Abstract
Multiple evidence indicates that perinatal factors make impact on immune development and affect offspring allergic rhinitis (AR) risk. In this systematic review and meta-analysis, we examined available published studies to clarify the relationship between cesarean section (C-section) and offspring AR in children. To explore the relationship between C-section, especially the special attention was paid to different cesarean delivery mode, and the risk of AR in children. Articles were searched using PubMed, Web of Science, EMBASE, Cochrane Library, China knowledge Network, Wanfang, and China Science and Technology Journal databases. A meta-analysis of 22 studies published before August 1, 2022, which included 1,464,868 participants, was conducted for statistical analysis with RevMan5.4. The correlation strength between C-section and offspring AR was determined by combining odds ratio (OR) and 95% confidence interval (95% CI). Meta-regression and subgroup analyses were used to explore potential sources of heterogeneity. Publication bias was detected using the funnel chart and Egger tests. Meta-analysis revealed that there was a significant correlation between C-section and children AR (OR = 1.19, 95% CI: 1.12–1.27, P < 0.001), especially C-section with a family history of allergy (OR = 1.82, 95% CI: 1.36–2.43, P < 0.001). Moreover, elective C-section (without genital tract microbe exposure) had the higher risk of offspring AR (OR = 1.24, 95% CI: 1.05–1.46, P = 0.010) compared with the whole study. Meta-regression demonstrated that sample size explained 38.0% of the variability between studies, and year of publication explained 18.8%. Delivery by C-section, particularly elective C-section and C-section with a family history of allergy can increase the risk of AR in children.
Similar content being viewed by others
Introduction
Allergic rhinitis (AR)1 is an IgE-mediated inflammation of the nasal mucosa, with symptoms including excess tear production, runny nose, and sneezing. Studies have reported that the global prevalence of AR is rising2 with a prevalence of 10%–30% in the United States. However, relatively few studies reported the incidence of AR in children. The International Childhood Asthma and Allergy in Childhood (ISAAC)3 estimated the prevalence of allergic diseases in different age groups through a multi-centered global survey, and reported prevalence rates of 8.3% and 15.1% for those aged 6–7 and 13–14 years, respectively. A meta-analysis of all the recent studies conducted under the ISAAC protocol (1,430,329 children aged 0–18 years) reported4 an overall AR prevalence of 12.66%. Currently, many regional studies reported the prevalence of childhood AR in China, varying from 12.08 to 30.04% in every city5. It is important to note that AR is more than just sneezing and a nuisance for the children. AR may contribute to sleep loss, learning impairment, decreased overall cognitive functioning, aggravation of underlying asthma, and increased propensity to develop asthma6,7.
According to the recommendation of the World Health Organization (WHO), the cesarean section (C-section) rate should be controlled at 10–15%8. However, women's preference for C-section has increased due to concerns of the short- and long-term risks related to vaginal delivery (VD)9. Presently, the C-section rate worldwide continuously exceeds the WHO recommendation10. The national epidemiological survey of the C-section rate in China reported an annual increase in the domestic C-section rate, reaching 34.9%11. C-section can be a life-saving intervention when medically indicated, but this procedure can also lead to short-term and long-term health effects for women and children. C-section is not only associated with an increased risk of uterine rupture, abnormal placentation, ectopic pregnancy, stillbirth, and preterm birth in woman, but also associated with altered immune development in children, like allergy, atopy, and asthma, and reduced intestinal gut microbiome diversity in children12.
Correlation between C-section and AR risk in future generations have attracted much attention. A previous meta-analysis of C-section and offspring AR was conducted in 200913, which included only seven studies and revealed a positive correlation between C-section and offspring AR. After that, more and more studies still further focus on this topic and have assessed the association between mode of delivery and AR in the following years. Many studies have explored the relationship between C-section and the incidence of AR in children. Because a discrepancy in quantity and degree, the results are not consistent. Some previous studies showed that C-section is not associated with the risk of AR in children14,15,16, Others showed that C-section17,18,19,20, especially C-section with a family history of allergy21,22,23 could increase the risk of AR in children. More recently, studies linking the risk of AR to mode of delivery have examined the roles of emergency C-section after the onset of labour, versus elective prelabour C-section. Therefore, whether C-section increases the risk of offspring AR remains controversial24. Moreover, the previous meta-analysis13 included people of all ages including adults and children. However, the risk of AR in adults is often related to smoking, long-term exposure to adverse environments, occupational factors and so on25,26. Our study concentrated on the effects of birth-mode changes on AR in children, and more on early-life observed effects. In line with the ‘hygiene hypothesis’, the intestinal microflora may be modified in those children who are not exposed to the vaginal microbiota, but the effect decreases with the age of the offspring27. The mode of delivery is an important factor affecting the composition of intestinal flora in newborns and infants. Caesarean section may negatively affect the normal development of the microbiota, leading to decreased amounts of lactobacilli and bifidobacteria and increased amounts of Clostridia28. Reduced diversity of the intestinal microbiota during infancy is associated with increased risk of allergic disease at children29. To address this problem, we conducted a systematic review and meta-analysis of epidemiological studies to explore the relationship between C-section, especially the special attention was paid to different cesarean delivery method, and the risk of AR in children.
Materials and methods
Protocol and registration
This systematic review was registered on PROSPERO (CRD: 42021251713) and conformed to the MOOSE30 and Preferred Reporting Items for Systematic Reviews and Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines31.
Information sources and search strategy
We searched international online databases, such as PubMed, EMBASE, Web of Science, CNKI, and VIP and Wanfang. In PubMed, the searches were carried out with a combination of subject and free words. The search strategy was designed and set up by an expert in database searches (Yide Yang) and two investigators (Zixin Liu and Junrong Chen).We have arranged the search terms according to PICO model to provide more details to get more accurate search results. According to the search subject of this article. Search terms used were as follows: Patient: pregnant women, pregnant, minors, adolescent, offspring , early life , toddler, child; Intervention: cesarean section, c-section, abdominal deliveries, post-cesarean section; Comparison: natural childbirth, vaginal delivery; Outcome: allergic rhinitis, hay fever, allergic rhinoconjunctivitis, seasonal allergic rhinitis. The titles and abstracts of the studies were screened, and full papers were retrieved if a decision could not be made based on the abstract alone. To identify all potentially eligible studies, the reference lists of all comprised studies were examined. We searched from the beginning of databases up to August 1, 2022.
Inclusion and exclusion criteria
The inclusion criteria were as follows: Study population: children aged 0–18 years old. Study types: cohort studies, cross-sectional studies, and case–control studies. Exposure factors: children delivery through C-section; Outcome: offspring diagnosed with AR; and relative risk (RR), hazard ratio (HR),or odds ratio (OR) and their confidence intervals can be obtained, or enough data to calculate them.
The exclusion criteria were as follows: literature with incomplete relevant data and when the original data could not be obtained from the author; non-Chinese and non-English literature; case studies, systematic reviews, dissertations, meta-analyses; and low-quality research.
Data extraction
All studies were included in this systematic review if they fulfilled the inclusion criteria. After an initial sweep, the two investigators (Zixin Liu and Junrong Chen) independently performed a thorough title and abstract screening. Articles were added to this shortlist of studies through reference cross-checking. Disagreements were resolved by a consensus discussion in a meeting with the senior author (Jian Li). After agreement between the two investigators, studies were merged into a final list for full-text analysis and data extraction. Both investigators agreed on the final shortlist for data analysis and extraction.
Data collection process
Two researchers (Zixin Liu, Junrong Chen) independently examined the literature and extracted the data. In the case of disagreement, a third researcher (Jian Li) was consulted if necessary. The final determination of the extracted literature was the result of a consensus reached by three individuals. If there were significant doubts regarding the data from a particular study, the corresponding authors were contacted for confirmation of the extracted data. Information extracted from the tables in the literature was as follows: study details (author, year of publication, and journal name); study methodology and characteristics; study design (case–control study, cross-sectional study, and cohort study); participant information (age, sex, country or territory, sample size, diagnosis, parental atopy history, and type of C-section); follow-up or study duration, relevant independent, and dependent variables; data analysis methods; adjustment for confounding factors; and effects obtained from study results (studies offered available data on the relevant risk estimates including an odds ratio [OR] and their 95% confidence interval [95% CI], or enough data to compute them). If studies reported relative risks (RR) or hazard ratios (HR) as the effect size, Hazard ratios were directly considered as RR32. RR (95% CI) were converted to OR (95% CI) by using the following formula: OR = [(1-P0) * RR/ (1-RR * P0)], where P0 indicates the incidence of the outcome of interest in the non-exposed/reference group. The standard error of the resulting converted relative risk was then determined with this formula: SElog (RR) = SElog (OR)*log (RR)/log (OR)33. This meta-analysis used OR as the effect size because some research reports only provided OR and did not include HR or RR. In such situations, using OR as the effect size allowed for better utilization of the data for comprehensive analysis. Adopting a unified algorithm and interpretation method could simplify the process of data integration and comparison, and improve the reliability and interpretability of the results. When several estimates were reported within the same study, the most adjusted model was used for the pooled analysis.
Quality assessment
For cohort and case–control studies, the Newcastle–Ottawa Scale (NOS)34 was used to assess the study quality, and the total NOS score was 10. NOS was used to evaluate case–control (selection, comparability, and exposure) and cohort studies (selection, comparability, and results). Those with scores of ≤ 5 and ≥ 6 were classified as low- and high-quality studies, respectively. For cross-sectional studies, 11 checklists recommended by the Agency for Healthcare Research and Quality35 were used to evaluate the studies36,37. A higher total score of each item corresponded to a higher-quality document; if the answer was "no" or "unclear,” the item score was "0," and if the answer was "yes," the item score was "1," of which the fifth was a reverse score. The evaluation of literature quality was as follows: low quality = 0–3, medium quality = 4–7, and high quality = 8–1138. The studies were independently evaluated by two researchers (Zixin Liu and Junrong Chen). In case of differences, a third experienced investigator (Jian Li) was consulted until a consensus was reached.
Data analysis
RevMan5.4 and STATA16.0 was used for statistical analysis, and OR was used as the effect index. The correlation strength between C-section and offspring AR was determined by combining OR and 95% CI. Cochran’s Q test and I2 index were used to evaluate the heterogeneity of the studies39. Regarding the heterogeneity test, a P-value of ≥ 0.1 and I2 of ≤ 50% suggested homogeneity among the studies, and a fixed effects model was used for merging; a P-value of ≤ 0.1 and I2 of ≥ 50% suggested heterogeneity among the studies, and a random effects model was used to estimate the combined effect. Prediction intervals were calculated in meta-analyses40 with at least four studies41 to take the large between-study heterogeneity into account. Meta-regression was used to investigate potential sources of heterogeneity between the studies. For analytical purposes, the studies’ characteristics were grouped as follows: study design; study areas; year of publication; sample size; age; diagnostic mode and follow-up period. According to the included studies, a subgroup analysis was also performed on type of C-section and C-section with or without history of atopy. Publication bias was assessed using funnel plot and Egger’s test correlation. Sensitivity analysis was performed by excluding the study one by one. P < 0.05 was deemed statistically significant.
Results
Study retrieval steps and screening results
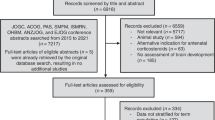
Two reviewers independently screened the studies included in the meta-analysis. We calculated the percentage agreement to evaluate the consistency of the two reviewers in the search process for the meta-analysis. The first round of percentage agreement was 86.7%. After discussing with a third experienced expert, the second round of percentage agreement improved to 100%. We retrieved 379 citations from multiple electronic databases. After excluding 158 repetitive studies, and reading titles and abstracts, 155 studies were excluded because they were systematic reviews, reviews, letters, case reports, and study factors were not relevant. After evaluating 66 full-text articles, studies with an outcome index of > 18 years (n = 9), lack of focus on the between C-section and childhood AR (n = 30), those that could not obtain specific data (n = 3), and low quality studies (n = 2) were excluded. Finally, 22 articles were included in this study14,15,16,17,18,19,20,21,22,23,42,43,44,45,46,47,48,49,50,51,52,53. The screening process and results are shown in Fig. 1.
Basic characteristics and quality evaluation of studies
This study included 22 studies involving 1,464,868 participants. Regarding research methods, there were 11 cohort studies14,16,18,19,20,23,42,43,44,48,51, 2 case–control studies21,49, and 9 cross-sectional studies15,17,22,45,46,47,50,52,53. For study areas, 6 were conducted in Europe14,17,18,42,43,44, 9 in Asia16,21,45,46,47,48,49,50,52, 5 in North America19,20,23,51,53, and 2 in South America15,22. In previous studies, Richards51 used RR as the effect value, Mitselou18 used HR. We ensured the comprehensiveness of analysis and the maximization of statistical ability by calculating OR54 as the combination effect value55.
The diagnosis of AR included the questionnaire survey and clinical diagnosis. In the questionnaire survey, 515,22,46,52,53 studies were conducted based on the ISAAC questionnaire. Clinical diagnosis was based on the physician's judgment based on the clinical history, using International Classification of Disease (ICD) diagnosis codes or Positive Skin Prick Test (SPT) reaction from the electronic health records.
Cohort studies were mainly from Europe and North America, while cross-sectional and case–control studies were concentrated in Asia. Pistiner 's study23 examined the relation between mode of delivery and the development of AR with a parental history of atopy, 321,22,45 studies used stratified analysis of C-section according to family history of allergies,318,19,51 studies reported the effect of different types of C-section on AR in children. The most adjusted covariates included age, preterm birth or neonatal complications, educational level, income, and birth order. The general characteristics and quality evaluation results were shown in Table 1.
Relationship between C-section and offspring AR
In this meta-analysis, the rate of heterogeneity was high (I2 = 68%, P < 0.001). A random effects model was used to demonstrate the results of the meta-analysis, and revealed that there was a significant correlation between C-section and children AR (OR = 1.19, 95% CI: 1.12–1.27, P < 0.001). The results were shown in Fig. 2.
Subgroup analysis and meta-regression
A series of random-effects subgroup analyses and meta-regression was conducted to examine whether the association between C-section and AR. For our study, the associations between C-section and AR were not substantially altered by study design (cohort study/cross-sectional study/ case–control study), study areas (Europe/Asia/North America/South America), age (< 6, ≥ 6, both), diagnostic mode (self-made questionnaire, ISAAC questionnaire, clinical diagnosis, questionnaire & diagnosis), and follow-up period (N/A, < 5, ≥ 5). However, meta-regressions confirmed the year of publication (2000–2009, 2010–2019, 2020–2022), and sample size (< 1000, 1000–9999, ≥ 10,000) may have been the possible causes of heterogeneity.
In the meta-regression analysis, sample size (38.0%) and year of publication (18.8%) explained the variability between studies. Subgroup analysis showed that, a greater effect size was observed for articles published before 2020 (1.32[1.09–1.60] and 1.24 [1.12–1.37]) compared with the whole study [1.19(1.12–1.27)], and the difference was statistically significant in the meta-regression (P = 0.031). A larger effect size of studies with sample sizes less than 10,000 (1.75 [1.42–2.15] and 1.30 [1.18–1.43]) was found in comparison with studies model assessment of sample size greater than 10,000 (1.10 [1.03–1.17]), and the difference was also significant (P = 0.001). Subgroup analysis indicated that for most of the variables evaluated, the association measurement continued to be associated with AR. Subgroup analysis and meta-regression results were shown in Table 2.
Results of the subgroup analysis according to the family history of allergy
Four studies were incorporated. The result demonstrated that the heterogeneity was low (I2 = 0%, P = 0.700). A fixed effects model was used to estimate the combined effect. The meta-analysis demonstrated that C-section with parental history of allergy had a higher risk of offspring AR (OR = 1.82, 95% CI: 1.36–2.43, P < 0.001) compared with C-section with No parental history of allergy (OR = 1.44, 95% CI: 1.06–1.94, P = 0.020) and the whole analysis (OR = 1.19, 95% CI: 1.12–1.27, P < 0.001) as shown in Fig. 3.
Results of the subgroup analysis according to C-section type
Emergent C-section, means a period of vaginal delivery and with genital tract microbe exposure but terminated because iatrogenic reason, was showed significantly higher risk for children AR than elective C-section in Mitselou’s18 study. But Richards’s51 study showed not significant (OR = 0.90, 95% CI: 0.78–1.04). So meta-analysis was conducted and showed that elective C-section (without genital tract microbe exposure) had the higher risk of offspring AR (OR = 1.24, 95% CI: 1.05–1.46, P = 0.010) compared with the whole analysis (OR = 1.19, 95% CI: 1.12–1.27, P < 0.001). In our research, no association between emergency cesarean (with genital tract microbe exposure) delivery and AR in children was found (OR = 1.02, 95% CI: 0.82–1.27, P = 0.870). The results were shown in Fig. 4.
Publication bias analysis and sensitivity test
The funnel chart was drawn using Stata16.0, and the trim-and-fill method was used to calculate the combined estimate after adjusting for publication bias. The resulting estimate did not change significantly. The statistical tests of publication bias were not significant for the OR of AR in the overall C-section (Egger’s test: p = 0.137), cohort study (Egger’s test: p = 0.390), cross-sectional study (Egger’s test: p = 0.675) and case–control study (Egger’s test: p = 0.752). Indicating no publication bias in this meta-analysis. Removing the included literatures one by one did not affect the results of this study, which means that the above results are stable and reliable.
Discussion
This is the first meta-analysis evaluating the effects of C-section on the AR risk of offspring in child. Our systematic review revealed 22 studies reporting on cesarean delivery and offspring AR risk. Findings suggest that the overall pooled OR for AR in children was 1.19(95% CI: 1.12–1.27, P < 0.001).
Results from the meta-regression analysis revealed that sample size and publication year affected the association between C-section and AR, which explained nearly 38% and 19% of the variability between studies. We observed larger effects of studies with sample sizes smaller than 10,000. The current analysis finds evidence of a negative correlation between sample size and effect size in studies that get included in meta-analyses56. In a large sample size study, there may be people whose children have AR but have not reported or lost to follow up, resulting in an effect value that is lower than the actual57,58. In the subgroup analysis, although the effect value of the cohort study is lower than the whole studies, the overall statistical difference can also be explained in the follow-up time. The study found that the study with a follow-up time of less than 5 years has a larger effect value than the overall study. The small trials were of low quality of methodology compared with large trials which may partly account for the small-study effects and overestimating effect sizes. Meanwhile, meta-regression on publication year revealed that the publication year was also a factor contributing to the higher heterogeneity in this study. Higher effect sizes were found in literature published in earlier years (2000–2009). There are several possible explanations for this finding. Firstly, it is possible that earlier literature relied more on self-made questionnaires for the assessment of AR. This could have led to the inclusion of children who had not yet developed AR, thus affecting the consistency of the results. With the passage of time, the diagnosis and assessment methods for allergic rhinitis have continuously improved and been standardized. This was also explained in our subgroup analysis of the diagnostic mode for AR. Additionally, it is likely that the heterogeneity in the results is also influenced by the advancements in research methods and medical technologies in different time periods. These changes may include more accurate and standardized data collection methods, as well as the utilization of more advanced medical technologies and treatment methods. These factors could contribute to increased heterogeneity in the results across different time periods. In conclusion, after incorporating the adjusted effect size finding. We had gotten a conclusion that the overall risk of AR in children delivered after C-section increased by 19% and increased by 82% after C-section with parental history of allergy. In addition, elective C-section had a higher risk of AR compared with the whole study.
We had observed that the overall risk of AR in children delivered through C-section increased. This is consistent with the results of previous studies on C-section and allergic diseases. Previous studies have shown that C-section increase the risk of asthma, food allergies, and allergic dermatitis in children of offspring59,60,61. In our subgroup analysis with a family history of allergies, we found that C-section in families with a history of allergies increased the risk of children developing AR, with heterogeneity reduced to 0%. One possible explanation for the reduced heterogeneity within this subgroup is the shared genetic predisposition to allergies among family members. This shared genetic background could contribute to a more consistent pattern of allergic reactions and therefore a lower heterogeneity in the subgroup. In addition to genetic factors, families often share similar living conditions, dietary habits, and exposure to potential allergens environmental influences within families which could contribute to the reduced heterogeneity. A family history of allergy is a common risk factor for allergic diseases in offspring. C-sections with a family history of allergy have a higher risk of AR in children than overall C-sections. This demonstrates that the interaction between genes and the environment plays an important role in the development of such diseases62.
Many studies have shown that delivery by C-section is associated with an increased risk of disease associated with allergic disease in the offspring, but these studies have generally not discriminated between the effect of acute and elective C-section. In our study, we observed that children who were born by elective C-section had a higher chance of developing AR. Similar results were found in other studies. A cohort study conducted in the Denmark included over 750,569 participants and showed that elective C-section was higher than the effect of emergency C-section on the risk of asthma63. In Behzad Darabi’s meta-analysis. They also found that children delivered by elective C-section have a higher risk of asthma than children delivered by emergency C-section64.
The etiology behind C-section leading to AR in children is not fully understood until now. Based on the hygiene hypothesis, the rapid progression of the epidemic was attributed to a reduced diversity of early environmental microbe exposure, which decreased the occurrence of allergic diseases, including eczema, AR, and asthma65. In a cohort study of allergic diseases in South Africa66, the incidence of AR in urban children (25.3%, 95% CI: 22.8–27.8%, P < 0.001) was significantly higher than that in rural children (40.5%, 95% CI: 37.7–43.4%, P < 0.001). The rate of C-section in urban areas was twice as high as that in rural areas. This may be because the antenatal and childhood contact with farm animals in a rural environment resulted in low prevalence of AR in children owing to multiple exposures that mediate strong protective epigenetic modification through the microflora diversity67,68,69. Healthy or ‘tolerant’ individuals often produce high amounts of allergen-specific IgG4 and/or IgA, and IgA-deficient individuals are at risk of developing an allergy70. Generally, the exposure to microorganisms results in lipopolysaccharide stimulation of Toll-like receptors to produce interleukin 12 (IL-12) and interferon λ (IFN-λ), which promote the development of the Thnaive cells into Th1 effector cells. An impaired regulation of this immune pathway to children born by C-section71. Factors that alter the composition and diversity of the microbiome, such as the mode of infant delivery have a strong effect on immune responses, altering the immune response from one what is consistent with a state of tolerance to a dysfunctional hyper-responsive state that is associated with allergic diseases. However, conflicting results have been observed in recent epidemiological investigations. First, the incidence of AR began to decline in some Western European countries, but there was no obvious evidence that it was due to the decline in sanitary condition or increase in the number of family members72. Second, in a cohort study by Weber73, on personal and family cleanliness and allergic disease, which included 399 participants, dust parameters in the household objectively reflected personal cleanliness, including hand washing and household cleanliness, although personal cleaning or household cleanliness was not associated with the risk of allergy. Other microbial components in the indoor dust that were not affected by personal hygiene might play an important role. Considering the reasons for the increasing prevalence of AR in children based on the hygiene hypothesis, further studies are needed to determine the underlying genetic and epigenetic mechanisms, focusing on the role of early allergens and differences in epidemiological studies74.
At the same time, some studies have also shown that differences in the colonization of neonatal intestinal flora caused by caesarean section are also related to allergic diseases in offspring. The developmental starting point of the infant gut microbiota is still unknown, but undoubtedly, the process of delivery seems to be a key point in the development of the neonatal microbiota. Babies born via C-section miss out on this initial exposure to the mother's microbiota. Instead, they are colonized primarily by the skin and environmental microorganisms present in the operating room. The intestinal microflora of infants born through VD had a higher abundance of Bifidobacterium, Lactobacillus, Escherichia coli, and Bacteroides; demonstrating greater intestinal microbial diversity75. Conversely, the microbiome of infants born via C-section shows an increased prevalence of either skin flora or potentially pathogenic microbial communities such as Klebsiella, Enterococcus, and Clostridium76. There are several explanations for this mechanism. Firstly, Shohei Akagawa77 points out that children born by C-section have characteristics such as reduced Bacteroides and decreased butyric acid concentration in the gut. The decrease in butyric acid concentration inhibits the differentiation of immature T cells into Tregs, which are crucial for suppressing excessive immune responses. This impairment of the immune system's ability to suppress excessive immune responses can lead to the development of allergic diseases. Secondly, there was evidence that low α-diversity and relative abundance of particular gut-commensal bacteria genera are associated with childhood respiratory diseases. However, the higher relative abundance of Bacteroidaceae, Clostridiaceae, and Enterobacteriaceae, and the lower relative abundance of Bifidobacteriaceae and Lactobacillaceae were associated with the development of allergies78. In addition, our study has shown that elective C-section is associated with a higher risk of AR. This might be because the newborns in contact with C-section related contaminants were exposed to the maternal genital tract microbiome due to factors, such as premature rupture of membranes, resulting in partial restoration of the newborn’s bacterial community to levels similar to those of babies born through VD79. In conclusion, the mode of delivery is a significance factor affecting the composition of intestinal flora in newborns and infants, which plays an important role in the development of the immune system.
A large number of studies have consistently observed that C-section could lead to an increased incidence of AR in children, therefore, most of the experiments were observational studies on the effects of C-section on allergic diseases in children. Several societal factors that may contribute to the rise in C-section rates include fear of VD among pregnant women, doctors' desire to avoid unknown medical risks, and cultural and religious differences in certain regions. Therefore, in order to reduce the C-section rate, it is recommended to consider the individual circumstances and medical indications for each delivery, including but not limited to strictly defining the indications for cesarean section, promoting VD, widespread adoption of painless delivery techniques, and encouraging women who have had a cesarean section to attempt vaginal birth after cesarean. Factors other than C-section might also affect the development of allergic diseases in children, including race, birth season, geographical location, social class or income level, tobacco exposure during pregnancy, changes in an indoor environment, adverse pregnancy outcomes, dietary changes, and antibiotic use15,18,80,81, which would cause the failure of immune tolerance and increase the risk of AR. In response to their preventive measures, Lunjani82 suggested that microbial exposure was the most effective way to prevent atopic diseases within 1–2 years after birth. For example, the orderly combination of infant intestinal microbial ecosystem was formed by an exposure to the maternal microflora, breastfeeding, and early exposure to various food, and environmental microorganisms during the prenatal period, childbirth, and delivery83. However, in a cohort study of 335 children by Jelding-Dannemand84, exclusive breastfeeding was observed to be ineffective in reducing early childhood sensitization or high-risk childhood related diseases at the age of 7 years. We found that most of the literature has corrected for breastfeeding, whether breastfeeding reduce the risk of C-section-related AR in our study remains unelucidated. However, the early introduction of supplementary foods, especially cereals, fish, and eggs, appeared to prevent asthma and allergies, and the introduction of supplementary foods as early as possible while continuing breastfeeding might be a more important strategy to prevent allergies in children85. These included studies did not specifically investigate and adjust the diet of mothers during pregnancy and children. It is necessary to study these factors in future research to explore whether diet can reduce the risk of AR in children born by C-section.
This study has several limitations. First, in the subgroup analysis regarding different types of C-section, we identified higher heterogeneity. Most of the studies investigating the relationship between C-section and AR in children were retrospective and did not include specific clinical indications for mothers choosing elective or emergency C-sections. Preterm delivery and low birth weight81 may affect the development of children's immune system, but some studies are aimed at term pregnancy, normal birth weight newborns as inclusion criteria, and the specific reasons for choosing C-section have not been described in the article, such as fetal distress, placenta previa, umbilical cord prolapse. Second, moderate-high heterogeneity across studies was detected for C-section and AR. Different issues could generate this heterogeneity, including different study population, study location, sample size, duration of follow-up, outcome diagnosis, publication areas, number of years, and treatment of confounding factors. Subgroup analysis and meta-regression were run to deal with the heterogeneity by using random-effect models. Third, this study included few small-scale studies and self-reported AR for the diagnosis, self-report or information from parental interviews may be liable to recall bias, which might have caused the actual prevalence to be higher than the present results, thereby reducing the universality of our findings, any deviations or inaccuracies in individual studies could have also influenced our analysis.
Therefore, C-section increase the risk of AR in children compared with VD. In the case of a high C-section rate worldwide, formulating specific clinical guidelines and implementing appropriate management plans helping reduce the AR risk in children after C-section need to be strengthened.
Conclusion
In conclusion, our study identifies and characterizes that C-section is associated with subsequent childhood AR. However, the confounding effects of delivery mode and Indications for C-section remain to be studied in more detail. Future well-designed studies are warranted to validate the findings of the current analysis. When the C-section rate remains high, it is suggested that the formulation of specific clinical guidelines and implementation of appropriate management plans can reduce the risk of AR.
Data availability
All data generated or analysed during this study are included in this published article.
Abbreviations
- AR:
-
Allergic rhinitis
- C-section:
-
Cesarean section
- OR:
-
Odds ratio
- 95% CI:
-
95% Confidence interval
- ISAAC:
-
International Childhood Asthma and Allergy in Childhood
- WHO:
-
World Health Organization
- VD:
-
Vaginal delivery
- NOS:
-
Newcastle–Ottawa Scale
- PRISMA:
-
Preferred reporting items for systematic reviews and meta-analyses
References
Weinberg, E. G. The WAO White Book on Allergy 2011–2012. Vol. 24 (2011).
Asher, M. I. et al. Worldwide time trends in the prevalence of symptoms of asthma, allergic rhinoconjunctivitis, and eczema in childhood: ISAAC Phases One and Three repeat multicountry cross-sectional surveys. The Lancet 368, 733–743. https://doi.org/10.1016/s0140-6736(06)69283-0 (2006).
Björkstén, B., Clayton, T., Ellwood, P., Stewart, A. & Strachan, D. Worldwide time trends for symptoms of rhinitis and conjunctivitis: Phase III of the International Study of Asthma and Allergies in Childhood. Pediat. Allergy Immunol. 19, 110–124. https://doi.org/10.1111/j.1399-3038.2007.00601.x (2008).
Pols, D. H. et al. Interrelationships between Atopic Disorders in Children: A Meta-Analysis Based on ISAAC Questionnaires. PloS One 10, e0131869. https://doi.org/10.1371/journal.pone.0131869 (2015).
Wang, M., Zheng, M., Wang, X. T. & Zhang, L. (2019) Research progress in epidemiology of allergic rhinitis in China. Chin. Arch. Otolaryngol. Head Neck Surg. 26, 415–420. https://doi.org/10.16066/j.1672-7002.2019.08.004.
Settipane, R. A. Complications of allergic rhinitis. Allergy Asthma Proc. 20, 209–213. https://doi.org/10.2500/108854199778339053 (1999).
Blaiss, M. S. Pediatric allergic rhinitis: physical and mental complications. Allergy Asthma Proc. 29, 1–6. https://doi.org/10.2500/aap2008.29.3072 (2008).
WHO. Appropriate technology for birth. Lancet (London, England) 2, 436–437 (1985).
Long, Q. et al. Prevalence of and reasons for women’s, family members’, and health professionals’ preferences for cesarean section in China: A mixed-methods systematic review. PLoS Med. 15, e1002672. https://doi.org/10.1371/journal.pmed.1002672 (2018).
Vogel, J. P. et al. Use of the Robson classification to assess caesarean section trends in 21 countries: A secondary analysis of two WHO multicountry surveys. Lancet Glob. Health 3, e260–e270. https://doi.org/10.1016/s2214-109x(15)70094-x (2015).
Li, H. T. et al. Geographic variations and temporal trends in Cesarean delivery rates in China, 2008–2014. JAMA 317, 69–76. https://doi.org/10.1001/jama.2016.18663 (2017).
Sandall, J. et al. Short-term and long-term effects of caesarean section on the health of women and children. Lancet (London, England) 392, 1349–1357. https://doi.org/10.1016/s0140-6736(18)31930-5 (2018).
Bager, P., Wohlfahrt, J. & Westergaard, T. Caesarean delivery and risk of atopy and allergic disease: Meta-analyses. Clin. Experim. Allergy 38, 634–642. https://doi.org/10.1111/j.1365-2222.2008.02939.x (2008).
Gerlich, J. et al. Pregnancy and perinatal conditions and atopic disease prevalence in childhood and adulthood. Allergy 73, 1064–1074. https://doi.org/10.1111/all.13372 (2018).
Gorris, A., Bustamante, G., Mayer, K. A., Kinaciyan, T. & Zlabinger, G. J. Cesarean section and risk of allergies in Ecuadorian children: A cross-sectional study. Immun. Inflamm. Dis. 8, 763–773. https://doi.org/10.1002/iid3.368 (2020).
Han, D. H. et al. Long-term breastfeeding in the prevention of allergic rhinitis: Allergic rhinitis cohort study for kids (ARCO-Kids Study). Clin. Experim. Otorhinolaryngol. 12, 301–307. https://doi.org/10.21053/ceo.2018.01781 (2019).
Krzych-Fałta, E. et al. The effect of selected risk factors, including the mode of delivery, on the development of allergic rhinitis and bronchial asthma. Adv. Dermatol. Allergol. 35, 267–273. https://doi.org/10.5114/ada.2018.76222 (2018).
Mitselou, N. et al. Adverse pregnancy outcomes and risk of later allergic rhinitis-Nationwide Swedish cohort study. Pediat. Allergy Immunol. 31, 471–479. https://doi.org/10.1111/pai.13230 (2020).
Renz-Polster, H. et al. Caesarean section delivery and the risk of allergic disorders in childhood. Clin. Experim. Allergy 35, 1466–1472. https://doi.org/10.1111/j.1365-2222.2005.02356.x (2005).
Salam, M. T. et al. Mode of delivery is associated with asthma and allergy occurrences in children. Ann. Epidemiol. 16, 341–346. https://doi.org/10.1016/j.annepidem.2005.06.054 (2006).
Jinmei, H., Jixian, L. & Xianhong, Z. Caesarean section and risk of allergic rhinitis and asthma in children. Chin. J. Pract. Pediat. 29, 713–717. https://doi.org/10.7504/ek2014090617 (2014).
Brandao, H. V. et al. Increased risk of allergic rhinitis among children delivered by cesarean section: A cross-sectional study nested in a birth cohort. BMC Pediatr. 16, 57. https://doi.org/10.1186/s12887-016-0594-x (2016).
Pistiner, M., Gold, D. R., Abdulkerim, H., Hoffman, E. & Celedon, J. C. Birth by cesarean section, allergic rhinitis, and allergic sensitization among children with a parental history of atopy. J. Allergy Clin. Immunol. 122, 274–279. https://doi.org/10.1016/j.jaci.2008.05.007 (2008).
Almqvist, C. & Oberg, A. S. The association between caesarean section and asthma or allergic disease continues to challenge. Acta Paediatr. 103, 349–351. https://doi.org/10.1111/apa.12562 (2014).
Saulyte, J., Regueira, C., Montes-Martínez, A., Khudyakov, P. & Takkouche, B. Active or passive exposure to tobacco smoking and allergic rhinitis, allergic dermatitis, and food allergy in adults and children: A systematic review and meta-analysis. PLoS Med. 11, e1001611. https://doi.org/10.1371/journal.pmed.1001611 (2014).
Hallit, S., Assi, T. B., Hallit, R. & Salameh, P. Allergic diseases, smoking, and environmental exposure among university students in Lebanon. J. Asthma 55, 35–42. https://doi.org/10.1080/02770903.2017.1306075 (2018).
Stokholm, J. et al. Delivery mode and gut microbial changes correlate with an increased risk of childhood asthma. Sci. Transl. Med. 12, 1. https://doi.org/10.1126/scitranslmed.aax9929 (2020).
Ficara, M. et al. Changes of intestinal microbiota in early life. J. Matern.-Fet. Neonatal. Med. 33, 1036–1043. https://doi.org/10.1080/14767058.2018.1506760 (2020).
Bisgaard, H. et al. Reduced diversity of the intestinal microbiota during infancy is associated with increased risk of allergic disease at school age. J. Allergy Clin. Immunol. 128, 646–652. https://doi.org/10.1016/j.jaci.2011.04.060 (2011).
Stroup, D. F. et al. Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA 283, 2008–2012. https://doi.org/10.1001/jama.283.15.2008 (2000).
Wang, X. et al. Reporting items for systematic reviews and meta-analyses of acupuncture: the PRISMA for acupuncture checklist. BMC Complem. Altern. Med. 19, 208. https://doi.org/10.1186/s12906-019-2624-3 (2019).
Miettinen, O. Estimability and estimation in case-referent studies. Am. J. Epidemiol. 103, 226–235. https://doi.org/10.1093/oxfordjournals.aje.a112220 (1976).
Zhang, J. & Yu, K. F. What’s the relative risk? A method of correcting the odds ratio in cohort studies of common outcomes. JAMA 280, 1690–1691. https://doi.org/10.1001/jama.280.19.1690 (1998).
Stang, A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur. J. Epidemiol. 25, 603–605. https://doi.org/10.1007/s10654-010-9491-z (2010).
Rostom, A., Dubé, C. & Cranne, A. Celiac Disease. https://www.ncbi.nlm.nih.gov/books/NBK35156/ (Evidence Reports/Technology Assessments, No. 104, 2004).
Ma, L. L. et al. Methodological quality (risk of bias) assessment tools for primary and secondary medical studies: What are they and which is better?. Mil. Med. Res. 7, 7. https://doi.org/10.1186/s40779-020-00238-8 (2020).
Zeng, X. et al. The methodological quality assessment tools for preclinical and clinical studies, systematic review and meta-analysis, and clinical practice guideline: a systematic review. J. Evid. Based Med. 8, 2–10. https://doi.org/10.1111/jebm.12141 (2015).
Hu, J. et al. Prevalence of suicide attempts among Chinese adolescents: A meta-analysis of cross-sectional studies. Compr. Psychiatry 61, 78–89. https://doi.org/10.1016/j.comppsych.2015.05.001 (2015).
Higgins, J. P., Thompson, S. G., Deeks, J. J. & Altman, D. G. Measuring inconsistency in meta-analyses. BMJ (Clin. Res. Ed.) 327, 557–560. https://doi.org/10.1136/bmj.327.7414.557 (2003).
Borenstein, M., Higgins, J. P., Hedges, L. V. & Rothstein, H. R. Basics of meta-analysis: I(2) is not an absolute measure of heterogeneity. Res. Synth. Methods 8, 5–18. https://doi.org/10.1002/jrsm.1230 (2017).
Higgins, J. P., Thompson, S. G. & Spiegelhalter, D. J. A re-evaluation of random-effects meta-analysis. J. R. Stat. Soc. Ser. A Stat. Soc. 172, 137–159. https://doi.org/10.1111/j.1467-985X.2008.00552.x (2009).
Nafstad, P., Magnus, P. & Jaakkola, J. J. Risk of childhood asthma and allergic rhinitis in relation to pregnancy complications. J. Allergy. Clin. Immunol. 106, 867–873. https://doi.org/10.1067/mai.2000.110558 (2000).
McKeever, T. M., Lewis, S. A., Smith, C. & Hubbard, R. Mode of delivery and risk of developing allergic disease. J. Allergy. Clin. Immunol. 109, 800–802. https://doi.org/10.1067/mai.2002.124046 (2002).
Negele, K. et al. Mode of delivery and development of atopic disease during the first 2 years of life. Pediat. Allergy Immunol. 15, 48–54. https://doi.org/10.1046/j.0905-6157.2003.00101.x (2004).
Park, Y. H. et al. Relationship between mode of delivery in childbirth and prevalence of allergic diseases in Korean children. Allergy Asthma Immunol. Res. 2, 28–33. https://doi.org/10.4168/aair.2010.2.1.28 (2010).
Li, Y. et al. Pre- and postnatal risk factors in relation to allergic rhinitis in school-aged children in China. PloS One 10, e0114022. https://doi.org/10.1371/journal.pone.0114022 (2015).
Chu, S. et al. Cesarean section without medical indication and risks of childhood allergic disorder, attenuated by breastfeeding. Sci. Rep. 7, 9762. https://doi.org/10.1038/s41598-017-10206-3 (2017).
Loo, E. X. L. et al. Associations between caesarean delivery and allergic outcomes: Results from the GUSTO study. Ann. Allergy Asthma Immunol. 118, 636–638. https://doi.org/10.1016/j.anai.2017.02.021 (2017).
Xiangying, Z., Chenguang, Q. & Guomin, N. Analysis of risk factors for allergic rhinitis in 4 to 14-year-old children in Wuhan. Chin. J. Child Health Care 26, 551–554. https://doi.org/10.11852/zgetbjzz2018-26-05-24 (2018).
Yu, B. et al. Prenatal and neonatal factors involved in the development of childhood allergic diseases in Guangzhou primary and middle school students. BMC Pediatr. 19, 479. https://doi.org/10.1186/s12887-019-1865-0 (2019).
Richards, M., Ferber, J., Li, D. K. & Darrow, L. A. Cesarean delivery and the risk of allergic rhinitis in children. Ann Allergy Asthma Immunol 125, 280–286. https://doi.org/10.1016/j.anai.2020.04.028 (2020).
Hu, Y. et al. Breastfeeding duration modified the effects of neonatal and familial risk factors on childhood asthma and allergy: a population-based study. Respir. Res. 22, 41. https://doi.org/10.1186/s12931-021-01644-9 (2021).
Meza-Lopez, C. et al. Prevalence of allergic diseases and their symptoms in schoolchildren according to the birth mode. Bol. Med. Hosp. Infant Mex. 78, 130–135. https://doi.org/10.2475/BMHIM.20000114 (2021).
Greenland, S. Quantitative methods in the review of epidemiologic literature. Epidemiol. Rev. 9, 1–30. https://doi.org/10.1093/oxfordjournals.epirev.a036298 (1987).
Siristatidis, C. et al. Controlled ovarian hyperstimulation for IVF: impact on ovarian, endometrial and cervical cancer—A systematic review and meta-analysis. Hum. Reprod. Update 19, 105–123. https://doi.org/10.1093/humupd/dms051 (2013).
Levine, T. R., Asada, K. J. & Carpenter, C. Sample Sizes and Effect Sizes are Negatively Correlated in Meta-Analyses: Evidence and Implications of a Publication Bias Against NonSignificant Findings. Commun. Monogr. 76, 286–302. https://doi.org/10.1080/03637750903074685 (2009).
Sigurdardottir, S. T. et al. Prevalence and early-life risk factors of school-age allergic multimorbidity: The EuroPrevall-iFAAM birth cohort. Allergy https://doi.org/10.1111/all.14857 (2021).
Kaplan, R. M., Chambers, D. A. & Glasgow, R. E. Big data and large sample size: a cautionary note on the potential for bias. Clin. Transl. Sci. 7, 342–346. https://doi.org/10.1111/cts.12178 (2014).
Thavagnanam, S., Fleming, J., Bromley, A., Shields, M. D. & Cardwell, C. R. A meta-analysis of the association between Caesarean section and childhood asthma. Clin. Experim. Allergy 38, 629–633. https://doi.org/10.1111/j.1365-2222.2007.02780.x (2008).
Papathoma, E., Triga, M., Fouzas, S. & Dimitriou, G. Cesarean section delivery and development of food allergy and atopic dermatitis in early childhood. Pediat. Allergy Immunol. 27, 419–424. https://doi.org/10.1111/pai.12552 (2016).
Koplin, J. et al. Is caesarean delivery associated with sensitization to food allergens and IgE-mediated food allergy: A systematic review. Pediatr. Allergy Immunol. 19, 682–687. https://doi.org/10.1111/j.1399-3038.2008.00731.x (2008).
Westman, M. et al. The link between parental allergy and offspring allergic and nonallergic rhinitis. Allergy 68, 1571–1578. https://doi.org/10.1111/all.12267 (2013).
Kristensen, K. & Henriksen, L. Cesarean section and disease associated with immune function. J. Allergy Clin. Immunol. 137, 587–590. https://doi.org/10.1016/j.jaci.2015.07.040 (2016).
Darabi, B., Rahmati, S., HafeziAhmadi, M. R., Badfar, G. & Azami, M. The association between caesarean section and childhood asthma: An updated systematic review and meta-analysis. Allergy Asthma Clin. Immunol. 15, 62. https://doi.org/10.1186/s13223-019-0367-9 (2019).
Strachan, D. P. Hay fever, hygiene, and household size. BMJ (Clin. Res. Ed.) 299, 1259–1260. https://doi.org/10.1136/bmj.299.6710.1259 (1989).
Levin, M. E. et al. Environmental factors associated with allergy in urban and rural children from the South African Food Allergy (SAFFA) cohort. J. Allergy Clin. Immunol. 145, 415–426. https://doi.org/10.1016/j.jaci.2019.07.048 (2020).
Han, Y.-Y., Forno, E., Badellino, H. A. & Celedón, J. C. Antibiotic use in early life, rural residence, and allergic diseases in Argentinean children. J. Allergy Clin. Immunol. Pract. 5, 1112–1118. https://doi.org/10.1016/j.jaip.2016.12.0254 (2017).
Morgan, B. W. et al. Asthma and allergic disorders in Uganda: A population-based study across urban and rural settings. J. Allergy Clin. Immunol. Pract. 6, 1580–1587. https://doi.org/10.1016/j.jaip.2017.11.032 (2018).
Campbell, B. et al. The effects of growing up on a farm on adult lung function and allergic phenotypes: an international population-based study. Thorax 72, 236–244. https://doi.org/10.1136/thoraxjnl-2015-208154 (2017).
Singh, K., Chang, C. & Gershwin, M. E. IgA deficiency and autoimmunity. Autoimmun. Rev. 13, 163–177. https://doi.org/10.1016/j.autrev.2013.10.005 (2014).
Cuppari, C. et al. Mode of delivery and risk for development of atopic diseases in children. Allergy Asthma Proc. 36, 344–351. https://doi.org/10.2500/aap.2015.36.3870 (2015).
Brooks, C., Pearce, N. & Douwes, J. The hygiene hypothesis in allergy and asthma: an update. Curr. Opin. Allergy Clin. Immunol. 13, 70–77. https://doi.org/10.1097/ACI.0b013e32835ad0d2 (2013).
Weber, J. et al. Asthma and the hygiene hypothesis: Does cleanliness matter?. Am. J. Respir. Crit. Care Med. 191, 522–529. https://doi.org/10.1164/rccm.201410-1899OC (2015).
Hunter, P. Public Health struggles to square hygiene with diversity: Research on the link between microbiomes and immune function puts the “hygiene hypothesis” to rest. EMBO Rep. 21, e51540. https://doi.org/10.1552/embr.202051540 (2020).
Shao, Y. et al. Stunted microbiota and opportunistic pathogen colonization in caesarean-section birth. Nature 574, 117–121. https://doi.org/10.1038/s41586-019-1560-1 (2019).
Montoya-Williams, D. et al. The neonatal microbiome and its partial role in mediating the association between birth by Cesarean section and adverse pediatric outcomes. Neonatology 114, 103–111. https://doi.org/10.1159/000487102 (2018).
Akagawa, S. & Kaneko, K. Gut microbiota and allergic diseases in children. Allergol. Int. 71, 301–309. https://doi.org/10.1016/j.alit.2022.02.004 (2022).
Alcazar, C.G.-M. et al. The association between early-life gut microbiota and childhood respiratory diseases: a systematic review. Lancet Microbe 3, e867–e880. https://doi.org/10.1016/s2666-5247(22)00184-7 (2022).
Dominguez-Bello, M. G. et al. Partial restoration of the microbiota of cesarean-born infants via vaginal microbial transfer. Nat. Med. 22, 250–253. https://doi.org/10.1038/nm.4039 (2016).
Zhou, Y. et al. Maternal tobacco exposure during pregnancy and allergic rhinitis in offspring: A systematic review and meta-analysis. Med. (Baltimore) 100, e26986. https://doi.org/10.1097/MD.0000000000026986 (2021).
Wooldridge, A. L. et al. Relationship between birth weight or fetal growth rate and postnatal allergy: A systematic review. J Allergy Clin Immunol 144, 1703–1713. https://doi.org/10.1016/j.jaci.2019.08.032 (2019).
Lunjani, N. et al. Recent developments and highlights in mechanisms of allergic diseases: Microbiome. Allergy 73, 2314–2327. https://doi.org/10.1111/all.13634 (2018).
Johnson, C. C. & Ownby, D. R. Allergies and Asthma: Do Atopic Disorders Result from Inadequate Immune Homeostasis arising from Infant Gut Dysbiosis?. Expert. Rev. Clin. Immunol. 12, 379–388. https://doi.org/10.1586/1744666X.2016.1139452 (2016).
Jelding-Dannemand, E., Malby Schoos, A. M. & Bisgaard, H. Breast-feeding does not protect against allergic sensitization in early childhood and allergy-associated disease at age 7 years. J Allergy Clin. Immunol. 136, 1302–1308. https://doi.org/10.1016/j.jaci.2015.02.023 (2015).
Nwaru, B. I. et al. Timing of infant feeding in relation to childhood asthma and allergic diseases. J. Allergy Clin. Immunol. 131, 78–86. https://doi.org/10.1016/j.jaci.2012.10.028 (2013).
Acknowledgements
We would like to acknowledge and thank the research team for Reproductive Health and Translational Medicine of Hunan Normal University (2023JC101) for their help in the codification process.
Funding
National Natural Science Foundation of China, Grant/Award Number: 81873861; Huxiang Young Talents project, Grant/Award Number: 2021RC3094; Hunan Provincial Science Fund for Distinguished Young Scholars, Grant/Award Number: 2022JJ10036;
Author information
Authors and Affiliations
Contributions
Z.L., J.L. and Y.D. designed the research protocol; Z.L. and J.C. selected studies to be included; Z.L., L.X. and X.L. curated the data; Z.L., Y.Z. and Y.Y. undertook the statistical analysis; Z.L. drafted the first manuscript. J.Z., H.S., M.T., Y.D. and J.L. reviewed and made contributions to the text. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liu, Z., Xie, L., Liu, X. et al. Cesarean section and the risk of allergic rhinitis in children: a systematic review and meta-analysis. Sci Rep 13, 18361 (2023). https://doi.org/10.1038/s41598-023-44932-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-44932-8
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.