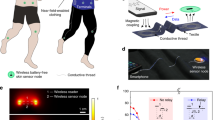
Abstract
A body area sensor network (bodyNET) is a collection of networked sensors that can be used to monitor human physiological signals. For its application in next-generation personalized healthcare systems, seamless hybridization of stretchable on-skin sensors and rigid silicon readout circuits is required. Here, we report a bodyNET composed of chip-free and battery-free stretchable on-skin sensor tags that are wirelessly linked to flexible readout circuits attached to textiles. Our design offers a conformal skin-mimicking interface by removing all direct contacts between rigid components and the human body. Therefore, this design addresses the mechanical incompatibility issue between soft on-skin devices and rigid high-performance silicon electronics. Additionally, we introduce an unconventional radiofrequency identification technology where wireless sensors are deliberately detuned to increase the tolerance of strain-induced changes in electronic properties. Finally, we show that our soft bodyNET system can be used to simultaneously and continuously analyse a person’s pulse, breath and body movement.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data that support the plots within this paper and other findings of this study are available from the corresponding authors upon reasonable request.
References
Yang, G. Z. Body Sensor Networks 2nd edn (Springer, 2014).
Hanson, M. A. et al. Body area sensor networks: challenges and opportunities. Computer 42, 58–65 (2009).
Chu, B., Burnett, W., Chung, J. W. & Bao, Z. Bring on the bodyNET. Nature 549, 328–330 (2017).
Gao, W. et al. Fully integrated wearable sensor arrays for multiplexed in situ perspiration analysis. Nature 529, 509–514 (2016).
Mukhopadhyay, S. C. Wearable sensors for human activity monitoring: a review. IEEE Sens. J. 15, 1321–1330 (2015).
Lee, H. et al. A graphene-based electrochemical device with thermoresponsive microneedles for diabetes monitoring and therapy. Nat. Nanotechnol. 11, 566–572 (2016).
Son, D. et al. Multifunctional wearable devices for diagnosis and therapy of movement disorders. Nat. Nanotechnol. 9, 397–404 (2014).
Yang, S. X. et al. ‘Cut-and-paste’ manufacture of multiparametric epidermal sensor systems. Adv. Mater. 27, 6423–6430 (2015).
Liu, Z. Y. et al. Thickness-gradient films for high gauge factor stretchable strain sensors. Adv. Mater. 27, 6230–6237 (2015).
Kim, J., Kumar, R., Bandodkar, A. J. & Wang, J. Advanced materials for printed wearable electrochemical devices: a review. Adv. Electron. Mater. 3, 1600260 (2017).
Liu, W., Song, M. S., Kong, B. & Cui, Y. Flexible and stretchable energy storage: recent advances and future perspectives. Adv. Mater. 29, 1603436 (2017).
Zamarayeva, A. M. et al. Flexible and stretchable power sources for wearable electronics. Sci. Adv. 3, e1602051 (2017).
Yi, F. et al. A highly shape-adaptive, stretchable design based on conductive liquid for energy harvesting and self-powered biomechanical monitoring. Sci. Adv. 2, e1501624 (2016).
Kaltenbrunner, M. et al. Ultrathin and lightweight organic solar cells with high flexibility. Nat. Commun. 3, 770 (2012).
Wang, S. H. et al. Skin electronics from scalable fabrication of an intrinsically stretchable transistor array. Nature 555, 83–88 (2018).
Andrews, J. B. et al. Patterned liquid metal contacts for printed carbon nanotube transistors. ACS Nano 12, 5482–5488 (2018).
Park, M. et al. Highly stretchable electric circuits from a composite material of silver nanoparticles and elastomeric fibres. Nat. Nanotechnol. 7, 803–809 (2012).
Larson, C. et al. Highly stretchable electroluminescent skin for optical signaling and tactile sensing. Science 351, 1071–1074 (2016).
Liang, J. J., Li, L., Niu, X. F., Yu, Z. B. & Pei, Q. B. Elastomeric polymer light-emitting devices and displays. Nat. Photon. 7, 817–824 (2013).
Keplinger, C. et al. Stretchable, transparent, ionic conductors. Science 341, 984–987 (2013).
Matsuhisa, N. et al. Printable elastic conductors by in situ formation of silver nanoparticles from silver flakes. Nat. Mater. 16, 834–840 (2017).
Trung, T. Q. & Lee, N. E. Flexible and stretchable physical sensor integrated platforms for wearable human-activity monitoring and personal healthcare. Adv. Mater. 28, 4338–4372 (2016).
Hartmann, F., Drack, M. & Kaltenbrunner, M. Meant to merge: fabrication of stretchy electronics for robotics. Sci. Robot. 3, eaat9091 (2018).
Sekitani, T. et al. Stretchable active-matrix organic light-emitting diode display using printable elastic conductors. Nat. Mater. 8, 494–499 (2009).
Kim, D. H. et al. Epidermal electronics. Science 333, 838–843 (2011).
Valentine, A. D. et al. Hybrid 3D printing of soft electronics. Adv. Mater. 29, 1703817 (2017).
van den Brand, J. et al. Flexible and stretchable electronics for wearable health devices. Solid State Electron. 113, 116–120 (2015).
Xu, S. et al. Soft microfluidic assemblies of sensors, circuits, and radios for the skin. Science 344, 70–74 (2014).
Huang, Z. et al. Three-dimensional integrated stretchable electronics. Nat. Electron. 1, 473–480 (2018).
Graz, I. M., Cotton, D. P. J., Robinson, A. & Lacour, S. P. Silicone substrate with in situ strain relief for stretchable thin-film transistors. Appl. Phys. Lett. 98, 124101 (2011).
Vanfleteren, J. et al. Printed circuit board technology inspired stretchable circuits. MRS Bull. 37, 254–260 (2012).
Han, S. et al. Battery-free, wireless sensors for full-body pressure and temperature mapping. Sci. Transl. Med. 10, eaan4950 (2018).
Kim, J. et al. Battery-free, stretchable optoelectronic systems for wireless optical characterization of the skin. Sci. Adv. 2, e1600418 (2016).
Bandodkar, A. J. et al. Battery-free, skin-interfaced microfluidic/electronic systems for simultaneous electrochemical, colorimetric, and volumetric analysis of sweat. Sci. Adv. 5, eaav3294 (2019).
Kim, J. et al. Epidermal electronics with advanced capabilities in near-field communication. Small 11, 906–912 (2015).
Paret, D. Antenna Designs for NFC Devices (Wiley, 2016).
Huang, X. et al. Stretchable, wireless sensors and functional substrates for epidermal characterization of sweat. Small 10, 3083–3090 (2014).
Brink, M., Muller, C. H. & Schierz, C. Contact-free measurement of heart rate, respiration rate, and body movements during sleep. Behav. Res. Methods 38, 511–521 (2006).
Chen, W. X. et al. Unconstrained monitoring of long-term heart and breath rates during sleep. Physiol. Meas. 29, N1–N10 (2008).
Dong, J. G. The role of heart rate variability in sports physiology. Exp. Ther. Med. 11, 1531–1536 (2016).
Bunde, A. et al. Correlated and uncorrelated regions in heart-rate fluctuations during sleep. Phys. Rev. Lett. 85, 3736–3739 (2000).
Sallen, R. P. & Key, E. L. A practical method of designing RC active filters. IEEE Trans. Circuit Theory 2, 74–85 (1955).
Acknowledgements
This research was supported by Samsung Electronics. X.C. acknowledges financial support from the Agency for Science, Technology and Research (A*STAR) under its AME Programmatic Funding Scheme (project no. A18A1b0045). N.M. acknowledges funding support from an overseas fellowship from the Japan Society for the Promotion of Science (JSPS). A.S.Y.P., Z.B. and N.M. acknowledge support from Stanford Precision Health and Integrated Diagnosis Center for seed funding support. The authors thank S. Taheri, W. Wang, J. Kim, B. Chu, Y. Zheng, J. Kang, Y. Kim, H.-C. Wu, J. Xu, T. Lei, Y. Liu, Z. Liu, G. Chen, Y. Jiang and B. Murmann for experimental assistance and insightful discussions. The authors also thank Dupont for providing the stretchable conductor inks.
Author information
Authors and Affiliations
Contributions
S.N., N.M., W.B. and Z.B. generated the design concept. S.N. designed the overall system architecture and verified this architecture through circuit simulation. S.N. designed, fabricated and tested the flexible readout circuits. S.N. and N.M. fabricated the intrinsically stretchable sensor tags, developed the Bluetooth user interfaces and performed all the system measurements and daily physiological signal monitoring. L.B. helped in the design and fabrication of strain sensors. N.M., S.N., Y.Y. and J.L. helped to prepare the three-dimensional schematics and carried out device photography. S.W., J.W., Y.J. and X.Y. contributed to the material choice of stretchable tags. A.S.Y.P. commented on the RFID system design. S.N., N.M., Z.B. and J.B.-H.T. wrote the manuscript. Z.B. and X.C. supervised the project. All authors reviewed and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
A patent based on this research has been submitted.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–16, Supplementary Tables 1–2 and Supplementary Notes 1–2.
Supplementary Movie 1
Display of pulse waves measured from our bodyNET by an oscilloscope.
Supplementary Movie 2
A demonstration of a bodyNET containing five sensing nodes, including one pulse node, one breathing node and three body movement nodes.
Supplementary Movie 3
A demonstration of a sensor node with a built-in seven-segment display to measure respiration.
Supplementary Movie 4
A demonstration of a sensor node located at the neck to measure head movement.
Rights and permissions
About this article
Cite this article
Niu, S., Matsuhisa, N., Beker, L. et al. A wireless body area sensor network based on stretchable passive tags. Nat Electron 2, 361–368 (2019). https://doi.org/10.1038/s41928-019-0286-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41928-019-0286-2
This article is cited by
-
In-depth correlation analysis between tear glucose and blood glucose using a wireless smart contact lens
Nature Communications (2024)
-
A physicochemical-sensing electronic skin for stress response monitoring
Nature Electronics (2024)
-
Digitally-defined ultrathin transparent wireless sensor network for room-scale imperceptible ambient intelligence
npj Flexible Electronics (2024)
-
Large-area, untethered, metamorphic, and omnidirectionally stretchable multiplexing self-powered triboelectric skins
Nature Communications (2024)
-
An asynchronous wireless network for capturing event-driven data from large populations of autonomous sensors
Nature Electronics (2024)