Abstract
Rheumatoid Arthritis (RA) is an increasingly prevalent inflammatory disorder worldwide. Its complex etiology has recently brought dietary factors, particularly fiber intake, into focus as potential influencers. Our study investigates the intricate relationship between various sources of dietary fiber and RA, emphasizing the mediating role of the Dietary Inflammatory Index (DII). Leveraging data from the National Health and Nutrition Examination Survey spanning 2011 to 2020. We meticulously assessed dietary fiber intake through dual 24 h dietary recall interviews, while RA diagnoses were established based on comprehensive medical surveys. The relationships between fiber intake, RA prevalence, and DII mediation were analyzed using sophisticated multivariate logistic regression and mediation analysis. Among our study cohort, 7% were diagnosed with RA. We observed a notable inverse correlation between increased total fiber intake, particularly 5 g/day increments, and the incidence of RA, with cereal fiber intake emerging as the primary mitigating factor. Intriguingly, the DII played a significant role in mediating this association, especially regarding cereal fiber. Our findings reveal a significant association between higher cereal fiber consumption and a reduced prevalence of RA. Additionally, the DII stands out as a pivotal mediator in this relationship, highlighting dietary management's critical role in preventing and managing RA.
Similar content being viewed by others
Introduction
Rheumatoid arthritis (RA) is a globally prevalent inflammatory disorder characterized by symmetric inflammatory polyarthritis, leading to significant joint damage and functional impairment1,2. Since 1990, there has been a concerning 8.2% annual increase in RA cases worldwide, highlighting the growing need for effective detection and prevention strategies in an aging global population3. The etiology of RA is complex and not fully understood, but recent studies have started to shed light on dietary factors as potential contributors to its development4.
Recent scholarly work suggests that dietary fiber intake may have a therapeutic effect against inflammatory conditions, with the role of gut microbiota and its byproducts becoming increasingly recognized5,6. Previous research has pointed to the potential benefits of increased fiber intake in reducing systemic inflammation in individuals with RA7. A groundbreaking study utilizing data from the UK Biobank has uncovered the potential protective effects of a diet rich in dietary fiber and polyunsaturated fatty acids against the onset of RA. However, the specific impacts of various fiber sources, such as grains, vegetables, and fruits, on RA risk are still not fully explored.
Inflammation is a key factor in the development of RA, and the Dietary Inflammatory Index (DII) serves as a vital tool for assessing the overall inflammatory potential of a diet by considering the inflammatory properties of different dietary components. A growing number of studies highlight the significant role of DII in influencing RA susceptibility and progression8. Current theories suggest that a diet rich in dietary fiber can mitigate inflammation and thus reduce the risk of RA. However, there is still a gap in research regarding the extent and nature of the DII’s mediating role in the relationship between dietary fiber intake and RA risk.
Considering these factors, we undertook a cross-sectional study using data from the 2011–2020 National Health and Nutrition Examination Survey (NHANES). Our objective was dual: to explore the relationship between various sources of dietary fiber and RA risk and to elucidate the potential mediating role of the DII in the interaction between dietary fiber intake and the onset of RA.
Methods
Study population
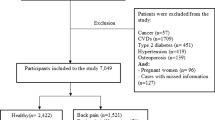
The National Center for Health Statistics (NCHS) conducted the NHANES, a nationally representative survey of the non-institutionalized population in the United States, as the basis for this study9,10,11. All study procedures were approved by the Research Ethics Review Board of NCHS, and informed consent was obtained from all participants. The present study complies with the guidelines for cross-sectional research provided by Strengthening the Reporting of Observational research in Epidemiology (STROBE). The dataset used in this study includes data from 2011 to 2020. In this study, after excluding participants without RA data (n = 12,820), and those with extreme total energy intakes (n = 93), a total of 15,114 samples were enrolled for the final analysis (Fig. 1).
We excluded participants who did not have self-reported RA data (n = 12,820), those with incomplete or missing dietary intake data (n = 13,011), those with missing covariate data (n = 4,424), and those with extreme total energy intakes (n = 93). The final sample for analysis included 15,114 participants (Fig. 1).
Variables definition
In alignment with prior research12,13, we enlisted a certified nutritionist to conduct two 24 h dietary recall sessions, assessing participants’ fiber intake. The initial session was conducted in person at the Mobile Examination Center (MEC), followed by a subsequent telephone session within 3 to 10 days. We calculated the average fiber consumption over these two days, adjusting for individual weight factors. Dietary fiber sources, including grains, vegetables, and fruits, were identified by collating relevant food codes (Fig. 2).
Professionals and two medical conditions accessed RA questionnaires to gather the data. When participants answered “yes” to the question, “Has your doctor ever told you to have arthritis,” we assumed these participants had arthritis. When this group of participants answered “rheumatoid arthritis” to the question “Which type of arthritis was it?”, we considered that these participants had RA. A meta-analysis that included 16 epidemiological studies demonstrated that self-reported RA is highly accurate and acceptable in large studies13.
The DII was employed as a critical metric to assess the inflammatory potential of various dietary components, ranging from vitamins to minerals14. We calculated the DII score by determining individual dietary markers and their corresponding z-scores, subsequently standardizing these into median percentiles. The standardized inflammatory effect of each percentile was then aggregated to form the final DII score for each participant. The selection of covariates for our analysis was informed by established links between fiber consumption, RA, and factors ranging from demographics to lifestyle habits.
Covariables were chosen based on prior knowledge as factors associated with fiber intake and RA. Covariates included age, gender, race, body mass index, family income-to-poverty ratio (PIR), menopause status (引用), moderate activities, waist circumference, energy intake, drinking alcohol status education level, and smoking status.
Statistical analysis
R software (version 4.3.0) alongside Empowerstats (version 4.1) powered all statistical evaluations. Participant demographics were segmented by RA status and gauged using the chi-square and t-test. Advanced logistic regression procedures aimed to discern the connections between fiber consumption, inflammation markers, and RA prevalence15,16,17. To ascertain the potential intermediary role of DII between fiber consumption and RA emergence, we deployed a parallel mediator approach, using distinct indicators as mediators18. Herein, the direct effect (DE) portrays inflammation’s influence on RA without intermediary factors, while indirect effects (IE) elucidate the mediator-influenced relationship between inflammation and RA. Mediator proportions were derived by equating IE with the overall effect (TE).
Ethical approval and consent to participate
The studies involving human participants were reviewed and approved by the NCHS Ethics Review Board. The patients/participants provided their written informed consent to participate in this study.
Results
Our analysis encompassed 15,114 subjects, with an average age of 46.37 (16.89) years during the evaluation. This cohort identified 1053 participants (approximately 7%) with RA. The fiber consumption median for the RA group stood at 13.6 g/day, spanning from 0 to 97.4 g/day, whereas for the non-RA counterparts, it was 14.7 g/day, ranging from 0 to 134.8 g/day. When juxtaposed against the non-RA group, RA-afflicted individuals predominantly comprised females, exhibited advanced age, possessed inferior educational backgrounds, and reported reduced income levels. Lifestyle-wise, RA subjects showcased heightened tendencies for smoking and alcohol consumption and diminished engagement in physical activities. Physiologically, they registered increased BMI and waist measurements, while their dietary charts reflected reduced caloric and fiber intakes (Table 1). Demographic attributes were scrutinized using chi-square and t-test methodologies.
Table 2 elaborates on the multivariate logistic regression analysis outcomes, highlighting an inverse correlation between augmented total fiber intake (5 g/day) and RA emergence [0.89 (0.86, 0.98)]. We employed a sensitivity analysis to refine this exploration, categorizing fiber consumption into tertiles. Those in the topmost fiber consumption tertile experienced a 25% reduction in RA prevalence relative to their lowest tertile peers [0.75 (0.63, 0.88)]. We further delved into the influence of fiber sources on RA incidence. Post multi-variable adjustments, only cereal fiber intake emerged as significantly inversely correlated with RA onset [0.85 (0.76, 1.02)], this inverse link being more pronounced in sensitivity analyses [0.72 (0.61, 0.85)]. Conversely, vegetable and fruit fiber consumption displayed tenuous and inconclusive links with RA emergence.
Table 3 delineates fiber consumption’s associations with DII. The weighted multivariate linear regression results underscored a significant inverse relationship between total fiber intake and both DII [− 2.82 (− 3.28, − 2.35)] and Hs-CRP [− 0.19 (− 0.32, − 0.07)]. Similar trends were observed for cereal and vegetable fiber intakes about DII. However, the relationship between fruit fiber intake and RA incidence remained inconclusive.
Table 4 elucidates the multivariate logistic regression analysis insights into the DII-RA incidence relationship. A notable positive correlation surfaced between DII and RA prevalence, indicating a 26% surge in RA cases for each unit increment in DII [1.26 (1.01, 1.58)]. Moreover, participants with the highest DII readings exhibited a 62% amplification in RA incidence relative to the lowest DII group [1.62 (1.02, 2.18)].
Our findings reveal that the DII plays a significant, albeit not predominant, mediating role in the association between total and cereal fiber intakes and RA incidence. Specifically, the mediation percentages of DII were found to be approximately 36.12% for total fiber intake and 40.35% for cereal fiber intake (P < 0.01), indicating a notable but not exclusive influence in this association (Fig. 2).
Discussion
This study unveils two critical insights into the dietary habits of the American adult population. Firstly, it establishes a clear correlation between suboptimal fiber intake and an increased prevalence of RA, identifying cereal fiber as the primary mitigating factor in this inverse relationship. Secondly, our analysis highlights the DII as a critical intermediary in the association between fiber consumption and the onset of RA. Notably, while the DII’s mediating role falls below 50%, its direct impact considerably surpasses the indirect effect, emphasizing its integral role in the fiber-RA incidence connection.
An expanding reservoir of scientific literature is emphasizing the influence of dietary elements and habits on RA onset19. Diet, as a pivotal external determinant, can influence RA’s development by modulating gut microbiota, antigenic expression, as well as the body’s inflammatory and antioxidant defense systems20,21. Typically, RA patients are counseled to augment their intake of anti-inflammatory nutrients22, such as dietary fiber and specific polyunsaturated fatty acids23, which harbor both anti-inflammatory and antioxidant attributes, potentially arresting inflammation escalation24. Regrettably, the present dietary quality among RA patients falls short, with our findings revealing that such patients presented markedly elevated DII scores compared to the general participants. Even though nutritional guidelines advocate a surge in fiber intake, the majority of Americans fall short, consuming less than half of the suggested daily fiber quota, with RA patients faring even worse25. An inferior diet correlates with the longevity and intensity of RA manifestations, potentially amplifying RA onset risks21,26. Our data underscore the significance of the fiber’s origin, pinpointing cereal fiber as the prime contender in inflammation reduction. While prior research has postulated anti-inflammatory roles for fiber, drawing from its ability to satiate, enhance gut health, modify dietary regimes, and optimize lipid and glucose metabolism27,28,29, the unique significance of cereal fiber over its vegetable or fruit counterparts remains a puzzle, warranting deeper probing.
Historically, heightened fiber consumption was touted as a remedy for alleviating RA symptoms30. Yet, the nexus between dietary fiber and RA vulnerability remains enigmatic and contentious, with scant research probing this connection. Contradictory findings, like those from the EPIC-Norfolk study linking reduced fruit and vegetable intake with escalated inflammatory polyarthritis31 or the Greek case-control study associating cooked vegetable and olive oil consumption with reduced RA risks32, further muddle the picture. Amidst such ambiguity, our study presents a robust framework, leveraging a comprehensive dataset and stringent control mechanisms, thereby bolstering the credibility and consistency of its conclusions.
The complex interplay between dietary fiber consumption and the onset of RA continues to be an area of active research. The prevailing consensus suggests that inflammation is at the heart of this relationship. Diets rich in fiber, like the Mediterranean and vegan diets, have been shown to reduce inflammation in RA patients30,33. Large-scale observational studies further reinforce the connection between fiber intake and inflammatory markers in diverse populations. The gut-joint axis, particularly the anti-inflammatory effects of fiber fermentation on gut microbiota, offers a plausible explanation for these findings34,35,36. Gut dysbiosis has been identified as a key factor in the development of RA, both in animal models and human studies37. Gut dysbiosis, a potential disruptor of intestinal barrier functionality, has emerged as a cornerstone of RA’s genesis in mice and human studies38,39,40. Our mediation analysis underscores the inverse association between high cereal fiber intake and RA prevalence, establishing DII as a critical mediator in this relationship.
However, our study’s strengths significantly contribute to its value. A key strength is the incorporation of the DII, a novel approach in RA research. The DII provided a comprehensive measure of the inflammatory potential of participants’ diets, allowing for a nuanced analysis of how dietary components, particularly fiber, relate to RA incidence. This incorporation presents a more holistic understanding of diet’s role in RA, beyond the traditional focus on single nutrients or food groups. Another noteworthy aspect of our study is its in-depth examination of the relationship between various sources of dietary fiber and RA incidence. By distinguishing between different fiber sources, such as cereal, vegetable, and fruit fibers, our study offers a more detailed understanding of how specific dietary choices might influence RA risk. This is particularly relevant given the diverse roles of different fiber types in modulating inflammation and gut microbiota.
In reflecting upon the methodological aspects of our study, we recognize certain limitations inherent in the tools we employed. Concerning the DII, while it offers a comprehensive assessment of the diet’s inflammatory potential, it is important to note that many dietary components influence the DII41. This complexity necessitates a cautious interpretation when attributing the effects of specific nutritional elements, such as cereal fiber, to the DII. It is plausible that there are other contributing dietary factors that our analysis may not have fully accounted for. Moreover, the use of the 24 h food recall method in our study, although a widely accepted tool in dietary research, presents its own set of limitations. One significant concern is its potential inability to accurately capture habitual dietary patterns, considering the day-to-day variations in food intake. Additionally, the reliability of this method is contingent upon the participant’s ability to accurately recall and report their food consumption, which introduces the possibility of under or over-reporting42,43. Acknowledging these limitations, future research endeavors might benefit from employing multiple food recalls or food frequency questionnaires to glean a more representative and comprehensive picture of long-term dietary habits. Despite these limitations, the extensive dataset, meticulous approach to dietary intake data collection, and innovative use of the DII in our study make a valuable contribution to the literature on the interplay between dietary fiber, inflammation, and RA onset. Our findings provide a foundation for future research and potential nutritional interventions to manage or reduce the risk of RA.
Conclusion
Our findings reveal a significant association between increased cereal fiber intake and a reduced prevalence of RA. This relationship is notably absent with vegetable or fruit fiber intake. Furthermore, the DII emerges as a crucial intermediary, elucidating the connection between higher cereal fiber consumption and lower RA occurrence.
Data availability
The survey data are publicly available on the internet for data users and researchers throughout the world ( www.cdc.gov/nchs/nhanes/).
Abbreviations
- RA:
-
Rheumatoid arthritis
- NHANES:
-
National health and nutrition examination survey
- NCHS:
-
National center for health statistics
- Hs-CRP:
-
High-sensitivity C-reactive protein
- DII:
-
Dietary inflammatory index
- DE:
-
Direct effect
- IE:
-
Indirect effect
- TE:
-
Total effect
References
England, B. R., Thiele, G. M., Anderson, D. R. & Mikuls, T. R. Increased cardiovascular risk in rheumatoid arthritis: Mechanisms and implications. BMJ 361, k1036 (2018).
Aletaha, D. & Smolen, J. S. Diagnosis and management of rheumatoid arthritis: A review. Jama 320(13), 1360–1372 (2018).
Safiri, S. et al. Global, regional and national burden of rheumatoid arthritis 1990–2017: A systematic analysis of the global burden of disease study 2017. Ann. Rheum. Dis. 78(11), 1463–1471 (2019).
Philippou, E. & Nikiphorou, E. Are we really what we eat? Nutrition and its role in the onset of rheumatoid arthritis. Autoimmun. Rev. 17(11), 1074–1077 (2018).
Swann, O. G., Kilpatrick, M., Breslin, M. & Oddy, W. H. Dietary fiber and its associations with depression and inflammation. Nutr. Rev. 78(5), 394–411 (2020).
Häger, J. et al. The role of dietary fiber in rheumatoid arthritis patients: A feasibility study. Nutrients 11(10), 2392 (2019).
Sparks, J. A. et al. Inflammatory dietary pattern and risk of developing rheumatoid arthritis in women. Clin. Rheumatol. 38(1), 243–250 (2019).
Marx, W. et al. The dietary inflammatory index and human health: An umbrella review of meta-analyses of observational studies. Adv. Nutr. 12(5), 1681–1690 (2021).
Zhang, Y., Xie, R. & Ou, J. A U-shaped association between serum albumin with total triiodothyronine in adults. J. Clin. Lab. Anal. 36(6), e24473 (2022).
Xie, R. & Zhang, Y. Index-based calculation or Transient Elastography to assess the degree of hepatic steatosis and fibrosis. J Nutr 153, 909 (2022).
Xie, R. & Zhang, Y. Is assessing the degree of hepatic steatosis and fibrosis based on index calculations the best choice for epidemiological studies?. Environ. Pollut. 317, 120783 (2022).
Frampton, J., Murphy, K. G., Frost, G. & Chambers, E. S. Higher dietary fibre intake is associated with increased skeletal muscle mass and strength in adults aged 40 years and older. J. Cachexia Sarcopenia Muscle 12(6), 2134–2144 (2021).
Peeters, G. M., Alshurafa, M., Schaap, L. & de Vet, H. C. Diagnostic accuracy of self-reported arthritis in the general adult population is acceptable. J. Clin. Epidemiol. 68(4), 452–459 (2015).
Shivappa, N., Steck, S. E., Hurley, T. G., Hussey, J. R. & Hébert, J. R. Designing and developing a literature-derived, population-based dietary inflammatory index. Public Health Nutr. 17(8), 1689–1696 (2014).
de Hair, M. J. et al. Smoking and overweight determine the likelihood of developing rheumatoid arthritis. Ann. Rheum. Dis. 72(10), 1654–1658 (2013).
Hagfors, L., Leanderson, P., Sköldstam, L., Andersson, J. & Johansson, G. Antioxidant intake, plasma antioxidants and oxidative stress in a randomized, controlled, parallel, Mediterranean dietary intervention study on patients with rheumatoid arthritis. Nutr. J. 2, 5 (2003).
Gioia, C., Lucchino, B., Tarsitano, M. G., Iannuccelli, C. & Di Franco, M. Dietary habits and nutrition in rheumatoid arthritis: Can diet influence disease development and clinical manifestations?. Nutrients 12(5), 1456 (2020).
Morrison, D. J. & Preston, T. Formation of short chain fatty acids by the gut microbiota and their impact on human metabolism. Gut Microbes 7(3), 189–200 (2016).
Evans, C. E. L. Dietary fibre and cardiovascular health: A review of current evidence and policy. Proc. Nutr. Soc. 79(1), 61–67 (2020).
Berube, L. T., Kiely, M., Yazici, Y. & Woolf, K. Diet quality of individuals with rheumatoid arthritis using the healthy eating index (HEI)-2010. Nutr. Health 23(1), 17–24 (2017).
Kaczmarczyk, M. M., Miller, M. J. & Freund, G. G. The health benefits of dietary fiber: Beyond the usual suspects of type 2 diabetes mellitus, cardiovascular disease and colon cancer. Metabolism 61(8), 1058–1066 (2012).
Satija, A. & Hu, F. B. Cardiovascular benefits of dietary fiber. Curr. Atheroscler. Rep. 14(6), 505–514 (2012).
Daïen, C. I., Pinget, G. V., Tan, J. K. & Macia, L. detrimental impact of microbiota-accessible carbohydrate-deprived diet on gut and immune homeostasis: An overview. Front. Immunol. 8, 548 (2017).
King, D. E. et al. Effect of a high-fiber diet vs a fiber-supplemented diet on C-reactive protein level. Arch. Intern. Med. 167(5), 502–506 (2007).
Pattison, D. J. et al. Vitamin C and the risk of developing inflammatory polyarthritis: Prospective nested case-control study. Ann. Rheum. Dis. 63(7), 843–847 (2004).
Linos, A. et al. Dietary factors in relation to rheumatoid arthritis: A role for olive oil and cooked vegetables?. Am. J. Clin. Nutr. 70(6), 1077–1082 (1999).
Smidowicz, A. & Regula, J. Effect of nutritional status and dietary patterns on human serum C-reactive protein and interleukin-6 concentrations. Adv. Nutr. 6(6), 738–747 (2015).
Ma, Y. et al. Association between dietary fiber and serum C-reactive protein. Am. J. Clin. Nutr. 83(4), 760–766 (2006).
Krishnamurthy, V. M. et al. High dietary fiber intake is associated with decreased inflammation and all-cause mortality in patients with chronic kidney disease. Kidney Int. 81(3), 300–306 (2012).
Qi, L. et al. Whole-grain, bran, and cereal fiber intakes and markers of systemic inflammation in diabetic women. Diabetes Care 29(2), 207–211 (2006).
Hills, R. D. Jr. et al. Gut microbiome: Profound implications for diet and disease. Nutrients 11(7), 1613 (2019).
Zaiss, M. M., Joyce Wu, H. J., Mauro, D., Schett, G. & Ciccia, F. The gut-joint axis in rheumatoid arthritis. Nat. Rev. Rheumatol. 17(4), 224–237 (2021).
Pianta, A. et al. Evidence of the immune relevance of prevotella copri, a gut microbe, in patients with rheumatoid arthritis. Arthritis Rheumatol. 69(5), 964–975 (2017).
Tajik, N. et al. Targeting zonulin and intestinal epithelial barrier function to prevent onset of arthritis. Nat. Commun. 11(1), 1995 (2020).
Krishnamurthy, V. M. et al. High dietary fiber intake is associated with decreased inflammation and all-cause mortality in patients with chronic kidney disease. Kidney Int. 81(3), 300–306 (2012).
Qi, L., van Dam RM, Liu, S., Franz, M., Mantzoros, C., Hu, F.B.: Whole-grain, bran, and cereal fiber intakes and markers of systemic inflammation in diabetic women. Diabetes Care 29(2), 207–211 (2006).
Hills, R.D., Jr., Pontefract, B.A., Mishcon, H.R., Black, C.A., Sutton, S.C., Theberge, C.R.: Gut Microbiome: Profound Implications for Diet and Disease. Nutrients 11(7) (2019).
Zaiss, M.M., Joyce Wu, H.J., Mauro, D., Schett, G., Ciccia, F.: The gut-joint axis in rheumatoid arthritis. Nat. Rev. Rheumatol. 17(4), 224–237 (2021).
Pianta, A., Arvikar, S., Strle, K., Drouin, E.E., Wang, Q., Costello, C.E., Steere, A.C.: Evidence of the Immune Relevance of Prevotella copri, a Gut Microbe, in Patients With Rheumatoid Arthritis. Arthritis Rheumatol 69(5), 964–975 (2017).
Tajik, N., Frech, M., Schulz, O., Schälter, F., Lucas, S., Azizov, V., Dürholz, K., Steffen, F., Omata, Y., Rings, A. et al:Targeting zonulin and intestinal epithelial barrier function to prevent onset of arthritis. Nat. Commun. 11(1), 1995 (2020).
Xie, R., Ning, Z., Xiao, M., Li, L., Liu, M., Zhang, Y.: Dietary inflammatory potential and biological aging among US adults: A population-based study. Aging Clin Exp Res 35(6),1273–1281 (2023).
Xie, R., Zhang, Y.: Association between 19 dietary fatty acids intake and rheumatoid arthritis: Results of a nationwide survey. Prostaglandins Leukot Essent Fatty Acids 188, 102530 (2023).
Xie, R., Zhang, Y.: Associations between dietary flavonoid intake with hepatic steatosis and fibrosis quantified by VCTE: Evidence from NHANES and FNDDS. Nutr. Metab. Cardiovasc. Dis. 33(6), 1179–1189 (2023).
Acknowledgements
We thank all the participants and staff in NHANES for their selfless dedication.
Author information
Authors and Affiliations
Contributions
H.W., S.Y., and Y.Z. designed the research. H.W., S.Y., Z.N., M.L., and Y.Z. collected, analyzed the data, and drafted the manuscript. H.W. and S.Y. revised the manuscript. All authors contributed to the article and approved the submitted version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wan, H., Zhang, Y., Ning, Z. et al. Associations of cereal fiber intake with rheumatoid arthritis mediated by dietary inflammatory index: insights from NHANES 2011–2020. Sci Rep 14, 2415 (2024). https://doi.org/10.1038/s41598-024-52806-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-52806-w
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.