Abstract
We aimed to compare two assessment methodologies (real-time vs. video-recorded) using the Unesp-Botucatu Pig Composite Acute Pain Scale (UPAPS) in piglets before and after castration. Twenty-nine male piglets were castrated. Four observers scored the UPAPS over three perioperative timepoints of castration following two assessment methodologies. In real-time assessments, the observers were in-person observing the piglets in front of the pen. After two weeks, the observers did video-recorded assessments randomizing piglets and timepoints. Modeling was conducted to compare the UPAPS and each pain-altered behavior between methodologies. Intraclass correlation coefficient (ICC), Bland–Altman, and Lin’s concordance correlation coefficient (CCC) were conducted to investigate agreement between methodologies. UPAPS was statistically equivalent between methodologies (P = 0.4371). The ICC for each method was very good (0.85 to 0.91). The agreement of the UPAPS assessed between methodologies had minimal bias (− 0.04), no proportion bias, and 53% of the assessments presented a perfect agreement. However, CCC of the UPAPS was moderate (0.65), and only one pain-altered behavior (“presents difficulty in overcoming obstacles or other animals”) occurred more in real-time assessments (P = 0.0444). In conclusion, piglet pain assessment by UPAPS can be conducted in real-time based on a suitable agreement between the real-time and video-recorded assessment methods.
Similar content being viewed by others
Introduction
From a global perspective, millions of pigs are raised in conditions that can result in individual’s experiencing pain1,2. In both commercial farm settings and research laboratories, painful situations can occur as a result of direct pain events inflicted on animals (e.g. tail docking, teeth resection, castration, ear tagging, and notching2,3) as well as indirect events resulting in pain experienced as a by-product of disease or management1,2.
In addition to the animal welfare implications associated with pain experienced by pigs, Sus scrofa is a common model used in translational biomedical research that frequently involves painful procedures such as organ transplantation, stem cell therapy, and endo- and laparoscopic procedures4. Failure to effectively identify, assess and treat pain in pigs can significantly influence research outcomes, resulting in potential bias and unpredictable results5,6,7.
Pain assessment in pigs has been studied over the last 30 years in detail8,9,10,11,12,13,14,15,16,17. Currently, there are five species-specific pain scales used in swine including the Piglet Grimace Scale18,19,20,21,22, Sow Grimace Scale23, Unesp-Botucatu Pig Composite Acute Pain Scale (UPAPS)8,24, and two additional behavioral pain scales25,26. In order to accurately assess pain, tools used must demonstrate a high evidence of validation based on robust scientific guidelines such as the Consensus-based Standards for the Selection of Health Measurement Instruments (COSMIN)27. According to these guidelines, the UPAPS represents the most robust assessment tool for measuring pain in pigs as demonstrated by its good repeatability and reproducibility, sensitivity, specificity, responsiveness, and excellent internal consistency. In addition, this tool has the discriminatory ability to diagnose pain across ages including newborn24 and mature pigs8.
Traditionally, the previous pain scales discussed, including the UPAPS, were developed using videos or photos8,18,22,23,24,25, because it is an essential step needed to conduct masked analyzes and intraobserver reliability28,29. However, relying on videos and photos in a research or farm setting is less practical given the logistical considerations such as equipment requirements, labor and time needed to collect, edit and assess videos7,30,31. Therefore, to implement pain assessments in a practical way, a real-time assessment approach is needed.
Real-time application of pain scales has been successful in rats7 and felines30, demonstrating accuracy in the tool and allowing clinicians to provide immediate analgesic intervention for the individuals expressing pain. However, although the UPAPS have been validated for pigs8,24, the efficacy of real-time assessment are still unknown.
Therefore, this study aimed to compare two assessment methodologies (real-time vs. video-recorded) using UPAPS in piglets before and after castration. The hypothesis is that there will be no differences between the real-time and video-recorded assessments and both assessments will have suitable agreement.
Results
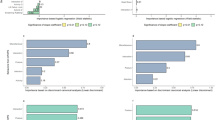
The UPAPS presented an overdispersion of zeros based on the histogram (Fig. S1A) and Cameron and Trivedi’s test (Lambda t-test score = 9.55 and P < 0.0001). The count of UPAPS zeros was higher at 1 h pre-castration (n = 154) and 3 h post-castration (n = 110) than immediately post-castration (n = 73), and this unbalance explains the best model considering timepoints in the model logistic component. Assessment methods showed a similar balance of count of UPAPS zeros (n = 140 in real-time and n = 143 in video-recorded) and were not included in the logistic component model (Fig. S1B). Multilevel zero-inflated negative binomial model parameters are depicted in the supplementary material (Table S1). In the post-hoc test, the UPAPS was lower at 1 h pre-castration and 3 h post-castration than immediately post-castration (P < 0.0001) (Fig. 1A) but was statistically equivalent between assessment methods (P = 0.4371) (Fig. 1B). Meanwhile, the UPAPS pain-altered behavior “difficulty overcoming obstacles” occurred more in real-time assessment than video-recorded (P = 0.0444) (Table 1).
The interobserver reliability of the UPAPS by intraclass correlation coefficient (ICC) was very good in real-time assessment (ICC = 0.92) and in video-recorded assessment (ICC = 0.85) (Table 2).
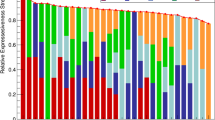
The agreement of the UPAPS assessed in real-time and video-recorded methods had minimal bias (− 0.04) and limit of agreement (LoA; − 4.40 to 4.30), and CCC of 0.65 (Fig. 2). The greater majority of the evaluations (53%) showed perfect agreement (no difference) between the two methods, some of the agreements (40%) had differences within the LoA, and only a few evaluations (7%) had differences beyond the LoA. The slope coefficient (β) of the mean between the two assessment methods (β = 0.05; P = 0.2690) was not significant (Table S2), suggesting no proportional bias. The model (χ2 = 185.89; P < 0.0001) showed variance not constant (heteroskedasticity behavior) by the Breusch Pagan test and by the pattern in ‘cone’ or ‘V’ format of data distribution in the Bland–Altman plot.
Bland–Altman test of Unesp-Botucatu Pig Composite Acute Pain Scale (UPAPS) assessed in real-time and video-recorded method (LoA is limit of agreement; CI is 95% confidence interval; solid line is the bias; dashed line is the lower and upper LoA; dotted lines is the 95% confidence interval for bias and 90% confidence interval for lower and upper LoA; CCC is Lin’s concordance correlation coefficient; green line is the simple linear model).
Discussion
Pain diagnosis is an essential step for pain management and relief in pigs and has the potential to improve the quality of life for millions of pigs experiencing painful procedures in farm and laboratory settings around the world2,3. In a translational biomedical research context, the untreated or under-treated pain may act as a confounding factor in the study contributing to poor quality of data and questioning ethical experimental practices5,6,7. As a response to this, objective approaches to assess pain, including the Unesp-Botucatu Pig Composite Acute Pain Scale (UPAPS) are needed to successfully identify and manage pigs experiencing pain27,28,29. However, assessing piglet pain has been traditionally conducted using video recordings which is a less practical approach given the logistical/financial challenges of placing video equipment in such settings and ethical concerns regarding the inability to provide real-time medical intervention and rescue analgesia in suffering animals. Therefore, the objective of this study was to compare two assessment methodologies (real-time vs. video-recorded) on a validated pain-altered behavior scale used in piglets before and after castration.
Behavioral assessment methodology (real-time vs. video-recorded) did not influence total pain scores across timepoints relative to castration. Both methodologies demonstrated sufficient agreement between one another with minimal bias, no evidence of proportional bias, and good interobserver reliability. In addition, most of assessments between methodologies presented perfect agreement, while most differences fell within the estimated limit of agreement (LoA). The results from the current study are in agreement with previous work evaluating real-time methodology assessment for pain evaluation grimace scales in felines30 and rats7, demonstrating sufficient agreement in pain assessment when utilizing real-time or video-recorded assessment. From a swine-specific perspective, the present study is beneficial as it can permit trained laboratory technicians and swine farm caretakers to assess pain in swine in real-time thus providing a means to assess, diagnose and treat pain conditions in a timely manner.
Although total pain score assessment was not different, it should be noted that the Lin’s concordance correlation coefficient (CCC) between methodologies was only moderate, and the variance of the data was inconsistent (heteroskedasticity). Based on bias, observers in this study slightly underestimated total pain scores using real-time assessment. These results are in agreement with previous work reported in felines30 and rats7. These deviations also mirror work conducted in mice with grimace scales that demonstrated lower total pain scores using real-time assessments compared to photographs edited from video-recordings31. These minor disagreements between assessment methodologies may be partially explained by observer fatigue and attention. Real-time assessment requires constant attention to the piglet in a distracting environment, which may be a potential source of fatigue as reported in previous studies7,30,31. In this situation, environmental conditions (e.g., sound, movement, other activities) could have influenced the observer making them less accurate in observing subtle individual behaviors. In contrast, video-recorded assessment was performed in the laboratory or personal office with minimal to no distractors and observers had the option to watch the video repeatedly and take short breaks in between video assessment, thus potentially explaining why real-time assessment underestimated scores compared to recorded. Additional work is needed to understand what additional factors influence this minor bias, including future studies looking at the impact of observer gender and experience on bias.
This study is not free of limitations. The number of times each observer watched each video was not recorded and the number of videos per day or hour was not standardized and these issues may have benefited the video-recorded assessments in comparison with real-time assessments. In addition, the presence of the human may have influenced the expression of pain-altered behaviors as a response to stress-induced analgesia33,34, thus influencing total pain scores for both methodologies.
Practical implications of the study include facilitating pain diagnosis by the use of the UPAPS in real-time in laboratory and farm settings, enabling immediate pain diagnosis and medical intervention. Also, the findings presented herein may benefit the approval process for drugs to relieve pain in swine that can only be labeled if they are proven efficacious using validated tools, and currently, in the United States there are no label-approved drugs to relieve pain in swine. Future studies may analyze the effect of experience, gender, training, and cultural aspects of the observers in the pain-altered behaviors in other painful conditions.
In conclusion, piglet pain assessment by UPAPS can be conducted in real-time based on suitable agreement between the real-time and video-recorded assessment methods, however, strategies need to be devised to overcome the smooth deficiencies in agreement between both methods.
Materials and methods
This study was approved by the Institutional Animal Care and Use Committee of North Carolina State University (IACUC protocol 20-113). Animals were cared for and handled in accordance with the Guide for the Care and Use of Agricultural Animals in Research and Teaching35. This study was conducted on a commercial sow farm located in the Southeastern United States as part of a larger study36. No animals were castrated exclusively for the purposes of this study, the piglets’ castration was a regular routine of the farm, which contributes with two of the four R’s of animal experimentation (reduce and responsibility37,38). Recognizing castration is painful, all enrolled piglets received pain management before the procedure. The study is reported in accordance with ARRIVE guidelines.
Animals, housing, and procedures
A total of 29 Large White x Duroc cross male piglets from 15 l (1.9 piglets per litter average) were enrolled in this study. Prior to enrollment, piglets were assessed using enrollment criteria described in Table 3. All male piglets were enrolled in each litter for the study (range 1–10 males enrolled per litter). Male piglets meeting all enrollment criteria were then weighed and individually identified using a permanent marker on the back. Castration was performed on each piglet by one trained farm employee. Twenty minutes prior to castration, all piglets received the following pain-control protocol: 3 ml of 2% buffered Lidocaine HCl injectable solution (Lidocaine Hydrochloride, Covetrus, Dublin, Ohio, US) administered intra-inguinal (1.5 ml per inguinal canal) and 2.2 mg/kg of flunixin meglumine (Banamine®, Merck Animal Health, Madison, NJ, US) administered intranasally. Piglets were then placed in dorsal recumbency, and two vertical incisions were made using a scalpel blade. Once the incisions were made, testicles were exposed, spermatic cords cut, and testicles were completely removed by traction.
Piglets were housed with sows on fully slatted, tunnel ventilated farrowing rooms. Room temperature was managed through a computerized control system at 22° ± 1.0 °C for the sow and heat mats for piglets were set to approximately 30–35 °C. Within each room, sows and litters were housed in individual farrowing crates (2.5 × 0.7 m) with additional space for piglets (2.5 × 1.3 m) surrounding the crates. Lighting was turned on between 06:00 h and 16:30 h.
Behavioral pain scale
Pain scores were quantified utilizing the previously validated Unesp-Botucatu Pig Composite Acute Pain Scale (UPAPS) for pre-weaned piglets8,24 (Table 4). The pain scale consisted of five behavioral items, with each item sub-categorized into four descriptive levels. A numerical score was designated from ‘0’ to ‘3’, with a ‘0’ representing normal behavior (free of pain) and '1’, ‘2’ and ‘3’ corresponding to proportional to pain intensity pronounced behavioral deviation. The total pain score of UPAPS (0–15) was considered to assess pain in the previous studies8,24. Behavior was assessed continuously for 4-min at the following three timepoints: 1 h before castration, immediately post-castration, and 3 h post-castration. The timepoints were chosen based on the pain intensity reported by previous studies assessing pain in swine8,24,36,39. Before castration, it is expected that pigs will not be experiencing pain. Moderate to intense pain is expected immediately post-castration, while at 3 h hours post-castration pigs are expected to be pain free or feel mild pain.
Observers and training
A total of four observers were used to collect pain assessment scores over the course of the trial. All observers were veterinary medicine students, currently enrolled at the university within the second or third year of the program. Two of the observers self-identified as females and two as males, one of each gender had familiarity with the swine industry and the other had no familiarity.
Prior to on-farm data collection, all observers underwent three 2 h training sessions conducted by one of the co-authors with previous experience with UPAPS (MLS). During this session, the trainer provided video examples of each behavior with written definitions and descriptions. Following this training, observers scored 20 4-min videos of piglets in pain (post-castration) and pain-free (pre-castration) using the UPAPS. Videos were assessed in random order and observers were masked to piglet state (pain or pain-free). These videos were different from the ones used for the assessments in the present study. The observers had a very good level of agreement (0.917 [0.839 to 0.964 95% confidence interval]) with the trainer based on the intraclass correlation coefficient.
Pain assessment method
The behavioral pain scale was assessed using two behavioral methodologies.
Real-time assessment
Observers stood within 30 cm of the perimeter of the farrowing crate to observe the piglets. Two observers were at the front and two at the back of the crate. Observers were quiet, had minimum movements and had little to no contact with the piglets. Each piglet was observed for 4 min per timepoint.
Video-recorded assessment
Video was recorded in parallel with real-time assessment using a high-definition camera (Sony HDR-CX405®; New York, NY, USA) placed on a tripod approximately 30 cm from the crate at a 122-cm height. The positioning of the camera was the position of the observers during real-time assessment at the front of the crate. A total of 177 videos were obtained (29 per timepoint, 348 min of video-recorded in total; each piglet was filmed for 4 min per timepoint). No video edition was performed. Video clips were assessed for quality and then masked by a senior researcher (MPG) who did not perform any video-recorded assessment. Video order was randomized for each observer and video-recorded assessments occurred 15 days following real-time assessment.
Statistical analysis
Data were analyzed using R software within the integrated RStudio environment (Version 4.1.0; 2021-06-29; RStudio, Inc., Boston, MA, USA). The functions and packages used were presented in the format ‘package::function’ corresponding to the computer programming language in R. A significance of 5% was considered for all tests. A palette of colors distinguishable by people with common forms of color blindness was used in all figures (ggplot2::scale_colour_viridis_d).
Modeling was conducted to compare real-time pain assessments versus video-recorded considering other effects of experimental design. The histogram plot (stats::hist) and Cameron and Trivedi’s test (overdisp::overdisp) proved the overdispersion (excess of zeros) in UPAPS, requiring a zero-inflated model. Zero-inflated models combine logistic and count distributions in the fixed effects of the same model for a better fit of the data40. Then, a multilevel zero-inflated negative binomial model (glmmTMB::glmmTMB) was identified as the best fit compared with other models according to the Bayesian information criterion (stats::BIC). The UPAPS was used as the response variable, while the assessment methods, and timepoints were used as explanatory variables in the model count component (negative binomial distribution). Timepoints were included as explanatory variables in the model logistic component (Bernoulli distribution). Piglets nested within each litter and observers were included as random effects of the model. In addition, the all behavioral items from the UPAPS were converted into dummy variables (0 = absence and 1 = presence of each level of each item) (fastDummies::dummy_columns) and used as response variable in a multilevel binomial logistic model (lme4::glmer) with the same fixed effects of the model count component and random effects described previously. For all models, the Bonferroni was used for adjusting the multiple comparisons in the post-hoc test (lsmeans::lsmeans and multcomp::cld).
Intraclass correlation coefficient (ICC), two-way random effects model, type agreement multiple observers/measurements, and its 95% confidence interval (CI) (irr::icc) were used to assess the interobserver reliability of the UPAPS.
Bland–Altman test for repeated measures and Lin’s concordance correlation coefficient (CCC) (SimplyAgree::agree_reps) were used to verify the agreement of UPAPS assessed in real-time and video-recorded method. Bland–Altman analysis is enabled to detect the bias referring to the difference between two measurement methods41. In addition, the Bland–Altman analysis provides the limit of agreement (LoA), which indicates the expected range that most differences between methods should occur41. A simple linear regression (stats::lm) was conducted to analyze the proportion bias between both assessment methods. Proportional bias represents an increase in the difference between the methods evaluated at higher or lower UPAPS. Then, the difference of UPAPS between the two assessment methods was used as a response variable and the mean of UPAPS between the two methods was used as an explanatory variable. Heteroskedasticity was tested by Breusch Pagan test (olsrr::ols_test_breusch_pagan).
Data availability
All data analysed during this study are included in its Supplementary Information files.
References
Guatteo, R. & Guémené, D. Sources of known and/or potential pain in farm animals. Adv. Anim. Biosci. 5, 319–332 (2014).
Ison, S. H., Eddie Clutton, R., Di Giminiani, P. & Rutherford, K. M. D. A review of pain assessment in pigs. Front. Vet. Sci. 3, 108 (2016).
Steagall, P. V., Bustamante, H., Johnson, C. B. & Turner, P. V. Pain management in farm animals: Focus on cattle, sheep and pigs. Animal 11, 1483 (2021).
Bergen, W. G. Pigs (Sus scrofa) in biomedical research. Adv. Exp. Med. Biol. 1354, 335–343 (2022).
Bertrand, H. G. M. J., Sandersen, C. & Flecknell, P. A. Reported analgesic and anaesthetic administration to non-human primates undergoing experimental surgical procedure: 2010–2015. J. Med. Primatol. 47, 217–225 (2018).
Carbone, L. Pain in laboratory animals: The ethical and regulatory imperatives. PLoS ONE 6, e21578 (2011).
Leung, V., Zhang, E. & Pang, D. S. J. Real-time application of the rat grimace scale as a welfare refinement in laboratory rats. Sci. Rep. 6, 1–12 (2016).
Luna, S. P. L. et al. Validation of the UNESP-Botucatu pig composite acute pain scale (UPAPS). PLoS ONE 15, e0233552 (2020).
Sutherland, M. A., Davis, B. L., Brooks, T. A. & McGlone, J. J. Physiology and behavior of pigs before and after castration: Effects of two topical anesthetics. Animal 4, 2071–2079 (2010).
Sutherland, M. A., Davis, B. L., Brooks, T. A. & Coetzee, J. F. The physiological and behavioral response of pigs castrated with and without anesthesia or analgesia. J. Anim. Sci. 90, 2211–2221 (2012).
Sutherland, M. A., Davis, B. L. & McGlone, J. J. The effect of local or general anesthesia on the physiology and behavior of tail docked pigs. Animal 5, 1237–1246 (2011).
Carroll, J. A., Berg, E. L., Strauch, T. A., Roberts, M. P. & Kattesh, H. G. Hormonal profiles, behavioral responses, and short-term growth performance after castration of pigs at three, six, nine, or twelve days of age. J. Anim. Sci. 84, 1271–1278 (2006).
Torrey, S., Devillers, N., Lessard, M., Farmer, C. & Widowski, T. Effect of age on the behavioral and physiological responses of piglets to tail docking and ear notching1. J. Anim. Sci. 87, 1778–1786 (2009).
Leidig, M. S., Hertrampf, B., Failing, K., Schumann, A. & Reiner, G. Pain and discomfort in male piglets during surgical castration with and without local anaesthesia as determined by vocalisation and defence behaviour. Appl. Anim. Behav. Sci. 116, 174–178 (2009).
Morrison, R. & Hemsworth, P. Tail docking of piglets 2: Effects of meloxicam on the stress response to tail docking. Animals 10, 1–9 (2020).
Hay, M., Vulin, A., Génin, S., Sales, P. & Prunier, A. Assessment of pain induced by castration in piglets: Behavioral and physiological responses over the subsequent 5 days. Appl. Anim. Behav. Sci. 82, 201–218 (2003).
McGlone, J. J., Nicholson, R. I., Hellman, J. M. & Herzog, D. N. The development of pain in young pigs associated with castration and attempts to prevent castration-induced behavioral changes. J. Anim. Sci. 71, 1441–1446 (1993).
Viscardi, A. V., Hunniford, M., Lawlis, P., Leach, M. & Turner, P. V. Development of a piglet grimace scale to evaluate piglet pain using facial expressions following castration and tail docking: A pilot study. Front. Vet. Sci. 4, 51 (2017).
Vullo, C. et al. Is the piglet grimace scale (PGS) a useful welfare indicator to assess pain after cryptorchidectomy in growing pigs? Animal 10, 412 (2020).
Viscardi, A. V. & Turner, P. V. Efficacy of buprenorphine for management of surgical castration pain in piglets. BMC Vet. Res. 14, 1–12 (2018).
Marchant-Forde, J. N., Rutherford, K., Colpoys, J. D., Viscardi, A. V. & Turner, P. V. Use of meloxicam or ketoprofen for piglet pain control following surgical castration pain evaluation in piglets post-castration. Front. Vet. Sci. 5, 299 (2018).
Di Giminiani, P. et al. The assessment of facial expressions in piglets undergoing tail docking and castration: Toward the development of the piglet grimace scale. Front. Vet. Sci. 3, 100 (2016).
Navarro, E., Mainau, E. & Manteca, X. Development of a facial expression scale using farrowing as a model of pain in sows. Animals 10, 1–11 (2020).
Robles, I. et al. Validation of the Unesp-Botucatu pig composite acute pain scale (UPAPS) in piglets undergoing castration. PLoS ONE 18, e0284218 (2023).
Dolores Contreras-Aguilar, M. et al. Application of a score for evaluation of pain, distress and discomfort in pigs with lameness and prolapses: Correlation with saliva biomarkers and severity of the disease. Res. Vet. Sci. 126, 155–163 (2019).
Nodari, R. S. et al. Validation of a behavioural pain scale in piglets undergoing castration. In Atti della Soceità Italiana di Patologia ed Allevamento dei Suini 117–125 (2011).
Tomacheuski, R. M. et al. Measurement properties of pain scoring instruments in farm animals: A systematic review using the COSMIN checklist. PLoS ONE 18, e0280830 (2023).
Streiner, D. L., Norman, G. R. & Cairney, J. Health Measurement Scales—A Practical Guide to Their Development and Use Vol. 60 (Oxford University Press, 2015).
Mokkink, L. B. et al. COSMIN Manual for Systematic Reviews of PROMs COSMIN Methodology for Systematic Reviews of Patient-Reported Outcome Measures (PROMs) User Manual (2018).
Evangelista, M. C. et al. Clinical applicability of the Feline Grimace Scale: Real-time versus image scoring and the influence of sedation and surgery. PeerJ 2020, e8967 (2020).
Miller, A. L. & Leach, M. C. The mouse grimace scale: A clinically useful tool? PLoS ONE 10, 1–10 (2015).
Altman, D. G. Practical Statistics for Medical Research (Chapman and Hall/CRC, 1991).
Pinho, R. H., Leach, M. C., Minto, B. W., Rocha, F. D. L. & Luna, S. P. L. Postoperative pain behaviours in rabbits following orthopaedic surgery and effect of observer presence. PLoS ONE 15, e0240605 (2020).
Sorge, R. E. et al. Olfactory exposure to males, including men, causes stress and related analgesia in rodents. Nat. Methods 11, 629–632 (2014).
FASS (The Federation of Animal Science Societies). Guide for the Care and Use of Agricultural Animals in Agricultural Research and Teaching 4th edn, 128–137 (FASS, 2020).
Lopez-Soriano, M. et al. Efficacy of inguinal buffered lidocaine and intranasal flunixin meglumine on mitigating physiological and behavioral responses to pain in castrated piglets. Front. Pain Res. 4, 1156873 (2023).
Banks, R. E. The 4th R of research. Contemp. Top. Lab. Anim. Sci. 34, 50–51 (1995).
Russell, W. M. S. & Burch, R. L. The Principles of Humane Experimental Technique (Methuen Publishing Ltd, 1959).
Lopez-Soriano, M., Rocha Merenda, V., Esteves Trindade, P. H., Loureiro Luna, S. P. & Pairis-Garcia, M. D. Efficacy of transdermal flunixin in mitigating castration pain in piglets. Front. Pain Res. 1, 184 (2022).
Fávero, L. P., Hair, J. F., de Souza, R. F., Albergaria, M. & Brugni, T. V. Zero-inflated generalized linear mixed models: A better way to understand data relationships. Mathematics 9, 1–28 (2021).
Altman, D. G. & Bland, J. M. Measurement in medicine: The analysis of method comparison studies. J. R. Stat. Soc. Ser. D 32, 307–317 (1983).
Author information
Authors and Affiliations
Contributions
P.H.E.T.: Methodology, Formal analysis, Data visualization, Investigation, Writing—original draft, and Writing—review & editing; M.L.S.: Conceptualization, Methodology, Project administration, Investigation, and Writing—original draft; V.R.M.: Conceptualization, Methodology, Investigation, and Writing—original draft; R.M.T.: Methodology, Investigation, and Writing—original draft; M.D.P.G.: Conceptualization, Methodology, Investigation, Writing—original draft, Supervision, and Funding acquisition. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Trindade, P.H.E., Lopez-Soriano, M., Merenda, V.R. et al. Effects of assessment method (real-time versus video-recorded) on a validated pain-altered behavior scale used in castrated piglets. Sci Rep 13, 18680 (2023). https://doi.org/10.1038/s41598-023-45869-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-45869-8
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.