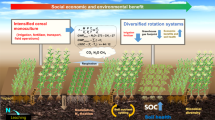
Abstract
Agriculture has played an important role in human health and welfare by producing large amounts of food to feed a growing world population, but this has also placed substantial pressures on natural resources and the environment. One of the most pressing challenges in agriculture is how to ensure food security and promote long-term social-economic development while maintaining healthy, sustainable ecosystems capable of quickly adapting to changing environments. Previous studies demonstrated the positive impact of mixed planting strategies on crop productivity as a consequence of reduced disease impact. Here we present data from a series of trials involving within-species diversification of potatoes grown under smallholding conditions, showing that the benefits of mixed planting strategies extend beyond increases in yield, production resilience and reductions in disease, to increased soil microbial diversity, improved soil nutrients and reduced evolution in the associated Phytophthora infestans pathogen. Taken together, these synergistic benefits provide a good opportunity for achieving sustainable agriculture.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Burdon, J. J., Barrett, L. G., Rebetzke, G. & Thrall, P. H. Guiding deployment of resistance in cereals using evolutionary principles. Evol. Appl. 7, 609–624 (2014).
Zhan, J., Thrall, P. H., Papaïx, J., Xie, L. & Burdon, J. J. Playing on a pathogen’s weakness: using evolution to guide sustainable plant disease control strategies. Annu. Rev. Phytopathol. 53, 19–43 (2015).
Garrett, K. A. & Mundt, C. C. Host diversity can reduce potato late blight severity for focal and general patterns of primary inoculum. Phytopathology 90, 1307–1312 (2000).
Newton, A. C., Ellis, R. P., Hackett, C. A. & Guy, D. C. The effect of component number on Rhynchosporium secalis infection and yield in mixtures of winter barley cultivars. Plant Pathol. 45, 930–938 (1997).
Zhu, Y. et al. Genetic diversity and disease control in rice. Nature 406, 718–722 (2000).
Carson, M. L. Crown rust development and selection for virulence in Puccinia coronata f. sp. avenae in an oat multiline cultivar. Plant Dis. 93, 347–353 (2009).
Mundt, C. C., Sackett, K. E. & Wallace, L. D. Landscape heterogeneity and disease spread: experimental approaches with a plant pathogen. Ecol. Appl. 21, 321–328 (2011).
Newton, A. C. et al. Soil tillage effects on the efficacy of cultivars and their mixtures in winter barley. Field Crop. Res. 128, 91–100 (2012).
Doring, T. F. et al. Comparative analysis of performance and stability among composite cross populations, variety mixtures and pure lines of winter wheat in organic and conventional cropping systems. Field Crop. Res. 183, 2345–245 (2015).
Cook, R. J. & Veseth, R. J. Wheat Health Management (American Phytopathological Society, St. Paul, 1991).
Smithson, J. B. & Lenne, J. M. Varietal mixtures: a viable strategy for sustainable productivity in subsistence agriculture. Ann. Appl. Biol. 128, 127–158 (1996).
Mundt, C. C. Use of multiline cultivars and cultivar mixtures for disease management. Annu. Rev. Phytopathol. 40, 381–410 (2002).
Creissen, H. E., Jorgensen, T. H. & Brown, J. K. Increased yield stability of field-grown winter barley (Hordeum vulgare L.) varietal mixtures through ecological processes. Crop. Prot. 85, 1–8 (2016).
Sommerhalder, R. J., McDonald, B. A., Mascher, F. & Zhan, J. Effect of hosts on competition among clones and evidence of differential selection between pathogenic and saprophytic phases in experimental populations of the wheat pathogen Phaeosphaeria nodorum. BMC Evol. Biol. 11, 188 (2011).
Zhan, J., Mundt, C. C., Hoffer, M. E. & McDonald, B. A. Local adaptation and effect of host genotype on the rate of pathogen evolution: an experimental test in a plant pathosystem. J. Evol. Biol. 15, 634–647 (2002).
Chin, K. M. & Wolfe, M. S. Selection on Erysiphe graminis in pure and mixed stands of barley. Plant Pathol. 33, 535–546 (1984).
Nei, M. Analysis of gene diversity in subdivided populations. Proc. Natl Acad. Sci. USA 70, 3321–3323 (1973).
Wricke, G. Cinc mehtodo zer ertussog der okojogischen streobrelte in Felder. Versochen Z. Oflanzenzucht 47, 92–96 (1962).
Finckh, M. et al. Cereal variety and species mixtures in practice, with emphasis on disease resistance. Agronomie 20, 813–837 (2000).
Cowger, C. & Mundt, C. C. Effects of wheat cultivar mixtures on epidemic progression of septoria tritici blotch and pathogenicity of Mycosphaerella graminicola. Phytopathology 92, 617–623 (2002).
Cox, C. M. et al. Cultivar mixtures for the simultaneous management of multiple diseases: tan spot and leaf rust of wheat. Phytopathology 94, 961–969 (2004).
Skelsey, P. et al. Influence of host diversity on development of epidemics: an evaluation and elaboration of mixture theory. Phytopathology 95, 328–338 (2005).
Andrivon, D., Lucas, J. M. & Ellisseche, D. Development of natural late blight epidemics in pure and mixed plots of potato cultivars with different levels of partial resistance. Plant Pathology 52, 586–594 (2003).
Garrett, K. A. et al. The effects of host diversity and other management components on epidemics of potato late blight in the humid highland tropics. Phytopathology 91, 993–1000 (2001).
Phillips, S. L., Shaw, M. W. & Wolfe, M. S. The effect of potato variety mixtures on epidemics of late blight, in relation to plot size and level of resistance. Ann. Appl. Biol. 147, 245–252 (2005).
Loreau, M. & Hector, A. Partitioning selection and complementarity in biodiversity experiments. Science 412, 72–76 (2001).
Tilman, D. et al. Diversity and productivity in a long-term grassland experiment. Science 294, 843–845 (2001).
Zak, D. R. et al. Plant diversity, soil microbial communities and ecosystem function: are there any links? Ecology 84, 2042–2050 (2003).
Dybzinski, R. et al. Soil fertility increases with plant species diversity in a long-term biodiversity experiment. Oecologia. 158, 85–93 (2008).
Richardson, A. E. & Simpson, R. J. Soil microorganisms mediating phosphorus availability update on microbial phosphorus. Plant Physiol. 156, 989–996 (2011).
Yang, G. et al. The interaction between arbuscular mycorrhizal fungi and soil phosphorus availability influences plant community productivity and ecosystem stability. J. Ecol. 102, 1072–1082 (2014).
Jacoby, R. et al. The role of soil microorganisms in plant mineral nutrition—current knowledge and future directions. Trends. Plant. Sci. 8, 1617 (2017).
Smith, S. E., Smith, F. A. & Jakobsen, I. Mycorrhizal fungi can dominate phosphate supply to plants irrespective of growth responses. Plant Physiol. 133, 16–20 (2003).
Tkacz, A. & Poole, P. Role of root microbiota in plant productivity. J. Exp. Botany 66, 2167–2175 (2015).
Liu, Z., Liu, G. H., Fu, B. J. & Zheng, X. Relationship between plant species diversity and soil microbial functional diversity along a longitudinal gradient in temperate grasslands of Hulunbeir, Inner Mongolia, China. Ecol. Res. 23, 511–518 (2008).
Berg, G. Plant-microbe interactions promoting plant growth and health: perspectives for controlled use of microorganisms in agriculture. Appl. Microbiol. Bio. 84, 11–18 (2009).
Wei, Z. et al. Trophic network architecture of root-associated bacterial communities determines pathogen invasion and plant health. Nat. Commun. 6, 8413 (2015).
Lambers, H., Mougel, C., Jaillard, B. & Hinsinger, P. Plant-microbe-soil interactions in the rhizosphere: an evolutionary perspective. Plant Soil 321, 83–115 (2009).
Heather, L. et al. Grassroots ecology: plant-microbe-soil interactions as drivers of plant community structure and dynamics. Ecology 83, 2281–2291 (2003).
Garrett, K. A. et al. Intraspecific functional diversity in hosts and its effect on disease risk across a climatic gradient. Ecol. Appl. 19, 1868–1883 (2009).
Zhu, W. et al. Increased frequency of self-fertile isolates in Phytophthora infestans may attribute to their higher fitness relative to the A1 isolates. Sci. Rep. 6, 29428 (2016).
Yang, L. et al. Trade-offs and evolution of thermal adaptation in the Irish potato famine pathogen Phytophthora infestans. Mol. Ecol. 25, 4047–4058 (2016).
Abang, M. et al. Differential selection on Rhynchosporium secalis during parasitic and saprophytic phases in the barley scald disease cycle. Phytopathology 96, 1214–1222 (1996).
Marshall, B., Newton, A. C. & Zhan, J. Quantitative evolution of aggressiveness of powdery mildew in a two cultivar barley mixture. Plant Pathol. 58, 378–388 (2009).
Lannou, C. & Mundt, C. C. Evolution of a pathogen population in host mixtures: rate of emergence of complex races. Theor. Appl. Genet. 94, 991–999 (1997).
Black, W., Mastenbroek, C., Mills, W. R. & Peterson, L. C. A proposal for an international nomenclature of races of Phytophthora infestans and of genes controlling immunity in Solanum demissum derivatives. Euphytica 2, 173–179 (1953).
Burdon, J. J., Zhan, J., Barrett, L. G., Papaïx, J. & Thrall, P. H. Addressing the challenges of pathogen evolution on the world’s arable crops. Phytopathology 106, 1117–1127 (2016).
Zhan, J. & McDonald, B. A. Experimental measures of pathogen competition and relative fitness. Annu.Rev. Phytopathol. 51, 131–153 (2013).
Zhan, J. et al. Achieving sustainable plant disease management through evolutionary principles. Trends. Plant. Sci. 19, 570–575 (2014).
Wang, B. et al. Potato viruses in China. Crop. Prot. 30, 1117–1123 (2011).
Wu, E. et al. Diverse mechanisms shape the evolution of virulence factors in the potato late blight pathogen Phytophthora infestans sampled from China. Sci. Rep. 6, srep26182 (2016).
Zhu, W. et al. Limited sexual reproduction and quick turnover in the population genetic structure of Phytophthora infestans in Fujian, China. Sci. Rep. 5, 10094 (2015).
Qin, C. et al. Comparative analyses of fungicide sensitivity and SSR marker variations indicate a low risk of developing azoxystrobin resistance in Phytophthora infestans. Sci. Rep. 6, 20483 (2016).
Lees, A. K. et al. Novel microsatellite markers for the analysis of Phytophthora infestans populations. Plant Pathol. 55, 311–319 (2006).
Armstrong, M. R. et al. An ancestral oomycete locus contains late blight avirulence gene Avr3a, encoding a protein that is recognized in the host cytoplasm. Proc. Natl Acad. Sci. USA 102, 7766–7771 (2005).
Chen, Y. & Halterman, D. A. Phenotypic characterization of potato late blight resistance mediated by the broad-spectrum resistance gene RB. Phytopathology 101, 263–270 (2011).
Zhu, S. et al. An updated conventional- and a novel GM potato late blight R gene differential set for virulence monitoring of Phytophthora infestans. Euphytica 202, 219–234 (2015).
Garland, J. L. & Mills, A. L. Classification and characterization of heterotrophic microbial communities on the basis of patterns of community-level sole-carbon-source utilization. Appl. Environ. Microb. 57, 2351–2359 (1991).
Barrow, N. J. Soil phosphate chemistry and the P-sparing effect of previous phosphate applications. Plant Soil 397, 401–409 (2015).
Walia, M. K. & Dick, W. A. Soil chemistry and nutrient concentrations in perennial ryegrass as influenced by gypsum and carbon amendments. J. Soil Sci. Plant Nut. 16, 832–847 (2016).
Ertiftik, H. & Zengin, M. Response of maize for grain to potassium and magnesium fertilizers in soils with high lime contents. J. Plant Nutr. 40, 98–103 (2017).
Jeger, M. J. & Viljanen-Rollinson, S. The use of the area under the disease-progress curve (AUDPC) to assess quantitative disease resistance in crop cultivars. Theor. Appl. Genet. 102, 32–40 (2001).
Zak, J. C., Willig, M. R., Moorhead, D. L. & Wildman, H. G. Functional diversity of microbial communities: a quantitative approach. Soil Biol. Biochem. 26, 1101–1108 (1994).
Everitt, B. S The Analysis of Contingency Tables. 2nd edn (Chapman & Hall/CRC Press: Boca Raton, 1992).
Tamura, K. et al. MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 28, 2731–2739 (2011).
Librado, P. & Rozas, J. DnaSPv5: a software for comprehensive analysis of DNA polymorphism data. Bioinformatics 25, 1451–1452 (2009).
Nei, M. & Gojobori, T. Simple methods for estimating the numbers of synonymous and nonsynonymous nucleotide substitutions. Mol. Biol. Evol. 3, 418–426 (1986).
Lawrence, I. & Lin, K. A concordance correlation coefficient to evaluate reproducibility. Biometrics 45, 255–268 (1989).
Ott, R. L. An Introduction to Statistical Methods and Data Analysis 5th edn (Duxbury Press, Belmont, 1992).
Acknowledgements
This work was supported by National Natural Science Foundation of China (grant Nos. U1405213, 31761143010 and 31460368) and the China Agriculture Research System (No. CARS-09-P20).
Author information
Authors and Affiliations
Contributions
L.N.Y. collected and genotyped pathogen isolates, generated sequence and pathogenicity data, analysed data and wrote the manuscript. Z.C.P. conducted field experiments, generated soil nutrition and microbial data and wrote the manuscript. W.Z and E.J.W. collected pathogen isolates, generated sequence and pathogenicity data and wrote the manuscript. D.C.H., X.Y., Y.Y.Q. and Y.W. conducted field experiments, collected pathogen isolates and pathogenicity data. R.S.C., L.P.S., P.H.T. and J.J.B. wrote the manuscript. Q.J.S. conceived and supervised the experiments. J.Z. conceived, designed and supervised the experiments, analysed the data and wrote the manuscript. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Tables 1–2, Supplementary Figures 1–3
Rights and permissions
About this article
Cite this article
Yang, LN., Pan, ZC., Zhu, W. et al. Enhanced agricultural sustainability through within-species diversification. Nat Sustain 2, 46–52 (2019). https://doi.org/10.1038/s41893-018-0201-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41893-018-0201-2
This article is cited by
-
Nitrogen uptake in lentil cultivar mixtures is not predictable from pure stands performance but is correlated with agronomic parameters and experimental conditions
Agriculture & Food Security (2024)
-
Cultivar mixtures increase crop yields and temporal yield stability globally. A meta-analysis
Agronomy for Sustainable Development (2024)
-
Host Diversification May Split Epidemic Spread into Two Successive Fronts Advancing at Different Speeds
Bulletin of Mathematical Biology (2022)
-
Plant-mediated rhizospheric interactions in intraspecific intercropping alleviate the replanting disease of Radix pseudostellariae
Plant and Soil (2020)
-
Diverse approaches to crop diversification in agricultural research. A review
Agronomy for Sustainable Development (2020)