Abstract
We present a general framework for developing a machine learning (ML) tool that supports clinician assessment of patient risk using electronic health record-derived real-world data and apply the framework to a quality improvement use case in an oncology setting to identify patients at risk for a near-term (60 day) emergency department (ED) visit who could potentially be eligible for a home-based acute care program. Framework steps include defining clinical quality improvement goals, model development and validation, bias assessment, retrospective and prospective validation, and deployment in clinical workflow. In the retrospective analysis for the use case, 8% of patient encounters were associated with a high risk (pre-defined as predicted probability ≥20%) for a near-term ED visit by the patient. Positive predictive value (PPV) and negative predictive value (NPV) for future ED events was 26% and 91%, respectively. Odds ratio (OR) of ED visit (high- vs. low-risk) was 3.5 (95% CI: 3.4–3.5). The model appeared to be calibrated across racial, gender, and ethnic groups. In the prospective analysis, 10% of patients were classified as high risk, 76% of whom were confirmed by clinicians as eligible for home-based acute care. PPV and NPV for future ED events was 22% and 95%, respectively. OR of ED visit (high- vs. low-risk) was 5.4 (95% CI: 2.6–11.0). The proposed framework for an ML-based tool that supports clinician assessment of patient risk is a stepwise development approach; we successfully applied the framework to an ED visit risk prediction use case.
Similar content being viewed by others
Introduction
The advent of widespread electronic health record (EHR) implementation coincided with healthcare advances that rapidly increased the volume and complexity of information clinicians can access about individual patients. Yet, from a clinical data synthesis perspective, the EHR and other electronic sources of clinical information still hold a wealth of untapped information that can benefit overall patient care. Unlocking the promise of electronically-stored healthcare data to improve healthcare across a population of patients requires better development and application of tools that collect and synthesize digitally stored data1.
The use of real-world data (RWD) from sources such as EHRs, registries, and claims data for the development of machine learning (ML)-based predictive risk models is an emerging research area2,3,4,5,6,7,8,9. However, to date the vast majority of research about the utility and value of ML approaches in the healthcare setting has been retrospective in nature, used historical data for model development and validation, and was limited to specific use cases10. There is a notable lack of prospective evaluation of ML tools in healthcare, which has hindered their widespread adoption into real-world clinical workflows in an evidenced-based fashion10,11. Indeed, in a recent systematic literature review of studies that used ML tools to address a clinical problem, just 2% of reviewed studies were prospective in design10. Additionally, there is a need for a consistent ML model evaluation framework in order to standardize approaches and facilitate comparisons across tools, data sources, and use cases.
The use of ML-based tools to aid the preemptive identification of patients who are at risk for an adverse clinical event could improve overall patient care and safety through more efficient healthcare delivery and the prompting of an early intervention to mitigate severity. The overall objective of this paper is to put forth a general framework to evaluate and deploy an ML-based clinical tool that supports a clinician’s independent assessment of patient risk for an adverse event by displaying medical information and predicted risk level using documented EHR-derived RWD. Then, in order to demonstrate the functionality and utility of the framework, we present an example use case in the oncology setting.
Results—use case
Retrospective evaluation
The retrospective evaluation included 28,433 encounters (2385 patients); 53% were women, the median age was 65 years, and 87% were White (Table 1). The most common cancers (excluding non-melanoma skin neoplasms) were breast, unspecified primary malignant neoplasms, prostate, and non-Hodgkin's lymphoma as defined by standard ICD mapping rules. The observed prevalence of one or more ED visit(s) within 60 days was 10% and the ML-based clinical tool classified 8% of encounters as high risk (pre-defined predicted probability ≥0.20). The positive predictive value (PPV) and negative predictive value (NPV) for future ED events was 26% and 91%, respectively. Patients identified as high risk by the tool had 3.5 times greater odds of a 60-day ED visit than those identified as low risk (95% CI: 3.4–3.5; Table 2).
Bias assessment
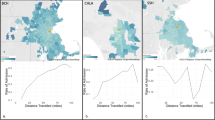
The observed calibration factors for the majority groups in the datasets were as follows: White race: 0.005 [−0.006, 0.014]; Female gender: −0.001 [−0.013, 0.011]; Non-Hispanic ethnicity: 0.004 [−0.006, 0.013] (Table 3). Patient demographic groups constituting minorities along the lines of race, gender, and ethnicity are also reported in Table 3. In all cases, 0 laid within the bootstrapped 95% confidence interval for the metric (i.e., ideal calibration). In the analysis, using calibration factors to assess the model’s fairness performance, the model appeared to be largely calibrated across racial, gender, and ethnic groups, although, as noted previously, the relevant point estimates for some groups have wide confidence intervals (Fig. 1).
Prospective evaluation
The prospective evaluation included 1236 patients; 53% were women, the median age was 65 years, and 84% were White (Table 4). The most common cancers (excluding non-melanoma skin cancer) were breast, prostate, lung, and multiple myeloma. The observed prevalence of an ED visit within 60 days was 7%. The ML-based clinical tool classified 10% of patients as high risk; of these higher risk patients, 76% were confirmed by a Huntsman at Home nurse practitioner review of the EHR that their clinical course would qualify them for admission to the Huntsman at Home program (95% CI: 0.62–0.89). The PPV and NPV for future ED events was 22% and 95%, respectively. Patients identified as high risk by the tool had 5.4 times greater odds of a 60-day ED visit than those identified as low risk (95% CI: 2.6–11.0; Table 2).
We also conducted exploratory stratified analysis by encounter dates before or after 01-13-2020 (60 days prior to national response date on 03-13-202012) to assess whether the model performance was affected by the impact of COVID-19 on patient behaviors and healthcare delivery. The outcome forecast precision (PPV) was lower after the date cut-off chosen to identify patient encounters whose 60 day “at risk” period overlapped the starting of the pandemic.
Discussion
We have developed a standardized framework to evaluate and deploy an ML-based clinical tool that supports a clinician’s independent assessment of patient risk for adverse clinical events by displaying medical information and predicted risk level using documented EHR-derived RWD. As a collaborative multidisciplinary team, we tested the ML-based clinical tool retrospectively on a representative target patient population, evaluated it for potential algorithmic bias, and—importantly—piloted its use in a prospective healthcare system quality improvement study that demonstrated clinically-useful model performance.
Several important learnings were derived from the development of the framework. A key takeaway was that prospective validation (framework step 5) was important for gaining clinician trust in the output of the ML-based adverse event risk prediction tool, which is critical for successful implementation of ML tools in a healthcare setting13,14. In our use case, results of the prospective validation did not indicate additional model changes were necessary for clinical usefulness beyond that suggested in the retrospective analysis. This finding raises the question of when prospective validation of an ML model is useful for deploying the model in different settings, for different patient populations, or for different endpoints. When previous prospective evaluation of the same (or a very similar) model was reassuring in a generally similar population, we believe appropriate rigorous prospective, ongoing monitoring of performance and adequate education to ensure user trust in the model are appropriate guardrails. However, it should be noted that this is speculative based on our experience deploying the ML model prospectively, and additional evaluations of ML-based tools in clinical practice can help to clarify the settings for which prospective research evaluation is necessary.
Additionally, groups responsible for assessing ML models to improve health outcomes should weigh both the upfront resources required for model development and validation, as well as the ongoing resources needed to responsibly monitor changes in performance and bias over time once embedded in the clinical workflow14. In the early stage of model development, it is critical to have upfront engagement from the clinical team who will utilize the tool, as well as to seek clinical feedback throughout the whole process. Discussions with all stakeholders on the practicality and clinical impact of each step will help to prioritize resources and steer the focus of model development. As we demonstrated in the use case, the ML-based clinical tool was focused on increasing the efficiency of surfacing high-risk patients to clinical staff in a timely manner, compared to no information provided by the ML-based clinical tool. Therefore, although the model did not perfectly predict all patients who truly had a 60-day ED visit, the PPV was largely improved compared to the baseline prevalence of risk. Additionally, the choice of modeling approach should be determined early on in the model development process. In this study, random forest and logistic regression models were both tested in the prototype phase. While the random forest model demonstrated slightly better performance than the logistic regression model (Supplementary Table 1), the logistic regression model was chosen for the prospective evaluation because it was better suited to the use case needs (i.e., easily interpretable and communicated to all users, including clinical partners involved in its development). Any modeling choice is accompanied by tradeoffs. In using a logistic regression, our model had high transparency and parsimony, but slightly lower performance. This could be because a logistic regression does not automatically capture interactions amongst features as a tree-based model can. Such tradeoffs between model performance, interpretability, and feature interactions and exhaustiveness should be considered in the context of the use case and different models should be tested prior to carrying out prospective evaluations using the final model.
There are few published articles that present a standardized framework or guidance for developing ML-based tools in a healthcare setting11,13,14, and we were unable to find any that included a prospective evaluation of the framework in a real-world use case study. Our proposed framework has several areas of strength. It is comprehensive, standardized, and versatile in its adaptability to various diseases and healthcare data types (e.g., EHR, claims, or registry data). A key strength is the pre-specification of the ML model design and evaluation plan. Additionally, the framework differentiates between retrospective and prospective validation steps, and it highlights the utility of a pilot program prior to full implementation, as demonstrated through our use case. Importantly, the framework emphasizes the necessity of bringing together a multidisciplinary team to develop the ML-tool collaboratively and iteratively. Integrating technical knowledge with clinical knowledge helps to break down information silos between team members. This approach improves human explainability of the ML model, so that all involved have a strong understanding of the tool in order to best serve patients, which is critical for early acceptance and broad adoption.
There are limitations to consider regarding the use of any ML tool in a healthcare setting14. First, ML models are not perfect tools for predicting the risk of an adverse clinical event. Accordingly, they should be used to support clinician assessment, not to replace it, and transparency is critical. Additionally, care would need to be taken to preserve model fairness and interpretability. In our use case, the PPV of 22% in the prospective study leaves an opportunity for improvement of the model's predictive performance. Potential directions for improvement of model performance include introducing additional clinical features to the model such as those extracted from unstructured clinical text, and exploring more sophisticated, nonlinear ML techniques. Second, an ML-based clinical tool may not include all possible clinical factors that may be predictive of the defined clinical event. Sensitivity analysis should be conducted during the retrospective evaluation to determine whether the inclusion and exclusion of certain factors affect the model performance. Third, an inherent limitation of using EHR-derived RWD to develop an ML-based clinical tool is the potential for missingness in the dataset; data not documented in the dataset cannot be taken into consideration by the risk model. Fourth, the model performance and real-world utility may vary with the time period of assessment. Our exploratory analysis stratified by COVID response date suggests that different factors (or a different impact of existing model factors) may improve model performance for near-term ED visits in the COVID setting. These performance changes can sometimes be mitigated through rigorous monitoring and model recalibration or retraining15,16; this updating could be more difficult in the face of large, sudden changes such as a pandemic. Fifth, there might be a trade-off between computational efficiency, practical usability, and clinical relevance. The ML-based tool demonstrated in this use case was built using retrospective encounter-level data and applied to weekly encounter-level data for computational efficiency and workflow fluency during clinical practice. However, the tool was prospectively evaluated using patient-level data for model validation and clinical impact on patients. Therefore, it is important to evaluate the ML-based tool from various aspects throughout the whole process. Finally, while the framework outlined in this paper is generalizable, the resulting algorithm may not be. Specific analyses and data used to build the model and analyze the performance should be localized to the specific healthcare setting in which it will be implemented. For instance, this model was trained using only data from patients from the HCI, and is thus applicable prospectively to that setting. Should this framework be used to create solutions for other patient populations, the steps of the framework should be repeated using target data from the relevant population and any models should be customized to meet the unique and specific objectives of the healthcare system.
In conclusion, we have developed a general framework to evaluate and deploy an ML-based clinical tool that supports a clinician’s independent assessment of patient risk and applied it in a real-world oncology setting retrospectively and prospectively. Future additional applications of the framework should be explored and can help to inform and refine approaches to developing ML tools for prospective use in healthcare settings.
Methods
In the following sections, the steps of the proposed ML-based clinical tool evaluation framework are described, followed by the methods for application of the framework to a healthcare system quality improvement use case at a large cancer center.
ML-based clinical tool evaluation general framework steps
The ML-tool evaluation general framework was developed by a multidisciplinary team of software engineers, data scientists, clinicians, researchers, administrators, and managers. Each step of the framework along with considerations is outlined in Table 5. In order to obtain value from the framework and to prevent wasted efforts, the following assumptions should be considered: (1) pursuit of the defined clinical quality improvement goal will meaningfully enhance patient care while maintaining or improving the efficiency and sustainability of healthcare delivery; (2) prediction of the defined clinical event enables care teams to make progress towards this goal; and (3) the ML-based clinical tool can be implemented in a way that improves technology utilization rather than contributing to provider burn-out.
Application of the framework to an oncology use case
To demonstrate the steps described in the general framework, we applied it to a real-world oncology setting. Together, the Huntsman Cancer Institute and Flatiron Health evaluated whether a Flatiron Health-developed supplemental ML-based clinical tool and monitoring framework could support the clinical program staff at the Huntsman Cancer Institute to enhance identification of potentially eligible patients for Huntsman at Home, a program providing acute level “hospital at home” care along with palliative and hospice services17. We followed best practices for model transparency and validation and the minimum information about clinical artificial intelligence modeling (MI-CLAIM) checklist is provided in Supplementary Table 218,19. This study complies with all relevant ethical regulations. The study protocol was submitted to the University of Utah Institutional Review Board (IRB_00127233), which determined that this quality improvement project did not meet the definitions of Human Subjects Research according to Federal regulations and therefore IRB oversight was not required or necessary for the study.
Use case step 1: Define healthcare system quality improvement goal met by predicting clinical event
A key healthcare system quality improvement goal for the Huntsman Cancer Institute is to reduce emergency department (ED) and hospital utilization for patients with cancer. To make progress towards this goal, the Huntsman Cancer Institute established the home-based Huntsman at Home program in 2018, which provides acute-level care to patients with cancer for conditions that commonly require ED evaluation and/or rehospitalization, such as poorly controlled symptoms, pain, dehydration, or infections. Episodic palliative or supportive care visits and/or end of life hospice care are also provided. The intent of this suite of comprehensive services is to improve patient quality of life, lengthen time at home, reduce avoidable ED visits and hospitalization stays, and improve family caregiver well-being17,20. Patients considered for enrollment to the Huntsman at Home program are identified by two pathways, direct clinician referral or as part of a hospital discharge plan either at the end of a stay or as part of an early discharge pathway. Since a large volume of unplanned hospitalizations occur through the ED, Huntsman at Home was interested in proactively identifying patients who were likely to have a near-term ED visit (60-day ED visit).
After aligning on key goals outlined in Step 1 of the framework, we were able to state the following: If the Huntsman at Home team anticipates that a patient who is being treated for cancer at Huntsman Cancer Institute may have a near-term ED visit, they will enroll the patient in Huntsman at Home, which will improve the patient experience and reduce the total cost of care.
Use case step 2: Build or acquire ML-based clinical tool that predicts defined clinical event
The ML-based clinical tool development was led by a multidisciplinary team at Flatiron Health. The probability of near-term ED visit was estimated based on demographic and clinical characteristics, using a logistic regression model with L2 regularization. We also tested model performance in the retrospective analysis (described in Step 3 below) using a random forest model in order to compare performance of the two models (Supplementary Table 1). The outcome of interest was narrowly defined in service of the broader quality improvement objective in Step 1. In defining the outcome of interest, care was taken to avoid any measurement error that could come from using proxies, as discussed in work by Mullainathan and Obermeyer21. Thus, near-term ED visits captured directly through the EHR were chosen as the model’s outcome of interest. In addition, this step presents as an opportunity to assess what qualitative factors such as model explainability, transparency, and fairness should be considered.
Demographic and clinical features were pre-determined based on Flatiron Health oncology data expertise and informed by Huntsman at Home clinical experience, and the final feature list included in the model was determined based on the data quality and feature importance (Supplementary Table 3). Demographic features included gender, race/ethnicity, and history of Medicaid enrollment. Clinical features included cancer diagnosis, comorbidities, lab test results for albumin, bilirubin, hematocrit and hemoglobin, weight loss, recency and frequency of prior visits, and prior medication orders and clinic administrations.
The model was trained on all patient encounters with the University of Utah Health System (e.g., office visits, diagnostic visits, emergency visits) from 01-01-2016 to 12-31-2018 using cross-validation to select model hyperparameters (Supplementary Material Notes). Prediction estimates were calculated only for patients who met the inclusion/exclusion criteria (Supplementary Table 4), (based on Huntsman at Home program guidelines) at the time of the index encounter for which a prediction was made. The model produces risk scores for visits, which represent the predicted probability of a subsequent ED visit by that patient within 60 days of the visit. A separate validation set of patients was used to assess initial model performance and to set a risk threshold such that visits with risk scores above it would be classified as “high risk”. A pragmatic risk threshold was chosen tailored to the Huntsman at Home program size and patient review capacity. A predicted probability of 0.20 was selected so that 10% of all visits were classified as high risk in order to make the manual effort associated with weekly review of predictions feasible for clinical experts in the Huntsman at Home program. The same risk threshold of 0.20 was applied in the prospective validation study (described below).
The model was trained on individual encounters, and thus included multiple observations per patient. This methodological decision was driven by the use case in which repeated risk assessments were needed over time because the patients' risk factors may change relatively quickly. Since the goal of the ML-based clinical tool was to predict an updated risk score for each eligible patient following any clinical interaction with the University of Utah Health System, we developed the model to capture a diversity of patient states along their care journeys. In other words, training a model on all encounters (e.g., office visits, diagnostic visits, emergency visits) meant that the team was able to learn from information that was gained between encounters (e.g., an updated lab result). This choice also necessitated the use of temporal features (e.g., patient had a visit to the ED in the 30 days before the current encounter). Including temporal features allowed the model to account for the recency of certain information, which was important because we made new predictions for each patient encounter over time rather than one prediction for all time. In determining what features were best suited to being coded temporally and what time bins to create for them, we consulted with clinical experts at Flatiron Health and HCI and performed a literature review. For example, we created binary features for albumin lab test results being abnormal in each of the recent windows (e.g., 5, 30, 90 days leading up to the encounter) in order to account for both very recent changes (e.g., what happened in the last 5 days before an encounter) that could elevate patient risk, as well as more long term features (e.g., 90 days) that could set a temporal baseline.
Use case step 3: Conduct retrospective evaluation of ML-based clinical tool
After determining that use of a logistic regression model with L2 regularization best suited the use case needs, we retrospectively evaluated the ML-based clinical tool on an independent test set of 28,433 samples representing encounters at the University of Utah Health System from 03-01-2019 to 09-30-2019 for the 2,385 patients who met the inclusion/exclusion criteria (Supplementary Table 4). Testing on data not used to train the model is common in ML to prevent model overfitting. Baseline metrics were calculated to describe the test set and included age (median [minimum–maximum; IQR]), gender (female, male), ethnicity (Hispanic/Latino), race (White, Asian, Black, Other), documented Medicaid payer prior to the index encounter date, and cancer diagnosis (categorical with categories based on ICD codes mapped to cancer groupings as per standard Flatiron Health data processing procedures22,23). An exploratory post-hoc analysis assessed model accuracy with standard metrics, as defined in Table 6.
Use case step 4: Conduct bias assessment of ML-based clinical tool
Different definitions of fairness within the context of racial/ethnic bias and ML have been proposed, including anti-classification, classification parity of specific metrics, and calibration (Corbett-Davies and Goel)24. Calibration measures the gap between predicted risk for an outcome and observed outcomes for a given group across different levels of predicted risk. An ideally fair model from this perspective would be one where this gap is non-existent for all groups of interest. Based on precedent in the literature for problems of risk prediction, such as the analysis in Obermeyer et al.25 we mitigated bias through assessment of calibration fairness. To assess calibration fairness, we produced calibration curves that compared, by group, the predicted risk of utilizing the ED against the observed rate of doing so. Using the same test set that was used to evaluate model performance, we also calculated a summary statistic for each group, a “calibration factor” that measured the difference between the mean predicted risk and the mean observed outcome for a group26. Models satisfying calibration fairness should have a calibration factor of zero (Supplementary Material Notes). We reviewed overall calibration curves for indications of subtler forms of bias that might not be captured by the calibration factor. In addition to avoiding measurement error and assessing the model for calibration fairness, we minimized other risks of bias by choosing transparent and interpretable modeling techniques (e.g., logistic regression). This was done to preclude other unforeseeable harm that could be a by-product of black box techniques27.
Use case step 5: Conduct prospective evaluation of ML-based clinical tool
Given the additional goal of evaluating impact on patient outcomes prospectively in the clinical setting, we assessed model performance at the patient level. A prospective quality improvement study of 1236 patients was conducted to evaluate the accuracy and the usability of this ML-based model among patients with a visit to the Huntsman Cancer Institute between 01-04-2020 and 02-07-2020. Otherwise applying the same inclusion/exclusion criteria as the retrospective portion of the study, patients were randomly selected, in a 1:1 ratio, to be in either the independent “hold out” sub-cohort to assess model accuracy, or the “deliverable” sub-cohort to assess real-world accuracy (Fig. 2). Patient-level index encounter was defined as the encounter that corresponded to the first high-risk classification for high-risk patients and first encounter for low-risk patients to ensure that each observation was statistically independent and capable of yielding valid, clinically meaningful inferences. Each patient was followed up for 60 days after the index encounter. We assessed model accuracy in the “hold-out” cohort using the metrics summarized in Table 6.
ED emergency department. aIf the patient’s predicted probability of risk is greater than the classification threshold the patient will be classified as “high risk”, otherwise the patient will be classified as “low risk”. The classification threshold is selected using retrospective data prior to the start of the prospective evaluation.
To evaluate prediction accuracy of the risk stratification model, we compared patients with predicted risk classification against their observed outcomes of ED visit within 60 days of the index encounter. For real-world accuracy, the clinical expert reviewed the charts of patients who were identified as high-risk by the risk stratification model and were in the “deliverable” cohort to classify their eligibility for the Huntsman at Home program based on Huntsman at Home standard protocol. We estimated the risk model’s clinical accuracy by the proportion of eligible patients among all the identified high-risk patients in the “deliverable” cohort. Baseline characteristics were described for both the “hold-out” and “deliverable” sub-cohorts to evaluate the success of randomization.
Use case step 6: Embed and monitor ML-based clinical tool in clinical workflow
Based on successful outcomes from preceding steps, the Huntsman at Home team is operationalizing the ML-based clinical tool, which will be used to supplement traditional referral pathways (e.g., clinician referrals) to the Huntsman at Home program by displaying relevant information. Once a patient is surfaced for evaluation (from any source), all decisions for Huntsman at Home enrollment are made by the clinical care team, based on standardized clinical evaluation processes, and not solely on the risk prediction result from the tool. Transparency was ensured through clear documentation and the clinician could independently review the basis of the risk prediction. To support the ML-based clinical tool in production, Flatiron Health developed a solution that continuously monitors the ML-based clinical tool predictions to ensure that the algorithms developed on retrospective data do not lead to unanticipated outcomes in a prospective real-world setting.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
The de-identified data that support the findings of this study may be made available upon request, and are subject to a license agreement; interested researchers should contact <DataAccess@flatiron.com> to determine licensing terms.
Code availability
The custom computer code used to generate the results of this study is available to the journal editors and study peer reviewers upon request for purposes of validating the results of the study. Because aspects of the custom computer code used to generate results reported in the paper is the proprietary confidential information of Flatiron Health, it cannot be made publicly available.
References
Rajkomar, A., Dean, J. & Kohane, I. Machine learning in medicine. N. Engl. J. Med. 380, 1347–1358 (2019).
Goldstein, B. A., Navar, A. M., Pencina, M. J. & Ioannidis, J. P. Opportunities and challenges in developing risk prediction models with electronic health records data: a systematic review. J. Am. Med. Inform. Assoc. 24, 198–208 (2017).
Pella, A. et al. Use of machine learning methods for prediction of acute toxicity in organs at risk following prostate radiotherapy. Med. Phys. 38, 2859–2867 (2011).
Frizzell, J. D. et al. Prediction of 30-day all-cause readmissions in patients hospitalized for heart failure: Comparison of machine learning and other statistical approaches. JAMA Cardiol. 2, 204–209 (2017).
Hong, J. C., Niedzwiecki, D., Palta, M. & Tenenbaum, J. D. Predicting emergency visits and hospital admissions during radiation and chemoradiation: an internally validated pretreatment machine learning algorithm. JCO Clin. Cancer Inform. 2, 1–11 (2018).
Morawski, K., Dvorkis, Y. & Monsen, C. B. Predicting hospitalizations from electronic health record data. Am. J. Manag. Care. 26, e7–e13 (2020).
Sahni, N., Simon, G. & Arora, R. Development and validation of machine learning models for prediction of 1-year mortality utilizing electronic medical record data available at the end of hospitalization in multicondition patients: a proof-of-concept study. J. Gen. Intern. Med. 33, 921–928 (2018).
Shahi, N. et al. Decision-making in pediatric blunt solid organ injury: a deep learning approach to predict massive transfusion, need for operative management, and mortality risk. J. Pediatr. Surg. 56, 379–384 (2021).
Lee, C. et al. Application of a novel machine learning framework for predicting non-metastatic prostate cancer-specific mortality in men using the surveillance, epidemiology, and end results (SEER) database. Lancet Digit. Health 3, e158–e165 (2021).
Ben-Israel, D. et al. The impact of machine learning on patient care: a systematic review. Artif. Intell. Med. https://doi.org/10.1016/j.artmed.2019.101785 (2020).
Mateen, B. A., Liley, J., Denniston, A. K., Holmes, C. C. & Vollmer, S. J. Improving the quality of machine learning in health applications and clinical research. Nat. Mach. Intell. 2, 554–556 (2020).
WhiteHouse.Gov. Notice on the continuation of the national emergency concerning the coronavirus disease 2019 (COVID-19) pandemic. https://www.whitehouse.gov/briefing-room/presidential-actions/2021/02/24/notice-on-the-continuation-of-the-national-emergency-concerning-the-coronavirus-disease-2019-covid-19-pandemic/ (2021).
Chen, P. C., Liu, Y. & Peng, L. How to develop machine learning models for healthcare. Nat. Mater. 18, 410–414 (2019).
Vollmer, S. et al. Machine learning and artificial intelligence research for patient benefit: 20 critical questions on transparency, replicability, ethics, and effectiveness. BMJ. https://doi.org/10.1136/bmj.l6927 (2020).
Davis, S. E. et al. A nonparametric updating method to correct clinical prediction model drift. J. Am. Med. Inform. Assoc. 26, 1448–1457 (2019).
Su, T. L., Jaki, T., Hickey, G. L., Buchan, I. & Sperrin, M. A review of statistical updating methods for clinical prediction models. Stat. Methods Med. Res. 27, 185–197 (2018).
Mooney, K. et al. Evaluation of oncology hospital at home: Unplanned health care utilization and costs in the huntsman at home real-world trial. JCO 39, 2586–2593 (2021).
Eddy, D. M. et al. Model transparency and validation: A report of the ISPOR-SMDM modeling good research practices task force-7. Med. Decis. Mak. 32, 733–743 (2012).
Norgeot, B. et al. Minimum information about clinical artificial intelligence modeling: The MI-CLAIM checklist. Nat. Med. 26, 1320–1324 (2020).
Mooney, K. et al. The oncology hospital at home: Health care utilization outcomes from the huntsman at home trial. JCO 38, 7000 (2020).
Mullainathan, S. & Obermeyer, Z. Does machine learning automate moral hazard and error? Am. Econ. Rev. 107, 476–480 (2017).
Birnbaum, B. et al. Model-assisted cohort selection with bias analysis for generating large-scale cohorts from the EHR for oncology research. Preprint at https://arxiv.org/abs/2001.09765 (2020).
Ma, X., Long, L., Moon, S., Adamson, B. J. S. & Baxi, S. S. Comparison of population characteristics in real-world clinical oncology databases in the US: Flatiron health, SEER, and NPCR. Preprint at https://www.medrxiv.org/content/10.1101/2020.03.16.20037143v2 (2020).
Corbett-Davis, S. & Goel, S. The measure and mismeasure of fairness: a critical review of fair machine learning. Preprint at https://arxiv.org/abs/1808.00023 (2018).
Obermeyer, Z., Powers, B., Vogeli, C. & Mullainathan, S. Dissecting racial bias in an algorithm used to manage the health of populations. Science 366, 447 (2019).
Van Calster, B., McLernon, D. J., van Smeden, M., Wynants, L. & Topic Group ‘Evaluating diagnostic tests and prediction models’ of the STRATOS initiative. Calibration: the Achilles heel of predictive analytics. BMC Med. 17, 230 (2019).
Bathaee, Y. The artificial intelligence black box and the failure of intent and causation. Harv. J. Law Technol. 31, 890–938 (2018).
Acknowledgements
The authors would like to acknowledge and thank Shannon Shepherd, MS, FNP-BC for participation in the clinical validation of the machine learning-based clinical tool. They would also like to thank Laura Koontz for review and critical input of this paper, and Cody Patton (Flatiron Health, Inc.) and Hannah Gilham (Flatiron Health, Inc.) for publication management and writing support. This study was sponsored by Flatiron Health Inc., which is an independent subsidiary of the Roche Group. L.C.’s contribution was supported by NIH NINR T32NR013456. Previous presentation: A portion of this study was presented at the 2020 ASCO Quality Care Symposium.
Author information
Authors and Affiliations
Contributions
A.O. contributed to study design (literature, background search), data collection, data analysis (including creation of figures/tables), data interpretation, and writing and review of paper. X.W. contributed to study design (literature, background search), data analysis (including creation of figures/tables), data interpretation, and writing and review of paper. P.S. contributed to study design (literature, background search), data collection, data interpretation, and writing and review of paper. A.S.R. contributed to study design (literature, background search), data analysis, data interpretation, and writing and review of paper. S.L. contributed to study design (literature, background search), data analysis (including creation of figures/tables), data interpretation, and writing and review of paper. B.A. contributed to study design (literature, background search), data analysis (including creation of figures/tables), data interpretation, and writing and review of paper. R.A.M. contributed to study design (literature, background search), data analysis, data interpretation, and writing and review of paper. L.C. contributed to study design (literature, background search), data collection, data interpretation, and writing and review of paper. K.M. contributed to study design (literature, background search), data collection, data interpretation, and writing and review of paper. K.T. contributed to study design (literature, background search), data collection, data interpretation, and writing and review of paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing non-financial interests but the following competing financial interests: A.O., X.W., P.S., A.S.R., S.L., B.A., and R.A.M. report employment at Flatiron Health, Inc., which is an independent subsidiary of the Roche Group. A.O., X.W., P.S., A.S.R., S.L., B.A., and R.A.M. also report stock ownership in Roche. A.O., P.S., R.A.M. also report equity ownership in Flatiron Health, Inc. L.C. reports employment at the University of North Carolina-Chapel Hill, Lineberger Cancer Institute. K.M. reports employment at the College of Nursing, University of Utah. K.T. reports employment at the Huntsman Cancer Institute, University of Utah.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Coombs, L., Orlando, A., Wang, X. et al. A machine learning framework supporting prospective clinical decisions applied to risk prediction in oncology. npj Digit. Med. 5, 117 (2022). https://doi.org/10.1038/s41746-022-00660-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41746-022-00660-3