Abstract
Rhabdomyolysis is a potentially life-threatening condition induced by diverse mechanisms including drugs and toxins. We aimed to investigate the incidence of rhabdomyolysis occurrence in intoxicated patients with psychoactive substances. In this review, three databases (PubMed, Scopus, Web of Science) and search engine (Google Scholar) were searched by various keywords. After the screening of retrieved documents, related data of included studies were extracted and analyzed with weighted mean difference (WMD) in random effect model. The highest incidence of rhabdomyolysis was observed in intoxication with heroin (57.2 [95% CI 22.6–91.8]), amphetamines (30.5 [95% CI 22.6–38.5]), and cocaine (26.6 [95% CI 11.1–42.1]). The pooled effect size for blood urea nitrogen (WMD = 8.78, p = 0.002), creatinine (WMD = 0.44, p < 0.001), and creatinine phosphokinase (WMD = 2590.9, p < 0.001) was high in patients with rhabdomyolysis compared to patients without rhabdomyolysis. Our results showed a high incidence of rhabdomyolysis induced by psychoactive substance intoxication in ICU patients when compared to total wards. Also, the incidence of rhabdomyolysis occurrence was high in ICU patients with heroin and amphetamine intoxication. Therefore, clinicians should anticipate this complication, monitor for rhabdomyolysis, and institute appropriate treatment protocols early in the patient’s clinical course.
Similar content being viewed by others
Introduction
Psychoactive substances affect thinking, emotion, mood and behavior, and consciousness after being consumed and are classified into central nervous system depressants (ethanol, opioids, cannabis), central nervous system stimulants (amphetamines, cocaine), hallucinogens (LSD), and empathogens (ecstasy)1. Nausea, vomiting, agitation, anxiety, and drowsiness are the most common adverse effects of psychoactive drugs but serious conditions such as psychosis, delirium, seizure, cardiotoxicity, severe lung injury, and acute kidney injury (AKI) due to rhabdomyolysis have seen reported in abusers2,3.
Previous studies have documented drug induced-rhabdomyolysis owing to the overuse of methanol, ethanol, methadone, opioid, cocaine, amphetamine, methamphetamine, ecstasy, synthetic cannabinoids, heroin, and tramadol4,5. Rhabdomyolysis is a syndrome characterized by muscle necrosis and the release of intracellular muscle constituents into circulation6. Creatine Phosphokinase (CPK) level commonly increases and muscle pain and myoglobinuria may be identified. The severity of rhabdomyolysis ranges from asymptomatic elevation in serum muscle enzymes to life-threatening conditions associated with extreme enzyme elevation, electrolyte imbalance, and acute kidney injury (AKI)7.
Rhabdomyolysis has been reported in a growing number of studies as one of the worst results of drug poisoning but with different incidences8. For instance, the incidence of cocaine-induced rhabdomyolysis is reported from 259 to 46.7%10 and this value for methadone is between 14.711 and 34.6%12.
Considering the increasing use of psychoactive substances in recent years and little known about drug-induced rhabdomyolysis in abusers, screening and assessing the incidence of rhabdomyolysis and proper management is essential. For this reason, we systematically reviewed the international databases in this study and the results of related papers were pooled regarding the incidence of rhabdomyolysis in hospitalized patients with intoxication.
Methods
Study design
This systematic review and meta-analysis study was according to the Preferred Reporting Items in Systematic Reviews and Meta-Analyses (PRISMA) guideline13, and protocol of the study was registered in PROSPERO (CRD42022326206).
Search strategy
To find related publications, a combination of related keywords was used in databases and search engine including PubMed, Scopus, Web of Science, and Google Scholar. In addition, we used a manual search to develop literature search, references of selected studies, and citations of studies. The final search was updated on July 15, 2022 before data analysis. The keywords used included a combination of suggested words by Medical Subject Heading (MeSH) and other related words, as represented in details in Table 1. Finally, three limitations in research including human studies, publication date (2000–2022), and English language studies were applied. All processes related to the literature search were done independently by two researchers (AA and ES).
Eligibility criteria
Eligibility criteria were elaborated based on the PICO structure (Population, Intervention\ Exposure, Comparator, and Outcomes), and the studied population under meta-analysis were intoxicated patients hospitalized in the intensive care unit (ICU) or general wards due to poisoning, overdose, and abuse and were either conscious or unconscious. Intoxicated patients following enteral, parenteral, and inhalational use of methadone, cocaine, heroin, tramadol, amphetamine, methamphetamine, ecstasy, MDMA (3,4-methylenedioxyamphetamine), opioid, synthetic cannabinoids, methanol, and ethanol were included in the study. Mono-intoxication means patients who were intoxicated with a single substance and multi-intoxication means patients who had co-exposure and concomitant substances were detected in their drug screen. The research question of the study was occurrence of rhabdomyolysis among hospitalized intoxicated patients. Exclusion criteria were case reports and case series with less than 5 samples, review or editorial articles, none English language manuscripts, and studies on children.
Study selection, data items, data collection
Retrieved observational studies from selected databases with relevant exposures were imported into EndNote citation management software. After removing duplicate studies, title and abstract of remained studies were screened and data extraction was done by two independent researchers (TB and SA). Data extraction forms contained the author's name, year, age, gender, country, continent, study design, type of psychoactive drug, sample size, type of hospitalization wards (ICU/total wards), multi-intoxication (yes/no), dose of substance, and patient’s medical history. Also, subgroup analyses for hospitalization ward, multi-intoxication, and geographical area (based on World Health Organization regions) was performed. In subgroup analysis, ICU means studies that included only ICU patients and total wards mean studies reporting total patients hospitalized in ICU and general wards. Also, poisoning with multiple drugs means studies reporting overall occurrence of rhabdomyolysis for all intoxicated patients but did not determine the incidence of rhabdomyolysis separately for each substance. Rhabdomyolysis occurrence was defined if included patients had CPK > 1000 IU/L/ or CPK > 5 × ULN14. Any disagreement at each stage was checked by a third researcher (AA).
Risk of bias of included studies
The risk of bias in studies was assessed independently by two researchers (MB and MS) and disagreements were discussed and checked by the third researcher (KE). For this purpose, Newcastle–Ottawa scale (NOS) was used to assess quality of nonrandomized studies in meta-analyses and the number of stars indicated methodological quality of articles.
Synthesis of results
The number of intoxicated patients in the studies was considered as denominator of the fraction and the number of samples containing occurrence of rhabdomyolysis was placed in the numerator. The effect size for rhabdomyolysis incidence in each subgroup was determined as pooled effect size with 95% confidence interval (CI). Weighted mean difference (WMD) was used to compare values of renal function indexes including blood urea nitrogen (BUN), creatinine (Cr), and CPK between intoxicated patients with and without rhabdomyolysis. The pooled effects size was estimated using random effect model by considering disparities between studies. Heterogeneity between studies was estimated by Cochrane Q test and I2 index. The type of hospitalization wards, geographical area, and multi-intoxication were considered to find the source of heterogeneity in the subgroup analysis. Publication bias was determined by Egger’s regression and Begg’s test. Sensitivity analysis was performed to assess the impact of a single study on the results. All statistical analyses for meta-analysis were done in Stata software (version 16.0; Stata Corporation).
Results
Study selection and characteristics
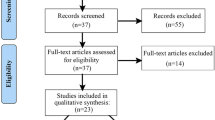
The process of study selection is depicted in the PRISMA flow diagram (Fig. 1). In the systematic search of electronic databases, 2493 nonredundant studies were found, of which 62 articles were potentially relevant. After reading their full texts, 5 articles were excluded due to lack of required data and 57 articles met the inclusion criteria for final analysis.
As described in Table 2, the percentage of hospitalized men in all included studies was high compared to women. The minimum and maximum mean age of patients was 19.4 and 46.0 years, respectively. The reported median age ranged from 19 to 50 years. A summary of included article characteristics is described in Table 2. Based on the types of psychoactive substances, a total of 57 articles containing 3,122,944 intoxicated patients were screened. Included articles were 11 studies for opioids, 6 for methadone, 11 for synthetic cannabinoids, 7 for cocaine, 17 for amphetamines, 5 for methanol, 4 for ethanol, 4 for heroin, 2 for tramadol, and 15 for multiple drugs. Most of the studies were done in the United States (n = 24), Iran (n = 13), Canada (n = 3), and Australia (n = 3).
Pooled estimate of rhabdomyolysis occurrence
The highest incidence of rhabdomyolysis occurrence was observed in heroin intoxication (57.2 [95% CI 22.6–91.8]), followed by amphetamines (30.5 [95% CI 22.6–38.5]) (Fig. 2), cocaine (26.6 [95% CI 11.1–42.1]), tramadol (17.07 [95% CI 10.6–23.5]), methadone (16.1 [95% CI 9.6–22.5]), synthetic cannabinoids (10.3 [95% CI 6.2–14.4]) (Fig. 3), and opioid (8.8 [95% CI 5.5–12.1]) (Fig. 4) (Table 3). The pooled incidence of rhabdomyolysis was low for intoxication with methanol (2.0 [95% CI 0.5–3.5]) and ethanol (3.0 [95% CI 0.3–5.7]) when compared with other psychoactive substances. In the amphetamine family, the pooled estimate of rhabdomyolysis for methamphetamine was 40.3 (95% CI 23.6–57.04), for amphetamine was 26.9 (95% CI 12.2–41.5), and for ecstasy was 19.9 (95% CI 3.3–36.5).
Subgroup analysis
Hospitalization ward and multi-intoxication
The pooled effect size in the subgroup of ICU patients was higher than in the total wards (Table 3). Incidence of rhabdomyolysis in ICU patients and total wards was respectively 13.6% vs 8.1% for opioids, 19.3% vs 16.5% for methadone, 25% vs 8.2% for synthetic cannabinoids, 40.0% vs 25.4% for cocaine, 71.5% vs 22.7% for amphetamines, 21% vs 1.75% for methanol, 100% vs 40.6% for heroin, and 30.5% vs 26.7% for multiple drug poisoning. In addition, pooled effect size in the subgroup of total wards was influenced by the severity of intoxication, which was different in the included studies. In comparison with multi-intoxication subgroup, pooled effect size of rhabdomyolysis occurrence was high in mono-intoxication subgroup except for synthetic cannabinoids (6.1% vs 13.2%) and amphetamines (24.1% vs 31.6%).
Geographic area
As it was showed in Table 3, according to the subgroup analysis based on the geographic area, the highest incidence of rhabdomyolysis occurrence was related to amphetamines in American region (65.6% [35.6–90.5]) and synthetic cannabinoids in European region (30.7% [18.9–43.8]), whereas the highest incidence of rhabdomyolysis occurrence in Eastern Mediterranean was related to other psychoactive substances. Subgrouping by geographic region reduced heterogeneity between studies.
Pooled mean effect size of renal function indexes
Table 4 shows the comparison of mean effect size of renal function indexes in patients with and without rhabdomyolysis. Accordingly, the value of BUN (WMD = 8.78, p = 0.002), Cr (WMD = 0.44, p < 0.001), and CPK (WMD = 2590.9, p < 0.001) was significantly high in patients with rhabdomyolysis compared to those patients without rhabdomyolysis.
Publication bias and sensitivity analysis
Table 3 represent specified p-values related to the publication bias in each type of psychoactive substances with Begg’s and Egger’s test, indicating that there was no publication bias in most of the intoxications. Sensitivity analysis was performed for all tests applied for meta-analysis and the results showed that none of the pooled effect size was influenced by a single study.
Discussion
The current study was a systematic review and meta-analysis of clinical data related to the incidence of rhabdomyolysis among intoxicated patients with psychoactive substances. To the best of our knowledge, this was the first systematic review conducted on rhabdomyolysis occurrence in psychoactive substance intoxication. Our results showed that pooled effect size for all categories of psychoactive substances was high in the subgroup of ICU patients compared with total wards. Also, intoxication with heroin (~ 100) and amphetamine (~ 71.5) showed the highest effect size for occurrence of rhabdomyolysis in ICU patients. In a study in Iran on 227 poisoned patients with refined opium extract, the majority of them (75.8%) were male. However, it has been documented that females have higher mitochondrial mass in skeletal muscle with greater oxidative phosphorylative capacities and therefore have greater protection against rhabdomyolysis66,67.
A broad range of neurological complications affecting both central nervous system (CNS) and peripheral nervous system (PNS) are encountered in heroin abusers24. CNS lesions include brain hypoxia and seizure, spongiform leukoencephalopathy, stroke, and myelopathy68 while PNS involvement commonly manifests as compressive neuropathy or focal rhabdomyolysis69. The etiology of this acute PNS complication is unclear. Some studies found immunological causes in patients who developed rhabdomyolysis. Also, mechanical trauma is considered a potential mechanism of focal nerve injury and localized rhabdomyolysis in heroin abusers70. The effect size of rhabdomyolysis in ICU patients with heroin intoxication (100 [95% CI 39.7–100]) was the highest in our study.
The second highest effect size of rhabdomyolysis in ICU patients was observed in intoxication with amphetamines (71.5 [95% CI 12.8–100]). The etiology of amphetamine-induced rhabdomyolysis has traditionally been attributed to agitation and/or physical restrain with intense isometric muscle contraction. However, many patients who use amphetamines are not agitated or restrained but experience rhabdomyolysis. Some of the indirect mechanisms and cofactors are gender difference (males at higher risk), monoamine receptor polymorphisms, cocaine and sedative co-injection, seizure, sepsis, and hyperthermia55. Hyperthermia is a major toxic reaction to amphetamines that can lead to rhabdomyolysis, hypotension, disseminated intravascular coagulation (DIC), and AKI. Hyperthermia occurs as a result of complex interactions between serotonin, dopamine, norepinephrine, and environmental conditions71. In some studies that reported a high frequency of rhabdomyolysis, there are several potential explanations for poor clinical outcomes and rhabdomyolyses like hyperthermia, concertation of amphetamines, and prolonged hypoxia. Most of the severe morbidity and mortality in these cases can be attributed to hyperthermia effects71.
In this study we found a higher effect size for patients with mono-intoxication than those with multi-intoxication following use of psychoactive substances. In many studies, alcohol abuse has been identified as a main cause of rhabdomyolysis8,38 and other studies regarding the etiology of rhabdomyolysis have reported opioid overdose as a significant contributing factor to rhabdomyolysis8. In this regard, Talaie et al. reported that opium poisoning is the most common cause of rhabdomyolysis (23.3%), followed by poisoning with benzodiazepines, phenobarbital, propranolol, aluminum phosphide, alcohol, and co-poisoning8. Also, Babak et al.in their study reported that rhabdomyolysis is mostly associated with methadone abuse, followed by opium abuse, and is more commonly correlated with poisoning in younger patients8,38.
Limitations
The final analysis in our study was substantially limited in the number and quality of studies available. Our review only included the published studies but we tried our best to contact researchers and obtain more information about their studies. Also, few studies in our review reported seizure induced by psychoactive intoxication that may contribute to rhabdomyolysis in intoxicated patients. The next limitation is that we had disparities in the distribution of intoxication severity in total wards subgroup affecting the effect size of the study. Furthermore, distribution of type of psychoactive substance in the subgroup of multiple poisoning was different between studies, which affects the effect size of each study. These limitations and lack of clarity in the studies caused high heterogeneity in our analysis. Another limitation of this study is that we searched only studies with English full text or at least English abstracts, and also subgroup analysis was not possible based on the dose and route of the substance used, blood levels of the drug, and other variables due to insufficient reported data in the studies. In addition, we did not perform any blinding process for all stages of study selection, quality assessment, and data extraction. Therefore, we propose running more comprehensive and original research in this regard to help make a better conclusion regarding the incidence of rhabdomyolysis in patients with psychoactive substance intoxication.
Conclusion
In conclusion, this systematic review and meta-analysis revealed high incidence of rhabdomyolysis occurrence in patients with heroin and amphetamine intoxication compared to other psychoactive substances. Clinicians should anticipate this complication, monitor for rhabdomyolysis particularly in the ICU, and institute appropriate treatment protocols early in the patient’s clinical course.
Data availability
All data generated or analyzed during this study are available from the corresponding author on reasonable request.
References
Parrott, A. C. Mood fluctuation and psychobiological instability: The same core functions are disrupted by novel psychoactive substances and established recreational drugs. Brain Sci. 8(3), 43 (2018).
Shukla, L., Ghadigaonkar, D. S. & Murthy, P. Poisoning with drugs of abuse: Identification and management. Indian J. Crit. Care Med. 23, S296 (2019).
Fudin, H. R., Babin, J. L., Hansen, A. L. & Ray, S. D. Side effects of drugs annual. Drugs 39, 31–55 (2017).
Thongprayoon, C. et al. Acute kidney injury in hospitalized patients with methanol intoxication: National Inpatient Sample 2003–2014. Hosp. Pract. 49, 203–208 (2021).
Petejova, N. & Martinek, A. Acute kidney injury due to rhabdomyolysis and renal replacement therapy: A critical review. Crit. Care. 18, 1–8 (2014).
Torres, P. A., Helmstetter, J. A., Kaye, A. M. & Kaye, A. D. Rhabdomyolysis: Pathogenesis, diagnosis, and treatment. Ochsner J. 15, 58–69 (2015).
Cervellin, G., Comelli, I. & Lippi, G. Rhabdomyolysis: Historical background, clinical, diagnostic and therapeutic features. Clin. Chem. Lab. Med. 48, 749–756 (2010).
Talaie, H. et al. Rhabdomyolysis among acute human poisoning cases. Hum. Exp. Toxicol. 26, 557–561 (2007).
Richards, J. R. et al. The association between cocaine use detected on drug screening and rhabdomyolysis. Toxicol. Commun. 4, 18–24 (2020).
Waldman, W. et al. Rhabdomyolysis related to acute recreational drug toxicity: A Euro-DEN study. PLoS ONE. 16, e0246297 (2021).
Sheibani, M. et al. Troponin, a predictor of mortality in methadone exposure: an observational prospective study. J. Am. Heart Assoc. 10, e018899 (2021).
Lavergne, V. et al. Why are we still dialyzing overdoses to tricyclic antidepressants? A subanalysis of the NPDS database. Semin. Dial. 1, 1–10 (2016).
Toews, L. C. Compliance of systematic reviews in veterinary journals with preferred reporting items for systematic reviews and meta-analysis (PRISMA) literature search reporting guidelines. J. Med. Libr. Assoc. 105, 233 (2017).
Stahl, K., Rastelli, E. & Schoser, B. A systematic review on the definition of rhabdomyolysis. J. Neurol. 267, 877–882 (2020).
Aghabiklooei, A., Edalatparvar, M., Zamani, N. & Mostafazadeh, B. Prognostic factors in acute methadone toxicity: A 5-year study. J. Toxicol. 2014, 1–6 (2014).
Arefi, M. et al. Renal failure prevalence in poisoned patients. Nephro-Urology. 6, 11910 (2014).
Armenian, P. et al. Multiple MDMA (Ecstasy) overdoses at a rave event: A case series. J. Intens. Care Med. 28, 252–258 (2013).
Azarakhsh, M., Rahimi, M., Zamani, N. & Arbabi, S. Refined opium extract (shireh) poisoning: A 10-year retrospective study in a referral poison control centre in Iran. Asia Pac. J. Med. Toxicol. 10, 12–17 (2021).
Brahmi, N. et al. Methanol poisoning in Tunisia: Report of 16 cases. Clin. Toxicol. 45, 717–720 (2007).
Bruggisser, M., Bodmer, M., Wilks, M., Kupferschmidt, H. & Liechti, M. Retrospective analysis of stimulant abuse cases reported to the Swiss Toxicological Information Centre during 1997–2009. in Swiss medical weekly. (2010).
Burton, B. N., Lin, T. C., Said, E. T. & Gabriel, R. A. National trends and factors associated with inpatient mortality in adult patients with opioid overdose. Anesthes. Analg. 128, 152–160 (2019).
Caldicott, D. G. et al. Dancing with “death”: p-Methoxyamphetamine overdose and its acute management. J. Toxicol. Clin. Toxicol. 41, 143–154 (2003).
Chhabra, N., Gimbar, R. P., Walla, L. M. & Thompson, T. M. Emergency department patient burden from an electronic dance music festival. J. Emerg. Med. 54, 435–439 (2018).
Dabby, R. et al. Acute heroin-related neuropathy. J. Periph. Nerv. Syst. 11, 304–309 (2006).
Forrester, M. B., Kleinschmidt, K., Schwarz, E. & Young, A. Synthetic cannabinoid exposures reported to Texas poison centers. J. Addict. Dis. 30, 351–358 (2011).
Gilley, M. et al. Synthetic cannabinoid exposure in adolescents presenting for emergency care. Pediatr. Emerg. Care. 37, e13–e16 (2021).
Greene, S. L., Dargan, P. I., O’connor, N., Jones, A. L. & Kerins, M. Multiple toxicity from 3, 4-methylenedioxymethamphetamine (“ecstasy”). Am. J. Emerg. Med. 21, 121–124 (2003).
Halpern, P. et al. Morbidity associated with MDMA (ecstasy) abuse: A survey of emergency department admissions. Hum. Exp. Toxicol. 30, 259–266 (2011).
Hermanns-Clausen, M., Kneisel, S., Szabo, B. & Auwärter, V. Acute toxicity due to the confirmed consumption of synthetic cannabinoids: Clinical and laboratory findings. Addiction. 108, 534–544 (2013).
Heyerdahl, F. et al. Acute poisonings treated in hospitals in Oslo: A one-year prospective study (II): Clinical outcome. Clin. Toxicol. 46, 42–49 (2008).
Imam, S. F. et al. Bath salts intoxication: A case series. J. Emerg. Med. 45, 361–365 (2013).
Isoardi, K. Z., Ayles, S. F., Harris, K., Finch, C. J. & Page, C. B. Methamphetamine presentations to an emergency department: Management and complications. Emerg. Med. Austral. 31, 593–599 (2019).
Isoardi, K. Z., Mudge, D. W., Harris, K., Dimeski, G. & Buckley, N. A. Methamphetamine intoxication and acute kidney injury: A prospective observational case series. Nephrology. 25, 758–764 (2020).
Kaewput, W. et al. Inpatient burden and mortality of methanol intoxication in the United States. Am. J. Med. Sci. 361, 69–74 (2021).
Kamijo, Y. et al. A multicenter retrospective survey of poisoning after consumption of products containing synthetic chemicals in Japan. Intern. Med. 53, 2439–2445 (2014).
Kasper, A. M. et al. Severe illness associated with reported use of synthetic cannabinoids: a public health investigation (Mississippi, 2015). Clin. Toxicol. 57, 10–18 (2019).
Katz, K. D. et al. Case series of synthetic cannabinoid intoxication from one toxicology center. West. J. Emerg. Med. 17, 290 (2016).
Babak, K., Mohammad, A., Mazaher, G., Samaneh, A. & Fatemeh, T. Clinical and laboratory findings of rhabdomyolysis in opioid overdose patients in the intensive care unit of a poisoning center in 2014 in Iran. Epidemiol. Health. 39, 4. https://doi.org/10.4178/epih.e2017050 (2017).
Kitchen, S. A. et al. Trends and outcomes of serious complications associated with non-fatal opioid overdoses in Ontario, Canada. Drug Alcohol Depend. 225, 108830 (2021).
Kourouni, I., Mourad, B., Khouli, H., Shapiro, J. M. & Mathew, J. P. Critical illness secondary to synthetic cannabinoid ingestion. JAMA Netw. Open. 3, e208516–e208516 (2020).
Lam, S.-M., Lau, A.C.-W. & Yan, W.-W. Over 8 years experience on severe acute poisoning requiring intensive care in Hong Kong, China. Hum. Exp. Toxicol. 29, 757–765 (2010).
Lund, C. et al. A one-year observational study of all hospitalized acute poisonings in Oslo: Complications, treatment and sequelae. Scand. J. Trauma Resuscit. Emerg. Med. 20, 1–11 (2012).
Mehrpour, O., Hoyte, C., Amirabadizadeh, A. & Brent, J. Clinical characteristics and time trends of hospitalized methadone exposures in the United States based on the Toxicology Investigators Consortium (ToxIC) case registry: 2010–2017. BMC Pharmacol. Toxicol. 21, 1–9 (2020).
Melli, G., Chaudhry, V. & Cornblath, D. R. Rhabdomyolysis: an evaluation of 475 hospitalized patients. Medicine. 84, 377–385 (2005).
Monte, A. A. et al. Characteristics and treatment of patients with clinical illness due to synthetic cannabinoid inhalation reported by medical toxicologists: A ToxIC database study. J. Med. Toxicol. 13, 146–152 (2017).
Mozafari, N., Talaie, H., Shoaei, S. D., Hashemian, M. & Mahdavinejad, A. Survey on hypothermia and hyperthermia in poisoned patients in a unique referral hospital, Tehran, Iran. Iran. Red Crescent Med. J. 18, 483 (2016).
Morrow, R. L., Bassett, K., Maclure, M. & Dormuth, C. R. Outcomes associated with hospital admissions for accidental opioid overdose in British Columbia: A retrospective cohort study. BMJ Open. 9, e025567 (2019).
Ng, F. Ten-year profile of acute poisoning patients presenting to an accident and emergency department requiring intensive care in a regional hospital of Hong Kong. Hong Kong J. Emerg. Med. 26, 3–14 (2019).
Nicol, J. J. et al. Deaths from exposure to paramethoxymethamphetamine in Alberta and British Columbia, Canada: A case series. Can. Med. Assoc. Open Access J. 3, E83–E90 (2015).
O’Connor, A. D., Padilla-Jones, A., Gerkin, R. D. & Levine, M. Prevalence of rhabdomyolysis in sympathomimetic toxicity: A comparison of stimulants. J. Med. Toxicol. 11, 195–200 (2015).
Oladunjoye, A. O., Oladunjoye, O. O., Olubiyi, O., Yee, M. R. & Espiridion, E. D. Predictors and outcomes of invasive mechanical ventilation in opioid overdose hospitalization in the United States. Cureus. 12, 9788 (2020).
Pajoum, A. et al. Rhabdomyolysis and acute poisoning; a brief report. Emergency 6, 1–10 (2018).
Rahimi, M. et al. Predictive factors of mortality in acute amphetamine type stimulants poisoning; a review of 226 cases. Emergency 6, 1–10 (2018).
Rahimi, M. et al. Planned Versus unplanned extubation in opioid overdose patients: Does it have any effect on the prognosis? A cohort study. Iran. J. Toxicol. 16, 185–194 (2022).
Richards, J. R. et al. Rhabdomyolysis, methamphetamine, amphetamine and MDMA Use: Associated factors and risks. J. Dual Diagn. 16, 429–437 (2020).
Riederer, A. M. et al. Acute poisonings from synthetic cannabinoids—50 US toxicology investigators consortium registry sites, 2010–2015. MMWR. Morbid. Mortal. Wkly. Rep. 65, 692 (2016).
Ridpath, A. et al. Illnesses and deaths among persons attending an electronic dance-music festival—New York City, 2013. MMWR. Morbid. Mortal. Wkly. Rep. 63, 1195 (2014).
Sporer, K. A. & Dorn, E. Heroin-related noncardiogenic pulmonary edema: A case series. Chest 120, 1628–1632 (2001).
Taheri, S. K., Afzali, S. & Torabian, S. Rhabdomyolysis syndrome in alcohol, psychotropic drugs, and illicit substance poisonings. Iran. J. Toxicol. 7, 866–870 (2013).
Talaie, H. et al. High sensitivity versus low level of vancomycin needs to be concern for another alternative anti-Staphylococcus aureus as the first-line antibiotic. Acta Bio Med. Atenei Parmensis 90, 498 (2019).
Talaie, H., Hosseini, S. M., Nazari, M., Esfahani, M. S. & Barari, B. Risk factors of hospital-acquired thrombocytopenia in toxicological intensive care unit. Int. J. Med. Toxicol. Forens. Med. 10, 32256–32256 (2020).
Tatusov, M., Mazer-Amirshahi, M., Abbasi, A. & Goyal, M. Clinical effects of reported synthetic cannabinoid exposure in patients admitted to the intensive care unit. Am. J. Emerg. Med. 37, 1060–1064 (2019).
Weng, T.-I. et al. Comparison of clinical characteristics between meth/amphetamine and synthetic cathinone users presented to the emergency department. Clin.Toxicol. 1, 1–7 (2022).
West, P. L., McKeown, N. J. & Hendrickson, R. G. Methamphetamine body stuffers: An observational case series. Ann. Emerg. Med. 55, 190–197 (2010).
Yalçın, M., Tunalı, N., Yıldız, H., Oğuz, A. & Gültekin, B. K. Sociodemographic and clinical characteristics of synthetic cannabinoid users in a large psychiatric emergency department in Turkey. J. Addict. Dis. 37, 259–267 (2018).
Boess, F., Ndikum-Moffor, F. M., Boelsterli, U. A. & Roberts, S. M. Effects of cocaine and its oxidative metabolites on mitochondrial respiration and generation of reactive oxygen species. Biochem. Pharmacol. 60, 615–623 (2000).
Dluzen, D. E. & McDermott, J. L. Estrogen, testosterone, and methamphetamine toxicity. Ann. N. Y. Acad. Sci. 1074, 282–294 (2006).
Büttner, A. The neuropathology of drug abuse. Neuropathol. Appl. Neurobiol. 37, 118–134 (2011).
Wang, I. K. et al. Rhabdomyolysis, acute renal failure, and multiple focal neuropathies after drinking alcohol soaked with centipede. Renal Fail. 26, 93–97 (2004).
Warner-Smith, M., Darke, S. & Day, C. Morbidity associated with non-fatal heroin overdose. Addiction 97, 963–967 (2002).
Green, A. R., O’shea, E. & Colado, M. I. A review of the mechanisms involved in the acute MDMA (ecstasy)-induced hyperthermic response. Eur. J. Pharmacol. 500, 3–13 (2004).
Author information
Authors and Affiliations
Contributions
The authors’ responsibilities were as follows S.S., E.K., A.A. and S.M. Conceptual design; A.A., S.E. literature search; A.A., B.T., J.G. and A.S. screened the studies and extracted the data; B.M., S.M. and E.K. assessed the risk of bias; M.Y. and A.A. analyzed the data; A.A., A.S., T.M., J.G. and S.M. drafted the manuscript; M.Y., S.S. and E.K. modified the final manuscript; and all authors: read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Amanollahi, A., Babeveynezhad, T., Sedighi, M. et al. Incidence of rhabdomyolysis occurrence in psychoactive substances intoxication: a systematic review and meta-analysis. Sci Rep 13, 17693 (2023). https://doi.org/10.1038/s41598-023-45031-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-45031-4
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.