Abstract
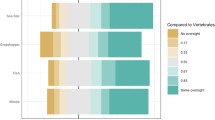
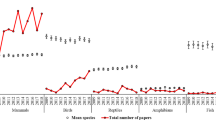
This national survey aimed to identify how biomedical researchers using vertebrate animals viewed issues of significance for translational science, including oversight and public engagement, and to analyze how researcher characteristics and animal model choice correlate with those views. Responses from 1,187 researchers showed awareness of, and concerns about, problems of translation, reproducibility and rigor. Surveyed scientists were nevertheless optimistic about the value of animal studies, were favorable about research oversight and reported openness with non-scientists in discussing their animal work. Differences in survey responses among researchers also point to diverse perspectives within the animal research community on these matters. Most significant was variability associated with the primary type of animal that surveyed scientists used in their work. Other significant divergence in opinion appeared on the basis of professional role factors, including the type of degree held, workplace setting, type of funding, experience on an institutional animal care and use committee and personal demographic characteristics of age and gender.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
We are sorry, but there is no personal subscription option available for your country.
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Processed survey data reported on in this article and information about the related analyses are deposited in UNC Libraries Digital Repository at https://cdr.lib.unc.edu/concern/data_sets/k643bb08t?locale=en. Data have been redacted to protect participant privacy. Researchers requiring access to removed data may contact R.L.W. at the provided email address.
References
Ioannidis, J. P. A. Why most published research findings are false. PLoS Med. 2, e124 (2005).
Baker, M. 1,500 scientists lift the lid on reproducibility. Nat. News 533, 452 (2016).
Munafò, M. R. et al. A manifesto for reproducible science. Nat. Hum. Behav. 1, 0021 (2017).
Begley, C. G. & Ellis, L. M. Drug development: raise standards for preclinical cancer research. Nature 483, 531–533 (2012).
Prinz, F., Schlange, T. & Asadullah, K. Believe it or not: how much can we rely on published data on potential drug targets? Nat. Rev. Drug Discov. 10, 712 (2011).
Seok, J. et al. Genomic responses in mouse models poorly mimic human inflammatory diseases. Proc. Natl Acad. Sci. USA 110, 3507–3512 (2013).
Mogil, J. S. Animal models of pain: progress and challenges. Nat. Rev. Neurosci. 10, 283–294 (2009).
Mak, I. W. Y., Evaniew, N. & Ghert, M. Lost in translation: animal models and clinical trials in cancer treatment. Am. J. Transl. Res. 6, 114–118 (2014).
Dawson, T. M., Golde, T. E. & Lagier-Tourenne, C. Animal models of neurodegenerative diseases. Nat. Neurosci. 21, 1370–1379 (2018).
van der Worp, H. B. et al. Can animal models of disease reliably inform human studies? PLoS Med. 7, e1000245 (2010).
Scott, S. et al. Design, power, and interpretation of studies in the standard murine model of ALS. Amyotroph. Lateral Scler. 9, 4–15 (2008).
Garner, J. P., Gaskill, B. N., Weber, E. M., Ahloy-Dallaire, J. & Pritchett-Corning, K. R. Introducing Therioepistemology: the study of how knowledge is gained from animal research. Lab Anim. (NY) 46, 103–113 (2017).
Muhlhausler, B. S., Bloomfield, F. H. & Gillman, M. W. Whole animal experiments should be more like human randomized controlled trials. PLoS Biol. 11, e1001481 (2013).
Peers, I. S., Ceuppens, P. R. & Harbron, C. In search of preclinical robustness. Nat. Rev. Drug Discov. 11, 733–734 (2012).
Kilkenny, C. et al. Survey of the quality of experimental design, statistical analysis and reporting of research using animals. PLoS ONE 4, e7824 (2009).
Beynen, A. C., Gärtner, K. & van Zutphen, L. F. M. In Principles of Laboratory Animal Science (eds. Zutphen, L. F. M., Baumans, V. & Beynen, A.C.) 103–110 (Elsevier, 2001).
Barbee, R. W. & Turner, P. V. Incorporating laboratory animal science into responsible biomedical research. ILAR J. 60, 9–16 (2019).
Kilkenny, C., Browne, W. J., Cuthill, I. C., Emerson, M. & Altman, D. G. Improving bioscience research reporting: the ARRIVE guidelines for reporting animal research. PLoS Biol. 8, e1000412 (2010).
Percie du Sert, N. et al. The ARRIVE Guidelines 2.0: updated guidelines for reporting animal research. PLoS Biol. 18, e3000410 (2020).
National Research Council. Guide for the Care and Use of Laboratory Animals 8th edn (The National Academies Press, 2011).
Russell, W. M. S. & Burch, R. L. The Principles of Humane Experimental Technique. (Universities Federation for Animal Welfare, 1959).
LaFollette, H. & Shanks, N. Brute Science: Dilemmas of Animal Experimentation (Routledge, 2020).
Pound, P., Ebrahim, S., Sandercock, P., Bracken, M. B. & Roberts, I. Where is the evidence that animal research benefits humans? Br. Med. J. 328, 514–517 (2004).
Greek, C. R. & Greek, J. S. Sacred Cows and Golden Geese: The Human Cost of Experiments on Animals. (A&C Black, 2000).
Jones, J. & Saad, L. Gallup Poll Social Series: Values and Beliefs. Available at https://www.gallup.com/201200/gallup-poll-social-series-work.aspx (2019).
Strauss, M. Americans Are Divided Over the Use of Animals in Scientific Research. Available at https://www.pewresearch.org/fact-tank/2018/08/16/americans-are-divided-over-the-use-of-animals-in-scientific-research/ (2018).
Lankau, E. W., Turner, P. V., Mullan, R. J. & Galland, G. G. Use of nonhuman primates in research in North America. J. Am. Assoc. Lab. Anim. Sci. 53, 278–282 (2014).
Magden, E. R., Mansfield, K. G., Simmons, J. H. & Abee, C. R. in Laboratory Animal Medicine 3rd edn (eds. Fox, J. G, Anderson, L. C., Otto, G. M. Pritchett-Corning, K. R. & Whary, M. T.) 771–930 (Academic Press, 2015).
Baker, K. C. & Dettmer, A. M. The well-being of laboratory non-human primates. Am. J. Primatol. 79, e22520 (2017).
Colman, R. J. et al. Marmosets: welfare, ethical use, and IACUC/regulatory considerations. ILAR J. https://doi.org/10.1093/ilar/ilab003 (2021).
Rader, K. Making Mice: Standardizing Animals for American Biomedical Research, 1900–1955 (Princeton University Press, 2004).
Perlman, R. L. Mouse models of human disease: an evolutionary perspective. Evol. Med. Public Health 1, 170–176 (2016).
Courtine, G. et al. Can experiments in nonhuman primates expedite the translation of treatments for spinal cord injury in humans? Nat. Med. 13, 561–566 (2007).
Colman, R. J. Non-human primates as a model for aging. Biochim. Biophys. Acta Mol. Basis Dis. 1864, 2733–2741 (2018).
Perrin, S. Preclinical research: make mouse studies work. Nat. News 507, 423–425 (2014).
Richter, S. H., Garner, J. P. & Würbel, H. Environmental standardization: cure or cause of poor reproducibility in animal experiments. Nat. Methods 6, 257–261 (2009).
Voelkl, B. et al. Reproducibility of animal research in light of biological variation. Nat. Rev. Neurosci. 21, 384–393 (2020).
Laukens, D., Brinkman, B. M., Raes, J., De Vos, M. & Vandenabeele, P. Heterogeneity of the gut microbiome in mice: guidelines for optimizing experimental design. FEMS Microbiol. Rev. 40, 117–132 (2016).
Willmann, R. et al. Enhancing translation: guidelines for standard pre-clinical experiments in mdx mice. Neuromuscul. Disordd 22, 43–49 (2012).
Eisner, D. A. Reproducibility of science: fraud, impact factors and carelessness. J. Mol. Cell. Cardiol. 114, 364–368 (2018).
Fang, F. C., Bennett, J. W. & Casadevall, A. Males are overrepresented among life science researchers committing scientific misconduct. Mbio 4, e00640–12 (2013).
Everitt, J. I. & Berridge, B. R. The role of the IACUC in the design and conduct of animal experiments that contribute to translational success. ILAR J. 58, 129–134 (2017).
Pritt, S., McNulty, J. A., Greene, M., Light, S. & Brown, M. Decreasing institutionally imposed regulatory burden for animal research. Lab Anim. (NY) 45, 297–300 (2016).
National Research Council. Toxicity Testing in the 21st Century: A Vision and a Strategy. (National Academies Press, 2007).
Festing, S. & Wilkinson, R. The ethics of animal research: talking point on the use of animals in scientific research. EMBO Rep. 8, 526–530 (2007).
DeGrazia, D. The ethics of animal research: what are the prospects for agreement? Camb. Q. Healthc. Ethics 8, 23–34 (1999).
Wadman, M. Hundreds of US scientists urge more transparency in animal research. Available at https://www.sciencemag.org/news/2018/06/hundreds-us-scientists-urge-more-transparency-animal-research (2018).
Basel Declaration Society. Basel Declaration. A Call for More Trust, Transparency and Communication on Animal Research. Available at https://www.basel-declaration.org/basel-declaration/ (2010).
MacArthur Clark, J., Clifford, P., Jarrett, W. & Pekow, C. Communicating about animal research with the public. ILAR J. 60, 34–42 (2019).
Waltz, M., Saylor, K. W., Fisher, J. A. & Walker, R. L. Biomedical researchers’ perceptions of the NIH’s Sex as a Biological Variable policy for animal research: results from a US national survey. J. Womens Health (Larchmt.) 30, 348–354 (2021).
Acknowledgements
The authors thank the following individuals associated with the University of North Carolina at Chapel Hill: Ryan Joseph Kramer, Molly Green, Lisa McManus and Megan Wood for research assistance; Julianne Kalbaugh for programming and administering the survey; and Teresa Edwards for input on the survey instrument. We thank those individual researchers who piloted the survey instrument and the additional members of our research team who offered feedback on the survey questions. Research reported in this article was supported under a grant from the National Institutes of Health, National Institute of General Medical Sciences award number R01GM099952, ‘Healthy Volunteers as Model Organisms: Comparative Research Ethics and Policy for Phase I Trials’ (principal investigators: J.A.F. and R.L.W.).
Author information
Authors and Affiliations
Contributions
J.A.F. and R.L.W. designed the survey. All authors contributed to the analysis plans and interpretation of the findings, and K.W.S. conducted the statistical analysis. All authors contributed to the writing and revising of the work for intellectual content, with R.L.W. taking the lead in drafting. All authors gave final approval of the submitted version and agreed to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review information
Lab Animal thanks Malcolm MacLeod and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Survey Text and Supplementary Institutions Searched
Rights and permissions
About this article
Cite this article
Walker, R.L., Saylor, K.W., Waltz, M. et al. Translational science: a survey of US biomedical researchers’ perspectives and practices. Lab Anim 51, 22–35 (2022). https://doi.org/10.1038/s41684-021-00890-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41684-021-00890-0