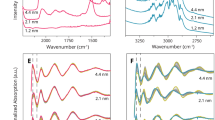
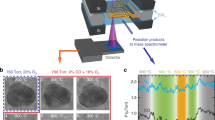
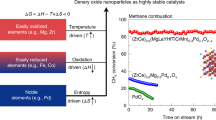
Abstract
In the high-temperature environments needed to perform catalytic processes, supported precious metal catalysts lose their activity severely over time. Generally, loss of catalytic activity is attributed to nanoparticle sintering or processes by which larger particles grow at the expense of smaller ones. Here, by independently controlling particle size and particle loading using colloidal nanocrystals, we reveal the opposite process as an alternative deactivation mechanism: nanoparticles rapidly lose activity for methane oxidation by high-temperature decomposition into inactive single atoms. This deactivation route is remarkably fast, leading to severe loss of activity in as little as 10 min. Importantly, this deactivation pathway is strongly dependent on particle density and the concentration of support defect sites. A quantitative statistical model explains how, for certain reactions, higher particle densities can lead to more stable catalysts.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Argyle, M. D. & Bartholomew, C. H. Heterogeneous catalyst deactivation and regeneration: a review. Catalysts 5, 145–269 (2015).
Bartholomew, C. H. Mechanisms of catalyst deactivation. Appl. Catal. A 212, 17–60 (2001).
Tollefson, J. Worth its weight in platinum. Nature 450, 334–335 (2007).
Robert, J. Farrauto Low-temperature oxidation of methane. Science 337, 659–661 (2012).
Liu, J., Ji, Q., Imai, T., Ariga, K. & Abe, H. Sintering-resistant nanoparticles in wide-mouthed compartments for sustained catalytic performance. Sci. Rep. 7, 41773 (2017).
Prieto, G., Zecevic, J., Friedrich, H., de Jong, K. & de Jongh, P. Towards stable catalysts by controlling collective properties of supported metal nanoparticle. Nat. Mater. 12, 34–39 (2013).
Prieto, G., Meeldijk, J. D., de Jong, K. P. & de Jongh, P. E. Interplay between pore size and nanoparticle spatial distribution: consequences for the stability of CuZn/SiO2 methanol synthesis catalysts. J. Catal. 303, 31–40 (2013).
Goodman, E. D., Schwalbe, J. A. & Cargnello, M. Mechanistic understanding and the rational design of sinter-resistant heterogeneous catalysts. ACS Catal. 7, 7156–7173 (2017).
Scott, S. L. A matter of life(time) and death. ACS Catal. 8, 8597–8599 (2018).
Hansen, T. W., Delariva, A. T., Challa, S. R. & Datye, A. K. Sintering of catalytic nanoparticles: particle migration or Ostwald ripening? Acc. Chem. Res. 46, 1720–1730 (2013).
Cargnello, M. et al. Efficient removal of organic ligands from supported nanocrystals by fast thermal annealing enables catalytic studies on well-defined active phases. J. Am. Chem. Soc. 137, 6906–6911 (2015).
Monai, M., Montini, T., Gorte, R. J. & Fornasiero, P. Catalytic oxidation of methane: Pd and beyond. Eur. J. Inorg. Chem. 2018, 2884–2893 (2018).
Willis, J. J. et al. Systematic structure–property relationship studies in palladium-catalyzed methane complete combustion. ACS Catal. 7, 7810–7821 (2017).
Zhu, G., Han, J., Zemlyanov, D. Y. & Ribeiro, F. H. The turnover rate for the catalytic combustion of methane over palladium is not sensitive to the structure of the catalyst. J. Am. Chem. Soc. 126, 9896–9897 (2004).
Schwartz, W. R. & Pfefferle, L. D. Combustion of methane over palladium-based catalysts: support interactions. J. Phys. Chem. C 116, 8571–8578 (2012).
Otto, K., Haack, L. P. & DeVries, J. E. Identification of two types of oxidized palladium on γ-alumina by X-ray photoelectron spectroscopy. Appl. Catal. B Environ. 1, 1–12 (1992).
Datye, A. K., Xu, Q., Kharas, K. C. & Mccarty, J. M. Particle size distributions in heterogeneous catalysts: what do they tell us about the sintering mechanism? Catal. Today 111, 59–67 (2006).
Kwak, J. H., Hu, J., Mei, D., Yi, C. & Kim, D. H. Coordinatively unsaturated Al3+ centers as binding sites for active catalyst phases of platinum on γ-Al2O3. Science 325, 1670–1673 (2009).
Zhang, Z. et al. Thermally stable single atom Pt/m-Al2O3 for selective hydrogenation and CO oxidation. Nat. Commun. 8, 16100 (2017).
Newton, M. A., Belver-Coldeira, C., Martínez-Arias, A. & Fernández-García, M. Dynamic in situ observation of rapid size and shape change of supported Pd nanoparticles during CO/NO cycling. Nat. Mater. 6, 528–532 (2007).
Peterson, E. J. et al. Low-temperature carbon monoxide oxidation catalysed by regenerable atomically dispersed palladium on alumina. Nat. Commun. 5, 4885 (2014).
Challa, S. R. et al. Relating rates of catalyst sintering to the disappearance of individual nanoparticles during Ostwald ripening. J. Am. Chem. Soc. 133, 20672–20675 (2011).
Campbell, C. T., Parker, S. C. & Starr, D. E. The effect of size-dependent nanoparticle energetics on catalyst sintering. Science 298, 811–814 (2002).
Huang, W., Goodman, E. D., Losch, P. & Cargnello, M. Deconvoluting transient water effects on the activity of Pd methane combustion catalysts. Ind. Eng. Chem. Res. 57, 10261–10268 (2018).
Schwartz, W. R., Ciuparu, D. & Pfefferle, L. D. Combustion of methane over palladium-based catalysts: catalytic deactivation and role of the support. J. Phys. Chem. C 116, 8587–8593 (2012).
Wischert, R., Laurent, P., Copéret, C., Delbecq, F. & Sautet, P. γ-Alumina: the essential and unexpected role of water for the structure, stability and reactivity of ‘defect’ sites. J. Am. Chem. Soc. 134, 14430–14449 (2012).
Ouyang, R., Liu, J. & Li, W. Atomistic theory of ostwald ripening and disintegration of supported metal particles under reaction conditions. J. Am. Chem. Soc. 135, 1760–1771 (2013).
Parker, S. C. & Campbell, C. T. Kinetic model for sintering of supported metal particles with improved size-dependent energetics and applications to Au on TiO2 (110). Phys. Rev. B 75, 035430 (2007).
Bruix, A. et al. Maximum noble-metal efficiency in catalytic materials: atomically dispersed surface platinum. Angew. Chem. Int. Ed. 53, 10525–10530 (2014).
Wu, L. et al. Tuning precursor reactivity toward nanometer-size control in palladium nanoparticles studied by in situ small angle X-ray scattering. Chem. Mater. 30, 1127–1135 (2018).
Schneider, C. A., Rasband, W. S. & Eliceiri, K. W. NIH image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671–675 (2012).
Hu, C. H. et al. Modulation of catalyst particle structure upon support hydroxylation: ab initio insights into Pd13 and Pt13/γ-Al2O3. J. Catal. 274, 99–110 (2010).
Kresse, G. & Hafner, J. Ab initio molecular dynamics for liquid metals. Phys. Rev. B 47, 558–561 (1993).
Kresse, G. & Hafner, J. Ab initio molecular-dynamics simulation of the liquid-metal–amorphous-semiconductor transition in germanium. Phys. Rev. B 49, 14251–14269 (1994).
Kresse, G. & Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132, 154104 (2010).
Digne, M., Sautet, P., Raybaud, P., Euzen, P. & Toulhoat, H. Use of DFT to achieve a rational understanding of acid–basic properties of γ-alumina surfaces. J. Catal. 226, 54–68 (2004).
Nell, J. & O’Neill, H. S. C. Gibbs free energy of formation and heat capacity of PdO: a new calibration of the Pd–PdO buffer to high temperatures and pressures. Geochim. Cosmochim. Acta 60, 2487–2493 (1996).
Rogal, J., Reuter, K. & Scheffler, M. Thermodynamic stability of PdO surfaces. Phys. Rev. B 69, 075421 (2004).
Larsen, A. H., Mortensen, J. J., Blomqvist, J. & Jacobsen, K. W. The atomic simulation environment—a Python library for working with atoms. J. Phys. Condens. Matter 29, 273002 (2017).
Acknowledgements
The authors acknowledge support from the US Department of Energy, Chemical Sciences, Geosciences and Biosciences Division of the Office of Basic Energy Sciences, via grant no. DE-AC02-76SF00515 to the SUNCAT Center for Interface Science and Catalysis. E.D.G. acknowledges support from the National Science Foundation Graduate Research Fellowship under grant no. DGE-1656518. M.C. acknowledges support from the School of Engineering at Stanford University and from a Terman Faculty Fellowship. Part of this work was performed at the Stanford Nano Shared Facilities (SNSF), supported by the National Science Foundation under award no. ECCS-1542152. Use of the Stanford Synchrotron Radiation Lightsource, SLAC National Accelerator Laboratory, is supported by the US Department of Energy, Office of Science, Office of Basic Energy Sciences under contract no. DE-AC02-76F00515. A.S.H. and S.R.B. acknowledge support from the Department of Energy, Basic Energy Sciences Funded Consortium for Operando and Advanced Catalyst Characterization via Electronic Spectroscopy and Structure (Co-ACCESS) at SLAC. O. Mueller is thanked for beamtime assistance. E.M.D. and P.N.P. acknowledge support from the state of Baden-Württemberg, Germany through bwHPC (bwunicluster and JUSTUS, RV bw16G001 and bw17D011) and financial support from the Helmholtz Association.
Author information
Authors and Affiliations
Contributions
E.D.G. and M.C. conceived and designed the experiments. E.D.G. performed catalyst synthesis and testing. A.C.J.-P. performed HAADF-STEM characterization. E.M.D. performed DFT calculations with support from P.N.P. C.J.W. performed X-ray absorption spectroscopy analysis with support from A.S.H. and S.R.B. F.A.-P. contributed to the discussion of atomic energetics. E.D.G. and M.C. wrote the manuscript with contributions and discussions from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–14; Supplementary Tables 1–3
Rights and permissions
About this article
Cite this article
Goodman, E.D., Johnston-Peck, A.C., Dietze, E.M. et al. Catalyst deactivation via decomposition into single atoms and the role of metal loading. Nat Catal 2, 748–755 (2019). https://doi.org/10.1038/s41929-019-0328-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-019-0328-1
This article is cited by
-
Nanoparticle proximity controls selectivity in benzaldehyde hydrogenation
Nature Catalysis (2024)
-
Generation of oxide surface patches promoting H-spillover in Ru/(TiOx)MnO catalysts enables CO2 reduction to CO
Nature Catalysis (2023)
-
Scalable synthesis of Cu clusters for remarkable selectivity control of intermediates in consecutive hydrogenation
Nature Communications (2023)
-
Dynamic and reversible transformations of subnanometre-sized palladium on ceria for efficient methane removal
Nature Catalysis (2023)
-
Designing main-group catalysts for low-temperature methane combustion by ozone
Nature Communications (2023)