Abstract
Non-small cell lung cancer (NSCLC) with epidermal growth factor receptor (EGFR) mutation is brain metastasis (BM)-prone. We determined the impact of this hallmark, along with EGFR subtype and generation of tyrosine kinase inhibitor (TKI) treatment, on patients’ outcome. 553 metastatic EGFR-mutant NSCLC patients received front-line EGFR-TKI treatment. Progression-free survival (PFS), overall survival (OS) and secondary T790M rate were analysed. BM was observed in 211 (38.2%) patients. BM (HR 1.20 [95% CI 0.99–1.48]; p = 0.053), ECOG PS 0–1 (HR 0.71 [95% CI 0.54–0.93]; p = 0.014) and afatinib treatment (HR 0.81 [95% CI 0.66–0.99]; p = 0.045) were associated with PFS. Afatinib-treated patients without BM demonstrated a significantly longer PFS (16.3 months) compared to afatinib-treated patients with BM (13.7 months) and to gefitinib/erlotinib-treated patients with (11.1 months) or without BM (14.2 months; p < 0.001). CNS-only progression trended higher in afatinib-treated patients. ECOG PS 0–1 (HR 0.41 [95% CI 0.31–0.56]; p < 0.001) and EGFR L858R mutation (HR 1.46 [95% CI 1.13–1.88]; p = 0.003), but not BM, were the predictors for OS. BM (OR 2.02 [95% CI 1.02–4.08]; p = 0.040), afatinib treatment (OR 0.26 [95% CI 0.12–0.50]; p < 0.001) and EGFR L858R mutation (OR 0.55 [95% CI 0.28–1.05]; p = 0.070) were associated with secondary T790M rate. In BM patients, gefitinib/erlotinib-treated ones with 19 deletion mutation and afatinib-treated ones with L858R mutation had the highest and the lowest T790M rate (94.4% vs. 27.3%, p < 0.001), respectively. BM and generation of EGFR-TKI jointly impact PFS and secondary T790M rate in patients with EGFR-mutant NSCLC, whereas OS was mainly associated with EGFR subtype.
Similar content being viewed by others
Introduction
Treatment of EGFR-TKI for advanced NSCLC patients with EGFR-sensitizing mutation is one of the supreme advances in lung cancer treatment today1. The first, second and third-generation EGFR-TKIs, with variable degree of therapeutic efficacies, are all currently standard of cares in this setting2. Although the survival outcome of EGFR-positive NSCLC patients has tremendously improved by these TKI treatments, the prognosis remains profoundly affected by certain clinical, molecular and therapeutic factors3,4.
Brain metastasis-prone nature is a hallmark of NSCLC with EGFR mutation; as earlier studies have revealed that EGFR-mutant patients demonstrated a higher rate of brain involvement compared to EGFR-wild type ones5,6,7,8. Presence of brain metastasis in EGFR-mutant patients may further impact the clinical outcome. This has been reported in front-line TKI-treated patients where, irrespective of first- or second-generation drugs administered, those with brain metastasis tended to have shorter PFS compared to those without9,10,11. Nevertheless, the impact of brain metastasis to OS has been relatively variable. Some studies have shown that it was associated with a worse OS in EGFR-positive patients12,13, partly due to a negative influence on patients’ performance status whereas some others have demonstrated that TKI-treated patients with brain metastasis experienced a similar OS as those without14,15,16, likely associated with the favorable intracranial efficacies of these drugs. Therefore, more clinical practice-based, brain metastasis-dedicated analyses from TKI-treated EGFR-mutant patients are warranted for elucidation.
In addition, generation of EGFR-TKI used also serves as an outcome-associated factor in EGFR-mutant patients. Notably, third-generation osimertinib treatment has exhibited a significantly longer PFS and OS compared to first-generation TKI treatment17. With good CNS penetration, osimertinib also significantly reduces BM not only in patients with advanced stage disease but also in those with early stage disease as an adjuvant treatment17,18. However, the wide use of front-line osimertinib remains unachievable in many countries due to the high cost. The second-generation afatinib has demonstrated superior efficacy over first-generation TKI in the LuxLung-7 trial19, whereas the finding did not appear to be consistently observed in real-world settings11,20,21. Dacomitinib, another second-generation TKI, further demonstrated a significantly improved OS in ARCHER 1050 trial comprising a cohort of patients without brain metastasis22,23. Recently, Jung et al. reported a different CNS efficacy between first- and second-generation TKIs in a real-world cohort of EGFR-mutant patients in which a lower CNS progression rate was observed in afatinib-treated patients24. However, given the higher treatment-related toxicity of afatinib than gefitinib/erlotinib, the frequent requirement of dose modification also raises the concern of suboptimal brain concentration secondary to the penetrant hurdle of blood brain barrier. As a result, Tan et al. has demonstrated that, in EGFR-mutant patients with brain metastasis, dose reduction of afatinib was associated with a reduced PFS25. Taken together, more data are still required to understand the joint effect regarding brain metastasis and generation of EGFR-TKI on the outcome of EGFR-mutant patients.
The two subtypes of common EGFR mutation, the L858R in exon 21 and the deletion in exon 19, are also closely implicated in patients’ outcome. Previous studies have revealed that EGFR L858R mutants usually harbour more co-occurring mutations, both within and beyond the EGFR gene, than EGFR 19 deletion mutants26,27. As a result, studies which involved gefitinib/erlotinib or osimertinib treatment for EGFR-mutant NSCLC frequently showed lower therapeutic efficacies in L858R patients compared to 19 deletion ones28,29. Nevertheless, this genotype-dependent treatment efficacy seems less evident in afatinib-treated patients22,30. In addition, the differences in terms of OS and secondary T790M mutation rate have also been reported between the two common EGFR genotypes31,32. Overall, how these clinical, molecular features and therapeutic parameter jointly influence the outcome of EGFR-mutant NSCLC requires the analysis from a large patient sample.
In this study, we analyzed a large, real-world cohort of metastatic EGFR-mutant NSCLC patients who received front-line treatment of gefitinib/erlotinib or afatinib. The survival outcome and secondary T790M mutation rate in relation to brain metastasis, EGFR subtypes and generation of EGFR-TKI administration were reported.
Methods
Patients and treatment
Patients diagnosed metastatic NSCLC with EGFR-sensitizing mutation, the exon 19 deletion or the exon 21 L858R mutation, who received first-line treatment of gefitinib, erlotinib or afatinib between January 2013 and December 2019 were included in present study. Patient received the treatment of gefitinib 250 mg, erlotinib 150 mg or afatinib 40 mg as the starting dose and those who received the treatment less than 3 weeks were excluded from the analysis. Acquired EGFR T790M mutation was tested by a therascreen® EGFR RGQ PCR kit (QIAGEN, Valencia, USA) for patients who had available tumor tissues and/or by plasma cell-free DNA (liquid biopsy) using a RainDrop™ Digital PCR System (RainDance Technologies, Boston, USA). The progression-free survival (PFS) was defined as the interval between the date of starting EGFR-TKI and the date of radiologically or clinically determined progression or death. The treatment response, including complete response (CR), partial response (PR), stable disease, and progressive disease, was evaluated according to the Response Evaluation Criteria in Solid Tumors (version 1.1). The study used data from the Chang Gung Research Database and the Ethics Committee of Chang Gung Memorial Hospital approved the study protocol and the waiver of informed consent form (No. 201801967B0).
Statistical analysis
The Mann–Whitney test was used to determine the statistical significance of continuous variables between the two groups and Chi-squared test was used for evaluating the categorical variables. The Kaplan–Meier survival curve was analysed using the R package survival, and the hazard ratio (HR) was analysed using the Cox regression model. The patterns of disease progression, a CNS progression alone or a systemic progression, were treated as competing risk events of which the cumulative incidence functions were calculated33. The modified Cox regression model for the subdistribution hazard of the cumulative incidence function was applied to calculate the disease progression hazard from a given pattern in the presence of competing events by using the R package cmprsk34. All the reported p values were two sided, and a p < 0.05 was considered statistically significant. Data were also analyzed using SPSS (version 10.1; SPSS, Chicago, IL, USA).
Ethics statement
The study was performed in accordance with the ethical standards of the 1964 Declaration of Helsinki. The Ethics Committee of Chang Gung Memorial Hospital approved the study (No. 201801967B0) and granted permission for access to the Chang Gung Research Database and the IRB approved the waiver of the informed consent form.
Results
Baseline patient characteristics by brain metastasis status
A total of 553 patients were included for analysis, in which 211 (38.2%) patients presented baseline brain metastasis and 342 (61.8%) patients were absent of baseline brain metastasis (Table 1). The clinical features including age, sex, smoking history, histology and EGFR mutation subtype were similar between the two groups. However, patients with baseline brain metastasis had a significantly higher frequency of co-occurring liver metastasis (17.1% vs. 9.6%, p = 0.012) and were significantly more likely to have an ECOG performance status ≥ 2 (21.8% vs. 14.0%, p = 0.020) compared to those without baseline brain metastasis. Notably, physician were more likely to prescribe first-generation EGFR-TKI (53.6% vs. 38.0%, p < 0.001) for patients presenting brain metastasis in clinical practice compared to those absent of brain metastasis (Table 1). The median follow-up duration was 33.6 months and 272 (49.2%) events of death were observed at the time of analysis.
Cox regression analysis of PFS
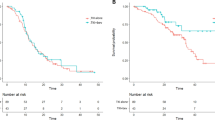
Univariate Cox regression analyses demonstrated that brain metastasis (HR 1.34 [95% CI 1.10–1.64]; p = 0.003), ECOG PS 0–1 (HR 0.58 [95% CI 0.45–0.74]; p < 0.001), second-generation EGFR-TKI afatinib treatment (HR 0.71 [95% CI 0.59–0.86]; p < 0.001) and liver metastasis (HR 1.56 [95% CI 1.19–2.04]; p = 0.001, Table 2) had significant impact on PFS. Multivariate regression adjustment exhibited that ECOG PS 0–1 (HR 0.71 [95% CI 0.54–0.93]; p = 0.014), second-generation EGFR-TKI afatinib treatment (HR 0.81 [95% CI 0.66–0.99]; p = 0.045) and liver metastasis (HR 1.31 [95% CI 1.00–1.77]; p = 0.047, Table 2) were independent predictors of PFS. In addition, a trend of negative impact on PFS remained observed for brain metastasis (HR 1.20 [95% CI 0.99–1.48]; p = 0.053, Table 2) where patients with brain metastasis showed a shorter median PFS (11.8 vs. 15.4 months; log-rank test p = 0.003), a lower 24-month PFS rate (22.3% [95% CI 17.0% to 29.3%] vs. 30.7% [95% CI 25.9% to 36.5%], Fig. 1A) compared to those without.
PFS and pattern of progression by brain metastasis status and generation of EGFR-TKI
We subsequently analysed the patient groups stratified by brain metastasis status and generation of EGFR-TKI treatment. Patients who received second-generation EGFR-TKI afatinib, irrespective of brain metastasis status, were generally younger, with better ECOG PS and had more EGFR 19 deletion genotype, compared to patients who received first-generation EGFR-TKI gefitinib/erlotinib (Table 3). Putting together, afatinib-treated patients without brain metastasis demonstrated a significantly longer PFS (16.3 months) compared to afatinib-treated patients with brain metastasis (13.7 months) and to gefitinib/erlotinib-treated patients with (11.1 months) or without brain metastasis (14.2 months; log-rank test p < 0.001, Fig. 1B). As to the pattern of disease progression: In brain metastasis group, afatinib-treated patients demonstrated a significantly lower rate of systemic progression (adjusted cause-specific HR, 0.54; 95% CI 0.38–0.77; p < 0.001) but a trend of higher CNS-alone progression (adjusted cause-specific HR, 2.28; 95% CI 0.88–5.87; p = 0.086, Fig. 2A), compared to gefitinib/erlotinib-treated patients. In non-brain metastasis group, similar systemic progression (adjusted cause-specific HR, 0.87; 95% CI 0.65–1.18; p = 0.370) and CNS-alone progression (adjusted cause-specific HR, 3.02; 95% CI 0.71–12.9; p = 0.140, Fig. 2B) were observed between afatinib- and gefitinib/erlotinib-treated patients.
Cumulative incidence of systemic progression without death (black) and CNS- alone progression without death (red) between first-generation EGFR-TKI gefitinib/erlotinib (solid line) and second-generation EGFR-TKI afatinib (broken line) in patients (A) with brain metastasis and (B) without brain metastasis.
OS by brain metastasis and Cox regression analysis
Patients with brain metastasis, compared to those without, had a numerically shorter OS (28.9 vs. 34.7 months; log-rank test p = 0.060, Fig. 3A) and a trend of increased risk of death (HR 1.26 [95% CI 0.99–1.60]; p = 0.063, Table 4). Univariate Cox regression demonstrated that older patients of age ≥ 65 (HR 1.33 [95% CI 1.05–1.69]; p = 0.019), ECOG PS 0–1 (HR 0.38 [95% CI 0.29–0.50]; p < 0.001), EGFR L858R mutation (HR 1.32 [95% CI 1.01–1.72]; p = 0.044), second-generation EGFR-TKI afatinib treatment (HR 0.69 [95% CI 0.54–0.87]; p = 0.002) and liver metastasis (HR 1.47 [95% CI 1.06–2.04]; p = 0.022, Table 4) also had significant impact on OS. Multivariate adjustment demonstrated that ECOG PS 0–1 (HR 0.41 [95% CI 0.31–0.56]; p < 0.001) and EGFR L858R mutation (HR 1.46 [95% CI 1.13–1.88]; p = 0.003, Table 4) were the independent factors predictive of OS. We later analysed the OS stratified by brain metastasis status and EGFR mutation subtype. Patients who had EGFR 19 deletion mutation and without brain metastasis exhibited a significantly longer OS (46.8 months) compared to EGFR 19 deletion patients with brain metastasis (34.3 months), EGFR L858R mutation with (28.3 months) and without (30.7 months; log-rank test p = 0.002, Fig. 3B) brain metastasis.
Brain metastasis status as a predictor of secondary T790M development
A total of 182 (32.9%) patients underwent an EGFR T790M mutation assay by tissue and/or liquid biopsy upon disease progression. A positive T790M status was determined in 101 (55.5%) patients and the T790M rate was similar between tissue (67/127, 52.7%) and liquid (34/67, 51.5%; Chi-squared p = 0.908) biopsy. Clinical factors including brain metastasis status, EGFR mutation subtype, generation of EGFR-TKI treatment and PFS longer than 12 months were all observed to be associated with the T790M positive rate by univariate logistic regression (Table 5). Multivariate analysis demonstrated that brain metastasis (OR 2.02 [95% CI 1.02–4.08]; p = 0.040), second-generation EGFR-TKI afatinib treatment (OR 0.26 [95% CI 0.12–0.50]; p < 0.001) and PFS longer than 12 months (OR 3.10 [95% CI 1.59–6.30]; p = 0.001, Table 5) were independent predictors to the secondary T790M rate. The EGFR L858R mutation (OR 0.55 [95% CI 0.28–1.05]; p = 0.070) was associated with a trend of lower T790M positivity after multivariate adjustment.
Determination of T790M rate by brain metastasis status-based subgroups
For patient groups stratified by brain metastasis status and length of PFS: The group without brain metastasis and PFS less than 12 months demonstrated a lowest secondary T790M rate compared to the others (31.9%; Chi-squared p = 0.002, Fig. 4A). For patient groups stratified by brain metastasis status, EGFR mutation subtype and generation of EGFR-TKI treatment: The group with brain metastasis and EGFR 19 deletion mutation receiving gefitinib/erlotinib demonstrated a highest T790M rate (94.4%) and the group with brain metastasis and L858R mutation receiving afatinib exhibited a lowest T790M rate (27.3%; Chi-squared p < 0.001, Fig. 4B), among others.
The secondary T790M rate between patients grouped by (A) with or without BM and PFS ≥ or < 12 months (B) with or without BM, EGFR mutation subtype and generation of EGFR-TKI treatment. BM brain metastasis, 19del EGFR 19 deletion mutation, L858R EGFR L858R mutation, 1G first-generation EGFR-TKI, 2G second-generation EGFR-TKI.
Discussion
The present study demonstrated that the presence of brain metastasis in EGFR-mutant NSCLC patients had clinical impact on the efficacy of front-line EGFR-TKI treatment, particularly for the first-generation one; whereas the impact toward OS was not significant. The generation of EGFR-TKI administration also jointly influenced the treatment efficacy. The EGFR mutation subtype mainly has an impact on the patients’ overall survival.
We have previously reported that the generation EGFR-TKI used was associated with therapeutic efficacy in EGFR-mutant NSCLC patients, in which gefitinib/erlotinib-treated patients presented a shorter PFS compared to afatinib-treated ones11. In this analysis, we demonstrated that brain metastasis further impacted the therapeutic efficacy of gefitinib/erlotinib whereas this impact on the efficacy of afatinib treatment seemed to be less significant. Thus, this finding indicated that afatinib may be a better treatment option than gefitinib/erlotinib in EGFR-positive patients with brain metastasis. Recently, a similar finding has also been reported by Jung et al. that patient who received afatinib treatment experienced a significantly longer CNS-PFS than those who received gefitinib or erlotinib24. These seemingly higher efficacies of afatinib in brain metastasis cohort may be partly explained by the previous studies which analyzed first- and second-generation EGFR-TKI concentrations in human cerebralspinal fluid35,36 where afatinib treatment was likely associated with a larger margin of differential between the measured drug concentration in cerebralspinal fluid and the reported data of in vitro IC50 against NSCLC cell lines with EGFR-sensitizing mutation37.
Nevertheless, the higher toxicity profile of afatinib treatment and thereby the related dose reduction or interruption also has been linked to a reduced efficacy previously25. Whether this can be associated with more CNS-alone progression, as a result of reduced serum concentration in the face of blood–brain barrier, was largely unknown. In the present study, a non-significantly higher hazard of CNS-alone progression was observed in afatinib-treated patients of whom 56.3% has undergone dose reduction during the treatment. This finding suggested that a specific attention may be paid to CNS-alone progression for afatinib-treated patients especially when they underwent dose modification even tumors were well-controlled in primary site or other non-CNS metastatic sites. Recently, dacomitinib has demonstrated excellent intracranial response with both standard and reduced dose scheme in a real-world cohort of EGFR-mutant patients38. However, whether dose reduction has an impact on the duration of CNS response and progression requires further investigation. Despite the status of brain metastasis implicated in the treatment efficacy of EGFR-TKI, it did not seem to impose significant impact on the patients’ overall survival in this analysis. This finding suggested that the efficacy of post-progression treatment was minimally compromised in the patients with brain metastasis likely because their disease burden in brain was successfully controlled. Following the line, the fact that approximately 60% of the patients with brain metastasis in the present study received EGFR-TKI alone without upfront brain radiotherapy might also support the mindset of deferring radiotherapy for selected patients without undermining their survival39.
Regarding to the prognostic impact of EGFR L858R and 19 deletion mutations, the issue remains pretty much unsettled as the findings from previous studies have been quite controversial40. In this study, we demonstrated that subtype of EGFR mutation had significant impact on OS in which patients with EGFR L858R mutation had less favorable OS compared to those with EGFR 19 deletion mutation. The explanation to this finding can likely be multi-faceted. Previous studies have revealed that NSCLC with L858R mutation tended to possess a higher number of co-occurring genomic alterations than NSCLC with 19 deletion mutation26,27. This increases the clonal complexity of the tumors and naturally places them in a higher likelihood to avert targeted therapies28,29. Other studies reported elsewhere further observed a higher frequency of primary EGFR T790M mutation in L858R patients compared to 19 deletion ones and thus associated with the primary resistance to first or second-generation EGFR-TKI treatments41,42. In contrary, the frequency of secondary EGFR T790M mutation was reported to be lower in L858R patients than in 19 deletion ones, both in previous studies and the present one43,44. This usually rendered patients whose disease progressed from first or second-generation EGFR-TKIs unavailable to the approved treatment of third-generation T790M-active drugs and thereby leading to a less favorable survival outcome. However, more data analysis from clinical trial and real world will be required to determine the true prognostic impact between the two common sensitizing mutations.
Along this line, identification of secondary EGFR T790M mutation in patients who progress from geftinib/erlotinib or afatinib treatment is of paramount importance. However, this is usually limited by unavailable/insufficient tumor tissue, unqualified nucleic acid, additional testing cost and, not infrequently, rapid disease progression45,46. In this analysis, several parameters were identified to be associated with secondary T790M rate including status of brain metastasis, PFS length of front-line EGFR-TKI treatment, generation of EGFR-TKI used and EGFR mutation subtype. We further observed differential secondary T790M rates in patients defined by two sets of brain metastasis-based parameters. The T790M rate was approximately 60% ~ 70% in patients with baseline brain metastasis irrespective of the PFS length of EGFR-TKI treatment (≥ or < 12 months). However, further dissection toward these patients revealed that EGFR 19 deletion cohort treated by gefitinib/erlotinib and EGFR L858R cohort treated by afatinib had the highest and the lowest T790M rates (94.4% vs. 27.3%), respectively. In patient without baseline brain metastasis, the T790M rates were similar (42% ~ 67%) among the four patient cohorts defined by EGFR mutation subtype and generation of EGFR-TKI used. However, the PFS length was significantly associated with T790M rate in these patients, where those who experienced a PFS < 12 months had a lower T790M rate compared to those who experienced a PFS ≥ 12 months (31.9% vs. 60.9%).
The limitation of this study, firstly, lies in its retrospective nature. Secondly, the front-line osimertinib has currently become the recommended standard of care for patients of advanced EGFR-mutant NSCLC, with the advantage for treatment or prevention of brain metastasis. However, the findings of the study would be important in places where osimertinib is not available to patients due to various reasons. Thirdly, clinical characteristics were imbalanced between patients with or without brain metastasis in the study. However, these biases reasonably reflected the different condition intrinsically existed between the two groups of patients and the exact impact against survival outcome was also clarified by statistical adjustment with regression-based method. Overall, this analysis demonstrated that status of brain metastasis and generation of EGFR-TKI have joint impact on the PFS of TKI treatment while the OS is mainly determined by EGFR mutation subtype. The differential secondary T790M rate may be further predicted by these three parameters. Additional prospective studies are warranted to validate these findings.
Data availability
The datasets generated and/or analysed in the current study are available from the corresponding author on reasonable request.
References
Hsu, W. H. et al. Overview of current systemic management of EGFR-mutant NSCLC. Ann. Oncol. 29(suppl_1), i3–i9 (2018).
Tsubata, Y., Tanino, R. & Isobe, T. Current therapeutic strategies and prospects for EGFR mutation-positive lung cancer based on the mechanisms underlying drug resistance. Cells 10(11), 3192 (2021).
Qin, K. et al. Prognostic value of TP53 concurrent mutations for EGFR- TKIs and ALK-TKIs based targeted therapy in advanced non-small cell lung cancer: A meta-analysis. BMC Cancer 20(1), 328 (2020).
Chan, S. W. S. et al. Systemic inflammatory markers of survival in epidermal growth factor-mutated non-small-cell lung cancer: Single-institution analysis, systematic review, and meta-analysis. Clin. Lung Cancer 22(5), 390–407 (2021).
Ge, M. et al. High probability and frequency of EGFR mutations in non-small cell lung cancer with brain metastases. J. Neurooncol. 135(2), 413–418 (2017).
Iuchi, T. et al. Frequency of brain metastases in non-small-cell lung cancer, and their association with epidermal growth factor receptor mutations. Int. J. Clin. Oncol. 20(4), 674–679 (2015).
Li, W. Y. et al. The role of EGFR mutation as a prognostic factor in survival after diagnosis of brain metastasis in non-small cell lung cancer: A systematic review and meta-analysis. BMC Cancer 19(1), 145 (2019).
Shin, D. Y. et al. EGFR mutation and brain metastasis in pulmonary adenocarcinomas. J. Thorac. Oncol. 9(2), 195–199 (2014).
Liang, S. K. et al. Real-world experience of afatinib as a first-line therapy for advanced EGFR mutation-positive lung adenocarcinoma. Oncotarget 8(52), 90430–90443 (2017).
Schuler, M. et al. First-line afatinib versus chemotherapy in patients with non-small cell lung cancer and common epidermal growth factor receptor gene mutations and brain metastases. J. Thorac. Oncol. 11(3), 380–390 (2016).
Huang, A. C. et al. First- or second-generation epidermal growth factor receptor tyrosine kinase inhibitors in a large, real-world cohort of patients with non-small cell lung cancer. Ther. Adv. Med. Oncol. 13, 17588359211035710 (2021).
Chang, C. Y. et al. Efficacy and prognosis of first-line EGFR-tyrosine kinase inhibitor treatment in older adults including poor performance status patients with EGFR-mutated non-small-cell lung cancer. Cancer Manag. Res. 13, 7187–7201 (2021).
Gijtenbeek, R. G. P. et al. Nationwide real-world cohort study of first-line tyrosine kinase inhibitor treatment in epidermal growth factor receptor-mutated non-small-cell lung cancer. Clin. Lung Cancer 21(6), e647–e653 (2020).
Payen, T. et al. Real world data of efficacy and safety of erlotinib as first-line TKI treatment in EGFR mutation-positive advanced non-small cell lung cancer: Results from the EGFR-2013-CPHG study. Respir. Med. Res. 80, 100795 (2021).
Lin, Y. T. et al. Clinical outcomes and secondary epidermal growth factor receptor (EGFR) T790M mutation among first-line gefitinib, erlotinib and afatinib-treated non-small cell lung cancer patients with activating EGFR mutations. Int. J. Cancer https://doi.org/10.1002/ijc.32025 (2018).
Yao, Z. H. et al. Real-world data on prognostic factors for overall survival in EGFR mutation-positive advanced non-small cell lung cancer patients treated with first-line gefitinib. Oncologist 22(9), 1075–1083 (2017).
Ramalingam, S. S. et al. Overall survival with osimertinib in untreated, EGFR-mutated advanced NSCLC. N. Engl. J. Med. 382(1), 41–50 (2020).
Tsuboi, M. et al. Overall survival with osimertinib in resected EGFR-mutated NSCLC. N. Engl. J. Med. 389(2), 137–147 (2023).
Park, K. et al. Afatinib versus gefitinib as first-line treatment of patients with EGFR mutation-positive non-small-cell lung cancer (LUX-Lung 7): A phase 2B, open-label, randomised controlled trial. Lancet Oncol. 17(5), 577–589 (2016).
Lin, Y. T. et al. Clinical outcomes and secondary epidermal growth factor receptor (EGFR) T790M mutation among first-line gefitinib, erlotinib and afatinib-treated non-small cell lung cancer patients with activating EGFR mutations. Int. J. Cancer 144(11), 2887–2896 (2019).
Fujiwara, A. et al. A retrospective comparison of the clinical efficacy of gefitinib, erlotinib, and afatinib in japanese patients with non-small cell lung cancer. Oncol. Res. 26(7), 1031–1036 (2018).
Paz-Ares, L. et al. Afatinib versus gefitinib in patients with EGFR mutation-positive advanced non-small-cell lung cancer: Overall survival data from the phase IIb LUX-Lung 7 trial. Ann. Oncol. 28(2), 270–277 (2017).
Wu, Y. L. et al. Dacomitinib versus gefitinib as first-line treatment for patients with EGFR-mutation-positive non-small-cell lung cancer (ARCHER 1050): A randomised, open-label, phase 3 trial. Lancet Oncol. 18(11), 1454–1466 (2017).
Jung, H. A. et al. The different central nervous system efficacy among gefitinib, erlotinib and afatinib in patients with epidermal growth factor receptor mutation-positive non-small cell lung cancer. Transl. Lung Cancer Res. 9(5), 1749–1758 (2020).
Tan, W. L. et al. Influence of afatinib dose on outcomes of advanced EGFR-mutant NSCLC patients with brain metastases. BMC Cancer 18(1), 1198 (2018).
Hong, S. et al. Concomitant genetic alterations with response to treatment and epidermal growth factor receptor tyrosine kinase inhibitors in patients with EGFR-mutant advanced non-small cell lung cancer. JAMA Oncol. 4(5), 739–742 (2018).
Barnet, M. B. et al. EGFR-co-mutated advanced NSCLC and response to EGFR tyrosine kinase inhibitors. J. Thorac. Oncol. 12(3), 585–590 (2017).
Sutiman, N. et al. EGFR mutation subtypes influence survival outcomes following first-line gefitinib therapy in advanced Asian NSCLC patients. J. Thorac. Oncol. 12(3), 529–538 (2017).
Goto, K. et al. A prospective, phase II, open-label study (JO22903) of first-line erlotinib in Japanese patients with epidermal growth factor receptor (EGFR) mutation-positive advanced non-small-cell lung cancer (NSCLC). Lung Cancer 82(1), 109–114 (2013).
Huang, C. H. et al. Afatinib treatment in a large real-world cohort of nonsmall cell lung cancer patients with common and uncommon epidermal growth factor receptor mutation. Int. J. Cancer 150(4), 626–635 (2022).
Li, W. Q. & Cui, J. W. Non-small cell lung cancer patients with ex19del or exon 21 L858R mutation: Distinct mechanisms, different efficacies to treatments. J. Cancer Res. Clin. Oncol. 146(9), 2329–2338 (2020).
Zhou, Y. et al. Survival outcomes and symptomatic central nervous system (CNS) metastasis in EGFR-mutant advanced non-small cell lung cancer without baseline CNS metastasis: Osimertinib vs. first-generation EGFR tyrosine kinase inhibitors. Lung Cancer 150, 178–185 (2020).
Satagopan, J. M. et al. A note on competing risks in survival data analysis. Br. J. Cancer 91(7), 1229–1235 (2004).
Fine, J. P. & Gray, R. J. A proportional hazards model for the subdistribution of a competing risk. J. Am. Stat. Assoc. 94(446), 496–509 (1999).
Tamiya, A. et al. Cerebrospinal fluid penetration rate and efficacy of Afatinib in patients with EGFR mutation-positive non-small cell lung cancer with leptomeningeal carcinomatosis: A multicenter prospective study. Anticancer Res. 37(8), 4177–4182 (2017).
Togashi, Y. et al. Cerebrospinal fluid concentration of gefitinib and erlotinib in patients with non-small cell lung cancer. Cancer Chemother. Pharmacol. 70(3), 399–405 (2012).
Cross, D. A. et al. AZD9291, an irreversible EGFR TKI, overcomes T790M-mediated resistance to EGFR inhibitors in lung cancer. Cancer Discov. 4(9), 1046–1061 (2014).
Peng, W. et al. Dacomitinib induces objective responses in metastatic brain lesions of patients with EGFR-mutant non-small-cell lung cancer: A brief report. Lung Cancer 152, 66–70 (2021).
Miyawaki, E. et al. Optimal sequence of local and EGFR-TKI therapy for EGFR-mutant non-small cell lung cancer with brain metastases stratified by number of brain metastases. Int. J. Radiat. Oncol. Biol. Phys. 104(3), 604–613 (2019).
Shah, R. & Lester, J. F. Tyrosine kinase inhibitors for the treatment of EGFR mutation-positive non-small-cell lung cancer: A clash of the generations. Clin. Lung Cancer 21(3), e216–e228 (2020).
Yu, H. A. et al. Poor response to erlotinib in patients with tumors containing baseline EGFR T790M mutations found by routine clinical molecular testing. Ann. Oncol. 25(2), 423–428 (2014).
Lee, Y. J., Lee, G. K. & Lee, Y.-S. Clinicopathologic features of lung cancer patients harboring de novo EGFR T790M mutation. J. Clin. Oncol. 32(15_suppl), 8075–8075 (2014).
Huang, Y. H. et al. The association of acquired T790M mutation with clinical characteristics after resistance to first-line epidermal growth factor receptor tyrosine kinase inhibitor in lung adenocarcinoma. Cancer Res. Treat. 50(4), 1294–1303 (2018).
Nosaki, K. et al. Re-biopsy status among non-small cell lung cancer patients in Japan: A retrospective study. Lung Cancer 101, 1–8 (2016).
Seto, T. et al. Real-world EGFR T790M testing in advanced non-small-cell lung cancer: A prospective observational study in Japan. Oncol. Ther. 6(2), 203–215 (2018).
Magios, N. et al. Real-world implementation of sequential targeted therapies for EGFR-mutated lung cancer. Ther. Adv. Med. Oncol. 13, 1758835921996509 (2021).
Acknowledgements
We thank to the contributions of research assistants Ms. Yen-Wen Wang and Ms. Yu-Chi Chiang to this study.
Funding
The study received funding support from Chang Gung Medical Foundation CORPG3J0332; CORPG3J0333; CMRPG5M0051.
Author information
Authors and Affiliations
Contributions
J.S.J. wrote and C.S.K. revised the manuscript; C.S.K., C.C.W. and C.T.Y. were responsible for study conception and design; A.C.H., J.S.J., T.H.C., P.H.T. and C.H.H. collected the data; C.S.K., C.C.W., H.W.K., F.T.C., Y.F.F. and C.T.Y. provided study materials and patients; C.S.K. and Y.K.G. analyzed and interpreted the data; all authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
CS-K received speaker honoraria from AstraZeneca, Boehringer Ingelheim, Roche, Pfizer, Eli Lilliy, Novartis, OnO Pharma, Chugai, Merck, Janssen Pharma, Takeda and Guardant Health. CS-K provided consultation for AstraZeneca, Boehringer Ingelheim, Eli Lilliy, Merck, Chugai, Takeda, Novartis and Guardant Health. None of the other authors have any conflict of interest to disclose.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ju, JS., Huang, A.CC., Tung, PH. et al. Brain metastasis, EGFR mutation subtype and generation of EGFR-TKI jointly influence the treatment outcome of patient with EGFR-mutant NSCLC. Sci Rep 13, 20323 (2023). https://doi.org/10.1038/s41598-023-45815-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-45815-8
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.