Abstract
A supercritical CO2 method was optimized to recover naringenin-rich extract from Mexican oregano (Lippia graveolens), a flavanone with high antioxidant and anti-inflammatory activity. The effect of the extraction parameters like pressure, temperature, and co-solvent on naringenin concentration was evaluated. We used response surface methodology to optimize the naringenin extraction from oregano; the chemical composition by UPLC-MS of the optimized extract and the effect of simulated gastrointestinal digestion on its antioxidant capacity and total phenolic content were also evaluated. The optimum conditions were 58.4 °C and 12.46% co-solvent (ethanol), with a pressure of 166 bar, obtaining a naringenin content of 46.59 mg/g extract. Also, supercritical optimized extracts yielded high quantities of cirsimaritin, quercetin, phloridzin, apigenin, and luteolin. The results indicated that the naringenin-rich extract obtained at optimized conditions had higher total phenolic content, antioxidant capacity by TEAC and ORAC, and flavonoid content, compared with the methanolic extract, and the simulated gastrointestinal digestion reduced all these values.
Similar content being viewed by others
Introduction
Oregano is a group of plants with common attributes such as aroma and flavor, Lippia graveolens, also known as Mexican oregano, is one the most widely distributed species. This plant has a variety of lipophilic compounds in its essential oil, and most studies are related to them1; however, the content of phenolic compounds has been of interest in recent studies2,3. Phenolic compounds are secondary metabolites ubiquitous in the plant kingdom; these molecules act as a defense mechanism against biotic and abiotic stress. Among them, flavonoids are some of the most important compounds in this category; they are synthesized by the phenylpropanoid pathway, forming a basic structure of two phenyl rings (A and B) united by a pyran ring (C)4. These compounds have been attributed with health benefits; for instance, quercetin has been shown to induce apoptosis in pancreatic cancer cell lines5, and cirsimaritin exhibited therapeutic effects in damaged beta cells6. Furthermore, naringenin (4ʹ,5,7-trihydroxyflavanone) has antioxidant, anti-inflammatory7, and anti-proliferative8 properties. Also, although commonly found in citric fruits, several studies indicate that naringenin is one of the most abundant flavonoids in Lippia graveolens2,3,9.
Commonly, flavonoids are extracted from plant matrixes using organic solvents, which can harm the environment and human health to various degrees; for example, the FDA classified solvents like methanol as class 2, with limited use because of its toxicity10. In this regard, alternative extraction methods have been employed to extract phenolic compounds, mitigating these drawbacks; microwave-assisted extraction, ultrasound-assisted extraction, pressurized liquid extraction, and supercritical CO2 extraction are the most common ones11,12. In this sense, supercritical CO2 extraction is an effective method to extract compounds of diverse polarity based on the behavior of CO2 in supercritical state (solvating like a liquid and diffusing like a gas). In this state, the CO2 can enter and disrupt the plant cell and extract desired compounds according to the process conditions; however, this solvent is non-polar, and adding a polar cosolvent allows the extraction of compounds of this nature13. The ability to tune parameters of the process like temperature, pressure, and cosolvent proportion allows this technology to have better characteristics compared to other techniques, including higher selectivity and yields of the desired compound, as well as short extraction times; furthermore, the supercritical conditions of CO2 are relatively low (31 °C and 74 bar), avoiding thermal degradation of phytochemicals and reducing power consumption14,15. Regarding extraction of phenolic compounds from plant matrixes, studies have determined conditions of 36–60 °C, pressure between 100 and 300 bar, and ethanol (cosolvent) proportion of up to 20% had been effective to obtain high quantities of these compounds3,16,17,18.
Furthermore, phenolic compounds must be bioaccessible and bioavailable to exert their bioactivities; in this context, bioaccessibility is defined as the proportion of compounds released from the food matrix and available for intestinal absorption19. In general, the gastrointestinal process induces changes in the structure of phenolic compounds by the changes in pH and the presence of digestive enzymes, affecting its bioaccessibility20,21.
Thus, this research aimed to optimize a supercritical CO2 process to obtain a naringenin-rich extract from Mexican oregano and evaluate the effect of a simulated gastrointestinal digestion on its antioxidant capacity and flavonoid content.
Results and discussion
Response surface methodology analysis and optimization
The model adjusted for the linear terms of temperature and cosolvent, and the quadratic term cosolvent2, for the response (naringenin content), without a lack of fit and a coefficient of determination (R2) of 0.8079 (Table 1); indicating that the adjusted terms explain 80.79% of the variability of the response. The prediction for naringenin content is shown in Eq. (1) for the codified values and in Eq. (2) for natural values.
The contour plot and response surface indicate a superior naringenin content at high temperatures and less than 15% of cosolvent, while pressure did not affect the extraction of this flavanone (Fig. 1).
The desirability optimization showed a predicted value of 46,590 µg/g extract at the optimum conditions of 58.4 °C (temperature) and 12.46% (cosolvent), with a pressure of 166 bar, showing a desirability value of 0.9384, being a value of 1 the optimal performance of the factors22.
After replicating the optimum conditions four times, the naringenin content was 51,457.79 ± 5831.36 µg/g extract in the supercritical extract at optimum condition (SE), indicating the model accurately predicted the response. Supercritical CO2 extraction is a highly sensitive method to process conditions, including (but not limited to) temperature, pressure, and cosolvent proportion. In this sense, our optimization allowed the extraction of high quantities of naringenin, related to the moderate temperature (less than 60 °C), suggesting that an increase in vapor pressure by increasing temperature affects the solubility of this flavanone in supercritical CO2 more than the CO2 density, which decreases while increasing temperature23. Moreover, low pressure is desired to improve selectivity, as well as reduce the extraction of oils, waxes, and other no desriables compounds24, and enhance flavonoid extraction25. In addition, naringenin solubility was enhanced by the addition of ethanol as cosolvent due to the polarity of the flavonoid; similarly, as seen in other plant matrixes, the use of this alcohol as cosolvents is effective in extracting this compound17,26; however, increasing the cosolvent proportion more than 12.46% reduced the naringenin extraction, attributed to a decrease in supercritical CO2 selectivity14, likewise, high quantities of cosolvent also decrease the influence of temperature in the extraction27; also, this effect can also be explained as an excess of cosolvent increase the interaction between it and the solute, reducing supercritical fluid-solute interaction28, and prevents supercritical CO2 from diffusing through the plant matrix29.
To the best of our knowledge, there are not studies about the specific extraction and optimization of naringenin from plants using supercritical CO2; although, it had been extracted from matrixes like spearmint (Mentha spicata L.) and Strobilanthes crispus leaves at similar process conditions (60 °C, 200 bar, 60 min; and 60 °C, 100 bar, 80 min; respectively)16,30.
Flavonoid quantification by UPLC-MS
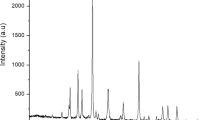
The content of flavonoids of the undigested and digested extracts is shown in Table 2, and their chromatograms in Supplementary Fig. S1. In the SE, the most abundant flavonoids were naringenin, cirsimaritin, and quercetin; while in the methanolic extract (ME) the major flavonoids were naringenin, phloridzin, and cirsimaritin. Furthermore, the flavonoid content of each flavonoid was higher in the SE, except for kaempferol, luteolin, and phloridzin. Interestingly, after the simulated digestion, the flavonoid content decreased significantly; in addition, most of the flavonoids only showed a bioaccessibility of 1% or lower, and only apigenin of the intestinal phase of the supercritical extract (ISE) and phloridzin of the intestinal phase of the methanolic extract (IME) showed a bioaccessibility of 4.83% and 7.09%, respectively; in this sense, the presence of a sugar substituent in the structure of phloridzin confers a more stable structure compared with the other aglycones31. Also, apigenin has better solubility at more alkaline pH (Supplementary Fig. S2), that allows better bioaccessibility at the intestinal phase, due to having a pKa of 7.02 at C5 of the A ring, with a deprotonated hydroxyl group and being more soluble. Compared with other flavonoids, naringenin (pKa = 7.86, at C7), cirsimaritin (pKa = 7.87, at C5), and quercetin (pKa = 7.58 at C7) are mostly in the protonated form at pH 7, reducing their solubility and, in consequence, their bioaccessibility in the intestinal phase.
The differences in flavonoids of both extracts are attributed to the different conditions of the processes; the supercritical extraction was carried out in conditions of polarity, solvent density, and viscosity adequate to extract higher quantities of naringenin and, at the same time, these conditions were adequate to the other flavonoids. Besides, the XLogP3-AA of the flavonoids in higher quantities (naringenin and cirsimaritin) is are similar, 2.4 and 2, respectively; compared with the rest of the compounds (ranged from 1.2 to 1.7); meaning the process conditions where adequate to extract compounds of these polarities. The presence of naringenin as the most abundant compound is in concordance with previous studies3, and other flavonoids have also been reported32.
Also, the low stability of the flavonoids during the simulated digestion can be attributed to the pH changes during the digestion steps. In general, flavonoids are more stable at gastric pH conditions, compared to the neutral pH of the intestine, favoring autoxidation reaction33. In addition, possible interactions between the compounds with the digestive enzymes could reduce their bioaccessibility34,35, as phenolic compounds precipitate proteins by binding to them36,37.
Our study is in concordance with other where naringenin, cirismaritin are ones of the major flavonoids2,9; also, a study of Bernal-Millán et al.32, showed that the different extraction methods did not affect the naringenin content in their extracts. In contrast, our study achieved the desired conditions to enhance the naringenin extraction.
Total phenolic content
The extraction method significantly affected the total phenolic content, as shown in Table 3, where the SE had a higher value than the ME (p < 0.05). This difference can be attributed to the better selectivity of the supercritical CO2, a process where the control of the variables enhances the phenolic compound extraction yield38, and this has also been reported for leaves and berries of myrtle39. The supercritical extraction process allowed to extract phenolic compounds in high quantities compared to the methanolic extraction, including naringenin; however, our results are lower compared to a previous study by Picos-Salas, et al.3, where the TPC was 354 mg GAE/g of extract; as well against the methanolic extract from Martínez-Rocha, et al.40, with a TPC of 270.25 mg GAE/g extract; which can be attributed to the plant growth conditions. In addition, the process in the present work is focused on enhancing the extraction of naringenin, probably avoiding the extraction of other compounds that can react to the Folin-Ciocalteu reagent; in addition, changes in extraction parameter are reflected in the total phenolic content value41.
Antioxidant capacity
There was no significant difference between the SE and ME TEAC values (Table 3). On the other hand, the SE showed significantly higher ORAC values than the ME (Table 3). The simulated digestion process caused a significant loss in antioxidant capacity; the TEAC values decreased to 5% for both extraction methods, while the ORAC results decreased to 1.87% for the ISE and 2.98% for the IME; this compared with the extract without simulated digestion.
It has been established that phenolic compounds suffer degradation during gastrointestinal digestion caused by the pH changes and presence of enzymes; in this sense, the simulated digestion provided harsh conditions to the compounds, causing changes in their structures by deprotonating their –OH radicals, thus, reducing their antioxidant capacity42,43.
The TEAC assay is based in a mixed mode, where electron transfer (ET), hydrogen atom transfer (HAT), and proton-coupled electron transfer mechanisms happen at different proportions depending on the solvent and pH of the reaction44; however, phenolic compounds respond better to assays related to HAT mechanism; this caused no significant differences between the antioxidant capacity of both undigested extracts. On the contrary, the ORAC assay is based on the HAT mechanism, which is predominant in phenolic compounds like flavonoids45.
These results indicate that the higher content of naringenin and other flavonoids, like cirsimaritin and quercetin, contributed to the better antioxidant capacity of the SE. Even if naringenin is the most abundant flavonoid in the extracts, its antioxidant power is rather low compared to other flavonoids due to the absence of a catechol group in the B ring and lack of C2–C3 double bond46; however, the relatively high presence of naringenin and its synergy with quercetin (with a catechol group in B ring) and cirsimaritin could influence the higher antioxidant values, and confer better antioxidant power to the extract. In this sense, the mixture of naringenin and quercetin has shown better antioxidant activity against the sum of their individual components47; also, the synergism phenomenon has been reported in leaves of crabapples, as the individual flavonoids showed lower values compared to mixtures of them48.
Compared to a previous study in our research group, the ORAC values were 1.4 times higher, which can be attributed to our specific process of extracting naringenin in high quantities. In contrast, in our previous work, the process was not optimized to a specific compound3. Also, compared to other supercritical extracts, our results were higher than both dry and fermented orange pomace extract (370–1260 µmol TE/g extract)49; as well against the peel oil extract from mandarin (Clementina orogrande) (433.46–738.68 µmol TE/g oil)50. In both cases, ethanol was used as cosolvent, enhancing the antioxidant response by increasing the flavanones like naringenin, naringin, and hesperetin. Also, previous studies in our research group showed lower TEAC values compared to current findings, this is due to less specific extraction condition, allowing of the extraction of a variety of compounds that can react to the ABTS radical3; but compared to other supercritical extracts rich in phenolic compounds, like from Sida rhombifolia leaves51 and cacado pod husk52, our results were higher.
Conclusion
The optimized conditions to obtain naringenin from oregano using supercritical CO2 were low pressure (166 bar) and cosolvent proportion (12.46%), and moderate temperature (58.4 °C). In addition, the optimized process yielded more cirsimaritin, quercetin, and apigenin, compared with a methanolic extraction. However, the bioaccessibility of these compounds was low, as seen in the reduction of their content and the antioxidant capacity of the extract. The use of supercritical CO2 enhances the extraction of naringenin from oregano (Lippia graveolens), and it was an effective method to obtain this flavanone; therefore, the naringenin-rich extract with health benefits can be used in different industries, and we suggest incorporating encapsulation techniques to increase the bioaccessibility of these extracts.
Materials and methods
Sample preparation
Wild Lippia graveolens were obtained in Santa Gertrudis, Durango, México (coordinates: N 23° 32′ 43.8″ W 104° 22′ 20.8″). The aerial parts (flowers, leaves, and small stems) were dried using an Excalibur Food Dehydrator Parallax Hyperware (Sacramento, CA) at 40 °C for 24 h. Dried samples were grounded to a fine powder using an Ika Werke M20 mill grinder (IKA, Germany) and were stored at − 20 °C for further experiments.
Reagents and chemicals
The nitrogen and CO2 were purchased from Linde (Culiacán, Sinaloa, Mexico), methanol and ethanol were obtained from Fermont (Mexico). The rest of the reagents were purchased from Sigma-Aldrich (St. Louis, MO, USA).
Conventional extraction
The conventional extraction was performed according to a previous report by Picos-Salas et al.3 with modifications. In brief, 0.1 g of oregano powder was mixed with 10 mL of methanol (100%) in constant shaking for 2 h. Afterward, the mixture was centrifugated at 11,627 g for 15 min at 4 °C using a Z 36 HK centrifuge (HERMLE, Germany). Then, the supernatant was collected and dried using a rotary evaporator R-300 (Buchi, Switzerland), followed by drying with nitrogen to ensure complete solvent remotion, and stored at − 20 °C until further analysis. Extraction was carried by triplicate (n = 3).
Supercritical CO2 extraction
Naringenin-rich extracts were obtained from Lippia graveolens using a MV-10 ASFE extractor (Waters Corporation, MA, USA), using CO2 as solvent and ethanol (99.9%) as cosolvent. The conditions used were selected based on literature research and preliminary studies. In brief, 2.5 g of sample was placed in a 10 mL vessel; the extraction was carried at the pressure, temperature, and cosolvent proportion according to the central composite design (Table 4). All extractions were carried at the same CO2 + cosolvent flow (5 mL/min), with static and dynamic extraction times (30 and 45 min each). All extracts were dried in a vacuum concentrator Multivapor P-12 (Buchi, Switzerland), resuspended in ethanol (99.9%), and stored at − 20 °C for further studies.
Simulated digestion
An in vitro digestion model was carried out following the protocol by Brodkorb et al.53, which consisted in a 3-step process that simulates oral, gastric, and intestinal digestion (see Supplementary Table S1). In brief, in a 50 mL tube, 1 mL of extract was mixed with 0.8 mL of oral solution, 5 µL of 0.3 M CaCl2, 0.1 mL of amylase (75 U/mL), 0.095 mL of distilled water, and incubated in an oscillator for 2 min at 37 °C. Afterward, the solution was mixed with 1.6 mL of gastric solution, and pH was adjusted to 3; then, 1 µL of 0.3 M CaCl2, 0.1 mL of pepsin (2000 U/mL), and 0.1 mL of lipase (60 U/mL) were added, and pH adjusted to 3; next, 0.199 mL of distilled water was added, and the solution was incubated during 2 h at 37 °C. Finally, 2.2 mL of gastric solution was added to the mix, and pH was adjusted to 7; then, 8 µL of 0.3 M CaCl2 and 1 mL of pancreatin (100 U/mL) were added, and pH was adjusted to 7; later, 0.792 mL of distilled water were added, and the solution was incubated for 2 h at 37 °C. After the simulated digestion process, methanol (100%) was added to precipitate proteins in 1:1 v/v, and the solution was placed at − 20 °C for 20 min; afterward, it was centrifugated at 11,627 g for 10 min and 4 °C. Finally, the supernatant was collected and named as supercritical extract intestinal phase (ISE) or methanolic extract intestinal phase (IME). Bioaccessibility (%) of each flavonoid was calculated as seen in Eq. (3):
where both flavonoid in intestinal phase and crude extract are expressed in µg of flavonoid/g extract.
Flavonoid content by UPLC-MS
The flavonoids in the extracts were evaluated based on the methodology by Bernal-Millán et al.54 using a UPLC class H (Waters Corporation, USA) coupled to a G2-XS QT mass analyzer (Quadrupole and Time of Flight). Flavonoids were separated using a UPLC BEH C18 column (1.7 μm × 2.1 mm × 100 mm) at 40 °C. Gradient elution was conducted with water-formic acid 0.1% (A) and acetonitrile (B) at the flow rate of 0.3 mL/min. The following gradient was used: 0 min, 95% (A); 5 min, 70% (A); 9 min, 30% (A); 14 min, 0% (A); 14.5 min, 0% (A); 15 min, 95% (A); and 16 min, 95% (A). Electrospray (ESI) was used for compound ionization, and the mass analysis condition was: capillary voltage of 1.5 kV, sampling cone of 30 V, desolvation gas of 800 L/h, and a temperature of 500 °C. A 0–30 V collision ramp was used. The flavonoids were identified and quantified by comparing them with a calibration curve using the corresponding standards (Supplementary Fig. S3).
Total phenolic content
The total phenolic content (TPC) was done as reported by Swain and Hillis55 with modifications. In brief, 10 µL of the extract was mixed with 230 µL of distilled water and 10 µL of the Folin–Ciocalteu reagent in a 96-well microplate. Afterward, 25 µL of 4 N Na2CO3 was added, and the mixture was incubated in darkness for 2 h. Then, absorbance was measured at 725 nm using a Synergy HT microplate reader (Bio-Tek Instruments, Inc., VT, USA). Results were expressed as mg of gallic acid equivalents per g of extract (mg GAE/g extract).
Oxygen radical absorbance capacity (ORAC)
The antioxidant capacity was measured by the ORAC assay described by Huang et al.56 using fluorescein as a fluorescent probe and 2,2′-azobis(2-methylpropionamidine) dihydrochloride (AAPH) as a peroxyl radical. A reaction mixture containing 25 µL of extract, 75 µL of 95.8 µM AAPH, and 200 µL of 0.96 µM fluorescein was put in a black-walled, clear-bottom 96-well microplate, and the reaction started when the AAPH was added. Phosphate buffer was used as a blank. The loss of fluorescence was measured every 70 s for 70 min at 485 nm for excitation and 580 nm for emission using a microplate reader. Results were expressed as µmol of Trolox equivalents per g of extract (µmol TE/g extract).
Trolox equivalent antioxidant capacity (TEAC)
The TEAC assay was carried out according to Thaipong et al.57 with some modifications. For this, the ABTS·+ radical was formed by mixing 7.4 mM ABTS with 2.4 mM potassium persulphate at a 1:1 ratio and incubated at room temperature in darkness during 12–16 h before use. Afterward, the radical was diluted with ethanol (99.9%) until an absorbance of 0.70 ± 0.02 at 734 nm. The reaction started after adding 10 µL of the extract with 190 µL of the ABTS·+ in a 96-well microplate, followed by incubation during 2 in darkness and absorbance measuring at 734 nm in a microplate reader. The ABTS·+ radical was used as a blank. Results were expressed as mmol of Trolox equivalents per g of extract (mmol TE/g extract).
XLogP3-AA and logD data obtention
XlogP3-AA values were obtained using PubChem database58, and the LogD values using the online platform Chemicalize59.
Experimental design
The naringenin supercritical CO2 extraction was optimized by the response surface methodology using the desirability function. A central composite design with three factors was used, namely pressure (X1, bar), temperature (X2, °C), and cosolvent proportion (X3, %) (Table 4), using the software Design-Expert 7.0 (Stat-Ease Inc., MN, USA). The naringenin content was used as a response variable and was measured by UPLC-MS using a naringenin standard as specified before. The optimum conditions were carried out in quadruplicate (n = 4).
Flavonoid quantification by UPLC-MS, TPC, ORAC, and TEAC were analyzed by an analysis of variance (ANOVA) with two factors (supercritical and methanolic extract) and two levels each one (extract without digestion and intestinal phase), and each analysis was performed by triplicate for the methanolic extracts, and by quadruplicate for the supercritical extract at optimized condition; also, mean comparisons were evaluated by Tukey’s HSD test using the software Minitab 19 (Minitab LLC, PA, USA). A level of p < 0.05 was considered a significant difference. Data were reported as mean ± standard error of the mean (SEM).
Data availability
All data generated or analyzed during this study is included in article and its supplementary information files.
References
Leyva-López, N., Gutiérrez-Grijalva, E., Vazquez-Olivo, G. & Heredia, J. Essential oils of oregano: Biological activity beyond their antimicrobial properties. Molecules 22, 989. https://doi.org/10.3390/molecules22060989 (2017).
Arias, J. et al. Optimization of flavonoids extraction from Lippia graveolens and Lippia origanoides chemotypes with ethanol-modified supercritical CO2 after steam distillation. Ind. Crop. Prod. 146, 112170. https://doi.org/10.1016/j.indcrop.2020.112170 (2020).
Picos-Salas, M. A. et al. Supercritical CO2 extraction of oregano (Lippia graveolens) phenolic compounds with antioxidant, α-amylase and α-glucosidase inhibitory capacity. J. Food Meas. Charact. 15, 3480–3490. https://doi.org/10.1007/s11694-021-00928-4 (2021).
Petrussa, E. et al. Plant flavonoids—Biosynthesis, transport and involvement in stress responses. Int. J. Mol. Sci. 14, 14950–14973. https://doi.org/10.3390/ijms140714950 (2013).
Angst, E. et al. The flavonoid quercetin inhibits pancreatic cancer growth in vitro and in vivo. Pancreas 42, 223–229. https://doi.org/10.1097/mpa.0b013e318264ccae (2013).
Lee, D. et al. Protective effect of cirsimaritin against streptozotocin-induced apoptosis in pancreatic beta cells. J. Pharm. Pharmacol. 69, 875–883. https://doi.org/10.1111/jphp.12719 (2017).
Picos-Salas, M. A. et al. Naringenin as a natural agent against oxidative stress and inflammation, and its bioavailability. Food Rev. Int. https://doi.org/10.1080/87559129.2022.2123502 (2022).
Rajappa, R. et al. Treatment with naringenin elevates the activity of transcription factor Nrf2 to protect pancreatic β-cells from streptozotocin-induced diabetes in vitro and in vivo. Front. Pharmacol. https://doi.org/10.3389/fphar.2018.01562 (2019).
Frías-Zepeda, M., Rosales-Castro, M., Escalona-Cardoso, G. & Paniagua, N. Ethanolic extract of Lippia graveolens stem reduce biochemical markers in a Murine Model with Metabolic Syndrome. Saudi J. Biol. Sci. 29, 103422. https://doi.org/10.1016/j.sjbs.2022.103422 (2022).
FDA. Q3C—Tables and List Guidance for Industry. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/q3c-tables-and-list-rev-4 (2018).
Lefebvre, T., Destandau, E. & Lesellier, E. Selective extraction of bioactive compounds from plants using recent extraction techniques: A review. J. Chromatogr. A 1635, 461770. https://doi.org/10.1016/j.chroma.2020.461770 (2021).
Chemat, F. et al. Green extraction of natural products. Origins, current status, and future challenges. TrAC rends Anal. Chem. 118, 248–263. https://doi.org/10.1016/j.trac.2019.05.037 (2019).
Khaw, K.-Y., Parat, M.-O., Shaw, P. N. & Falconer, J. R. Solvent supercritical fluid technologies to extract bioactive compounds from natural sources: A review. Molecules 22, 1186. https://doi.org/10.3390/molecules22071186 (2017).
Temelli, F., Saldaña, M. D. A. & Comin, L. Application of supercritical fluid extraction in food processing. in Comprehensive Sampling and Sample Preparation (ed. J. Pawliszyn) 415–440 (Academic Press, 2012).
Tyśkiewicz, K., Konkol, M. & Rój, E. The application of supercritical fluid extraction in phenolic compounds isolation from natural plant materials. Molecules 23, 2625. https://doi.org/10.3390/molecules23102625 (2018).
Bimakr, M. et al. Comparison of different extraction methods for the extraction of major bioactive flavonoid compounds from spearmint (Mentha spicata L.) leaves. Food Bioprod. Process. 89, 67–72. https://doi.org/10.1016/j.fbp.2010.03.002 (2011).
Santos, S. A. O., Villaverde, J. J., Silva, C. M., Neto, C. P. & Silvestre, A. J. D. Supercritical fluid extraction of phenolic compounds from Eucalyptus globulus Labill bark. J. Supercrit. Fluid. 71, 71–79. https://doi.org/10.1016/j.supflu.2012.07.004 (2012).
Kus, P., Jerkovic, I., Jakovljevic, M. & Jokic, S. Extraction of bioactive phenolics from black poplar (Populus nigra L.) buds by supercritical CO2 and its optimization by response surface methodology. J. Pharm. Biomed. Anal. 152, 128–136. https://doi.org/10.1016/j.jpba.2018.01.046 (2018).
Cardoso, C., Afonso, C., Lourenço, H., Costa, S. & Nunes, M. L. Bioaccessibility assessment methodologies and their consequences for the risk–benefit evaluation of food. Trends Food Sci. Technol. 41, 5–23. https://doi.org/10.1016/j.tifs.2014.08.008 (2015).
Chen, G.-L. et al. Total phenolic, flavonoid and antioxidant activity of 23 edible flowers subjected to in vitro digestion. J. Funct. Foods 17, 243–259. https://doi.org/10.1016/j.jff.2015.05.028 (2015).
Gutiérrez-Grijalva, E. P., Antunes-Ricardo, M., Acosta-Estrada, B. A., Gutiérrez-Uribe, J. A. & Heredia, J. B. Cellular antioxidant activity and in vitro inhibition of α-glucosidase, α-amylase and pancreatic lipase of oregano polyphenols under simulated gastrointestinal digestion. Food Res. Int. 116, 676–686. https://doi.org/10.1016/j.foodres.2018.08.096 (2019).
Derringer, G. & Suich, R. Simultaneous optimization of several response variables. J. Qual. Technol. 12, 214–219. https://doi.org/10.1080/00224065.1980.11980968 (1980).
Núñez, G. A., del Valle, J. M. & de la Fuente, J. C. Solubilities in supercritical carbon dioxide of (2E,6E)-3,7,11-trimethyldodeca-2,6,10-trien-1-ol (farnesol) and (2S)-5,7-dihydroxy-2-(4-hydroxyphenyl)chroman-4-one (naringenin). J. Chem. Eng. Data 55, 3863–3868. https://doi.org/10.1021/je900957v (2010).
Al-Rawi, S. S. et al. The effect of supercritical fluid extraction parameters on the nutmeg oil extraction and its cytotoxic and antiangiogenic properties. Procedia Food Sci. 1, 1946–1952. https://doi.org/10.1016/j.profoo.2011.09.286 (2011).
Atwi-Ghaddar, S., Destandau, E. & Lesellier, E. Optimization of supercritical fluid extraction of polar flavonoids from Robinia pseudoacacia L. heartwood. J. Utilization. 70, 102440. https://doi.org/10.1016/j.jcou.2023.102440 (2023).
Bimakr, M. et al. Optimization of supercritical carbon dioxide extraction of bioactive flavonoid compounds from spearmint (Mentha spicata L.) leaves by using response surface methodology. Food Bioprocess Tech. 5, 912–920. https://doi.org/10.1007/s11947-010-0504-4 (2012).
Bitencourt, R. G., Palma, A. M., Coutinho, J. A. P., Cabral, F. A. & Meirelles, A. J. A. Solubility of caffeic acid in CO2+ ethanol: Experimental and predicted data using Cubic Plus Association Equation of State. J. Supercrit. Fluid. 138, 238–246. https://doi.org/10.1016/j.supflu.2018.04.008 (2018).
Michielin, E. M. Z. et al. Chemical composition and antibacterial activity of Cordia verbenacea extracts obtained by different methods. Bioresour. Technol. 100, 6615–6623. https://doi.org/10.1016/j.biortech.2009.07.061 (2009).
Putra, N. R. et al. Extraction rate of valuable compounds from peanut skin waste by ethanol-assisted supercritical carbon dioxide: modelling and optimization. Malay. J. Fundamental Appl. Sci. 18, 157–170. https://doi.org/10.11113/mjfas.v18n2.2237 (2022).
Liza, M. S. et al. Supercritical carbon dioxide extraction of bioactive flavonoid from Strobilanthes crispus (Pecah Kaca). Food Bioprod. Process. 88, 319–326. https://doi.org/10.1016/j.fbp.2009.02.001 (2010).
Xie, L. et al. Comparison of flavonoid O-glycoside, C-glycoside and Their Aglycones On Antioxidant Capacity And Metabolism During In Vitro Digestion And In Vivo. Foods 11, 882. https://doi.org/10.3390/foods11060882 (2022).
Bernal-Millán, M. D. J. et al. Green extracts and UPLC-TQS-MS/MS profiling of flavonoids from Mexican Oregano (Lippia graveolens) using natural deep eutectic solvents/ultrasound-assisted and supercritical fluids. Plants 12, 1692. https://doi.org/10.3390/plants12081692 (2023).
Morzel, M., Canon, F. & Guyot, S. Interactions between salivary proteins and dietary polyphenols: Potential consequences on gastrointestinal digestive events. J. Agric. Food Chem. 70, 6317–6327. https://doi.org/10.1021/acs.jafc.2c01183 (2022).
Friedman, M. & Jürgens, H. S. Effect of pH on the stability of plant phenolic compounds. J. Agric. Food Chem. 48, 2101–2110. https://doi.org/10.1021/jf990489j (2000).
Lamothe, S., Azimy, N., Bazinet, L., Couillard, C. & Britten, M. Interaction of green tea polyphenols with dairy matrices in a simulated gastrointestinal environment. Food Funct. 5, 2621–2631. https://doi.org/10.1039/C4FO00203B (2014).
He, Q., Lv, Y. & Yao, K. Effects of tea polyphenols on the activities of α-amylase, pepsin, trypsin and lipase. Food Chem. 101, 1178–1182. https://doi.org/10.1016/j.foodchem.2006.03.020 (2006).
Gutiérrez-Grijalva, E. P., Angulo-Escalante, M. A., León-Félix, J. & Heredia, J. B. Effect of in vitro digestion on the total antioxidant capacity and phenolic content of 3 species of Oregano (Hedeoma patens, Lippia graveolens, Lippia palmeri). J. Food Sci. 82, 2832–2839. https://doi.org/10.1111/1750-3841.13954 (2017).
Junior, M. R. M., Leite, A. V. & Dragano, N. R. V. Supercritical fluid extraction and stabilization of phenolic compounds from natural sources—Review (supercritical extraction and stabilization of phenolic compounds). Open Chem. Eng. J. 4, 51–60. https://doi.org/10.2174/1874123101004010051 (2010).
Pereira, P., Cebola, M.-J., Oliveira, M. C. & Bernardo-Gil, M. G. Supercritical fluid extraction vs conventional extraction of myrtle leaves and berries: Comparison of antioxidant activity and identification of bioactive compounds. J. Supercrit. Fluid. 113, 1–9. https://doi.org/10.1016/j.supflu.2015.09.006 (2016).
Martínez-Rocha, A., Puga, R., Hernández-Sandoval, L., Loarca-Piña, G. & Mendoza, S. Antioxidant and antimutagenic activities of Mexican Oregano (Lippia graveolens Kunth). Plant Foods Hum. Nutr. 63, 1–5. https://doi.org/10.1007/s11130-007-0061-9 (2008).
Uwineza, P. A., Gramza-Michałowska, A., Bryła, M. & Waśkiewicz, A. Antioxidant activity and bioactive compounds of lamium album flower extracts obtained by supercritical fluid extraction. Appl. Sci. 11, 7419. https://doi.org/10.3390/app11167419 (2021).
Odriozola-Serrano, I. et al. Stability and bioaccessibility of phenolic compounds in rosehip extracts during in vitro digestion. Antioxidants 12, 1035. https://doi.org/10.3390/antiox12051035 (2023).
Tagliazucchi, D., Verzelloni, E., Bertolini, D. & Conte, A. In vitro bio-accessibility and antioxidant activity of grape polyphenols. Food Chem. 120, 599–606. https://doi.org/10.1016/j.foodchem.2009.10.030 (2010).
Munteanu, I. G. & Apetrei, C. Analytical methods used in determining antioxidant activity: A review. Int. J. Mol. Sci. 22, 3380. https://doi.org/10.3390/ijms22073380 (2021).
Galano, A. et al. Food antioxidants: Chemical insights at the molecular level. Annu. Rev. Food Sci. Technol. 7, 335–352. https://doi.org/10.1146/annurev-food-041715-033206 (2016).
Ohkatsu, Y., Sakurai, T. & Sato, T. Relationship between chemical structure and antioxidant function of flavonoids. J Jpn. Petrol. Inst. 53, 213–221. https://doi.org/10.1627/jpi.53.213 (2010).
Baranowska, M. et al. Interactions between polyphenolic antioxidants quercetin and naringenin dictate the distinctive redox-related chemical and biological behaviour of their mixtures. Sci. Rep. 11, 12282. https://doi.org/10.1038/s41598-021-89314-0 (2021).
Qin, X., Lu, Y., Peng, Z., Fan, S. & Yao, Y. Systematic chemical analysis approach reveals superior antioxidant capacity via the synergistic effect of flavonoid compounds in red vegetative tissues. Front. Chem. https://doi.org/10.3389/fchem.2018.00009 (2018).
Espinosa-Pardo, F. A., Nakajima, V. M., Macedo, G. A., Macedo, J. A. & Martínez, J. Extraction of phenolic compounds from dry and fermented orange pomace using supercritical CO2 and cosolvents. Food Bioprod. Process. 101, 1–10. https://doi.org/10.1016/j.fbp.2016.10.002 (2017).
Quispe-Fuentes, I., Uribe, E., López, J., Contreras, D. & Poblete, J. A study of dried mandarin (Clementina orogrande) peel applying supercritical carbon dioxide using co-solvent: Influence on oil extraction, phenolic compounds, and antioxidant activity. J. Food Process. Preserv. https://doi.org/10.1111/jfpp.16116 (2022).
Ferro, D. M., Mazzutti, S., Vitali, L., Müller, C. M. O. & Ferreira, S. R. S. Integrated extraction approach to increase the recovery of antioxidant compounds from Sida rhombifolia leaves. J. Supercrit. Fluid. 149, 10–19. https://doi.org/10.1016/j.supflu.2019.03.013 (2019).
Valadez-Carmona, L., Ortiz-Moreno, A., Ceballos-Reyes, G., Mendiola, J. A. & Ibáñez, E. Valorization of cacao pod husk through supercritical fluid extraction of phenolic compounds. J. Supercrit. Fluid. 131, 99–105. https://doi.org/10.1016/j.supflu.2017.09.011 (2018).
Brodkorb, A. et al. INFOGEST static in vitro simulation of gastrointestinal food digestion. Nat. Protoc. 14, 991–1014. https://doi.org/10.1038/s41596-018-0119-1 (2019).
Bernal-Millán, M. D. J. et al. Spray-dried microencapsulation of Oregano (Lippia graveolens) polyphenols with maltodextrin enhances their stability during in vitro digestion. J. Chem.-NY 2022, 1–10. https://doi.org/10.1155/2022/8740141 (2022).
Swain, T. & Hillis, W. E. The phenolic constituents of Prunus domestica. I.—The quantitative analysis of phenolic constituents. J. Sci. Food Agric. 10, 63–68. https://doi.org/10.1002/jsfa.2740100110 (1959).
Huang, D., Ou, B., Hampsch-Woodill, M., Flanagan, J. A. & Prior, R. L. High-throughput assay of oxygen radical absorbance capacity (ORAC) using a multichannel liquid handling system coupled with a microplate fluorescence reader in 96-well format. J. Agric. Food Chem. 50, 4437–4444. https://doi.org/10.1021/jf0201529 (2002).
Thaipong, K., Boonprakob, U., Crosby, K., Cisneros-Zevallos, L. & Hawkins Byrne, D. Comparison of ABTS, DPPH, FRAP, and ORAC assays for estimating antioxidant activity from guava fruit extracts. J. Food Compost. Anal. 19, 669–675. https://doi.org/10.1016/j.jfca.2006.01.003 (2006).
PubChem. https://pubchem.ncbi.nlm.nih.gov/ (2023).
Chemicalize. https://chemicalize.com/app/calculation (2023).
Acknowledgements
This paper is included in the Cátedras CONAHCYT Project #397.
Author information
Authors and Affiliations
Contributions
M.A.P.S. conceptualized the idea, conducted the research, and prepared the manuscript; N.L.L. reviewed the manuscript; P.d.J.B.B. did the UPLC-MS analysis; M.A.R. reviewed the manuscript; J.B.H. obtained funding and reviewed the manuscript; L.A.C.B. supervised the statistical analysis, reviewed the manuscript; M.A.A.E. reviewed the manuscript; E.P.G.G. conceptualized the idea, obtained funding, supervised the research, and reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Picos-Salas, M.A., Leyva-López, N., Bastidas-Bastidas, P.d. et al. Supercritical CO2 extraction of naringenin from Mexican oregano (Lippia graveolens): its antioxidant capacity under simulated gastrointestinal digestion. Sci Rep 14, 1146 (2024). https://doi.org/10.1038/s41598-023-50997-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-50997-2
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.