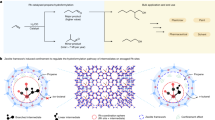
Abstract
Hydrogen peroxide (H2O2) is a valuable chemical with a wide range of applications, but the current industrial synthesis of H2O2 involves an energy-intensive anthraquinone process. The electrochemical synthesis of H2O2 from oxygen reduction offers an alternative route for on-site applications; the efficiency of this process depends greatly on identifying cost-effective catalysts with high activity and selectivity. Here, we demonstrate a facile and general approach to catalyst development via the surface oxidation of abundant carbon materials to significantly enhance both the activity and selectivity (~90%) for H2O2 production by electrochemical oxygen reduction. We find that both the activity and selectivity are positively correlated with the oxygen content of the catalysts. The density functional theory calculations demonstrate that the carbon atoms adjacent to several oxygen functional groups (–COOH and C–O–C) are the active sites for oxygen reduction reaction via the two-electron pathway, which are further supported by a series of control experiments.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Myers, R. L. The 100 Most Important Chemical Compounds (Greenwood Press, London, 2007).
Fukuzumi, S., Yamada, Y. & Karlin, K. D. Hydrogen peroxide as a sustainable energy carrier: electrocatalytic production of hydrogen peroxide and the fuel cell. Electrochim. Acta 82, 493–511 (2012).
Campos‐Martin, J. M., Blanco‐Brieva, G. & Fierro, J. L. Hydrogen peroxide synthesis: an outlook beyond the anthraquinone process. Angew. Chem. Int. Ed. 45, 6962–6984 (2006).
Edwards, J. K. et al. Switching off hydrogen peroxide hydrogenation in the direct synthesis process. Science 323, 1037–1041 (2009).
Solsona, B. E. et al. Direct synthesis of hydrogen peroxide from H2 and O2 using Al2O3 supported Au–Pd catalysts. Chem. Mater. 18, 2689–2695 (2006).
Freakley, S. J. et al. Palladium–tin catalysts for the direct synthesis of H2O2 with high selectivity. Science 351, 965–968 (2016).
Samanta, C. Direct synthesis of hydrogen peroxide from hydrogen and oxygen: an overview of recent developments in the process. Appl. Catal. A Gen. 350, 133–149 (2008).
Edwards, J. K., Freakley, S. J., Lewis, R. J., Pritchard, J. C. & Hutchings, G. J. Advances in the direct synthesis of hydrogen peroxide from hydrogen and oxygen. Catal. Today 248, 3–9 (2015).
Rankin, R. B. & Greeley, J. Trends in selective hydrogen peroxide production on transition metal surfaces from first principles. ACS Catal. 2, 2664–2672 (2012).
Solsona, B. E. et al. Direct synthesis of hydrogen peroxide from H2 and O2 using Al2O3 supported Au−Pd catalysts. Chem. Mater. 18, 2689–2695 (2006).
Wood, K. N., O’Hayre, R. & Pylypenko, S. Recent progress on nitrogen/carbon structures designed for use in energy and sustainability applications. Energy Environ. Sci. 7, 1212–1249 (2014).
Drogui, P., Elmaleh, S., Rumeau, M., Bernard, C. & Rambaud, A. Hydrogen peroxide production by water electrolysis: application to disinfection. J. Appl. Electrochem. 31, 877–882 (2001).
Yamanaka, I. & Murayama, T. Neutral H2O2 synthesis by electrolysis of water and O2. Angew. Chem. Int. Ed. 47, 1900–1902 (2008).
Siahrostami, S. et al. Enabling direct H2O2 production through rational electrocatalyst design. Nat. Mater. 12, 1137–1143 (2013).
Verdaguer-Casadevall, A. et al. Trends in the electrochemical synthesis of H2O2: enhancing activity and selectivity by electrocatalytic site engineering. Nano Lett. 14, 1603–1608 (2014).
Fellinger, T.-P., Hasché, F., Strasser, P. & Antonietti, M. Mesoporous nitrogen-doped carbon for the electrocatalytic synthesis of hydrogen peroxide. J. Am. Chem. Soc. 134, 4072–4075 (2012).
Choi, C. H. et al. Hydrogen peroxide synthesis via enhanced two-electron oxygen reduction pathway on carbon-coated Pt surface. J. Phys. Chem. C 118, 30063–30070 (2014).
Park, J., Nabae, Y., Hayakawa, T. & Kakimoto, M. Highly selective two-electron oxygen reduction catalyzed by mesoporous nitrogen-doped carbon. ACS Catal. 4, 3749–3754 (2014).
Choi, C. H. et al. Tuning selectivity of electrochemical reactions by atomically dispersed platinum catalyst. Nat. Commun. 7, 10922 (2016).
Chen, Z. et al. Development of a reactor with carbon catalysts for modular-scale, low-cost electrochemical generation of H2O2. React. Chem. Eng. 2, 239–245 (2017).
Jirkovský, J. S. et al. Single atom hot-spots at Au–Pd nanoalloys for electrocatalytic H2O2 production. J. Am. Chem. Soc. 133, 19432–19441 (2011).
Perazzolo, V. et al. Nitrogen and sulfur doped mesoporous carbon as metal-free electrocatalysts for the in situ production of hydrogen peroxide. Carbon 95, 949–963 (2015).
Sidik, R. A., Anderson, A. B., Subramanian, N. P., Kumaraguru, S. P. & Popov, B. N. O2 reduction on graphite and nitrogen-doped graphite: experiment and theory. J. Phys. Chem. B 110, 1787–1793 (2006).
Hasché, F., Oezaslan, M., Strasser, P. & Fellinger, T.-P. Electrocatalytic hydrogen peroxide formation on mesoporous non-metal nitrogen-doped carbon catalyst. J. Energy Chem. 25, 251–257 (2016).
Liu, Y., Quan, X., Fan, X., Wang, H. & Chen, S. High‐yield electrosynthesis of hydrogen peroxide from oxygen reduction by hierarchically porous carbon. Angew. Chem. Int. Ed. 54, 6837–6841 (2015).
Li, N. et al. A novel carbon black graphite hybrid air-cathode for efficient hydrogen peroxide production in bioelectrochemical systems. J. Power Sources 306, 495–502 (2016).
Moraes, A. et al. Surface and catalytical effects on treated carbon materials for hydrogen peroxide electrogeneration. Electrocatalysis 7, 60–69 (2016).
Rosca, I. D., Watari, F., Uo, M. & Akasaka, T. Oxidation of multiwalled carbon nanotubes by nitric acid. Carbon 43, 3124–3131 (2005).
Liang, Y. et al. Co3O4 nanocrystals on graphene as a synergistic catalyst for oxygen reduction reaction. Nat. Mater. 10, 780–786 (2011).
Assumpção, M. et al. A comparative study of the electrogeneration of hydrogen peroxide using Vulcan and Printex carbon supports. Carbon 49, 2842–2851 (2011).
Barros, W. R. Oxygen reduction to hydrogen peroxide on Fe3O4 nanoparticles supported on Printex carbon and graphene. Electrochim. Acta 162, 263–270 (2015).
Mehta, V. & Cooper, J. S. Review and analysis of PEM fuel cell design and manufacturing. J. Power Sources 114, 32–53 (2003).
Lu, Z. Superaerophilic carbon‐nanotube‐array electrode for high‐performance oxygen reduction reaction. Adv. Mater. 28, 7155–7161 (2016).
Datsyuk, V. et al. Chemical oxidation of multiwalled carbon nanotubes. Carbon 46, 833–840 (2008).
Kundu, S., Wang, Y., Xia, W. & Muhler, M. Thermal stability and reducibility of oxygen-containing functional groups on multiwalled carbon nanotube surfaces: a quantitative high-resolution XPS and TPD/TPR study. J. Phys. Chem. C 112, 16869–16878 (2008).
Andrews, R., Jacques, D., Qian, D. & Rantell, T. Multiwall carbon nanotubes: synthesis and application. Acc. Chem. Res. 35, 1008–1017 (2002).
Huang, W., Wang, Y., Luo, G. & Wei, F. 99.9% purity multi-walled carbon nanotubes by vacuum high-temperature annealing. Carbon 41, 2585–2590 (2003).
Yue, Z., Jiang, W., Wang, L., Gardner, S. & Pittman, C. U. Surface characterization of electrochemically oxidized carbon fibers. Carbon 37, 1785–1796 (1999).
Bahn, S. R. & Jacobsen, K. W. An object-oriented scripting interface to a legacy electronic structure code. Comput. Sci. Eng. 4, 56–66 (2002).
Giannozzi, P. et al. QUANTUM ESPRESSO: a modular and open-source software project for quantum simulations of materials. J. Phys. Condens. Matter 21, 395502 (2009).
Adllan, A. A. & Dal Corso, A. Ultrasoft pseudopotentials and projector augmented-wave data sets: application to diatomic molecules. J. Phys. Condens. Matter 23, 425501 (2011).
Wellendorff, J. et al. Density functionals for surface science: exchange-correlation model development with Bayesian error estimation. Phys. Rev. B 85, 235149 (2012).
Nørskov, J. K. et al. Origin of the overpotential for oxygen reduction at a fuel-cell cathode. J. Phys. Chem. B 108, 17886–17892 (2004).
Acknowledgements
This work was initiated by the support of the Materials Sciences and Engineering Division of the Basic Energy Sciences office at the US Department of Energy, under contract DEAC02-76-SFO0515. We acknowledge support from SUNCAT seed funding in SLAC. We also gratefully acknowledge support from the Chemical Sciences, Geosciences, and Biosciences Division of the Office of Sciences at the US Department of Energy to the SUNCAT Center for Interface Science and Catalysis under award number DE-SC0004993.
Author information
Authors and Affiliations
Contributions
Z.L., G.C., S.S. and Y.C. conceived the research. Z.L., G.C., Z.C., K.L., J.X., L.L., T.W., D.L. and Y.L. performed the experiments. S.S. and J.K.N. performed the theoretical calculation. Z.L., G.C., Z.C., T.F.J. and Y.C. contributed new reagents and analytical tools. Z.L., G.C., S.S., Z.C., T.F.J., J.K.N. and Y.C. analysed the data. Z.L., G.C., S.S., Z.C., T.F.J., J.K.N. and Y.C. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures 1–17, Supplementary Tables 1–4, Supplementary Note 1, Supplementary Reference
Rights and permissions
About this article
Cite this article
Lu, Z., Chen, G., Siahrostami, S. et al. High-efficiency oxygen reduction to hydrogen peroxide catalysed by oxidized carbon materials. Nat Catal 1, 156–162 (2018). https://doi.org/10.1038/s41929-017-0017-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-017-0017-x
This article is cited by
-
Durable CO2 conversion in the proton-exchange membrane system
Nature (2024)
-
Constructing sulfur and oxygen super-coordinated main-group electrocatalysts for selective and cumulative H2O2 production
Nature Communications (2024)
-
Mesoporous carbon spheres with programmable interiors as efficient nanoreactors for H2O2 electrosynthesis
Nature Communications (2024)
-
In-situ study of the hydrogen peroxide photoproduction in seawater on carbon dot-based metal-free catalyst under operation condition
Nano Research (2024)
-
Selective oxygen electroreduction to hydrogen peroxide in acidic media: The superiority of single-atom catalysts
Nano Research (2024)