Abstract
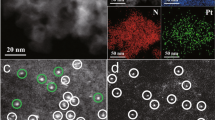
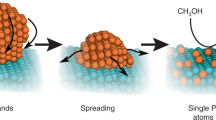
Single-atom catalysts offer a pathway to cost-efficient catalysis using the minimal amount of precious metals. However, preparing and keeping them stable during operation remains a challenge. Here we report the synthesis of double transition metal MXene nanosheets—Mo2TiC2Tx, with abundant exposed basal planes and Mo vacancies in the outer layers—by electrochemical exfoliation, enabled by the interaction between protons and the surface functional groups of Mo2TiC2Tx. The as-formed Mo vacancies are used to immobilize single Pt atoms, enhancing the MXene’s catalytic activity for the hydrogen evolution reaction. The developed catalyst exhibits a high catalytic ability with low overpotentials of 30 and 77 mV to achieve 10 and 100 mA cm−2 and a mass activity about 40 times greater than the commercial platinum-on-carbon catalyst. The strong covalent interactions between positively charged Pt single atoms and the MXene contribute to the exceptional catalytic performance and stability.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding authors upon reasonable request.
References
Zou, X. & Zhang, Y. Noble metal-free hydrogen evolution catalysts for water splitting. Chem. Soc. Rev. 44, 5148–5180 (2015).
Niether, C. et al. Improved water electrolysis using magnetic heating of FeC–Ni core–shell nanoparticles. Nat. Energy 3, 476–483 (2018).
Liu, Y. et al. Self-optimizing, highly surface-active layered metal dichalcogenide catalysts for hydrogen evolution. Nat. Energy 2, 17127 (2017).
Chen, C. et al. Highly crystalline multimetallic nanoframes with three-dimensional electrocatalytic surfaces. Science 343, 1339–1343 (2014).
Morales-Guio, C. G., Stern, L.-A. & Hu, X. Nanostructured hydrotreating catalysts for electrochemical hydrogen evolution. Chem. Soc. Rev. 43, 6555–6569 (2014).
Jones, J. et al. Thermally stable single-atom platinum-on-ceria catalysts via atom trapping. Science 353, 150–154 (2016).
Cheng, N. et al. Platinum single-atom and cluster catalysis of the hydrogen evolution reaction. Nat. Commun. 7, 13638 (2016).
Deng, J. et al. Triggering the electrocatalytic hydrogen evolution activity of the inert two-dimensional MoS2 surface via single-atom metal doping. Energy Environ. Sci. 8, 1594–1601 (2015).
Li, X. et al. Single-atom Pt as co-catalyst for enhanced photocatalytic H2 evolution. Adv. Mater. 28, 2427–2431 (2016).
Lin, L. et al. Low-temperature hydrogen production from water and methanol using Pt/α-MoC catalysts. Nature 544, 80–83 (2017).
Zhang, Z. et al. Thermally stable single atom Pt/m-Al2O3 for selective hydrogenation and CO oxidation. Nat. Commun. 8, 16100 (2017).
Zhang, L., Han, L., Liu, H., Liu, X. & Luo, J. Potential-cycling synthesis of single platinum atoms for efficient hydrogen evolution in neutral media. Angew. Chem. Int. Ed. 56, 13694–13698 (2017).
Qiao, B. et al. Single-atom catalysis of CO oxidation using Pt1/FeOx. Nat. Chem. 3, 634–641 (2011).
Li, H. et al. Synergetic interaction between neighbouring platinum monomers in CO2 hydrogenation. Nat. Nanotech. 13, 411–417 (2018).
Gao, C., Wang, J., Xu, H. & Xiong, Y. Coordination chemistry in the design of heterogeneous photocatalysts. Chem. Soc. Rev. 46, 2799–2823 (2017).
Yang, H. B. et al. Atomically dispersed Ni(I) as the active site for electrochemical CO2 reduction. Nat. Energy 3, 140–147 (2018).
Liu, G. et al. MoS2 monolayer catalyst doped with isolated Co atoms for the hydrodeoxygenation reaction. Nat. Chem. 9, 810–816 (2017).
Fei, H. et al. General synthesis and definitive structural identification of MN4C4 single-atom catalysts with tunable electrocatalytic activities. Nat. Catal. 1, 63–72 (2018).
Yang, X.-F. et al. Single-atom catalysts: a new frontier in heterogeneous catalysis. Acc. Chem. Res. 46, 1740–1748 (2013).
Zhang, C. et al. Single-atomic ruthenium catalytic sites on nitrogen-doped graphene for oxygen reduction reaction in acidic medium. ACS Nano 11, 6930–6941 (2017).
Yang, M. et al. Catalytically active Au-O(OH)x-species stabilized by alkali ions on zeolites and mesoporous oxides. Science 346, 1498–1501 (2014).
Liu, P. et al. Photochemical route for synthesizing atomically dispersed palladium catalysts. Science 352, 797–800 (2016).
Wan, J. et al. Defect effects on TiO2 nanosheets: stabilizing single atomic site Au and promoting catalytic properties. Adv. Mater. 30, 1705369 (2018).
Anasori, B., Lukatskaya, M. R. & Gogotsi, Y. 2D metal carbides and nitrides (MXenes) for energy storage. Nat. Rev. Mater. 2, 16098 (2017).
Shahzad, F. et al. Electromagnetic interference shielding with 2D transition metal carbides (MXenes). Science 353, 1137–1140 (2016).
Naguib, M. et al. New two-dimensional niobium and vanadium carbides as promising materials for Li-ion batteries. J. Am. Chem. Soc. 135, 15966–15969 (2013).
Xie, X. et al. Porous heterostructured MXene/carbon nanotube composite paper with high volumetric capacity for sodium-based energy storage devices. Nano Energy 26, 513–523 (2016).
Liang, X., Garsuch, A. & Nazar, L. F. Sulfur cathodes based on conductive MXene nanosheets for high-performance lithium-sulfur batteries. Angew. Chem. Int. Ed. 54, 3907–3911 (2015).
Lukatskaya, M. R. et al. Cation intercalation and high volumetric capacitance of two-dimensional titanium carbide. Science 341, 1502–1505 (2013).
Ma, T. Y., Cao, J. L., Jaroniec, M. & Qiao, S. Z. Interacting carbon nitride and titanium carbide nanosheets for high-performance oxygen evolution. Angew. Chem. Int. Ed. 55, 1138–1142 (2016).
Seh, Z. W. et al. Two-dimensional molybdenum carbide (MXene) as an efficient electrocatalyst for hydrogen evolution. ACS Energy Lett. 1, 589–594 (2016).
An, X. et al. The synergetic effect of Ti3C2 MXene and Pt as co-catalysts for highly efficient photocatalytic hydrogen evolution over g-C3N4. Phys. Chem. Chem. Phys. 20, 11405–11411 (2018).
Pandey, M. & Thygesen, K. S. Two-dimensional MXenes as catalysts for electrochemical hydrogen evolution: a computational screening study. J. Phys. Chem. C 121, 13593–13598 (2017).
Satheeshkumar, E. et al. One-step solution processing of Ag, Au and Pd@MXene hybrids for SERS. Sci. Rep. 6, 32049 (2016).
Ming, M. et al. Promoted effect of alkalization on the catalytic performance of Rh/alk-Ti3C2X2 (XO, F) for the hydrodechlorination of chlorophenols in base-free aqueous medium. Appl. Catal. B 210, 462–469 (2017).
Li, Z. et al. Reactive metal–support interactions at moderate temperature in two-dimensional niobium-carbide-supported platinum catalysts. Nat. Catal. 1, 349–355 (2018).
Karlsson, L. H., Birch, J., Halim, J., Barsoum, M. W. & Persson, P. O. Atomically resolved structural and chemical investigation of single MXene sheets. Nano Lett. 15, 4955–4960 (2015).
Tao, Q. et al. Two-dimensional Mo1.33C MXene with divacancy ordering prepared from parent 3D laminate with in-plane chemical ordering. Nat. Commun. 8, 14949 (2017).
Hunt, S. T., Milina, M., Wang, Z. & Román-Leshkov, Y. Activating earth-abundant electrocatalysts for efficient, low-cost hydrogen evolution/oxidation: sub-monolayer platinum coatings on titanium tungsten carbide nanoparticles. Energy Environ. Sci. 9, 3290–3301 (2016).
Huang, X. et al. High-performance transition metal-doped Pt3Ni octahedra for oxygen reduction reaction. Science 348, 1230–1234 (2015).
Gao, G., O’Mullane, A. P. & Du, A. 2D MXenes: a new family of promising catalysts for the hydrogen evolution reaction. ACS Catal. 7, 494–500 (2016).
Ling, C., Shi, L., Ouyang, Y. & Wang, J. Searching for highly active catalysts for hydrogen evolution reaction based on O-terminated MXenes through a simple descriptor. Chem. Mater. 28, 9026–9032 (2016).
Liu, Y. et al. Low overpotential in vacancy-rich ultrathin CoSe2 nanosheets for water oxidation. J. Am. Chem. Soc. 136, 15670–15675 (2014).
Lian, P. et al. Alkalized Ti3C2 MXene nanoribbons with expanded interlayer spacing for high-capacity sodium and potassium ion batteries. Nano Energy 40, 1–8 (2017).
Stamenkovic, V. R. et al. Improved oxygen reduction activity on Pt3Ni(111) via increased surface site availability. Science 315, 493–497 (2007).
Tavakkoli, M. et al. Electrochemical activation of single-walled carbon nanotubes with pseudo-atomic-scale platinum for the hydrogen evolution reaction. ACS Catal. 7, 3121–3130 (2017).
Zhang, H. et al. Dynamic traction of lattice-confined platinum atoms into mesoporous carbon matrix for hydrogen evolution reaction. Sci. Adv. 4, eaao6657 (2018).
Nørskov, J. K. et al. Trends in the exchange current for hydrogen evolution. J. Electrochem. Soc. 152, J23–J26 (2005).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953 (1994).
Perdew, J. P. Density-functional approximation for the correlation energy of the inhomogeneous electron gas. Phys. Rev. B 33, 8822 (1986).
Grimme, S. Semiempirical GGA-type density functional constructed with a long-range dispersion correction. J. Comput. Chem. 27, 1787–1799 (2006).
Acknowledgements
The authors thank P. Li from Nanjing University of Aeronautics and Astronautics, Nanjing for performing DFT calculations. Y. Zheng from the University of Adelaide and Y.-C. Huang from the National Synchrotron Radiation Research Centre are acknowledged for their valuable discussions. The authors acknowledge use of the JEOL 2010 and JEOL JEM-ARM200F STEM within the University of Wollongong (UoW) Electron Microscopy Centre. This project was financially supported by the Australian Research Council (ARC) through ARC Discovery projects (DP160104340 and DP170100436) and a Rail Manufacturing CRC (RMCRC) project.
Author information
Authors and Affiliations
Contributions
G.W., J.Z. and Y.Z. conceived the idea. J.Z. and Y.Z. performed the electrochemical experiments. X.G. synthesized the Mo2TiC2Tx MXene. Y.G. provided the MAX materials for this work. J.Z., Y.Z. and X.G. carried out characterizations. C.-L.D., R.-S.L. and C.-P.H. performed and analysed EXAFS and XANES analysis. J.Z., Y.Z., C.C., Y.L., Y.G. and G.W. proposed the mechanism research and discussions. All authors contributed to writing of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs 1–31, Supplementary Tables 1–4, Supplementary Notes 1–5 and Supplementary References
Supplementary Data 1
Atomic coordinates of the optimized (3x3) Mo2TiC2O2–PtSA model
Supplementary Data 2
Atomic coordinates of the optimized (3x3) Mo2TiC2O2 model
Supplementary Data 3
Atomic coordinates of the optimized (4x4) Mo2TiC2O2–PtSA model
Supplementary Data 4
Atomic coordinates of the optimized (4x4) Mo2TiC2O2 model
Rights and permissions
About this article
Cite this article
Zhang, J., Zhao, Y., Guo, X. et al. Single platinum atoms immobilized on an MXene as an efficient catalyst for the hydrogen evolution reaction. Nat Catal 1, 985–992 (2018). https://doi.org/10.1038/s41929-018-0195-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-018-0195-1
This article is cited by
-
Comprehensive synthesis of Ti3C2Tx from MAX phase to MXene
Nature Protocols (2024)
-
Facilitating alkaline hydrogen evolution reaction on the hetero-interfaced Ru/RuO2 through Pt single atoms doping
Nature Communications (2024)
-
Stable hydrogen evolution reaction at high current densities via designing the Ni single atoms and Ru nanoparticles linked by carbon bridges
Nature Communications (2024)
-
Single-atom platinum with asymmetric coordination environment on fully conjugated covalent organic framework for efficient electrocatalysis
Nature Communications (2024)
-
Nickel and/or platinum modified crystalline silicon–carbon composites and their electrochemical behaviour towards the hydrogen evolution reaction
Journal of Applied Electrochemistry (2024)