Abstract
Hepatocellular carcinoma (HCC) is the most common primary malignant liver tumor and a leading cause of cancer-related deaths worldwide. However, current diagnostic tools are often invasive and technically limited. In the last decade, non-invasive liquid biopsies have transformed the field of clinical oncology, showcasing the potential of various liquid-biopsy derived analytes, including extracellular vesicles (EVs), to diagnose and monitor HCC progression and metastatic spreading, serving as promising novel biomarkers. A prospective single-center cohort study including 37 HCC patients and 20 patients with non-malignant liver disease (NMLD), as a control group, was conducted. Serum EVs of both groups were analyzed before and after liver surgery. The study utilized microbead-based magnetic particle sorting and flow cytometry to detect 37 characteristic surface proteins of EVs. Furthermore, HCC patients who experienced tumor recurrence (R-HCC) within 12 months after surgery were compared to HCC patients without recurrence (NR-HCC). EVs of R-HCC patients (n = 12/20) showed significantly lower levels of CD31 compared to EVs of NR-HCC patients (p = 0.0033). EVs of NMLD-group showed significantly higher expressions of CD41b than EVs of HCC group (p = 0.0286). The study determined significant short-term changes in CD19 dynamics in EVs of the NMLD-group, with preoperative values being significantly higher than postoperative values (p = 0.0065). This finding of our pilot study suggests EVs could play a role as potential targets for the development of diagnostic and therapeutic approaches for the early and non-invasive detection of HCC recurrence. Further, more in-depth analysis of the specific EV markers are needed to corroborate their potential role as diagnostic and therapeutic targets for HCC.
Similar content being viewed by others
Introduction
Hepatocellular carcinoma (HCC) is the most common primary liver cancer, with an increasing incidence and high recurrence rate, which negatively impacts prognosis and overall survival1. Currently, hepatic resection, radiofrequency ablation, and liver transplantation represent curative therapeutic options for HCC at early-stages. Recurrence rates of early-stage HCC remain high by 35% within the first year2 and resemble a crucial prognostic factor since early recurrence is associated with developing incurable disease3,4. The liver represents the most frequent site of HCC recurrence, lately reviewed by 66%2, leading to the question, whether therapeutic options can be optimized. Currently available diagnostics include radiographic differentiation of indeterminate or hazardous lesions, and alpha-fetoprotein level measurement, which remain limited regarding the analysis of tumor biology5.
Molecular profiling could lead to earlier detection of HCC recurrence at a stage, where curative resection and ablation modalities are amenable5. Hence, innovative early biomarkers to detect micro metastases are awaited to improve curative therapeutic options in early recurrence after HCC resection.
To optimize diagnosis and monitoring of cancer, non-invasive methods such as liquid biopsies have revolutionized the field of clinical oncology offering ease in tumor sampling and continuous monitoring by repeated sampling6. Thereby, tumor-derived extracellular vesicles (EVs) extracted from liquid biopsies are generally present in the blood circulation at early stages of the disease and persist across all disease stages, thus, represent promising potential biomarkers7,8,9. EVs are a heterogenous population of particles released from cells and found in biofluids that consist of lipid-bilayer membrane-coated vesicles10,11. EVs cargo consist of miRNAs, RNAs, lipids and proteins4. The composition can be altered due to external stimuli such as the blood-ph and hypoxia4. Regarding systemic and local tumor dynamics, it was recently shown, that hepatic tumors prepare a comforting microenvironment via release of EVs, initiate tumor angiogenesis and even might evade the immune cell recognition through EV mediated mechanisms12.
It has been shown that EVs play a crucial role in cellular communication and metastasis of HCC10,12 and could therefore enhance early recurrence diagnosis and thus optimize HCC treatment. As EVs are not only highly heterogeneous in molecular composition but also in their structure of surface proteins, the aims of this study was to characterize the dynamics of EV surface markers in recurrent and non-recurrent HCC patients up to 12 months after surgery. Patients with non-malignant liver disease (NMLD) were used as controls.
Material and methods
Patient characteristics and sample collection
The prospective single-center cohort study was conducted at the Johann Wolfgang Goethe University Frankfurt between 2016 and 2019. This study was approved by the ethics committee of the University of Frankfurt (approval number: 321/16). Informed consent forms were signed and obtained from all patients. All experiments were performed in accordance with relevant guidelines and regulations. Blood samples were collected from 36 patients with HCC and 20 patients with non-malignant liver disease (NMLD, control group) before and after surgery. Patients with HCC received segmental resection or hemihepatectomy. After the follow up period of up to 12 months, blood sample and recurrence analysis was taken from 20 HCC patients. Follow up was conducted during routine examination 6–12 months after the surgery. Overall, 16 patients were excluded after 12 months due to loss of follow up and incomplete data set. Out of these 20 patients, 12 (60%) patients were diagnosed with recurrent HCC after a median follow-up period of 12 months.
Isolation of extracellular vesicles (EVs)
Highly purified EVs were isolated by using EV Isolation Kit Pan Human (Miltenyi Biotec, Bergisch Gladbach, DE). EVs from each patient were analyzed separately. EV isolation was performed by positive selection using MicroBeads recognizing the tetraspanin proteins CD9, CD63, or CD81. The procedure was conducted as follows: 2 ml of Plasma from each sample was pre-cleared by serial centrifugation at 300× g for 10 min, 2000× g for 30 min, 10,000× g for 45 min. Supernatant was collected and transferred into a new tube after each centrifugation step. Next, 50 μL of EV Isolation MicroBeads was added to 2 ml of pre-cleared plasma and incubated for 1 h at room temperature. The labeled EVs are loaded onto a μ Column attached in the μMACS™ Separator. Columns were then washed 4 × with 200 µl isolation buffer and finally the magnetically labeled EVs were eluted by flushing the columns with 100 µl isolation buffer.
Multiplex surface marker analysis
Serum EV surface expression was analysed by a bead based multiplex EV analysis using MACSPlex Exosome Kit, human (Miltenyi Biotec, Bergisch Gladbach, DE). Detection of 37 EV surface epitopes (CD1c, CD2, CD3, CD4, CD8, CD9, CD11c, CD14, CD19, CD20, CD24, CD25, CD29, CD31, CD40, CD41b, CD42a, CD44, CD45, CD49e, CD56, CD62p, CD63, CD69, CD81, CD86, CD105, CD133.1, CD142, CD146, CD209, CD326, HLA-ABC, HLA-DR DP DQ, MCSP, ROR1 and SSEA-4), including two isotype controls was conducted according to manufacturer’s instructions. Isolated serum EV were stained as follows: 120 µl of each EV sample was incubated with 15 µl of MACSPlex exosomes capture beads overnight at room temperature using a tube rotator on permanent run (12 rpm). Samples were washed with 500 μL MACSPlex Buffer and centrifuged at room temperature at 3000 × g for 5 min. The supernatant was aspirated, 5 μL MACSPlex Exosome Detection Reagent CD9, CD63, and CD81 were added to each sample and incubated for 1 h at room temperature in a tube rotator (12 rpm). Thereafter, 500 μL MACSPlex Buffer was added to each sample and incubated for 15 min in a tube rotator at room temperature. Finally, EV-containing samples were centrifuged at 3000× g for 5 min and EV was collected by aspirating the supernatant, leaving about 150 μL in the tube. Samples were analysed by FACSCanto™ II.
(BD Bioscience). 120 µl isolation buffer was used as background control. The 39 bead populations can be distinguished by different fluorescence intensities detected in the FITC and PE channel.
Nanoparticle tracking analysis (NTA)
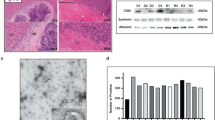
Isolated EVs were evaluated by NanoSight NS500 instrument (Malvern Panalytical, Malvern UK) equipped with a red laser (642 nm). The laser illuminated the Nanoparticles and their movement under Brownian motion was recorded for 30 s. The NanoSight particle tracking program (NTA 3.2) was then used to perform nanoparticle tracking analysis (NTA) on six videos. Captures settings were set to the following: camera level of 16, 1300 for the slider shutter, and 512 for the gain slider. The NTA-obtained size distribution profiles were averaged within each sample throughout the six videos, the distribution size profiles were then normalized to total nanoparticle concentrations. Every experiment was run at a 1:100 dilution ((Fig. 1A, Figure S1).
Characterization of EVs: (A) NTA analysis showing concentrations and size distribution of Evs isolated from serum of HCC (Pre and Post, n = 5 each)), NMLD patients (n = 5) and resuspension buffer (0.02 µM PBS) used for EVs extraction procedure. (B) Example of Protein expression of exosomes markers CD9 and CD63 by Wes protein simple. (C) Representative dotplot cluster distribution of 37 EVs markers including 2 controls. (D) Representative histograms of EVs stained with CD9 and CD63 detected by MACSPlex EV kit. EVs: Extracellular Vesicles.
Western blot analysis of lysed EVs
Wes Simple Western System, a capillary-based electrophoresis (ProteinSimple, Bio-Techne, Minneapolis, USA) was performed according to the manufacturer’s instruction (https://proteinsimple.com). The isolated EVs were directly lysed and identified using primary antibodies (CD9, clone D3H4P and CD63, clone E1W3T), Cell Signaling Technology Europe, Leiden, Netherlands) and (CD81, clone M38), Invitrogen, Thermo Fisher Scientific, Massachusetts, USA). All primary antibodies were diluted at 1:20. Anti-rabbit and anti-mouse detection module served as secondary antibodies. The resulting chemiluminescent signal was detected and analyzed by Compass software for (SW) Simple Western (Fig. 1B, Figure S2).
Flow cytometry
Measurement of 37 surface proteins was achieved using BD FACSCanto II and analysed by FACSDiva (Becton Dickinson, USA). The median APC fluorescence of each specific bead population obtained from control (buffer only) was subtracted from the signal intensities of the respective beads incubated in the samples. Surface protein expression was measured as fluorescence intensity, the corresponding control antibody was considered as measurement threshold and regarded protein expression below the threshold as negative (Fig. 1C/D, and Figure S3).
Statistical analysis
Statistical analysis was performed with SPSS (IBM SPSS Statistics, New York, USA) and GraphPad Prism (GraphPad Prism 8.0.0, San Diego, USA) for graph creation. All data showed asymmetric distribution. Statistical analysis included Mann Whitney U test for patient characteristics. Analysis of protein expression was conducted by Friedman test for dependent and Kruskal Wallis test for inter group comparisons plus Bonferroni post hoc test. Results were visualized by median and 95% confidence interval. A level of p < 0.05 was considered statistically significant.
Results
Patients characteristics
HCC and NMLD
The group of HCC patients had a significantly higher age (64.05 ± 10.20 year) than patients with NMLD (48.40 ± 14.13, p < 0.0001) and a significantly higher percentage of males (HCC 58.33; NMLD 15%; p < 0.0006, Table 1). Percentage of open surgery was comparable within the two groups (HCC 66.67%, NMLD 65%, Table 1).
HCC: recurrence and non-recurrence
In our study, 60% of patients were diagnosed with recurrent HCC following a median follow-up period of 12 months. When examining the recurrence rates after 1 year, we observed a range of numbers in the literature, spanning from 15%13 to 48.1%14. Notably, "early recurrence," defined as occurring within 2 years of resection, accounts for over 70% of tumor recurrences15. Our findings align with these patterns, which places the recurrence rate in our study within the expected range when compared to other investigations.
The age between patients with recurrence (R-HCC) and non-recurrence (NR-HCC) was comparable (R-HCC 61.88 ± 10.66; NR-HCC 67.31 ± 8.48). 50% of HCC patients had liver fibrosis (R-HCC 58.33%; NR-HCC 37.5%), 38.89% liver cirrhosis (R-HCC 25%; NR-HCC 62.5%) and 22.22% non-alcoholic steatohepatitis (NASH) (R-HCC 16.67%; NR-HCC 12.5%).
The median tumor volume at the time of resection for the HCC group was 187.71 ± 247.15 and showed comparable results between R-HCC (183.86 ± 143.97) and NR-HCC (211.70 ± 243.83) Similarly R Status was comparable between R-HCC (R0 = 66.67%) and NR-HCC (R = 87.5%). The AFP level before and after surgery was similar between the R-HCC and NR-HCC (p = ns, Table 1).
EV characteristics
Within the representative cohort of 5 patients per group (HCC preoperative/HCC postoperative and NMLD), EVs size distributions were comparable (Fig. 1A, Figure S1) in the NTA analysis. The concentration of particles in HCC preoperatively was : 2.08e + 009 (+ /− 1.27e + 008) particles/ml, and in HCC postoperatively: 2.35e + 009 + /− 7.46e + 007 particles/ml whereas plasma of NMLD group had a higher concentration of : 4.35e + 009 + /− 2.32e + 008 particles/ml. The mean diameter was in HCC preoperatively 165.2 + /− 23.2 nm and for HCC postoperatively 186.3 + /− 4.3 nm. EVs of the NMLD group had the mean diameter of 155.8 + /− 3.8 nm. We confirmed CD9 and CD63 expression as EVs markers in the groups of our study by using Western Blot for analysis of lysed EVs (Fig. 1B, Figure S2).
EV dynamics in NMLD patients
In the NMLD patients, the expression of CD19 was significantly upregulated postoperatively (p = 0.0065; Fig. 2). Additionally, the NMLD patients there was a trend of increased protein expressions of MSCP and CD62P postoperatively compared to preoperatively (p = ns).
Surface protein expression of EVs in the NMLD group (A) for preoperative and postoperative probes in blue and red, respectively. CD19 expression is postoperatively significantly increased in the NMLD group (p = 0.0065). (B) CD19 expression in EVs of NMLD pre- and postoperatively (**p < 0.01). NMLD: Non-malignant liver disease.
EV dynamics in HCC patients
The short-term expression of EV surface proteins in the HCC group remained stable from pre- to postoperative with no significant differences. However, protein expressions of MSCP and CD62P were decreased pre- to postoperative with no significant differences (Fig. 3). Regarding the long-term comparison of the R-HCC and NR-HCC group (n = 20), CD31 expression was significantly higher in the NR-HCC group than in the R-HCC group up to 12 months after surgery (p = 0.0031; Fig. 4). Interestingly, the NR-HCC group showed a tendency of higher expressions of CD9, CD62P, CD42a and CD29 up to 12 months postoperativly compared to the R-HCC group, but these differences were not statistically significant (Fig. 4).
(A) Development of EV expression in the R-HCC and the NR-HCC group up to 12 months postoperative. CD31 expression was significantly higher in the NR-HCC group than in the R-HCC group up to 12 months after surgery (p = 0.0031). (B) CD31 expression in EVs of HCC, NR-HCC and R-HCC (**p < 0.01). HCC, Hepatocellular carcinoma; R-HCC, Recurrence hepatocellular carcinoma. NR-HCC, Non-recurrence hepatocellular carcinoma.
Comparision of EV dynamics between HCC and NMLD patients
In this study, a notable difference in the expression of CD41b was observed postoperatively in the HCC patient group compared to the NMLD group, with a statistically significant difference (p = 0.0286). However, when considering the aggregate preoperative and postoperative EV expressions, there were no significant disparities between the two patient cohorts. Intriguingly, both HCC and NMLD groups demonstrated a pronounced inclination towards elevated expressions of several markers, namely CD9, CD41b, CD42a, CD29, and CD31 (Fig. 5).
(A) Comparison of postoperative EV expression between the HCC and the NMLD group. The HCC group showed a significantly decreased expression of CD41b compared to the NMLD group (p = 0.0286). (B) CD41b expression in EVS of HCC and NMLD. NMLD: Non-malignant liver disease (*p < 0.05). HCC: Hepatocellular carcinoma.
Discussions
Utilizing liquid biopsy as a promising diagnostic tool for Hepatocellular Carcinomas (HCCs) and predicting their recurrence has been a focus in recent studies16,17,18. While previous research has concentrated on circulating tumor DNAs and microRNAs19, our study uniquely aimed to assess extracellular vesicle (EV) surface protein expression as potential novel biomarkers. The analysis of EV surface protein expressions extended up to 12 months post-resection to evaluate their dynamic changes. A significant challenge in HCCs lies in the absence of specific markers for predicting or diagnosing recurrence. This pilot study, for the first time, identified variations in EV surface protein patterns among HCC patients, both in comparison to the Nonalcoholic Fatty Liver Disease (NMLD) group and within HCC patients experiencing recurrence and non-recurrence. The findings contribute novel insights to the quest for effective markers in HCC recurrence prediction and diagnosis.
Recently, surface markers of EVs have gained attention since they indicated presence of HCC and cholangiocarcinoma with increased levels of CD147 and CD13320. Additionally, EVs transfer proteins and different types of RNAs, including miRNAs, from highly malignant cells to surrounding cells, promoting HCC migratory and metastatic capacities21. This is of particular interest because tumor invasion and migration leads to early and high HCC recurrence rates after resection22. Advances in this field may lead to earlier diagnosis and more efficient targeted therapies for HCC. Recent investigations, from Sun et al. (2020) showed promising results regarding HCC specific EV purification7.
In the NMLD group, the surgical procedure led to a significant increase in the CD19 rate postoperatively, which is consistent with current literature, as CD19 increment has already been reported in benign hepatic surgery23. CD19 is a protein primarily associated with B cells, which are a type of white blood cell involved in the immune response24. It is well-known in the context of certain haematological malignancies, particularly B-cell leukemias and lymphomas, and is a target for certain cancer therapies like CAR T-cell therapy25. Interestingly, no significant changes in the EVs dynamics were observed due to the surgical resection of the liver in the HCC group. However, decreased granzyme B (+) CD19 (+) B cells were correlated with early recurrence in HCC patients after liver transplantation with poor tumor differentiation, microvascular invasion, increased total tumor diameter, and tumor beyond Milan criteria26.
12 months post-surgery, the study unveiled a significant correlation between decreased levels of CD31 and HCC recurrence. CD31, a marker present on both white blood cells and endothelial cells, plays a vital role in angiogenesis—the formation of new blood vessels27. This process is crucial for tumor growth and spread and has been linked to promoting HCC metastasis28,29,30. It's noteworthy that CD31 has also been associated with cirrhosis18, aligning with our study's results. Based on these findings, we strongly advocate for a more extensive study to reevaluate the significance of CD31 in HCC recurrence.
The surface protein CD41b, similar to a recently discovered platelet-derived exosome marker associated with nonalcoholic fatty liver disease (NAFLD), appears to be elevated in the NMLD group, potentially explaining the substantial increase of CD41b compared to the HCC group in our study. There is existing evidence for CD41-positive EVs in alcoholic liver disease1. Despite aligning with other studies, caution should be exercised regarding the findings on CD41b due to reports of its instability in liquid biopsies1,23.
One limitation of this conducted study is the small number of patients in the cohort. Furthermore, our analysis focused solely on the surface markers of EVs. Exploring other biomolecules within EVs, such as miRNA, would be valuable for a more in-depth analysis of EV characteristics in correlation with clinical data.
Conclusions
In conclusion, the present study delved into the comprehensive examination of EV surface patterns in HCC and NMLD. Through our research, we have successfully demonstrated a straightforward and efficient technique for isolating EVs, shedding light on discernible differences in EV surface characteristics between individuals with HCC and those with NMLD. These findings not only contribute to our understanding of the intricate molecular landscape associated with these liver conditions but also provide a crucial foundation for subsequent investigations into EV profiling, especially in the context of HCC recurrence. The insights gained from this study hold the potential to pave the way for the identification of pertinent biomarkers, facilitating the development of more precise and optimized therapeutic strategies in the future.
Data availability
The data that support the findings of this study are available from the corresponding author, [MAJ], upon reasonable request.
References
Villanueva, A. Hepatocellular carcinoma. N. Engl. J. Med. 380(15), 1450–1462 (2019).
Tabrizian, P., Jibara, G., Shrager, B., Schwartz, M. & Roayaie, S. Recurrence of hepatocellular cancer after resection: Patterns, treatments, and prognosis. Ann. Surg. 261(5), 947–955 (2015).
Llovet, J. M. et al. Immunotherapies for hepatocellular carcinoma. Nat. Rev. Clin. Oncol. 13, 151–172 (2022).
Kobayashi, T. et al. Patients with early recurrence of hepatocellular carcinoma have poor prognosis. Hepatobiliary Pancreat. Dis. Int. 16(3), 279–288 (2017).
D’Agnano, I. & Berardi, A. C. Extracellular vesicles, a possible theranostic platform strategy for hepatocellular carcinoma-an overview. Cancers (Basel) 12(2), 261 (2020).
Lone, S. N. et al. Liquid biopsy: A step closer to transform diagnosis, prognosis and future of cancer treatments. Mol. Cancer 21(1), 79 (2022).
Sun, N. et al. Purification of HCC-specific extracellular vesicles on nanosubstrates for early HCC detection by digital scoring. Nat. Commun. 11(1), 4489 (2020).
van Niel, G., D’Angelo, G. & Raposo, G. Shedding light on the cell biology of extracellular vesicles. Nat. Rev. Mol. Cell Biol. 19(4), 213–228 (2018).
Liu, J. et al. Extracellular vesicles in liquid biopsies: Potential for disease diagnosis. Biomed. Res. Int. 2021, 6611244 (2021).
Han, Q., Zhao, H., Jiang, Y., Yin, C. & Zhang, J. HCC-derived exosomes: Critical player and target for cancer immune escape. Cells 8(6), 558 (2019).
Hirsova, P. et al. Extracellular vesicles in liver pathobiology: Small particles with big impact. Hepatology 64(6), 2219–2233 (2016).
Lapitz, A., Azkargorta, M., Milkiewicz, P., Olaizola, P., Zhuravleva, E., Grimsrud MM, et al. Liquid biopsy-based protein biomarkers for risk prediction, early diagnosis and prognostication of cholangiocarcinoma. J. Hepatol. (2023).
Hong, Y. M. et al. Risk factors of early recurrence after curative hepatectomy in hepatocellular carcinoma. Tumour. Biol. 39(10), 1010428317720863 (2017).
Jung, S.-M. et al. Characteristics of early recurrence after curative liver resection for solitary hepatocellular carcinoma. J. Gastrointest. Surg. 23(2), 304–311 (2019).
Chan, A. W. H. et al. Development of pre and post-operative models to predict early recurrence of hepatocellular carcinoma after surgical resection. J. Hepatol. 69(6), 1284–1293 (2018).
Roy, D., Pascher, A., Juratli, M. A. & Sporn, J. C. The potential of aptamer-mediated liquid biopsy for early detection of cancer. Int. J. Mol. Sci. 22(11), 5601 (2021).
Vogl, T. J. et al. Early dynamic changes in circulating tumor cells and prognostic relevance following interventional radiological treatments in patients with hepatocellular carcinoma. PLoS One. 16(2), e0246527 (2021).
Juratli, M. A., Struecker, B., Katou, S., Morguel, M. H. & Pascher, A. Einfluss der Molekularpathologie auf die onkologische Chirurgie von Leber- und Gallengangstumoren. Chirurg. 92(11), 1003–1010 (2021).
Thietart, S. & Rautou, P.-E. Extracellular vesicles as biomarkers in liver diseases: A clinician’s point of view. J. Hepatol. 73(6), 1507–1525 (2020).
Julich-Haertel, H. et al. Cancer-associated circulating large extracellular vesicles in cholangiocarcinoma and hepatocellular carcinoma. J. Hepatol. 67(2), 282–292 (2017).
Wang, J. et al. Extracellular vesicles and hepatocellular carcinoma: Opportunities and challenges. Front. Oncol. 12, 884369 (2022).
Chen, L. et al. HCC-derived exosomes elicit HCC progression and recurrence by epithelial-mesenchymal transition through MAPK/ERK signalling pathway. Cell Death Dis. 9(5), 513 (2018).
Couri, T. & Pillai, A. Goals and targets for personalized therapy for HCC. Hepatol. Int. 13(2), 125–137 (2019).
Li, X. et al. CD19, from bench to bedside. Immunol. Lett. 183, 86–95 (2017).
Sotillo, E. et al. Convergence of acquired mutations and alternative splicing of CD19 enables resistance to CART-19 immunotherapy. Cancer Discov. 5(12), 1282–1295 (2015).
Li, H. et al. Decreased granzyme B(+)CD19(+)B cells are associated with tumor progression following liver transplantation. Am. J. Cancer Res. 11(9), 4485–4499 (2021).
Qian, H., Yang, L., Zhao, W., Chen, H. & He, S. A comparison of CD105 and CD31 expression in tumor vessels of hepatocellular carcinoma by tissue microarray and flow cytometry. Exp. Ther. Med. 16(4), 2881–2888 (2018).
Zhang, Y.-Y. et al. CD31 regulates metastasis by inducing epithelial-mesenchymal transition in hepatocellular carcinoma via the ITGB1-FAK-Akt signaling pathway. Cancer Lett. 429, 29–40 (2018).
Wang, J., Xu, X., Jia, J., Wu, C. & Ge, R. Epressions of SE-1, CD31 and CD105 in the vascular endothelial cells and serum of rat with hepatocellular carcinoma. Chin. Med. J. 17 (2010)
Bösmüller, H. et al. Microvessel density and angiogenesis in primary hepatic malignancies: Differential expression of CD31 and VEGFR-2 in hepatocellular carcinoma and intrahepatic cholangiocarcinoma. Pathol. Res. Pract. 214(8), 1136–1141 (2018).
Acknowledgements
We thank Larissa Laufer and Scarlett Bather for helping with data curation.
Funding
Open Access funding enabled and organized by Projekt DEAL. This work was supported by the Rudolf Geißendoerfer-Stiftung in the form of a grant awarded to MAJ (2015); Frankfurter Forschungsfoerderung in the form of a grant awarded to MAJ [FFF 2017]; B. Braun-Stiftung in the form of a grant awarded to MAJ (2018).
Author information
Authors and Affiliations
Contributions
Conceptualization: M.A.J, E.O., B.S., T.V., W.O.B. Data curation: N.P., E.O. Formal analysis: N.P., E.O., S.K., S.M., F.B., A.A., D.R. Funding acquisition: M.A.J., A.P., W.O.B. Project administration: M.A.J., E.O., B.S. J.H., P.H. Supervision: M.A.J., E.O., A.S., A.P., W.O.B. Prepared Figures: M.A.J., D.R., A.M. Writing—original draft: M.A.J., N.P. Writing—review & editing: M.A.J., E.O., D.R., B.S., F.B., A.M., N.S., A.S., A.A., A.P., W.O.B., P.H. Revision: M.A.J., E.O., D.R., B.S., A.M., N.S. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Juratli, M.A., Pollmann, N.S., Oppermann, E. et al. Extracellular vesicles as potential biomarkers for diagnosis and recurrence detection of hepatocellular carcinoma. Sci Rep 14, 5322 (2024). https://doi.org/10.1038/s41598-024-55888-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-55888-8
Keywords
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.