (Grant No. 21978147). Y.Z. thanks the National Natural Science Foundation of China (Grant No. 21878008) and the Fundamental Research Funds for the Central Universities (BUCTRC201807). X.M. thanks the National Natural Science Foundation of China (21902182) and the Fundamental Research Funds for the Central Universities (2019QH01). J.D. acknowledges support from the Youth Innovation Promotion Association CAS. X.Z. thanks the National Natural Science Foundation of China (Grant No. 11875258). J.-C.B. thanks SCG Chemicals (Thailand) for funding. We also thank the Hefei Light Source and Shanghai Light Source for use of instruments and the Diamond Light Source for access and support in the use of the electron Physical Science Imaging Centre (EM16969, 17397), which contributed to the results presented here. J.L. thanks the National Natural Science Foundation of China (Grand Nos. 21433005, 91645203 and 21590792). The calculations were performed using the supercomputers at Tsinghua National Laboratory for Information Science and Technology. We also thank Y. Li for providing characterization resources and S. Ji for performing ICP analysis. We thank C. Chen for performing TPR measurement, W.-C. Lin for helping with the fixed-bed reactions and Y. Yang for assisting with the FTIR measurements.
Author information
Authors and Affiliations
Contributions
H.D. conceived the idea, designed and carried out the synthesis, characterizations and catalytic reactions, analysed the data and wrote the manuscript. J.L. and X.M. performed DFT calculations and wrote the manuscript. Y.Z. carried out catalytic reactions and analysed the data. M.X. performed the in situ XPS and FTIR measurements and analysed the data. J.D, X.Z. and D.C. performed the in situ XPS measurements and data analysis. J.Z. helped design the experiments and analysed the data. C.A., M.D. and A.K. performed the TEM and STEM measurements. Y.-K.P. performed acidity analysis. T.I. conceived the idea and anticipated discussion. J.-C.B. regulated the experiments and anticipated discussion. J.L., S.C.E.T. and D.O.H. supervised the project, helped design the experiments, analysed the data and wrote the manuscript. All the authors commented on the manuscript and have given approval to the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures 1–28, Tables 1–3, Notes 1 and 2 and References.
Rights and permissions
About this article
Cite this article
Duan, H., Liu, JC., Xu, M. et al. Molecular nitrogen promotes catalytic hydrodeoxygenation. Nat Catal 2, 1078–1087 (2019). https://doi.org/10.1038/s41929-019-0368-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-019-0368-6
This article is cited by
-
Recent Progress on the Hydrodeoxygenation of Lignin-Derived Pyrolysis Oil Using Ru-Based Catalysts
Korean Journal of Chemical Engineering (2024)
-
Dispersed surface Ru ensembles on MgO(111) for catalytic ammonia decomposition
Nature Communications (2023)
-
Lignin Depolymerization for Its Valorization
BioEnergy Research (2023)
-
Photoelectrocatalytic C–H halogenation over an oxygen vacancy-rich TiO2 photoanode
Nature Communications (2021)
-
Coupling N2 and CO2 in H2O to synthesize urea under ambient conditions
Nature Chemistry (2020)